Abstract
Background and Aims:
To establish the role of imaging in the diagnosis, staging, treatment planning, and surveillance of musculoskeletal tumours in orthopaedic oncology.
Methods:
A thorough literature search was carried out into the applications of conventional radiography, ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI), and nuclear medicine techniques. The strengths, limitations, and complementary contributions of each modality were reviewed in relation to diagnosis, staging, biopsy guidance, treatment response assessment, and surveillance.
Results:
A multidisciplinary approach—integrating clinicopathological assessment with multimodality imaging, maximises diagnostic accuracy and guides patient-specific management. Radiography remains the first-line investigation, offering initial lesion characterisation and narrowing the differential. US provides high-resolution evaluation of superficial soft tissue lesions and enables real-time image-guided biopsy. CT offers superior depiction of cortical integrity, osseous destruction, and matrix mineralisation, with advanced dual-energy techniques improving material characterisation and reducing metal artefacts. MRI is the gold standard for local staging, with advanced sequences such as diffusion-weighted imaging, dynamic contrast-enhanced MRI, and chemical shift imaging enhancing lesion characterisation and treatment response assessment. Established and emerging nuclear medicine techniques provide complementary functional information, enabling early detection of metastases and metabolic treatment response evaluation.
Conclusion:
This review outlines the strengths, limitations, and complementary contributions of these modalities in optimising the diagnostic and treatment pathways for orthopaedic oncology patients. It therefore highlights how imaging in orthopaedic oncology is inherently multimodal, with each modality contributing unique and complementary diagnostic insights.
Keywords
Introduction
Imaging is an essential tool in the management of patients in the orthopaedic oncological setting. The combination of comprehensive clinical assessment and patient-specific imaging can facilitate accurate diagnosis and staging, guide management, aid in evaluating for treatment response, as well as surveil for tumour recurrence.[1] There are various imaging modalities available, each with its own unique strengths, offering complementary diagnostic information. Following thorough history-taking and clinical examination, radiographs are usually the first-line imaging investigation, with cross-sectional modalities, magnetic resonance imaging (MRI) and computed tomography (CT), sometimes subsequently indicated, based on the radiographic findings.[2] Ultrasonography (US) is increasingly being used as initial imaging, particularly for palpable superficial lesions, due to its excellent soft tissue resolution; however, its role in the accurate characterisation and diagnosis of deep lesions remains limited.[3] Nuclear medicine, comprising scintigraphy and positron emission tomography (PET), is a functional imaging method with a multitude of uses. Depending on the particular radiotracer selected, locations of different metabolic processes can be demonstrated and quantified, including osteoblastic activity, glucose metabolism and sodium-potassium pump activity.[2,4] This review will systematically review the current roles, indications, and complementary diagnostic contributions of these modalities in the evaluation and management of musculoskeletal tumours.
Imaging Modalities in Orthopaedic Oncology
Radiography
Conventional radiography, or plain film, is recommended as the initial investigation for assessing suspected bone tumours or musculoskeletal pathology in the broader sense. This modality is able to demonstrate the anatomical location of a lesion, depict its margin, density and any internal matrix mineralisation, as well as the presence of any periosteal reaction.[5] The radiographic appearance of a lesion, coupled with patient demographic data, clinical history and whether the lesion is solitary or there are multiple, can hugely narrow the list of differential diagnoses. Through this widely available and relatively inexpensive initial examination, the need for further imaging, follow-up and management can be planned in a more focused way.[1,6] Radiography is not without limitation, however, as it is relatively insensitive at detecting lesions in anatomic regions with multiple overlapping structures, such as the spine and pelvis. Moreover, in the context of osseous metastases, small or early metastatic lesions may go unrecognised, given that a 30%-50% bone density loss must occur for their radiographic demonstration.[1,7] Similarly, radiographs have poor soft tissue contrast, only rarely differentiating between the densities of water and fat if the projection allows.[5]
Different lesions have predilections for either flat or long bones, and the radiographic location of a lesion within the latter of these two can narrow the differential. Many lesions tend to arise in haematopoietic marrow regions, such as Ewing sarcoma, lymphoma, myeloproliferative processes and metastases.[5] In the skeletally mature population, the majority of red marrow is present in the axial skeleton, which is less sensitively assessed by radiographs. For this reason, cross-sectional modalities, such as CT or MRI, are often utilised to assess lesions in these regions. The transverse and longitudinal locations of a lesion along a long bone, however, can be accurately assessed by radiographs obtained in orthogonal planes, generally anterior-posterior and lateral. With regard to the transverse location, a bone lesion can be juxtacortical, intracortical, eccentric intramedullary or central intramedullary, and it can be epiphyseal, metaphyseal or diaphyseal longitudinally.[5] The majority of lesions arise in the metaphyses of long bones, and therefore the differential is most narrowed when posed with an epiphyseal or, to a lesser extent, diaphyseal lesion. Radiographic characteristics of the lesions which suggest a non-aggressive process include: a narrow zone of transition, absence of periosteal reaction and lack of an extraosseous soft tissue component [Figure 1]. A wide zone of transition or the presence of either a periosteal reaction or soft tissue component are radiographic features concerning for an aggressive process.[8]
Leiomyosarcoma of bone. Sequential sagittal radiographs of the proximal lower leg demonstrating a mildly expansile, well-defined lucent lesion with endosteal scalloping and no soft tissue component (arrows), within the proximal third of the tibial diaphysis (a). This lesion increased in size over time (b) and was subsequently prophylactically nailed (c) due to metastases precluding curative surgery
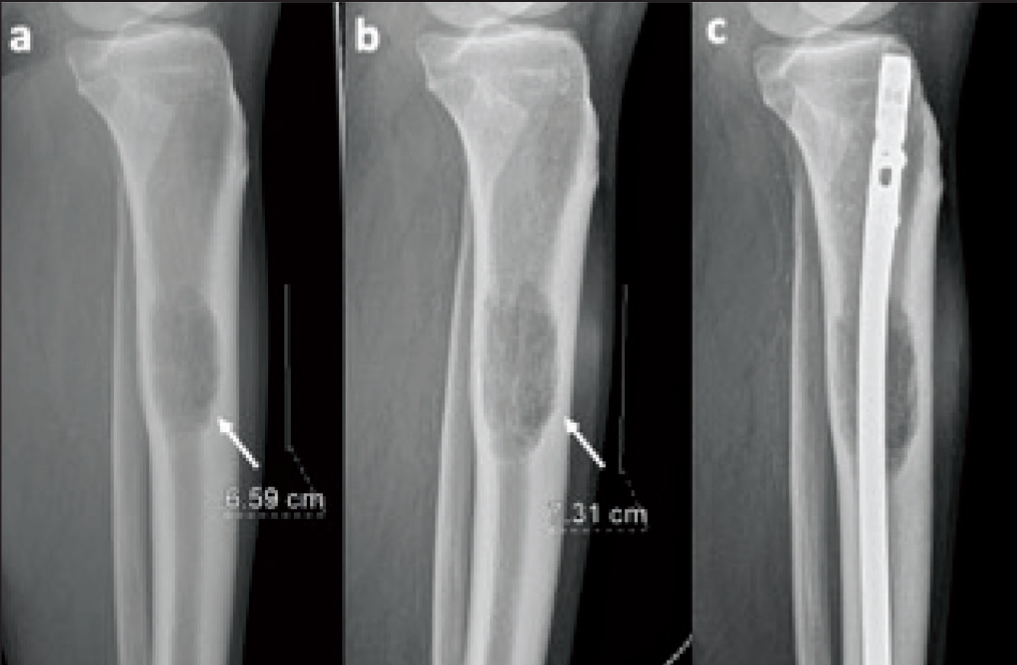
Sarcoma has a propensity for metastasising to the lungs, and therefore chest radiographs (CXR) are a major tool in surveillance for pulmonary disease. While radiographs are less sensitive than CT for small pulmonary nodules, it has been suggested that surveilling patients post-sarcoma resection with CXR does not result in worse overall survival, with the additional benefits of reduced radiation burden and optimisation of resources.[9]
Ultrasonography
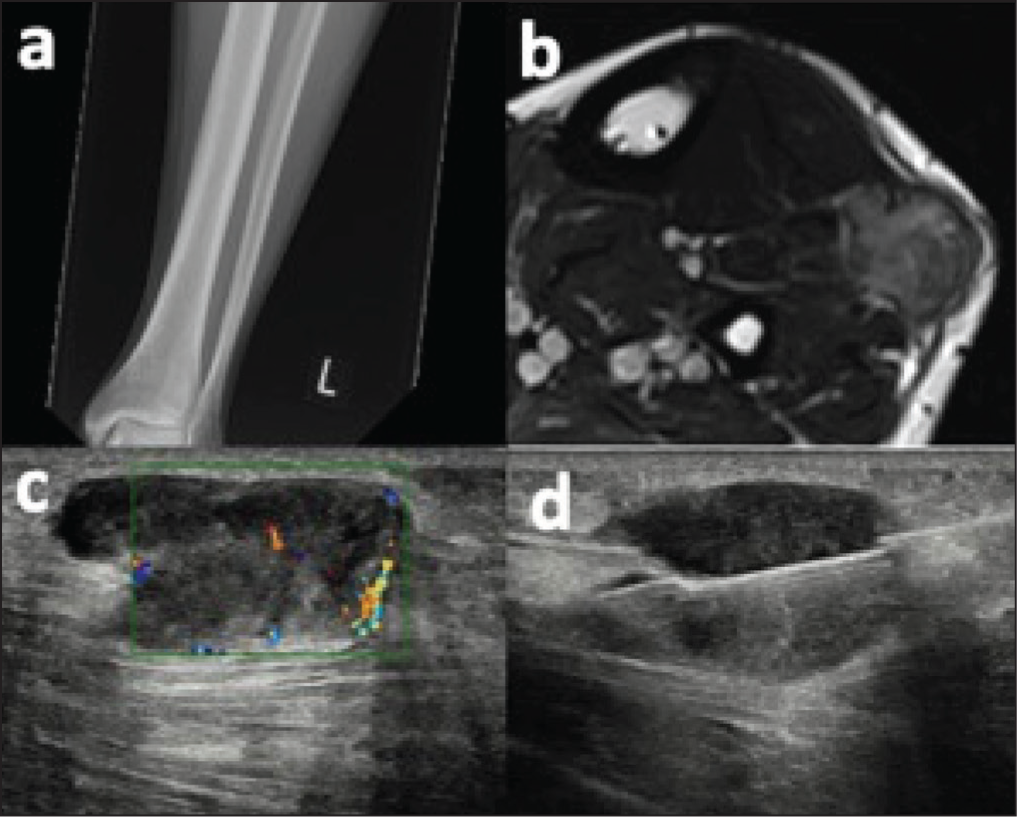
US can either be used diagnostically as a standalone modality, particularly in superficial soft tissue lesions [Figure 2], or in conjunction with others, in cases of deeper or calcified lesions.[3] In addition to its low cost and accessibility, other benefits of US include its ability to accurately differentiate between solid and cystic lesions, along with its dynamic capabilities, allowing assessment for compressibility and vascularity.[2] Additionally, US can be used to guide percutaneous closed soft tissue biopsy, particularly in cases where the biopsy target is small or close to other critical structures, such as neurovascular bundles or tendons.[4] In dedicated sarcoma clinics, US permits prompt diagnostic workup and streamlines patient care. When US is performed by experienced musculoskeletal radiologists, it can achieve high sensitivity (89%), specificity, and negative predictive value (97%) in distinguishing benign from malignant lesions, as well as facilitating immediate ultrasound-guided biopsy when necessary [Figure 3]. This one-stop-shop approach reduces diagnostic delays, unnecessary appointments, and patient anxiety, all while supporting efficient triage to the appropriate care pathways.[10] While the utility of US in the soft tissue setting cannot be overstated, it is a user-dependent modality in that the diagnostic utility of the scan depends on the proficiency of the individual scanning. Furthermore, inherent physical features of the US limit its use in assessing deep or calcified lesions, along with bone tumours, due to the inability of the ultrasonographic waves to penetrate to the tissues of interest.
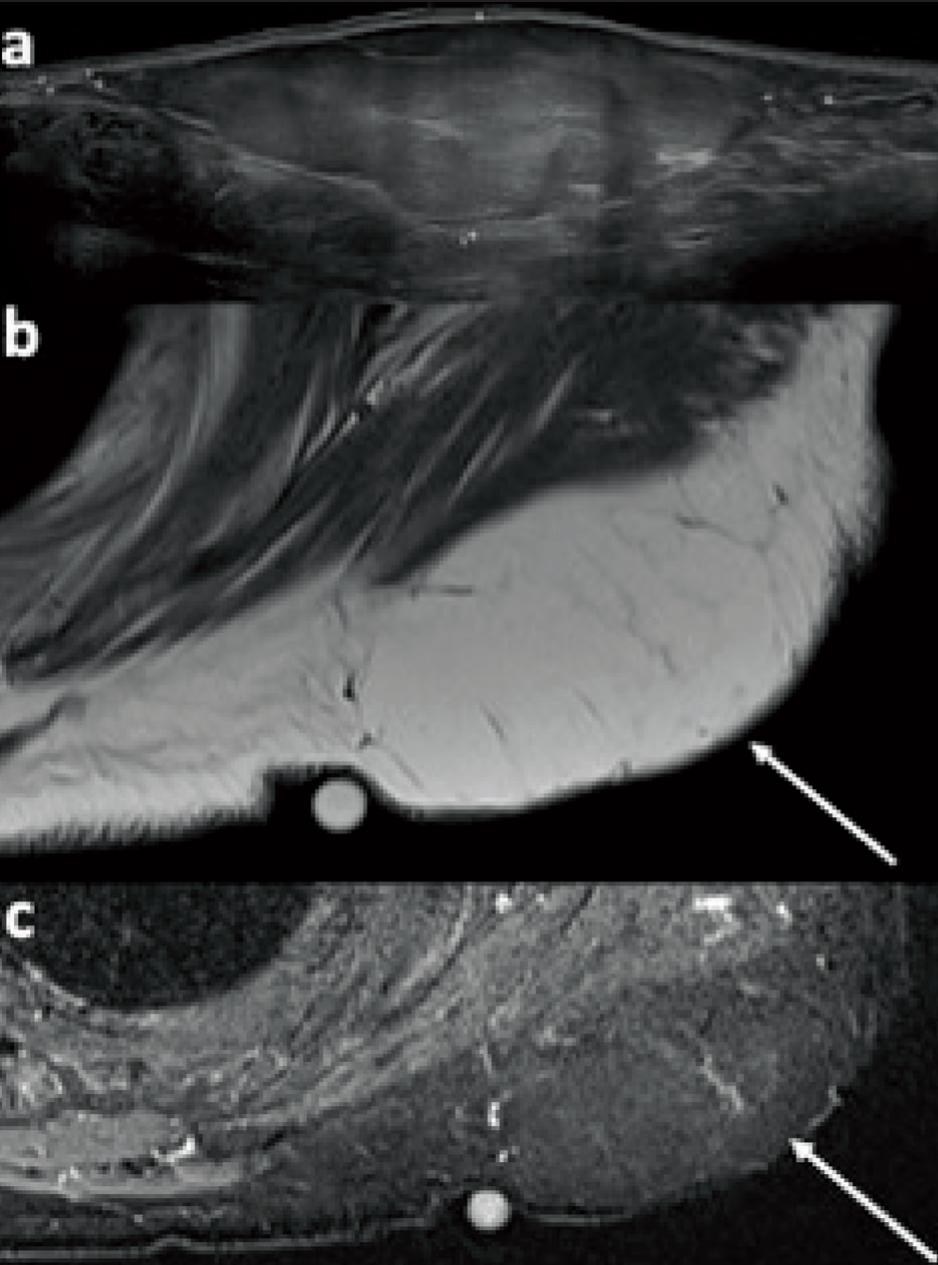
Subcutaneous lipoma. Selected ultrasound (a), transverse T1-weighted (b) and STIR (c) MR images demonstrate a large subcutaneous lipoma (arrows) which abuts and exerts mass effect on the underlying posterior deltoid without invasion. No non-lipomatous components or thickened septae are present, and there is homogeneous fat suppression
Synovial sarcoma. Anteroposterior radiograph of the distal lower leg (a) demonstrates no abnormality. Transverse T2-weighted MRI (b) demonstrates a heterogeneous solid lesion at the anterior compartment of the mid-lower leg, involving the extensor digitorum longus and peroneus longus tendons. Ultrasound demonstrates a solid hypoechoic lesion with internal vascularity (c), which was biopsied under ultrasound guidance (d)
Computed Tomography
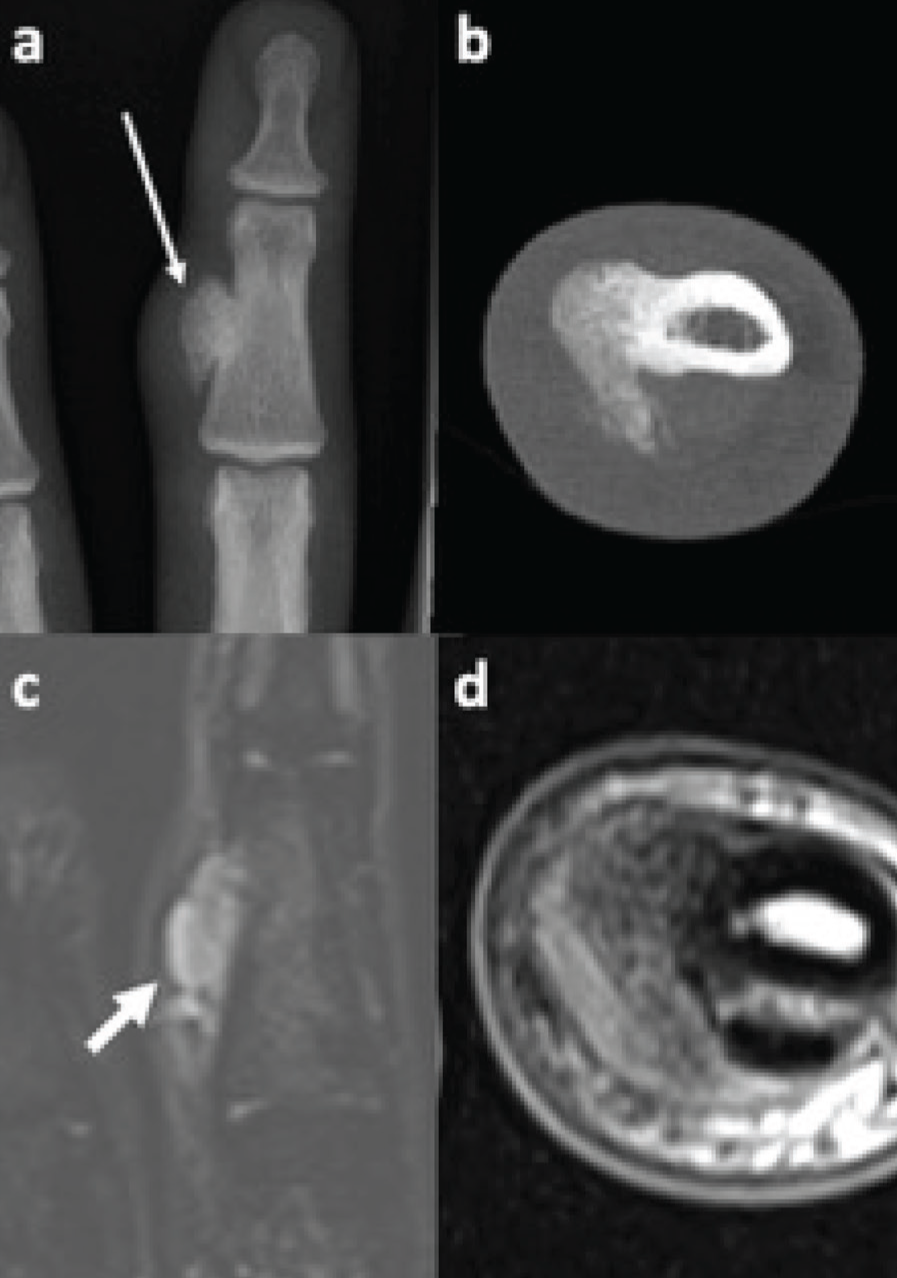
CT remains a useful adjunct to US and MRI in the radiological evaluation of musculoskeletal tumours, due to its ability to provide high-resolution cross-sectional detail. CT is particularly of value in delineating the degree of osseous destruction, any cortical involvement, and the presence of matrix mineralisation, all of which are key features for tumour characterisation [Figure 4]. Volumetrically acquired CT images can also be reconstructed in three dimensions, which is of use in surgical planning. The sensitivity of CT in the detection of subtle matrix mineralisation is superior to that of radiographs and MRI.[11] Dual-energy CT (DECT), which is becoming increasingly available, uses photons at two different energy levels when obtaining images. Following this, via post-processing algorithms, information about the composition of lesions can be provided by exploiting the different linear attenuation coefficients of materials at different photon energy levels.[1,12] The most commonly used post-processing algorithms are those for monosodium urate crystal detection and tophaceous gout quantification, a process which is commonly mistaken for tumour, and to create bone marrow oedema maps, which otherwise are completely imperceptible on conventional CT images.[13] An additional use of post-processed DECT is for metal artefact reduction, which is of particular use in patients with endoprosthetic reconstructions, following limb salvage surgeries as part of their orthopaedic oncological management.[13] The use of CT in distant staging of orthopaedic oncology will be discussed in the subsequent nuclear medicine section, given its use alongside PET imaging.
Bizarre parosteal osteochondromatous proliferations. Dorsopalmar radiograph and transverse CT of the middle finger (a, b) demonstrate a pedunculated ossified lesion arising from the radial cortex of the middle phalanx (long arrow). The lesion displays high signal on the coronal fluid-sensitive MRI (c) and intermediate signal on the T1-weighted MRI (d)
Aside from the diagnostic uses of CT, this modality can also be used to guide image-guided intervention. By performing intermittent limited scans, reducing the dose to the patient, a needle can be accurately positioned into locations which would otherwise be inaccessible through closed techniques. Orthopaedic oncological interventions, which can be performed under CT guidance, include biopsy, ablation and cementoplasty.[14]
The limitations of CT are its relatively poor soft tissue contrast compared to US and MRI, and limited sensitivity for early marrow infiltration. While DECT shows promise in the assessment of bone marrow oedema, this technique in isolation has not yet become a standard for locoregional assessment.
Magnetic Resonance Imaging
MRI is widely accepted as the imaging modality of choice for both diagnosis and local staging in orthopaedic oncology when an indeterminate or aggressive lesion is identified due to its exceptional soft tissue contrast, multiplanar capabilities and absence of ionising radiation.[3] MRI can delineate tumour margins as well as identify bone marrow involvement and extraosseous soft tissue components [Figure 5]. Involvement of adjacent structures, such as the neurovascular bundle, different muscle compartments and physis, can also be accurately depicted by MRI.[4] Regarding local staging, intra-articular extension of a tumour has been reported to be overestimated around the knee by MRI, and must be correlated with clinical features, given the differing surgical methods for intra- and extra-articular tumours.[15] MRI is the primary modality utilised in assessing for osseous oedema, due to its sensitivity for changes in water content. By using different pulse sequences, acquired in different anatomical planes, the differing physical properties of a lesion and the surrounding normal structures can be exploited to produce images of high diagnostic value.[12]
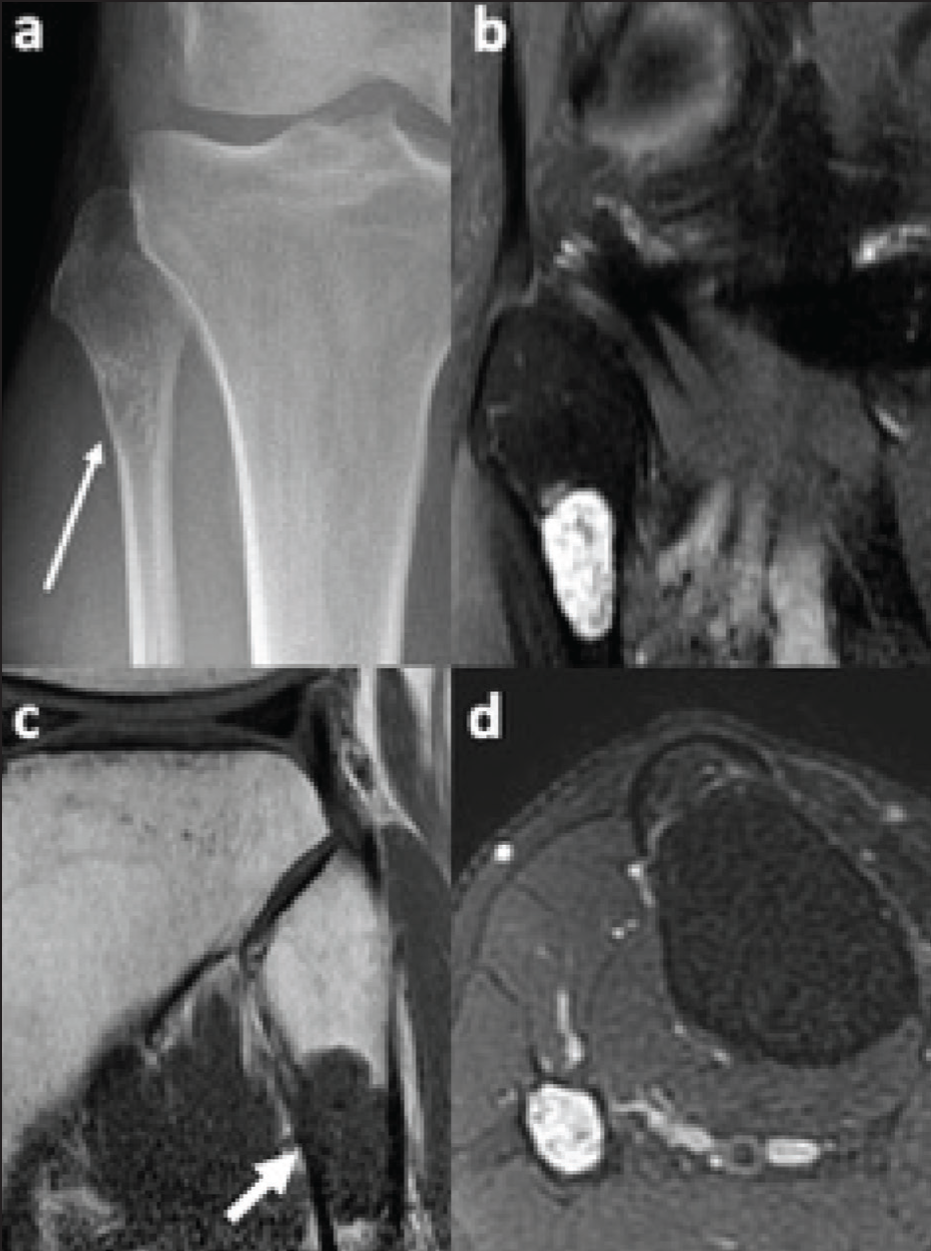
Central cartilage tumour. Anteroposterior radiograph (a) of the proximal lower leg demonstrating a lucent lesion at the proximal fibular metaphysis, with a narrow zone of transition, internal chondroid matrix calcification and endosteal scalloping (long arrow). Sagittal fluid-sensitive (b) and T1-weighted (c) and transverse fluid-sensitive (d) MRI images confirm the chondroid nature of this lesion and more accurately demonstrate the diffuse endosteal scalloping. No cortical breach or adjacent soft tissue oedema is demonstrated
Advanced MRI techniques, which also add value in orthopaedic oncology, include diffusion-weighted imaging (DWI), dynamic contrast-enhanced (DCE) MRI and chemical shift imaging. DWI measures the amount of Brownian motion water molecules display in their local environments, a principle which is exploited to characterise the cellularity of various lesions.[16] This information is useful in the initial diagnostic setting, when taken in combination with the clinical picture and other radiological findings, to narrow the differential. DWI, along with its complementary apparent diffusion coefficient (ADC) map, is also valuable in the assessment of tumour response to treatment. Tumours with areas of increased ADC values following treatment are suggestive of regions of necrosis, secondary to a response to treatment.[16]
DCE-MRI is a dynamic imaging technique in which multiple MR sequences are acquired at different time points, following the administration of an intravenous contrast agent, allowing for calculation of rates of enhancement and washout of regions of interest, usually set about the lesion in question.[17] While there is an overlap, in general, benign lesions demonstrate late contrast enhancement, owing to their relative lack of neovascularity, and malignant lesions demonstrate early arterial enhancement, followed by plateau or washout.[17] It has been suggested that, in addition to characteristics demonstrated by conventional MRI, DCE patterns are able to suggest whether a soft tissue sarcoma is low- or high-grade.[18,19] Chemical shift imaging is a non-contrast technique which employs the difference in precession frequencies of protons associated with water and fat molecules, to identify the presence of microscopic fat in lesions. This is achieved by acquiring in- and out-of-phase images, relating to the water and fat protons phase of precession, and assessing for a decreased signal intensity on the out-of-phase image.[20] This is useful for identifying whether a lesion has replaced normal fat-containing marrow constituents, suggesting malignancy, or contains microscopic fat, suggesting benignity.[20,21]
One of the limitations of MRI is its poor demonstration of cortical bone and calcification, due to the highly organised ultrastructure of these substances, resulting in very short relaxation times (TR). While radiographs and CT serve as more sensitive modalities for assessment of these tissues, novel MRI sequences with ultrashort- (UTE) or zero-echo-times (ZTE) are able to produce near-isotropic CT-like images in relatively short scanning times.[22] Given the agreeability between these novel MRI sequences and CT images, and the lack of ionising radiation associated with the former, it is likely that UTE/ZTE techniques will be employed in the future, particularly in situations where radiation burden is of particular concern, for example, in paediatric and pregnant populations.
Whole-body MRI (WB-MRI) is an alternative distant staging modality to bone scintigraphy and FDG-PET/CT, discussed subsequently, in which coronal short-tau inversion recovery and T1-weighted and transverse diffusion images are obtained of the whole body [Figure 6], across multiple large-field-of-view sequences.[7] Studies show WB-MRI as being at least comparable to bone scintigraphy, without the associated radiation burden; however, the sensitivity of WB-MRI for identifying rib lesions specifically has been shown to be poorer than both scintigraphy and conventional CT, likely relating to respiratory motion.[23]
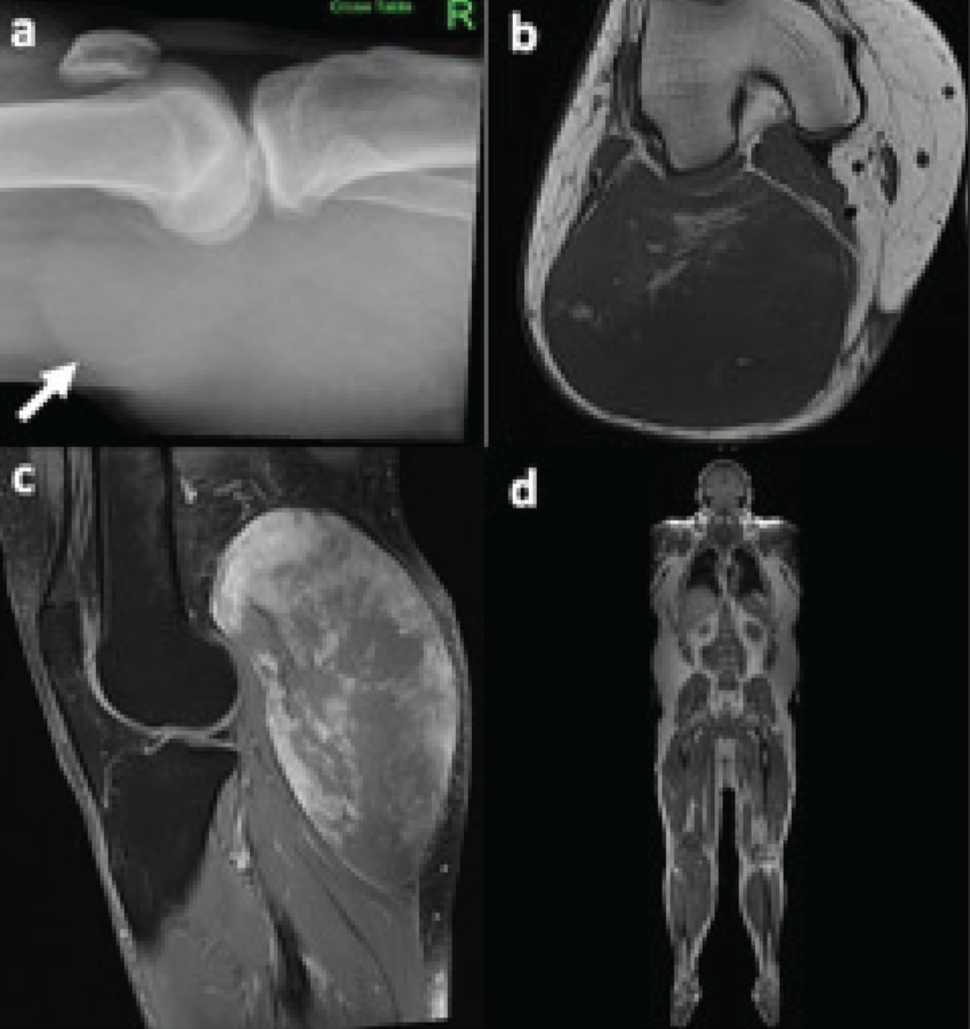
Myxoid liposarcoma. Horizontal beam lateral radiograph of the knee (a) demonstrates a large soft tissue mass in the popliteal fossa (arrow), without evidence of underlying osseous involvement. Transverse T1-weighted (b) and sagittal fluid-sensitive MRI (c) demonstrate a large solid heterogeneous lesion with internal foci of fat, superficial to the lateral head of gastrocnemius origin. A whole-body MRI (d) was performed for staging, which demonstrated no evidence of distant metastases
Nuclear Medicine
Nuclear medicine is a facet of radiology which combines anatomical and functional imaging techniques. Depending on the selected radiotracer administered, studies can identify regions with increased osteoblastic activity, glucose metabolism or sites of specific metastases, for example, in prostate cancer, to name but a few of the examinations under the nuclear medicine umbrella. Bone scintigraphy, or bone scan, is a study in which methylene diphosphonate is radiolabelled with technetium 99-m and administered intravenously to the patient. This tracer accumulates at sites of increased osteoblastic activity, and as the accumulated radiotracer decays, photons are emitted, which display as corresponding regions of high tracer uptake on planar images acquired by a gamma camera.[24] Bone metastases are the most common malignant bone tumour, and scintigraphy is much more sensitive than radiographs in identifying osteoblastic lesions, only requiring between 5% and 10% change in the ratio of lesion to normal bone, in order to be demonstrated.[24] Historically, in the context of bone sarcoma, bone scintigraphy and thoracic imaging, either by radiograph or CT, formed the mainstay of evaluation for distant sites of disease; however, bone scintigraphy has now been largely supplanted by FDG-PET/CT and whole-body MRI for staging and surveillance.[25] Scintigraphy remains the most cost-efficient of these modalities, but its relatively poor specificity and the superior anatomic detail provided by the cross-sectional hybrid techniques have contributed to this paradigm shift.
Fluorodeoxyglucose (FDG)-PET/CT is complementary to MRI and anatomic CT in the sarcoma setting, and is able to identify both the primary lesion as well as distant sites of disease, which may be occult on conventional CT [Figures 7 and 8]. Additionally, FDG-PET/CT can also be used to identify the most metabolically active area of a lesion, allowing for this to be targeted during biopsy, in order to maximise the likelihood of obtaining a diagnostic sample.[26] The degree of FDG uptake, or standard uptake value (SUV), is generally greater in higher-grade lesions within the same histological type; for example, enchondromas and atypical cartilaginous tumours show lower SUVs than high-grade and dedifferentiated chondrosarcomas for example.[27] Given that metabolic changes occur sooner than anatomic changes during treatment, FDG-PET/CT has been reported as useful in early treatment response assessment, with a reduction in activity reflecting a response to treatment.[26] Rather than being of use in the initial diagnostic stages, FDG-PET/CT has emerged as a key modality at the point of distant staging, with its increased sensitivity compared with conventional CT.[27]
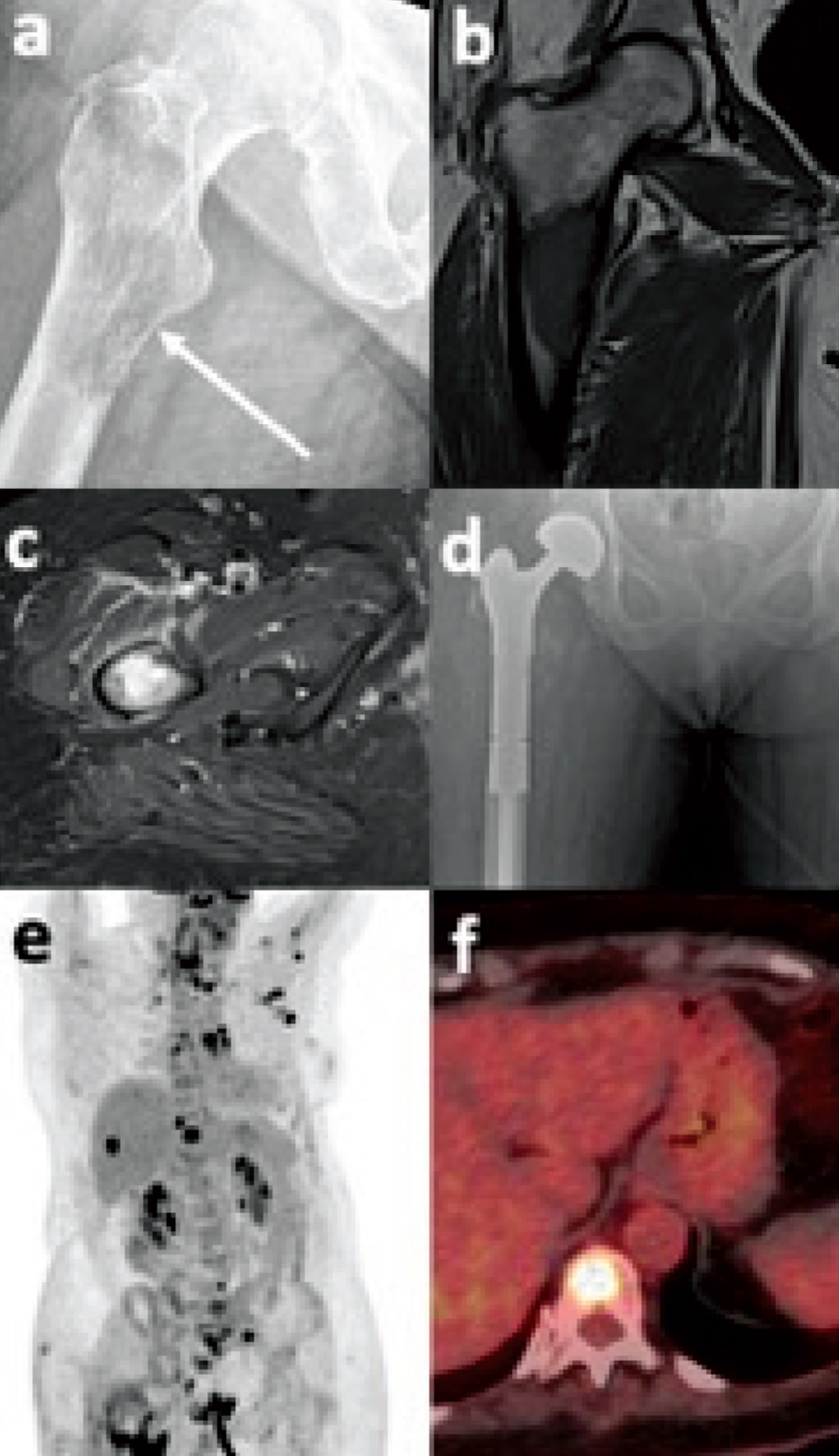
Metastatic adenocarcinoma of the lung. Anteroposterior radiograph of the proximal femur (a) demonstrates an ill-defined lucent metaphyseal lesion with a permeative pattern of medial cortical destruction (arrow). Coronal T1-weighted (b) and transverse fluid-sensitive images demonstrate the lesion involving greater than two-thirds of the bone diameter and eroding the medial calcar. The patient subsequently underwent a proximal femoral replacement (d) following a pathological fracture through this lesion. Maximum intensity projection (e) and fused PET/CT (f) demonstrate multifocal nodal and osseous metastases, and a T11 vertebral body metastasis, respectively, as areas of increased tracer uptake
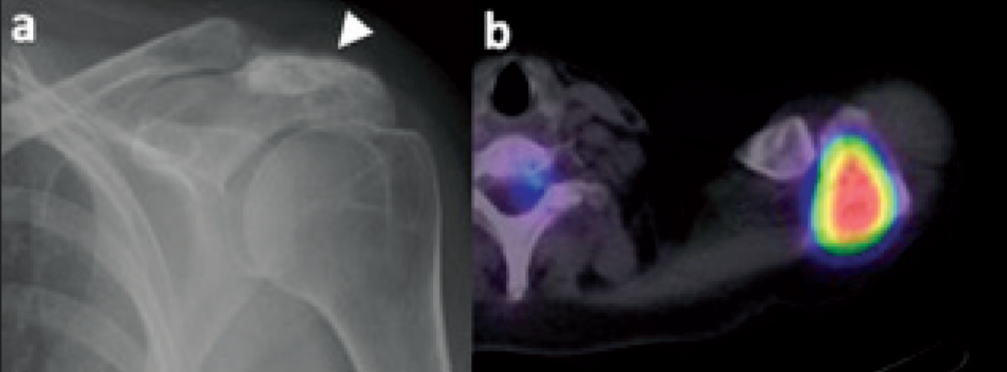
Osteoblastic acromial metastasis. Anteroposterior radiograph of the shoulder (a) demonstrates increased sclerosis and expansion of the acromion process of the scapula (arrowhead). Fused transverse PET/CT image (b) demonstrates markedly increased tracer uptake at the scapular acromion process
FDG-PET/MRI is a relatively new hybrid modality, having been in routine use for the last decade, which combines whole-body MRI with the functional component of PET. The main benefit of FDG-PET/MRI compared with its CT counterpart is the absence of radiation from the anatomical imaging (MRI) component, making it useful for young patients who require long-term whole-body surveillance.[27] In terms of local staging of soft tissue sarcoma, it has been shown that FDG-PET/MRI, in comparison to conventional MRI, offers information regarding the relationship of tumour to adjacent bone or nerves, the invasion of which could lead to a drastically different surgical approach with increased morbidity.[26] While there are reported benefits of this modality, image acquisition is much longer than that of FDG-PET/CT, and it is not widely available at the majority of centres, limiting its accessibility. There is limited literature regarding the value of FDG-PET/MRI for the staging and follow-up of primary osseous malignancies.[28]
Conclusions
The optimal imaging strategy in orthopaedic oncology is inherently multimodal, utilising the complementary strengths of each modality with the aim of establishing an understanding of tumour biology, behaviour, and anatomy. While radiography remains, and will likely continue as, the gold standard for initial bone lesion evaluation, advances in imaging technology and the advent of artificial intelligence will likely have increasingly important roles in the evaluation of bone lesions. US and CT offer valuable targeted insights, particularly in guiding interventions and the latter in assessing osseous architecture, and MRI remains indispensable for local staging and characterisation. Functional imaging has a key role in staging, restaging, and surveilling, and is able to offer earlier detection of disease progression than anatomical imaging alone. An integrated, patient-specific imaging approach, coordinated via the multidisciplinary team, remains essential for achieving accurate diagnosis, ratified surgical planning, and optimal outcomes long-term in orthopaedic oncology.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Not required.
Informed consent
Informed consent was obtained from the patient.
Credit author statement
Conception and design, or acquisition of data, or analysis and interpretation of data = NR, WJR.
Design, or acquisition of data, or analysis and interpretation of data = NR, WJR.
Drafting the article or revising it critically for important intellectual content = NR, HU, SK, SM, WJR.
Final approval of the version to be published = NR, HU, SK, SM, WJR.
Data availability
Data are available to share on request.
Use of artificial intelligence
No AI was used.
