Abstract
Objective:
This case report demonstrates the potential of dual tracer imaging (fluorodeoxyglucose + dihydroxyphenylalanine (FDG + DOPA)) positron emission tomography/magnetic resonance imaging (PET/MRI) brain in diagnosing and distinguishing the patients with multiple system atrophy-Parkinsonian type (MSA-P) from atypical Parkinsonism.
Methods:
A 65-year-old male presented with postural instability, urinary symptom in form of urgency, hesitancy, precipitancy followed by incontinence, slowness of all activity of daily living (ADL) with stiffness of all limbs, slurred speech and difficulty in swallowing for four years. It was clinically suspected as atypical Parkinson’s disease. The DOPA PET/MRI revealed asymmetric reduced patchy uptake in bilateral striatum qualitatively and quantitatively more prominently in bilateral putamen (right> left—maximum standardised uptake value (SUVmax): putamen left 2.18; right 1.91; caudate nucleus left 2.71; right 2.62). Bilateral putamen is atrophic with heterogeneous signal changes. Midbrain Pons ratio measures 0.6 (reported threshold of 0.52).
Results:
All of these features when put together help us to point it towards the MSA-P which was further confirmed by 18F-FDG study which showed regional FDG hypometabolism in bilateral putamen with relative sparing bilateral caudate, palladium and thalamus.
Conclusion:
Dual tracer PET/MRI will be the key to reliable uses in the diagnosis of clinically suspected neurodegenerative disorder and accurate prediction of therapeutic intervention.
Objective
The aim of this study was to demonstrate the clinical utility of dual-tracer imaging (fluorodeoxyglucose + dihydroxyphenylalanine (FDG + DOPA)) positron emission tomography magnetic resonance imaging (PET/MRI) brain in the diagnosis and management of multiple system atrophy-Parkinsonian type (MSA-P) and differentiation from other atypical parkinsonian syndromes (APS).
Introduction
MSA-P manifests with parkinsonian motor symptoms and additional features such as cortical dysfunction (e.g., cognitive deficits, apraxia), pseudobulbar signs and cerebellar or pyramidal involvement.[1] Fluorodopa (F-DOPA) PET serves as an indicator of dopaminergic neuron loss within the substantia nigra.[2,3] In contrast, FDG PET/MRI identifies metabolic deficits that may emerge before structural brain atrophy is detectable on conventional imaging.[4] The topography of hypometabolism aids in differentiating different APS, such as corticobasal degeneration (CBD), dementia with Lewy bodies (DLB), progressive supranuclear palsy (PSP) or MSA.[5,6]
This case report illustrates the clinical utility of dual-tracer (FDG and DOPA) PET/MRI brain imaging in diagnosing MSA‑P and differentiating it from other APS.
Case Presentation
A 65-year-old male presented with postural instability for the past four years. The patient developed progressive urinary dysfunction (urgency, hesitancy, followed by incontinence) over the last three years. Tremulousness of the bilateral upper limbs, slowness of daily activities and generalised limb stiffness were present for the last three years. Slurred speech was noted over the same period. He developed rapid eye movement (REM) sleep behaviour disorder (acting out dreams) and dysphagia (choking on both solids and liquids) over the last two years.
On examination (O/E):

Discussion
This case demonstrates how FDOPA and FDG PET/MRI can enhance differentiation of MSA-P from other APS. In our patient, the asymmetric reduction in DOPA uptake (right > left) within bilateral putamen and relative preservation of caudate nucleus uptake, along with midbrain-pons ratio of 0.6, pointed towards MSA-P. Previous studies demonstrate that MSA-P typically shows bilateral, symmetric or mildly asymmetric reduction in putaminal uptake, associated with putaminal atrophy and hypointensity on MRI [Figure 1].[5]
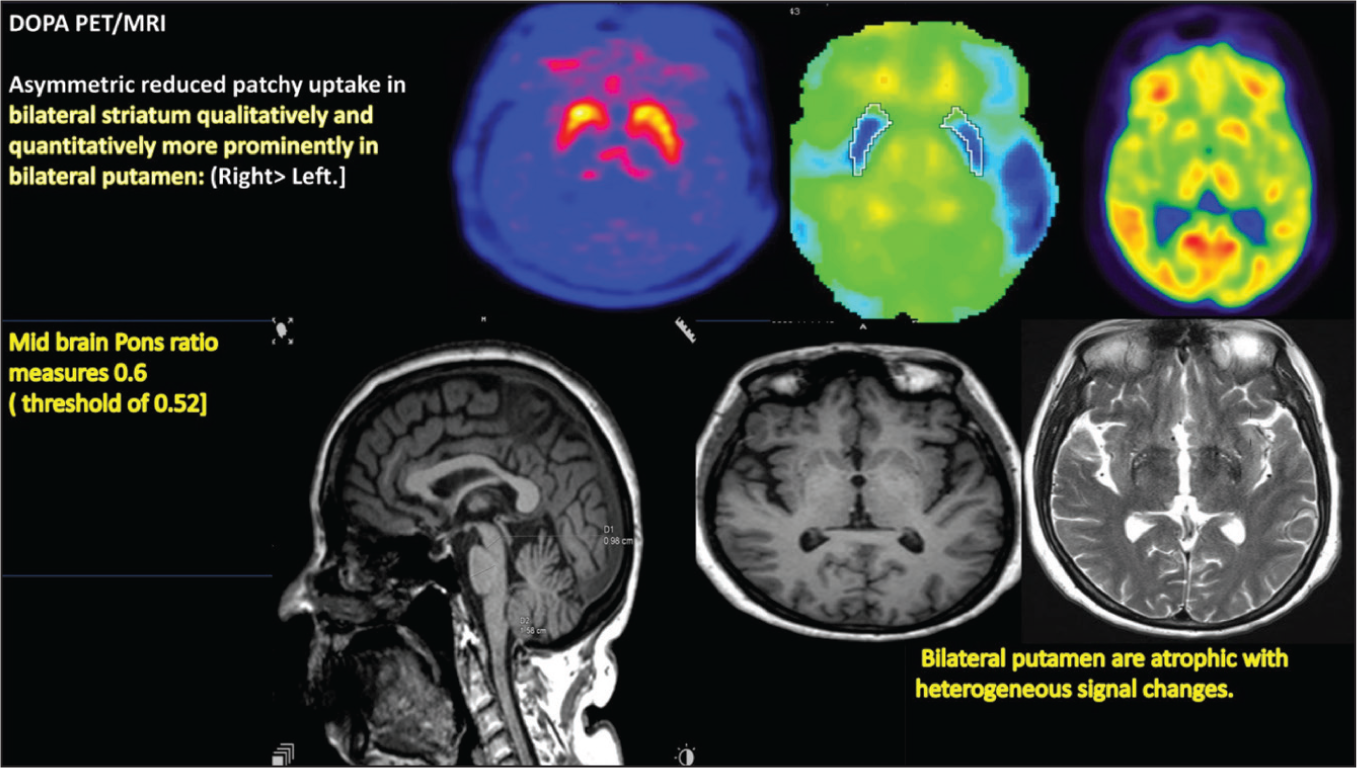
A comparative pattern of dual tracer PET/MRI in atypical Parkinsonism is summarised in Table 1. Compared to other reports, our findings align with FDOPA PET indicating presynaptic dopaminergic loss,[2,3] while FDG PET highlights cortical metabolic changes. Our case’s isolated bilateral putaminal hypometabolism without diffuse cortical involvement is characteristic of MSA-P,[5,6] whereas CBD exhibits asymmetric cortical hypometabolism involving frontal and parietal lobes.[5]
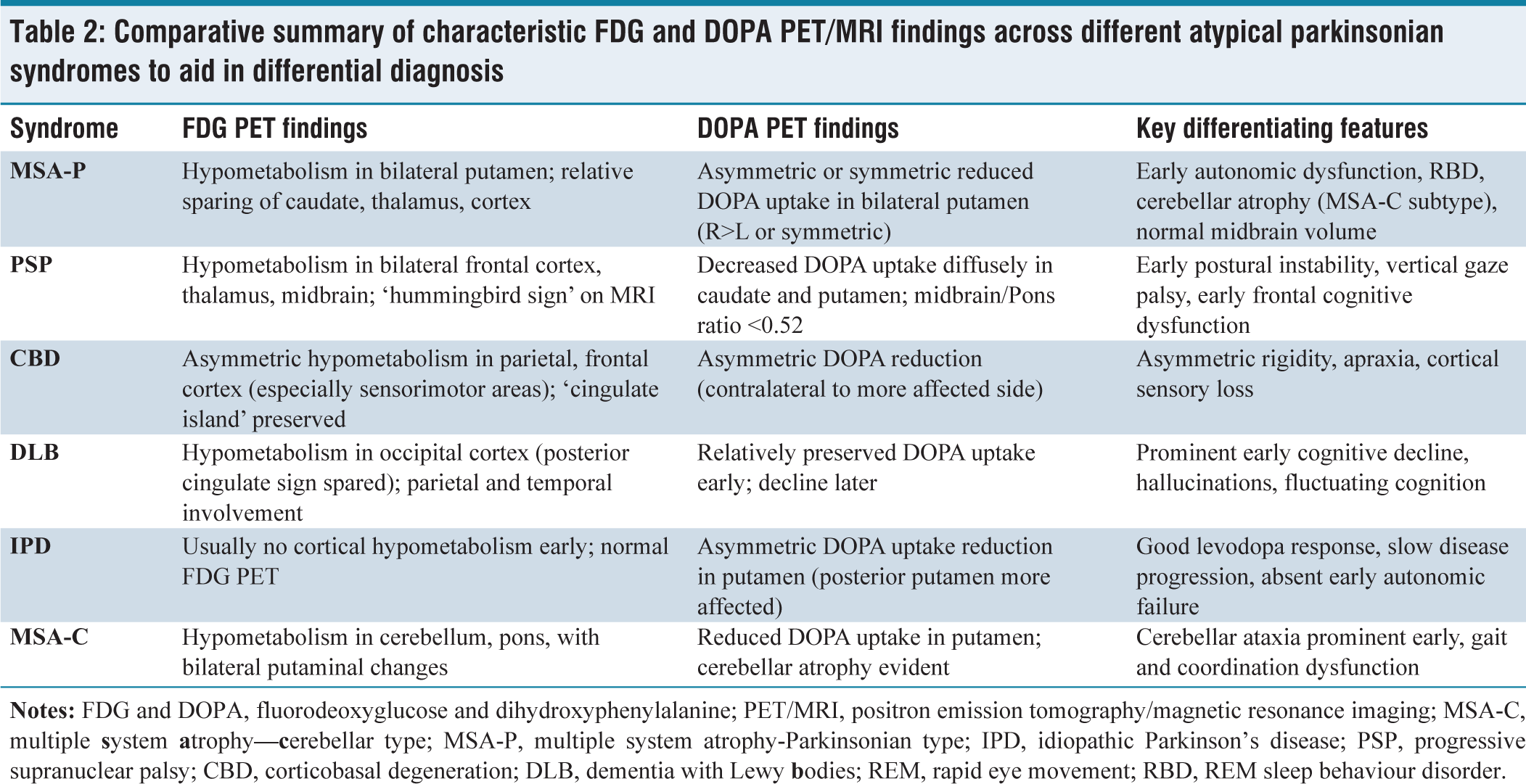
The combination of early-onset autonomic symptoms, REM sleep behaviour disorder and asymmetric striatal FDOPA uptake strongly supports MSA-P.[5,6] Preservation of thalamic and caudate metabolism helped differentiate it from PSP and DLB. Table 2 highlights key PET/MRI differentiators across atypical Parkinsonian syndromes. Midbrain-to-pons ratio of 0.6 was above the PSP threshold (<0.52), effectively ruling out PSP despite some overlapping clinical features. Unlike idiopathic Parkinson’s disease (IPD), where early cortical involvement is minimal,[5] MSA-P shows distinct striatal metabolic patterns. Incorporating dual-tracer imaging enhances diagnostic confidence, as demonstrated by Meyer et al. and Zhao et al.[5,6] Our case shows how FDG and DOPA PET/MRI complement clinical assessment, allowing early diagnosis, prognostication and optimal management planning.
Pattern of dual tracer (FDG + DOPA) PET/MRI brain in atypical Parkinsonism

Comparative summary of characteristic FDG and DOPA PET/MRI findings across different atypical parkinsonian syndromes to aid in differential diagnosis
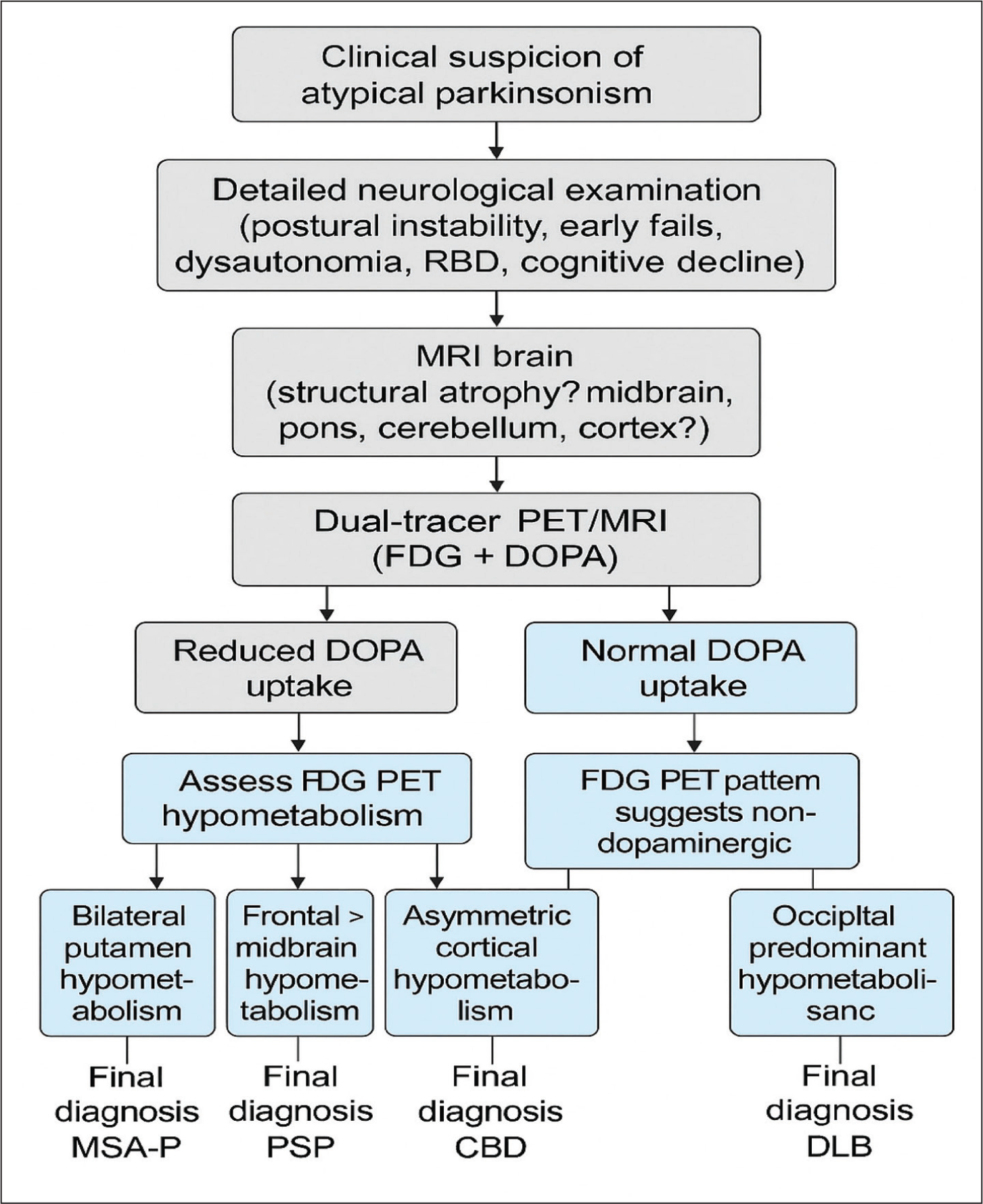
Taken together, these observations strongly point to MSA‑P. This diagnosis was supported by 18F‑FDG PET/MRI, which revealed pronounced hypometabolism confined to the bilateral putamina, with relative preservation of the caudate nuclei, pallidia and thalami. FDOPA PET/MRI further demonstrated presynaptic dopaminergic denervation, effectively excluding Alzheimer’s disease (AD).[7,8]. By contrast, APS often show diffuse cortical hypometabolism on FDG PET/MRI,[9,10] whereas our case displayed a primarily striatal metabolic defect, facilitating accurate differentiation. A diagnostic approach integrating clinical and imaging findings is illustrated in Figure 2.
Diagnostic pathway integrating clinical features with dual-tracer PET/MRI findings for accurate differentiation of atypical parkinsonism
Conclusion
This case highlights the value of dual-tracer imaging with FDOPA and FDG PET/MRI in guiding clinical evaluation of suspected MSA‑P. The presence of mild cognitive deficits on FDG PET/MRI may serve as a prognostic marker for future cognitive decline or dementia. Early and precise diagnosis through this imaging approach supports tailored therapeutic planning and underscores the importance of incorporating cognitive rehabilitation into the management of APS.
Footnotes
Acknowledgements
I would like to express my deep gratitude to Pushpendra Nath Renjen, A. N. Jena and Dinesh Mohan Chaudhari for their invaluable contributions to this study. Their expertise and guidance were instrumental in shaping this research. Special thanks to the technicians in the neurology and nuclear medicine departments for their support in data collection and imaging analysis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval
Indraprastha Apollo Hospitals Ethics Committee, Approval No.: IAH-NEURO-2025/014.
Credit author statement
Ramesh Krishnan was involved in conceptualisation, study design, manuscript writing and case analysis.
Pushpendra Nath Renjen helped in supervision, neurological assessment and critical review.
Amar Nath Jena helped in imaging interpretation, PET/MRI analysis and review.
Dinesh Mohan Chaudhari was involved in data collection, literature review, manuscript editing.
Credit author statement
Written informed consent was obtained from the patient for participation and publication of relevant clinical details and imaging.
Data availability
Data supporting the findings of this study are available upon request.
Use of artificial intelligence
No AI tools were used in the preparation of this manuscript.
