Abstract
Background:
Once considered a vestigial remnant, the left atrial appendage (LAA) is now understood to play a critical role in cardiac hemodynamics, thrombogenesis, and arrhythmogenesis. Its clinical relevance is particularly evident in patients with atrial fibrillation (AF), where it is the predominant site for thrombus formation.
Anatomy and Risk Factors:
The morphological variants of the LAA—such as chicken wing, cactus, windsock, and cauliflower—have direct implications for stroke risk stratification. The chicken wing morphology is associated with lower stroke risk due to higher flow velocities, while the cauliflower type is more thrombogenic due to its complex structure and reduced flow. Other factors like LAA size, trabeculations, and degree of fibrosis also contribute significantly to thrombosis risk.
Diagnostic Advances:
Imaging modalities for LAA evaluation have evolved considerably. While transesophageal echocardiography (TEE) remains standard, newer techniques such as cardiac computed tomography (CT) and magnetic resonance imaging (MRI) offer superior sensitivity and specificity in detecting thrombi, while also providing detailed assessment of LAA anatomy and flow dynamics.
Management and Treatment Options:
Anticoagulation therapy, including warfarin and Direct Oral Anticoagulants (DOACs), remains the mainstay for stroke prevention. However, issues such as bleeding risk and patient noncompliance have led to the development of LAA occlusion strategies. Devices like WATCHMAN and AMULET, as well as surgical options like the AtriClip, are increasingly utilized in patients with contraindications to long-term anticoagulation.
Clinical Evidence:
Large clinical trials such as ATLAS (Apixaban for the Reduction of Thromboembolism in Patients With Device-Detected Subclinical Atrial Fibrillation) and LAAOS III (Left Atrial Appendage Occlusion Study) have demonstrated the efficacy of surgical LAA closure in reducing the risk of thromboembolic events in selected patient populations.
Conclusion:
The LAA is a dynamic and clinically significant structure with substantial implications in stroke prevention. Its anatomical diversity, enhanced imaging assessment, and evolving therapeutic approaches make it a crucial target in managing patients with atrial fibrillation.
Introduction
The left atrial appendage (LAA), a small projection of the left atrium (LA), is situated mainly between the lateral and anterior walls of the atrium, aligned with the left pulmonary veins. Studies show that characteristics of LAA, such as its morphology, number of lobes, volume, LAA size, orifice diameter, degree of trabeculations, flow velocity, strain rate, degree of fibrosis, susceptibility to increased thrombogenesis are independently associated with an increased risk of stroke, particularly in patients with atrial fibrillation (AF).[1]
The LAA is widely recognised as a primary site for thrombus formation and plays a crucial role in thromboembolic events, especially in patients with AF. Its anatomy is highly variable, and several classification systems have been proposed to categorise its distinct morphologies. Research utilising computed tomography has demonstrated a strong correlation between the risk of stroke or transient ischaemic attack (TIA) and LAA morphology in individuals with AF.[2]
The CHA2DS2-VASc scoring system is commonly used to estimate stroke risk in such patients but does not account for LAA characteristics. In cases where the CHA2DS2-VASc score is low and anticoagulation may not be indicated, incorporating LAA features into risk assessment could have clinical value. Some studies suggest that factors such as LAA flow and morphology could enhance stroke risk stratification. Furthermore, LAA morphology may also play a role in refining risk assessment for patients who require anticoagulation, as specific morphologies contribute to residual stroke risk despite treatment.[3]
The utility of LAA characteristics has been depicted in the pathogenesis of thromboembolism, irrespective of AF’s valvular or nonvalvular origin.
Left Atrial Appendage: More Than a Vestigial Organ
LAA is trabeculated and is the remnant of the original embryonic LA, which develops during the third week of gestation.[4] The LAA points anterosuperiorly and overlaps the left circumflex artery or left coronary artery from which it gets its blood supply. Additionally, the pulmonary trunk and the left margin of the right ventricular outflow tract are overlapped. The orifice is usually oval-shaped and less frequently round, triangular or water-drop-shaped.[5] Veinot et al. in a comprehensive postmortem heart study, described a lobe of the LAA as a protrusion from its main tubular body. The most common configuration was found to be two lobes, with the number of lobes ranging from one to four. The prevalence of one, two, three and four lobes was 20%, 54%, 23% and 3% respectively.[6]
Based on cardiac magnetic resonance imaging (MRI) and computed tomography (CT) findings, Wang et al. classified the morphology of LAA into four types, namely chicken wing, cauliflower, cactus and windsock[7] [Figure 1].
Wang et al. classification of the LAA morphology into four types, namely, chicken wing, cauliflower, cactus and windsock [7]
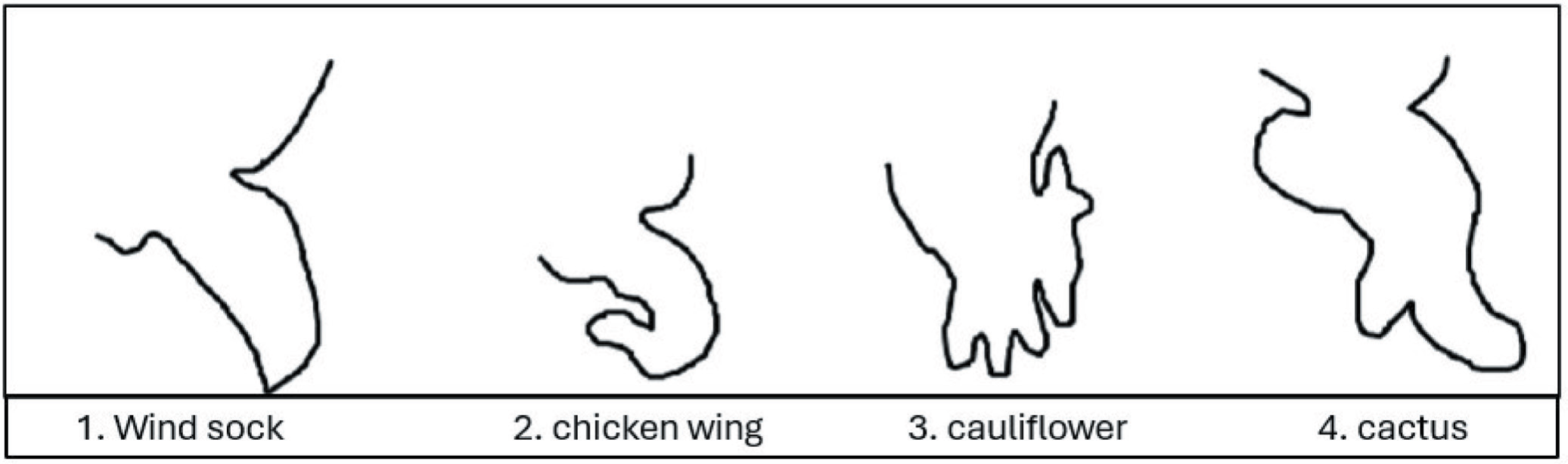
The morphologies play a key role in the percentage of strokes established.
Chicken-wing, found in 48% of cases, being the most common type, has a central lobe with a bend in the proximal section, often with additional secondary lobes. The cactus-shaped LAA accounts for 30% of cases and has a dominant central lobe with secondary lobes. The windsock LAA, present in 19% of cases, is characterised by a long primary lobe. The cauliflower LAA being the rarest has an irregular ostium and a complex internal structure with several lobes but no dominant lobe.[8]
Physiology and Pathogenesis Linking Stroke and LAA
Although previously believed to be nonfunctional, LAA is more distensible than the LA, and its endothelial cells produce natriuretic peptides, such as atrial natriuretic peptide (ANP) and B-type natriuretic peptide (BNP). Its greater extensibility and higher ANP concentration also regulate LA pressure. However, cardiac output is not significantly affected, so occluding or removing the LAA has no substantial haemodynamic effects.[9] Elevated circulatory neurohormones such as ANP, BNP, angiotensin II and transforming growth factor-β1 establish a link between atrial cardiomyopathy and arrhythmias.[10] Paracrine factors such as endothelin stimulates ANP secretion and nitric oxide (NO) reduce it.[11]
LAA orifice of >4.5 cm2 ± 1.5 potentially increases the incidence of stroke.[8] In some studies conducted on postmortem hearts, enlarged LAA body and ostium were more associated with thrombus formation. The risk of thrombus formation is also attributed to the stasis of blood due to disorganised atrial rhythm. This could also be due to derangements such as endothelial dysfunction, increased fibrosis of LAA and increased surface area.[12] Other characteristics implicated in thrombogenesis include increased LAA volume, decreased flow velocity and increased trabeculations of the LAA. Morphologically, the cauliflower type LAA is majorly associated with thrombus formation, which is attributed to its decreased flow velocity and large ostium.[2] Simultaneously, high flow velocity could be a reason for chicken wing morphology to be least associated with ischaemic strokes.[13]
Importance of ‘Left Atrial Appendage in Atrial Fibrillation’
The LAA is believed to be the origin of 90% of thrombi in cases of nonvalvular AF (NVAF) and 57% in valvular AF.[2] In a study, transoesophageal echocardiography (TEE) was conducted on 317 patients with AF and a recent thromboembolic event; 20% of patients were found to have thrombi in the LAA. In addition to this, a pooled analysis of 23 studies, encompassing 4,792 AF patients who underwent cardiac surgery, TEE or autopsy, revealed that left atrial thrombi were present in at least 14% of cases, which were majorly situated in the LAA.[13] The anatomical orientation of the LAA in relation to surrounding cardiac structures has been recognised as a contributing factor to thrombus formation. Nedios et al. observed that a higher LAA take-off, located above the left superior pulmonary vein, was linked to tachycardia-driven thrombogenic flow and an increased risk of thromboembolism in patients with AF and a low CHA2DS2-VASc score.[1] Reduced flow velocity in LAA is an indicator of functional left atrial remodelling, which affects structural remodelling of the LA. Hence, in AF, the loss of contractility LAA results in blood stagnation, favouring thrombus formation. Several other factors contributing to thrombogenesis in AF include endocardial damage caused by endocardial denudation, atrial dilation, fibroelastic infiltration of extracellular matrix, platelet activation and activation of coagulation system and alterations in growth factors.[9]
Cardiac Imaging of LAA: Evolving from TEE to MRI
Identifying LAA thrombi is essential prior to catheter ablation, cardioversion and LAA closure procedures, as well as in patients presenting with clinical signs of embolic events. Detecting thrombi in these cases often necessitates initiating anticoagulant therapy.[14] The structure, morphology and detection of thrombosis in the LAA can be evaluated using TEE, intracardiac echocardiography, cardiac CT and cardiac MRI. Doppler imaging via TEE is the most effective method for assessing LAA haemodynamic velocity.[8] TEE was the traditional gold standard investigation for the detection of LAA thrombi; being semi-invasive, it requires pre-procedural sedation and monitoring. Additionally, complications like bleeding risk, oesophageal perforation, laryngospasm and arrhythmias were often encountered with TEE. However, the thrombus in the LAA can be detected using other modalities like cardiac computed tomography and cardiac MRI. Cardiac CT has shown excellent sensitivity and specificity in identifying LAA thrombus compared to TEE. However, no significant difference in diagnostic accuracy was observed between cardiac CT and MRI when evaluating such cases.[14] A comparative study was also carried out, in which 43 patients (21 patients with AF and 22 with sinus rhythm) who were diagnosed to have LAA thrombus on TEE were subjected to undergo multisequence cardiac MRI. The median CHA2DS2-VASc score among patients with AF was 2.52 ± 1.12, with 76.7% on anticoagulant therapy. Cardiac MRI effectively visualised LAA in all cases, with a sensitivity of 97.44%, specificity of 75%, positive predictive value of 97.44%, negative predictive value of 75% and overall accuracy of 95.35%. Most importantly, MRI detected additional conditions such as Libman–Sacks endocarditis and fibroelastoma, which had been misdiagnosed as thrombus by TEE, which led to significant changes in patient management. Such statistically significant results and the noninvasive and radioprotective nature of cardiac MRI emphasise its critical role in LAA imaging.[15]
Managing LAA Thrombus: Pathway to Decrease Stroke Risk
Stroke prevention has been the utmost goal while managing AF and cases of suspected left atrial thrombus or LAA thrombus. Although anticoagulants are effective in the prevention of ischaemic strokes, they are used cautiously or even discontinued in about 50% of eligible patients due to concerns about bleeding risk, complications or noncompliance. Looking at the increasing prevalence of AF and the notable morbidity and mortality associated with AF-associated ischaemic strokes, LAA occlusion has emerged as a crucial alternative.[16]
Oral vitamin K antagonists (VKAs), such as warfarin, reduce stroke risk by about 60% compared to a placebo. Nonvitamin K antagonist oral anticoagulants (NOACs), like apixaban, offer an additional 19% reduction in the risk of stroke or emboli compared to VKAs and are linked to a 10% relative reduction in mortality. While NOACs decrease the risk of intracranial haemorrhage by 50% and major bleeding by 14% compared to VKAs, they are also associated with a higher incidence of gastrointestinal bleeding.[17] In such a case, the preferred approach could be transitioning to a different NOAC, with a subsequent switch to VKAs or low-molecular-weight heparin if needed. Left atrial appendage closure (LAAC) is reserved as a last resort, especially for patients who experience embolic strokes despite being given optimum anticoagulation therapy.[18]
The approval of catheter-based LAAC devices, such as the WATCHMAN and AMULET, by the U.S. Food and Drug Administration (US FDA), has become an alternative to oral anticoagulation. A 2020 EHRA/EAPCI expert consensus statement outlined indications for catheter-based LAAC in patients with atrial fibrillation and higher thromboembolic risk (CHA2DS2-VASc score ≥2 for males or ≥3 for females, considering factors like heart failure, hypertension, age ≥75, diabetes, prior stroke or TIA, vascular disease or age 65–74). These indications include patients with an increased bleeding risk on chronic oral anticoagulants (e.g., post-Intracerebral Hemorrhage [ICH] or recurrent bleeding), those unwilling or unable to take oral anticoagulants despite counselling, or those with contraindications to oral anticoagulant therapy. Hence, a thorough individualised risk-benefit analysis is recommended. Surgical LAAC can be performed endoscopically with the help of devices such as the AtriClip (Atri-Cure) or Penditure (Medtronic), either as an independent procedure or in combination with endoscopic atrial ablation. Invasive technique, like endocardial suture ligation, involves opening the appendage and using cardiopulmonary bypass, but it is associated with a significant risk of bleeding and incomplete occlusion in 10%–30% of cases. Conversely, epicardial suture closure, which can be performed without cardiopulmonary bypass, involves either sewing the appendage directly or securing pre-tied suture loops around the LAA base. The success rate of complete closure with this method varies widely from 23% to 100%, depending on the operator and technique. Surgical LAAC may be a valuable option for atrial fibrillation patients undergoing cardiac surgery, as evidenced by findings from the recent Apixaban for the Reduction of Thromboembolism in Patients With Device-Detected Subclinical Atrial Fibrillation (ATLAS) and Left Atrial Appendage Occlusion Study (LAAOS) III trials. The ATLAS feasibility trial showed that LAA exclusion during cardiac surgery had a 99% success rate with minimal adverse events. At one year, appendage exclusion reduced thromboembolic events (3.4% vs. 5.6% without LAAE), but postoperative anticoagulation increased bleeding risk (16.1% vs. 5.4% in case of no oral anticoagulants).[17]
Other randomised controlled trials (RCTs) were conducted to evaluate the efficacy of the LAA occlusion as an alternative to oral anticoagulants for stroke prevention in NVAF.
These include a multicentric randomised, unblinded Bayesian-designed study, namely, PROTECT-AF Trial, which enrolled 707 NVAF patients (with a CHADS2-VASc score of more > = 1) for a period of 2005–2008 and followed up for four years. Its main objective was to determine the efficacy of mechanical LAA closure as compared to warfarin and to access its noninferiority or superiority to latter. Among the total number of participants, 463 received percutaneous LAA closure (with WATHCHMAN device) and 244(control group) received warfarin (International Normalized Ratio [INR]: 2–3). Results showed that LAA closure had a lower primary event rate (2.3 vs. 3.8 events per 100 patient years) as compared to warfarin. Moreover, LAA closure group showed lower cardiovascular mortality (1.0 vs. 2.4 events per 100 patient-years; hazard ratio = 0.40; P = .005) and all-cause mortality (3.2 vs. 4.8 events per 100 patient-years; hazard ratio = 0.66; P = .04). Henceforth percutaneous LAA closure met the primary efficacy end-point for prevention of stroke, systemic embolism and cardiovascular and all-cause mortality. It was shown to be superior and noninferior alternative to warfarin but it had limitations in terms of safety due to high pre-procedural complications.[19]
As a follow-up to PROTECT-AF and to address the safety and efficacy of Watchman LAA occlusion device for stroke prevention in NVAF, a randomised trial, namely, PREVAIL Trial was conducted. Here 407 participants with NVAF were enrolled (with CHADS2 ≥ 2 or 1+ additional risk factor). Subjects were randomly allocated in 2:1 ratio, with 269 receiving intervention with Watchman device and rest 138(control group) being on warfarin, and were followed up for 18 months. The first composite primary efficacy end-point (composite of stroke, systemic embolism, cardiovascular/unexplained death) was 6.4% in the intervention group compared to 6.3% in control group, hence not achieving noninferiority. The secondary efficacy end-point (with stroke/systemic embolism >7 days post-procedure) had a rate of 2.53% in intervention group as compared to 2.00% in control group, hence achieving noninferiority. Moreover, Watchman device demonstrated improved safety performance (2.2%) as compared to the PROTECT AF Trial, along with significant reduction in pericardial effusion and notably reduced overall adverse events (4.2% in PREVAIL vs. 8.7% in PROTECT AF). Henceforth, these results suggested that LAA occlusion is a reasonable alternative to warfarin for the prevention of stroke in patients with NVAF, who do not pose contraindications to short-term warfarin treatment.[20]
Later on, a five-year meta-analysis of these two demonstrated noninferiority of LAAC to the warfarin group along with reduced stroke rates and lower cardiovascular complications and mortality in the LAAC group. However, the overall bleeding risk was similar in both groups, but nonprocedure-related bleeding was lower in the LAAC group.[17]
Conclusion
Over time, it has been clear that the LAA is more than just a vestigial organ. LAA is a functional structure that plays a significant role in cardiac haemodynamics, thrombogenesis and arrhythmogenesis. Cauliflower morphology is the most common one associated with thrombus formation and eventually is a risk for stroke. On the other hand, chicken wing morphology is known to be the safest. Several diagnostic modalities like TEE, intracardiac echocardiography, cardiac CT and cardiac MRI have evolved in imaging LAA. Out of these, CT and MRI have shown exceptional detection of thrombus and nearly similar sensitivity and specificity when compared to the traditional counterpart, that is, TEE.
Furthermore, managing LAA thrombus has become crucial to stroke prevention in such patients. This has led to the emergence of LAA occlusion devices namely WATCHMAN and others, that have proven to be beneficial in vast majority of subjects. Although various RCTs demonstrate the noninferiority of LAA closure in comparison to oral anticoagulants, the effectiveness is still debatable in the case of long-duration follow-ups.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Credit author statement
PNR, DMC and AG were involved in concept building and data review.
NS and MAM were involved in manuscript drafting.
Data availability
None.
Use of artificial intelligence
No AI was used.
