Abstract
Background:
Metabolic syndrome (MetS) comprises a cluster of cardiometabolic risk factors, including abdominal obesity, hypertension, dyslipidaemia and dysregulated glucose metabolism. It significantly increases the risk of ischaemic stroke and adversely affects recovery outcomes.
Objective:
This study aims to examine the relationship between MetS and stroke risk, with a focus on the underlying pathophysiological mechanisms, the impact on stroke outcomes, and the importance of managing modifiable components of MetS.
Methods:
A comprehensive literature review was conducted, evaluating epidemiological data and mechanistic insights into the role of insulin resistance, chronic inflammation and visceral fat accumulation in stroke pathophysiology. Studies addressing MetS components, stroke recurrence and recovery outcomes were critically analysed.
Results:
Individuals with MetS have a 1.6–5 times higher risk of stroke, with hyperglycaemia exacerbating neuronal injury and blood-brain barrier dysfunction. While some studies present inconsistent findings on MetS and stroke recurrence, managing modifiable factors—such as obesity, hypertension and dysglycaemia—demonstrates potential in reducing stroke incidence and enhancing recovery.
Conclusion:
MetS is a significant risk factor for ischaemic stroke and poor recovery. Addressing its components through targeted interventions is crucial. Further research is required to delineate the long-term impact of MetS on stroke outcomes and to refine preventive strategies.
Introduction
Metabolic syndrome (MetS) refers to a group of interconnected cardiometabolic risk factors that pose significant public health challenges. Various organisations have established criteria to define MetS, including the US National Cholesterol Education Program (NCEP), the International Diabetes Federation (IDF) and the Chinese Diabetes Society (CDS). Consequently, the prevalence of MetS can differ widely between studies and even within the same population, depending on the criteria used for its definition.[1] In 1988, Reaven introduced the idea that insulin resistance (IR) contributes not only to the development of Type 2 diabetes mellitus (T2DM) but also to cardiovascular disease (CVD). He identified a pattern of abnormalities often linked to IR and named it syndrome X.[2] To differentiate this from an existing cardiological syndrome X, the term ‘metabolic’ was added, creating what is now known as MetS X. Even in the absence of T2DM, this condition is recognised as a significant risk factor for CVD. It includes several components such as IR, hyperinsulinemia, dysglycaemia, dyslipidaemia and hypertension. The diagnosis of MetS is based on five indicators: waist circumference, fasting blood glucose, triglycerides, high-density lipoprotein (HDL) cholesterol and blood pressure. A diagnosis requires the presence of at least three of these five criteria.[3]
Pathophysiology
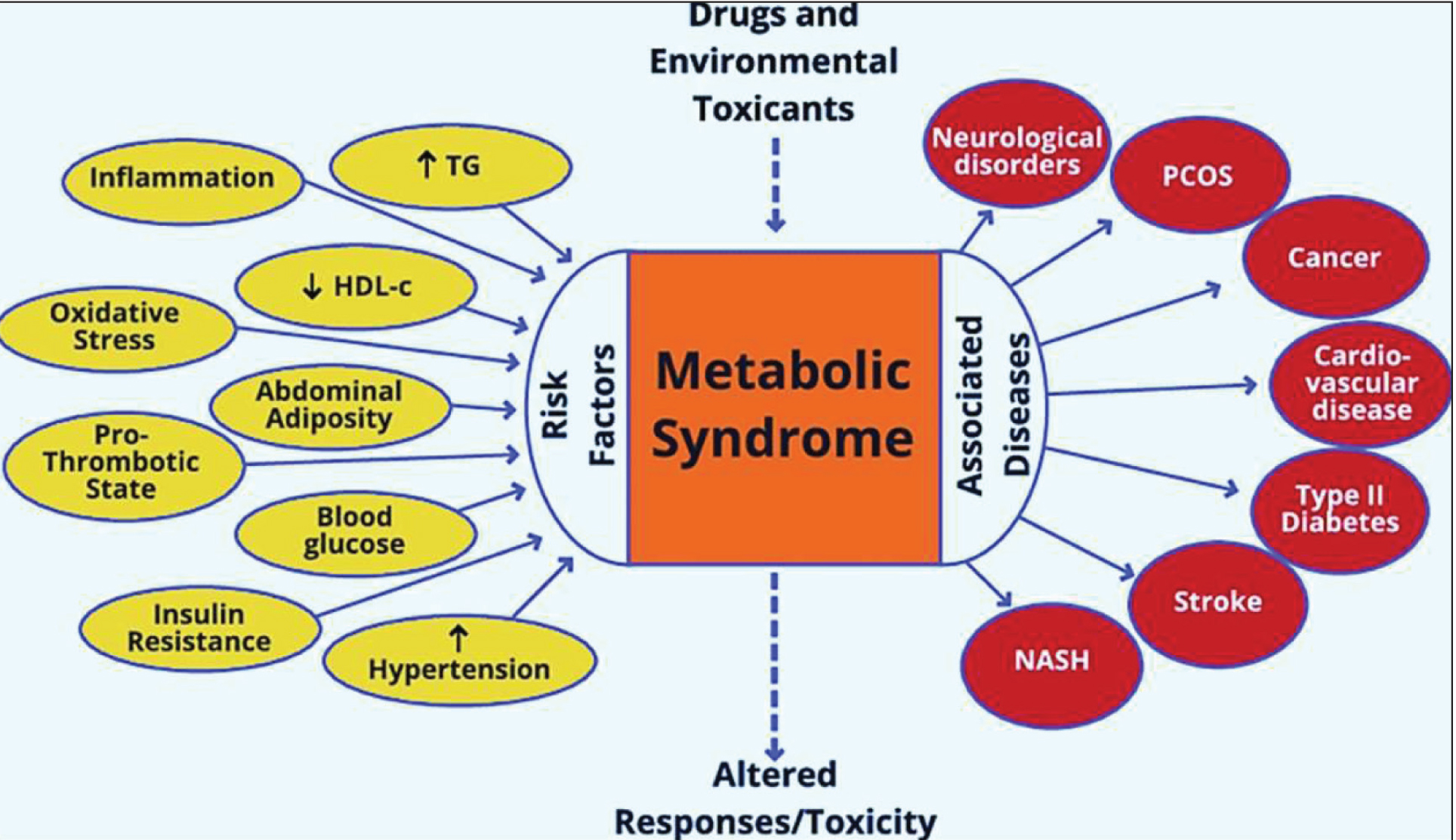
The underlying mechanisms of MetS are complex and not yet fully understood. There is ongoing debate about whether the various components of MetS represent separate conditions or stem from a unified pathogenic process. Alongside genetic and epigenetic influences, lifestyle and environmental factors, such as overeating and physical inactivity, play a significant role in its development.[3] Excessive calorie consumption, in particular, is thought to drive the process, as visceral fat accumulation is a key factor that activates many pathways associated with MetS. Key mechanisms implicated in its progression include IR, chronic inflammation and neurohormonal dysregulation, which collectively contribute to the increased risk of CVDs and T2DM [Figure 1].[4]
Figure 1 illustrates the risk factors and associated diseases of metabolic syndrome (MetS). Key risk factors include inflammation, oxidative stress, abdominal adiposity, insulin resistance, elevated blood glucose, hypertension and dyslipidaemia (↑triglycerides, ↓HDL). These factors contribute to MetS and its progression to conditions like cardiovascular disease, type 2 diabetes, stroke, non-alcoholic steatohepatitis (NASH), cancer, neurological disorders and PCOS. Environmental factors, including toxicant exposure, further exacerbate these outcomes
MetS and Stroke Risk
MetS has been linked to a heightened risk of stroke in several studies. Data from the National Health and Nutrition Examination Survey, involving 10,357 participants, revealed that MetS was significantly more prevalent in individuals with a self-reported history of stroke (43.5%) compared to those without any history of vascular disease (22.8%). MetS was independently associated with stroke history across all ethnic groups and genders (OR: 2.16; 95% CI: 1.48-3.16).[5] Additional studies focusing on older populations have also confirmed a higher frequency of MetS in patients with a history of atherothrombotic or non-embolic ischaemic strokes. These findings highlight the importance of recognising MetS as a clinical tool for identifying individuals at increased stroke risk.[6]
The elevated risk of ischaemic stroke in patients with MetS is largely attributed to its role in promoting atherosclerosis in arteries supplying the brain. IR, a key feature of MetS, contributes to this link by inducing proatherothrombotic effects on the fibrinolytic system and vascular endothelium.[7]
Long-term population studies show that individuals with MetS face a significantly higher risk of major cardiovascular events, including stroke and cardiovascular mortality. Adjusted risk ratios for ischaemic stroke in prospective studies range from 2.1 to 2.47, with hazard ratios reported as high as 5.15.[8] This increased risk is consistent across different definitions of MetS, as well as across sex, age and ethnic groups. Additionally, the likelihood of ischaemic stroke rises with the number of MetS components, each independently linked to an elevated risk of stroke.[9]
MetS and Acute Stroke Outcomes
The prevalence of MetS has been steadily rising in various populations, with studies showing its significant role in stroke risk and prognosis. In China, MetS prevalence has reached 58.1% among the elderly, and similar trends are observed globally, with notable associations between MetS and an increased risk of stroke.[10] A meta-analysis reported that individuals with MetS were 1.6 times more likely to experience a stroke compared to those without the syndrome.[11] In acute ischaemic stroke studies, MetS was present in over half of the patients, with certain components like hyperglycaemia showing a strong link to poor short-term outcomes.[11]
A Chinese trial also showed that hyperglycaemia emerged as an independent predictor of severe disability at 30 and 90 days post-stroke. It is thought to exacerbate ischaemic damage by aggravating neuronal injury, disrupting the blood-brain barrier and promoting haemorrhagic infarct conversion. Dyslipidaemia, characterised by low HDL-C and high triglycerides, was also associated with negative short-term outcomes, although some results were inconsistent due to potential confounding factors.[12] Interestingly, no significant link was found between MetS as a whole and stroke recurrence or poor short-term prognosis in this study, possibly due to offsetting effects of individual MetS components. Limitations, including a small, hospital-based sample and lack of long-term outcome data, suggest caution in generalising these findings.[12]
Conclusion
MetS is a significant contributor to the risk of ischaemic stroke, driven by its components such as hyperglycaemia, IR, hypertension and dyslipidaemia. Among these, hyperglycaemia plays a particularly important role in worsening stroke outcomes by exacerbating neuronal damage, disrupting the blood-brain barrier and increasing the likelihood of haemorrhagic transformation. Addressing these modifiable risk factors is essential for reducing stroke incidence and improving recovery in affected individuals. MetS also underscores the need for preventive strategies focused on managing excess adiposity and IR while exploring its connections to conditions like MASLD (Metabolism Associated Steatotic Liver Disease), sleep disorders and microvascular complications. These insights pave the way for more effective interventions in stroke care and prevention.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Not applicable.
Informed consent
Not applicable.
Credit author statement
This manuscript is based on a review of existing literature. P. N. Renjen was responsible for the conceptualization, methodology and formal analysis. Dinesh Chaudhari contributed to the original draft preparation. All other authors were involved in writing, reviewing and editing the manuscript. P. N. Renjen also performed the final validation of the manuscript.
Data availability
Not applicable.
Use of artificial intelligence
Artificial intelligence was not used for this study.
