Abstract
Background and Aim:
Pseudotumor cerebri (PTC), also called benign intracranial hypertension, is characterised by increased intracranial pressure (ICP), optic nerve damage, vision problems, and papilledema. First identified by Heinrich Quincke in 1893, this condition can arise from various causes, such as venous stenosis, cerebrospinal fluid (CSF) abnormalities, hormonal imbalances, excess vitamin A, and certain medications like tetracycline.
Materials and Methods:
A systematic review was conducted following PRISMA guidelines, focusing on PTC and idiopathic intracranial hypertension (IIH). The review analysed diagnosis, surgical management, complications, and affected populations. Databases such as PubMed and ScienceDirect were searched using terms like ‘pseudotumor cerebri’, ‘IIH’ and ‘intracranial pressure’. Data from relevant studies were analysed using SPSS and Excel.
Results:
This review included data from 12,307 patients, presented in several tables and figures. Notably, a review of Cushing’s disease surgeries at the University of Virginia (1982–2008) found seven cases of PTC as a late complication in a cohort of 941 surgeries, affecting both children and adults.
Conclusion:
PTC and IIH require tailored management. Medications such as acetazolamide, topiramate, and diuretics like furosemide are recommended as first-line treatments. Weight loss, including bariatric surgery, is advised for overweight patients to help reduce ICP. Surgery is considered when medical treatments are ineffective.
Keywords
Introduction
Pseudotumor cerebri (PTC), also called benign intracranial hypertension or pseudotumor brain syndrome, presents with symptoms like increased intracranial pressure (ICP), optic nerve damage, inflammation, vision loss, and swelling of the optic disc (papilledema). First described by German physician Heinrich Quincke in 1893, this condition can be caused by factors such as venous stenosis, cerebrospinal fluid (CSF) imbalance, choroid plexus dysfunction, endocrine disorders, excessive vitamin A, and reactions to drugs like tetracycline.[1-3]
The global incidence of idiopathic intracranial hypertension (IIH) is about 1 in 100,000 people, with higher rates (1–5 per 100,000) in women aged 15–65. Geographic variation is noted, with lower rates (0.3/100,000) in Japan and higher rates (2.3/100,000) in Libya, influenced by factors like obesity.[3] The exact cause of IIH is unclear, but it may involve lymphatic system dysfunction, abnormal CSF production, or venous sinus thrombosis or stenosis due to elevated ICP.
New treatments aim to address these mechanisms, including cerebral venous stenting and monitoring visual changes with optical coherence tomography.[4] In severe cases of optic neuropathy—especially when rapidly worsening or resistant to conservative treatments—surgical options like ventriculoperitoneal, ventriculoatrial, or lumboperitoneal shunts may be considered. Optic nerve decompression (OND) through subtemporal decompression may also be used, with treatment choice depending on the patient’s specific symptoms.[5]
This review aims to better understand the underlying mechanisms of PTC/IIH by exploring its causes and focusing on both surgical and non-surgical treatments to improve patient outcomes.
Materials and Methods
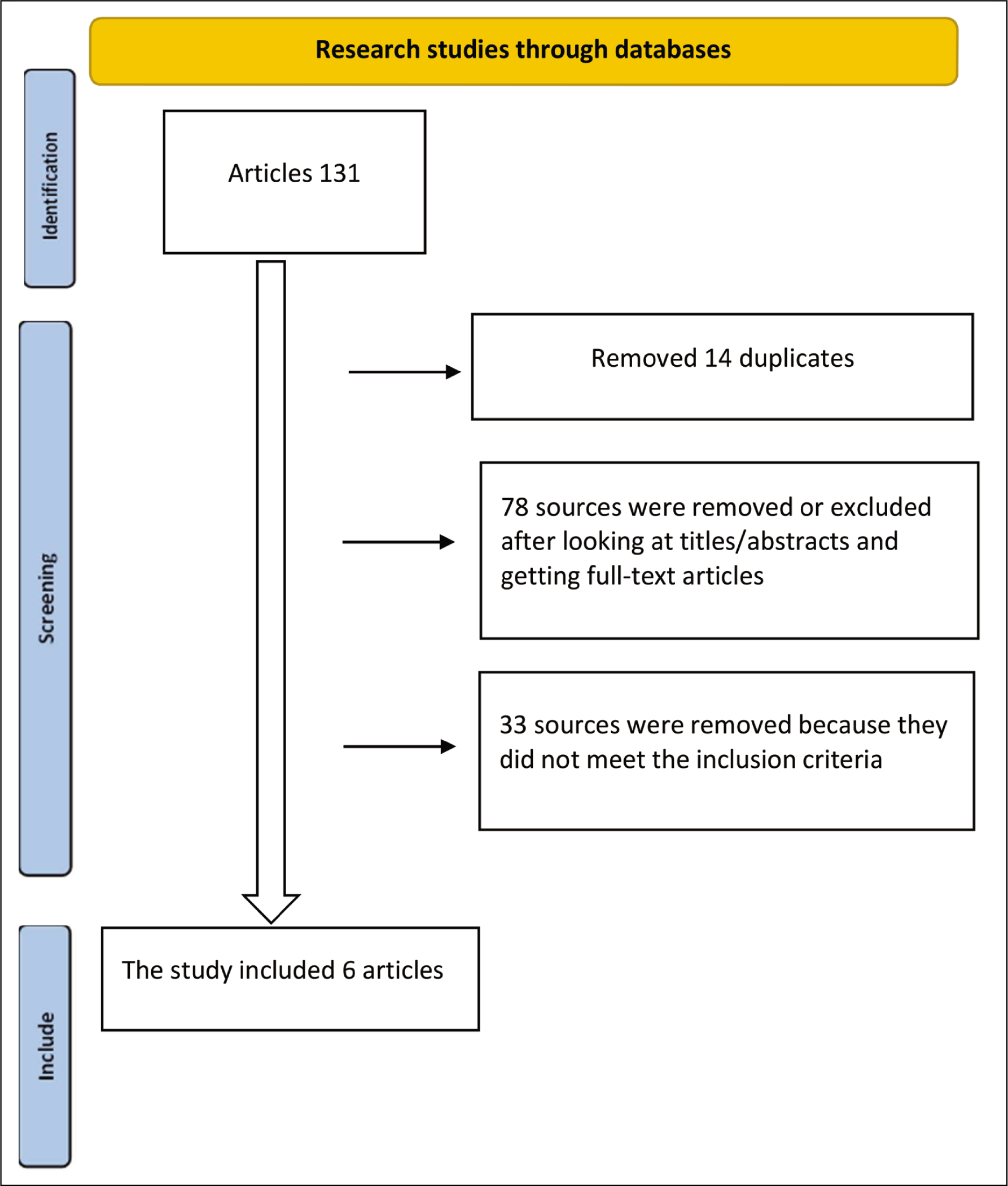
Using the PRISMA criteria, a comprehensive review of the scientific literature on PTC, also known as IIH, was conducted. This review focused on its surgical management, diagnosis, complications, and common demographic profiles. Databases such as ScienceDirect and PubMed were searched using keywords like ‘pseudotumor cerebri’, ‘IIH’, ‘ICP’, ‘intracranial pressure’ and ‘IIH management’, including terms related to ventriculoperitoneal shunting (Figure 1). Articles from the earliest available records up to May 2024 were considered. Manual searches of reference lists were also performed to identify additional relevant studies. The review adhered to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and employed Excel and SPSS Statistics for Windows, Version 26.0 (IBM Corp., Armonk, New York, USA) for data analysis.
The PICO (Population, Intervention, Comparison, Outcome) framework was applied to define the study population, with a focus on patients aged 18–85 years. The goal was to systematically assess and summarise the management, interventions, outcomes, and treatments related to PTC and IIH.
Flowchart of the Pseudotumor Cerebri [PTC or IIH] according to the PRISMA protocol detailed
Comprehensive Search Strategy Keywords
The comprehensive search strategy incorporated the following MeSH terms:
(“Pseudotumor Cerebri/classification”[MeSH] OR “Pseudotumor Cerebri/complications”[MeSH] OR “Pseudotumor Cerebri/congenital”[MeSH] OR “Pseudotumor Cerebri/diagnosis”[MeSH] OR “Pseudotumor Cerebri/diagnostic imaging”[MeSH] OR “Pseudotumor Cerebri/diet therapy”[MeSH] OR “Pseudotumor Cerebri/drug therapy” [MeSH] OR “Pseudotumor Cerebri/enzymology”[MeSH] OR “Pseudotumor Cerebri/ethnology”[MeSH] OR “Pseudotumor Cerebri/etiology”[MeSH] OR “Pseudotumor Cerebri/genetics” [MeSH] OR “Pseudotumor Cerebri/history”[MeSH] OR “Pseudotumor Cerebri/immunology”[MeSH] OR “Pseudotumor Cerebri/metabolism”[MeSH] OR “Pseudotumor Cerebri/microbiology”[MeSH] OR “Pseudotumor Cerebri/mortality” [MeSH] OR “Pseudotumor Cerebri/physiopathology”[MeSH] OR “Pseudotumor Cerebri/psychology”[MeSH] OR “Pseudotumor Cerebri/radiotherapy”[MeSH] OR “Pseudotumor Cerebri/rehabilitation”[MeSH] OR “Pseudotumor Cerebri/surgery” [MeSH] OR “Pseudotumor Cerebri/therapy”[MeSH] )
Inclusion Criteria
Patients presenting with symptoms of intracranial hypertension (PTC).
Patients diagnosed with PTC who are candidates for OND.
Patients with PTC for whom surgery and stenting are considered treatment options.
Adult patients aged 18–85 diagnosed with PTC (IIH).
Exclusion Criteria
Patients misdiagnosed with PTC based on MRI findings.
Patients with symptoms of intracranial hypertension caused by a space-occupying mass unrelated to PTC.
Paediatric patients with symptoms of PTC but with other differential diagnoses.
Data Extraction
Research on PTC, including the use of corticosteroid therapy, was examined based on the inclusion criteria. The extracted data included demographic information, interventions, controls, and the most prevalent affected population (primarily women, though paediatric cases were also considered).
Risk of Bias Assessment
A methodological quality assessment of six systematic reviews was conducted using a risk-of-bias assessment tool. After an extensive evaluation, the management and treatment of PTC, ranging from conservative options like acetazolamide to surgical interventions (such as optic nerve fenestration, shunts, or venous sinus stenting), were examined. Each study’s risk of bias and applicability concerns were assessed, with a focus on the pathophysiology of ICP. The selected studies highlighted the clinical characteristics and pathophysiology of PTC. This review specifically evaluated IIH’s pathophysiology, conservative management, and decision-making regarding surgical intervention, including diagnosis, prognosis, and disease progression.
Analytical Statistics
Summary statistics, including mean differences and odds ratios (OR), were used for key outcomes. Weighted mean differences (WMD) and 95% confidence intervals (CI) were calculated to assess outcomes across the included studies. A significance level of P < .05 was used for all primary and subgroup analyses.
Results
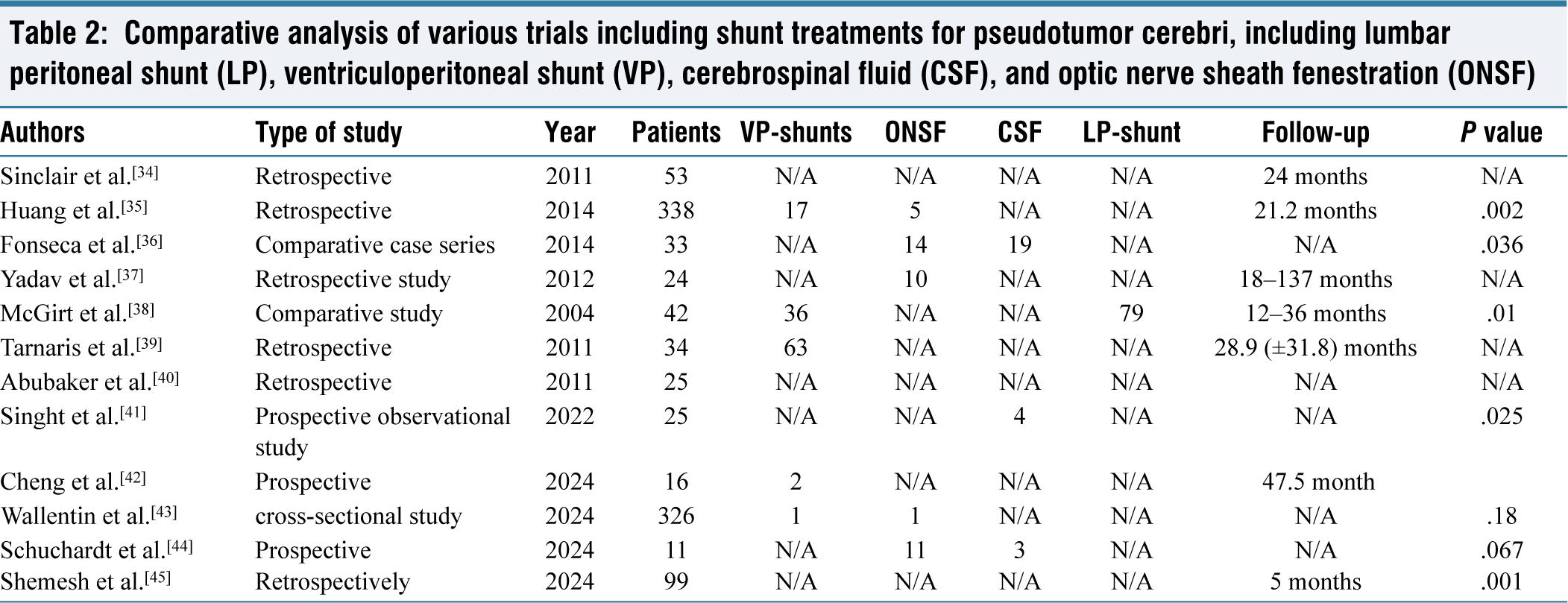
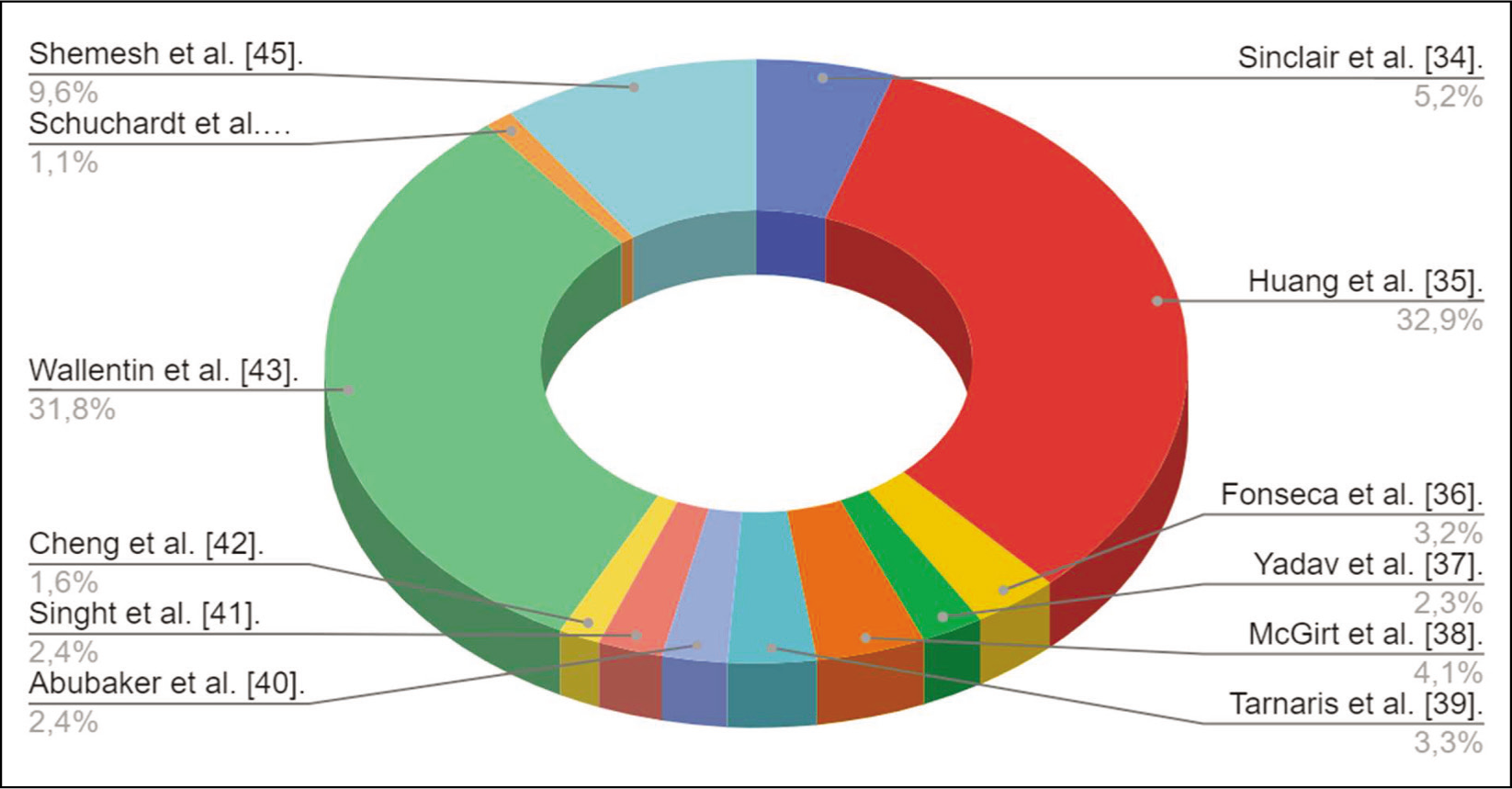
A total of 12,307 patients were included in our study, as shown in Table 1 and Figure 2, which account for 11,281 patients. Additionally, 1,026 patients are represented in Tables 2 and 3, and Figures 3 and 4. According to the National Institutes of Health (NIH), a review of surgical cases for Cushing’s disease performed at the University of Virginia between 1982 and 2008 identified seven cases of PTC. These cases, occurring as a late complication, involved four men and three women out of 941 surgeries performed on 218 children and 198 other patients.[5] Another retrospective longitudinal cross-sectional study examined 10,720 patients with PTC who underwent surgery between 2010 and 2016, within the age range of 18 to 65. Of these, 297 patients required optic nerve sheath fenestration (ONSF), with 92% of them being female[6] (Table 1).
Studies pertaining to patients with idiopathic intracranial hypertension (IIH), obstructive sleep apnoea (OSA), optic nerve sheath fenestration (ONSF), intracranial pressure (ICP), and pseudotumor cerebri (PCT)
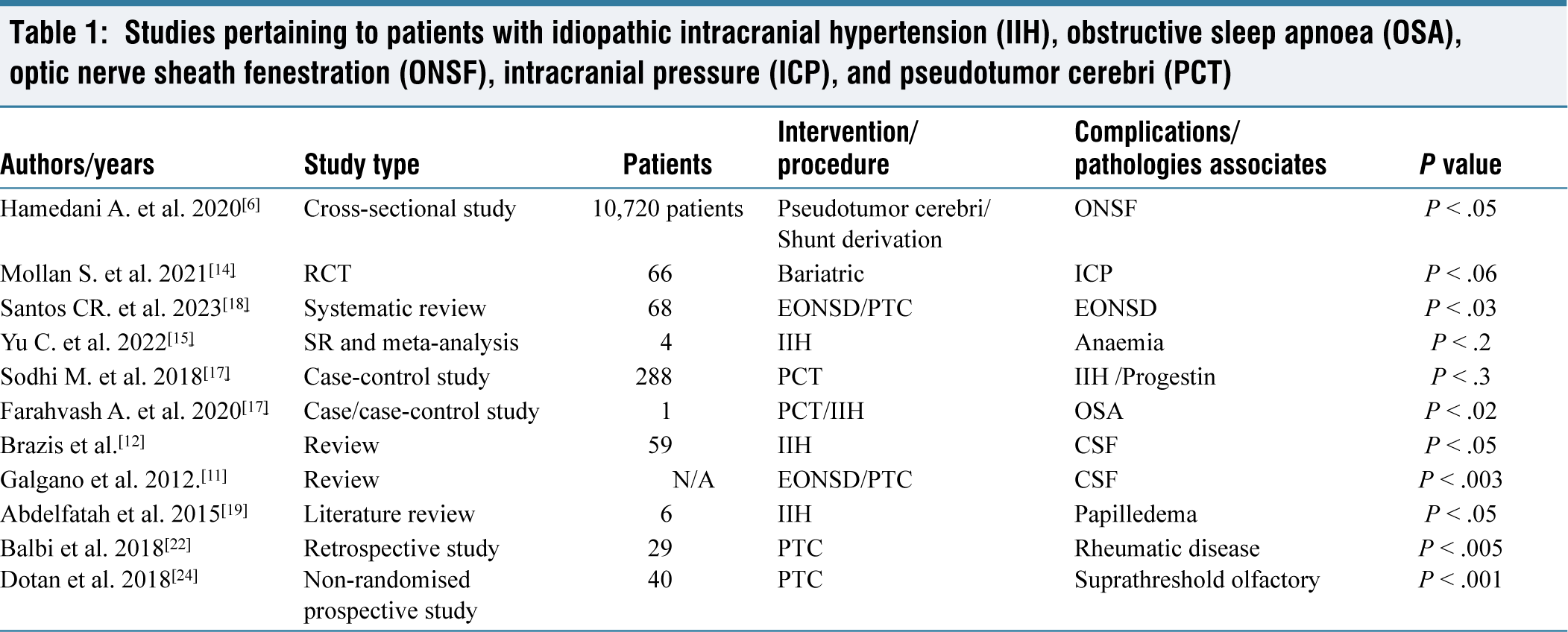
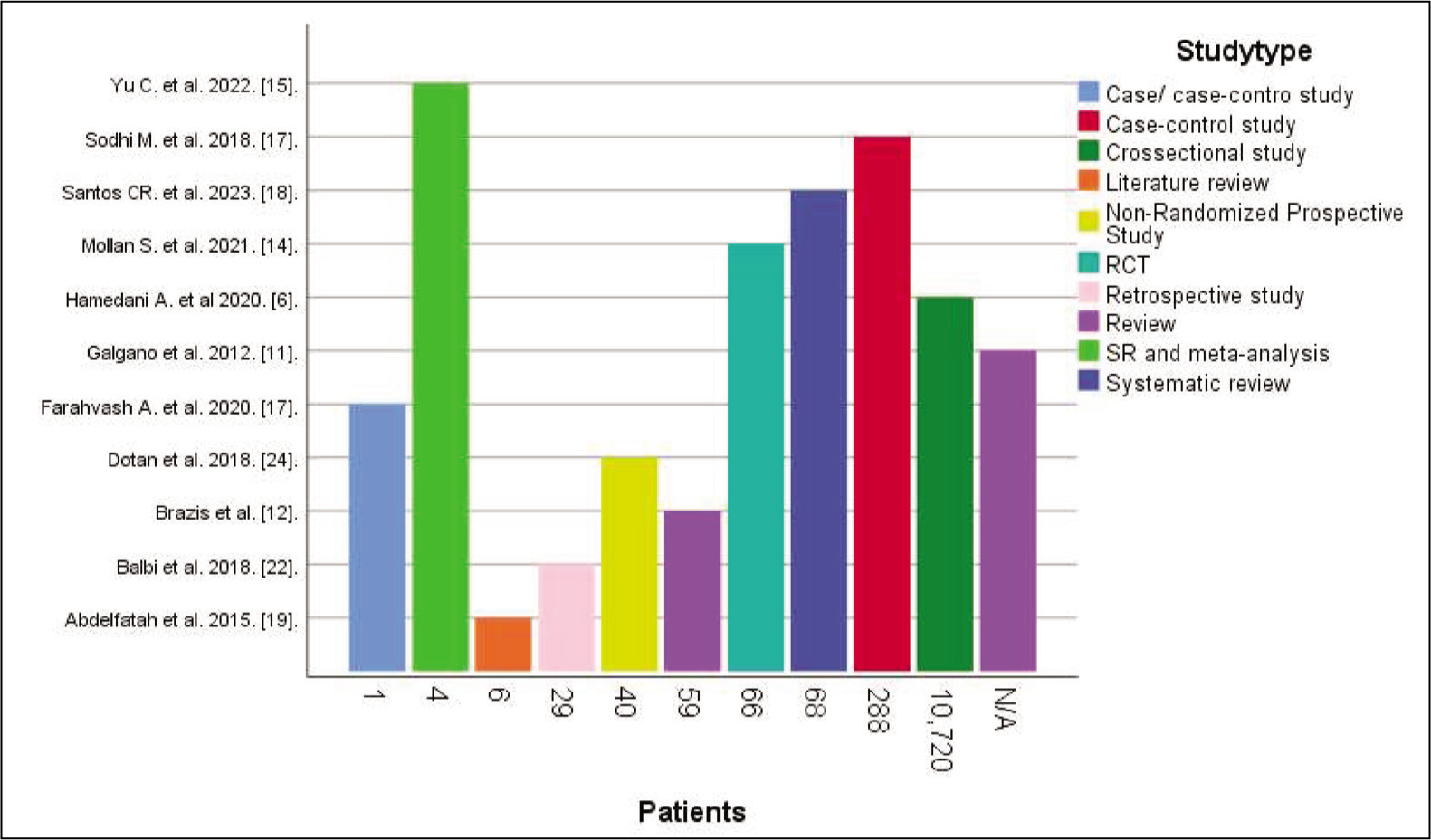
Graph showing patients with related diseases and pseudotumor cerebri
Comparative analysis of various trials including shunt treatments for pseudotumor cerebri, including lumbar peritoneal shunt (LP), ventriculoperitoneal shunt (VP), cerebrospinal fluid (CSF), and optic nerve sheath fenestration (ONSF)
Modified Dandy criteria

Surgical management of pseudotumor cerebri, VP-shunts, ONSF, CSF, LP-Shunt
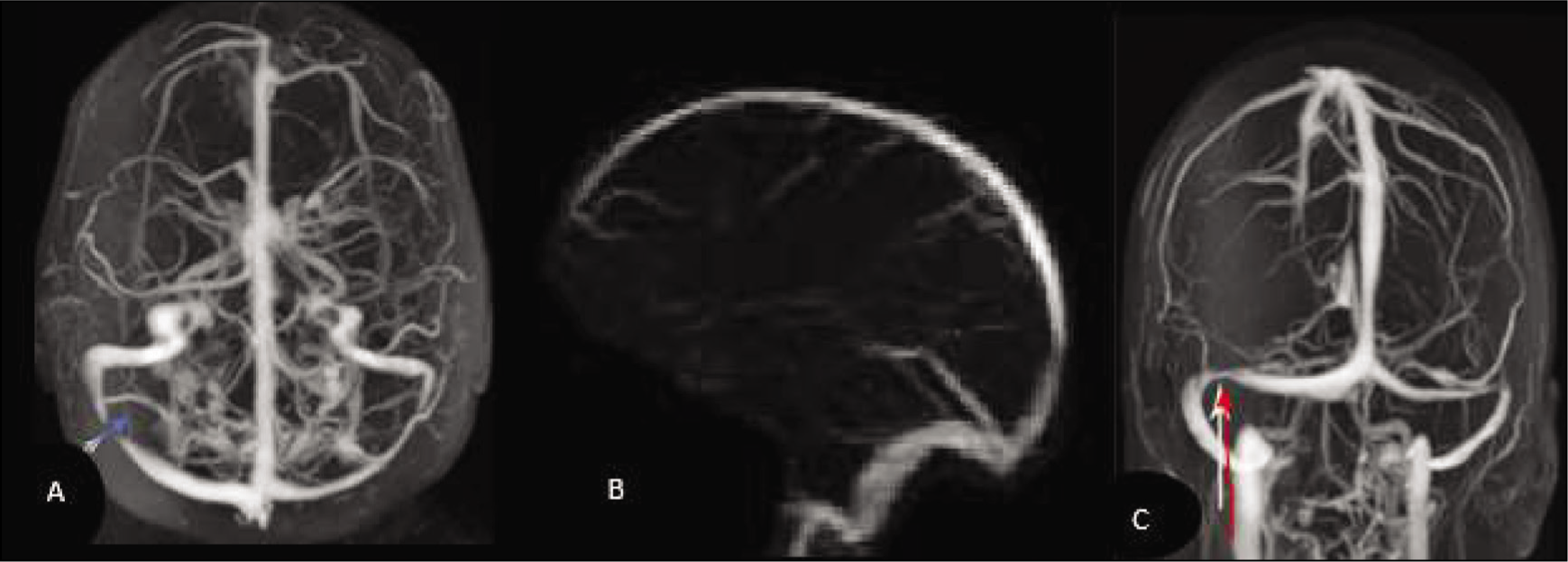
MRI (magnetic resonance venography) Stent of the right transverse sinus with normal sinus pressure, (A) with idiopathic intracranial hypertension, (B and C) with stenosis of the transverse venous sinus
A novel method combining cisternotomy and decompressive craniotomy has shown promising benefits in reducing ICP by relieving symptoms such as headache, vomiting, and papilledema. This procedure enhances CSF outflow and lowers ICP, and is believed to be associated with the lymphatic system and Virchow-Robin spaces within the cerebral parenchyma. However, its application has been primarily limited to conditions unrelated to head trauma.[7]
A multi-institutional study conducted in the United States and Canada included 165 patients, predominantly women from diverse ethnic backgrounds, and a control group of 123 men. Using Illumina BeadChips’ Human Core Exome v1.0, the study identified 301,908 single nucleotide polymorphisms (SNPs). Notable SNPs included rs2234671 on chromosomes 2, 6, and 12, along with potentially significant SNPs on chromosomes 5, 13, and 14, which demonstrated meaningful associations.[20] Dandy explored subtemporal decompression as a treatment for PTC, reporting favourable clinical outcomes in 22 patients. He attributed sudden increases in ICP to vascular changes rather than CSF alterations.[12]
Since ventriculoperitoneal shunting is often utilised alongside other successful stenting methods for treating IIH, a retrospective study involving 52 patients revealed that only 6% required stenting. The lumboperitoneal shunt has a lower revision rate compared to the ventriculoperitoneal shunt and has been shown to alleviate headaches and improve visual acuity, resulting in shorter hospital stays. However, complications such as ipsilateral headaches are common but usually resolve a few days after the procedure. Cerebral haemorrhage, although less frequent, can occur due to persistent venous flow obstruction.[13,29]
Procedure for PTC
Dandy was the first to document various subtemporal decompression techniques that proved effective for PTC, long before the development of CSF shunt methods. In his study, 22 PTC patients who underwent surgical subtemporal decompression exhibited favourable clinical outcomes. Postoperative examinations revealed papilledema in 14 patients; however, no fatalities were reported. A 1998 study supporting Dandy’s findings noted that eight patients with PTC did not require reoperation. Of these, eight individuals experienced visual field loss, but acuity improved after one month. Following subtemporal decompression, CSF shunting was employed to alleviate headaches, and nearly all patients did not report ongoing or recurring visual acuity impairment.[11]
Cisternotomy: Anatomical Development and Procedure
The anterior clinoid process is situated within the temporal and frontal lobes, which are characterised by adherent dura mater. This anatomical configuration allows for the creation of an extradural corridor leading to the carotid optic cistern. By opening these lobes, we can approach the cisterns more closely through a proper incision at the orbito-meningeal gap. After performing a frontotemporal craniotomy with an incision in the lateral orbital-meningeal band, a plane will be established where the dura of the frontal lobe can be separated from the temporal dura over the lateral wall of the cavernous sinus and the anterior clinoid process. The procedure typically involves performing a decompressive hemicraniectomy, opening the lateral and optical-carotid cisterns, and incising the double-layered Liliequist’s membrane, through which the third nerve and the communicating artery traverse from the anterior to the posterior cerebral fossa.[8]
Dural Venous Sinus Stenting for PTC
Stent implantation in the dural venous sinus has been shown to be beneficial for treating patients with PTC. For instance, a case of PTC, initially misdiagnosed due to bilateral stenosis of the transverse sinus observed on venography, was successfully treated with the placement of a self-expanding stent through the stenosis of the right transverse sinus. However, some studies suggest that the underlying cause of the transverse sinus disease was bilateral transverse sinus stenosis rather than thrombosis. Ultimately, direct retrograde cerebral venography revealed a filling defect related to venous sinus blockage.[12]
Conservative Treatment
While there is a lack of comprehensive data to support their efficacy, carbonic anhydrase inhibitors are recommended as a first-line treatment for patients with persistent headaches despite vision impairment. Both methazolamide (Neptane) and acetazolamide (Diamox) act as moderate diuretics by inhibiting carbonic anhydrase in the choroid plexus, which reduces CSF production. For adults, acetazolamide is typically initiated at a dose of 1 g, taken as 250 mg once daily or 500 mg twice daily, with a maximum dosage of 4 g. Potential adverse effects include taste changes, fatigue, and paraesthesia, necessitating dose adjustments. Although corticosteroids can provide immediate ICP reduction, their use is generally discouraged in PTC due to adverse effects such as weight gain and fluid retention. Topiramate is frequently employed for headache management unrelated to ICP, and it has shown comparable efficacy to acetazolamide in treating headaches and reducing papilledema, with the added benefit of weight loss. While octreotide is not typically used as a primary treatment, studies indicate that it may effectively lower ICP as an insulin factor 1 and growth hormone inhibitor.[4,5]
Discussion
Individuals with a body mass index (BMI) meeting the modified Dandy criterion and less than 30 kg/m² have shown improvement in PTC symptoms through conservative and bariatric treatments.[10] Brazis et al. discovered a low frequency of Chiari malformation among PTC cases, which was sometimes mistakenly linked to abnormalities in venous flow, necessitating a careful distinction.[12,13]
Recent studies indicate that acetazolamide is the primary treatment for IIH; however, if tolerance to acetazolamide is not achieved, surgical intervention may be required.[21,22] The use of pseudocetorol in rheumatic illnesses, prothrombotic events, and antiphospholipid syndrome has been associated with the development of PTC.[23] Stent placement in venous sinuses, particularly the transverse sinus, has been shown to improve visual acuity, reduce headaches and papilledema, and significantly benefit patients with bilateral transverse sinus stenosis.[24]
For less invasive diagnostics and functional improvements, other treatments, such as sniffing sticks, Ishihara colour vision testing, and tomography, have been proposed.[25-27] For patients severely affected by PTC in both eyes, unilateral ONSF is recommended.[28]
Antiplatelet medications should be managed carefully around the time of stent implantation. Weight loss therapies have shown promise for symptom relief and ICP management, and clinical follow-up is critical for PTC patients as the disease progresses.[29] Conditions such as Addison’s disease and steroid withdrawal may contribute to IIH.[30] In cases of refractory pathologies, anatomical interventions such as cisternotomy—incisions that create conduits for CSF flow—have shown potential.[8,31] Patients with PTC may benefit from topiramate and tretinoin arsenic after failing acetazolamide, particularly those undergoing treatment for acute promyelocytic leukaemia.[32]
Limitations
The study’s discoverability across multiple databases is hindered by its contentious and imprecise nature, characterised by ambiguous descriptors. A significant number of articles fail to pinpoint the exact cause of PTC. Each study presents a plausible explanation, contributing to the complexity of understanding this condition and complicating efforts to identify a singular causative factor.
Conclusion
The study of PTC, also known as IIH, is marked by considerable debate due to the diverse findings and varying research methodologies. Based on these insights, several recommendations can be made:
Sequential treatment approach: Initiate a conservative treatment regimen involving medications such as acetazolamide, topiramate, furosemide, and carbonic anhydrase inhibitors. This conservative approach should be exhausted before considering surgical options. Weight management strategies: For overweight patients, exploring bariatric therapy may be beneficial, as it has shown potential in reducing ICP. Consideration of surgical intervention: In cases where conservative treatments are unsuccessful, a decompressive craniotomy combined with a cisternotomy is recommended. This approach aims to effectively alleviate brain tension and lower ICP. Shunt placement: Ventriculoperitoneal, ventriculoatrial, or lumboperitoneal shunts are suggested due to their proven effectiveness in reducing ICP. ONSF: ONSF is recommended as it has demonstrated efficacy in reducing ICP and can aid in managing IIH.
These recommendations provide a multi-faceted approach to managing PTC, emphasising conservative treatments initially while outlining surgical options for cases resistant to non-invasive interventions.
Supplemental material
Supplemental material for this article available online.
Supplemental material
Supplemental material for this article available online.
Footnotes
Abbreviations
PCT: Pseudotumor cerebri.
ONSF: Optic nerve sheath fenestration.
ICP: Intracranial pressure.
IIH: Idiopathic intracranial hypertension.
OSA: Obstructive sleep apnoea.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Ethics approval was not required for this systematic review and may not be required for this article.
Credit author statement
Conceptualisation: BC and DES.
Writing: Preparing the Original Draft: DAES.
Writing: Review and Editing DAES.
Supervision: GC.
Data availability
The data are available on reasonable request.
Use of artificial intelligence
Artificial intelligence was not use in this project.
