Abstract
Background and Aims:
Meningiomas have different subtypes, including the rare and indolent metaplastic meningioma, which are characterised by the presence of mesenchymal components. The histopathological features of osseous metaplastic meningiomas include the existence of bone tissue and tumour cells within areas devoid of psammoma bodies. These tumours can have devastating symptoms affecting the quality of life of the patient severely. Tumours in proximity to critical structures pose a unique challenge in management, making it essential to address these issues for effective local control and symptom relief.
Materials and Methods:
Here we present a rare case report of a gentleman diagnosed to have intracranial osseous metaplastic meningioma who experienced severe visual deficits, for whom postoperative radiotherapy was administered and excellent outcome was achieved.
Results:
At a 12-month follow-up, patient had dramatic improvement in vision.
Discussion:
Stereotactic radiotherapy is a safe and effective method for achieving excellent local control, improving symptoms and enhancing quality of life of the patient. The proximity of critical structures and their dose tolerances should be key considerations while deciding on the fractionation scheme.
Keywords
Introduction
Meningiomas are the most common primary intracranial tumours. Of which, 81.1% of which are typical (grade I), 16.9% are atypical (grade II) and 1.7% are anaplastic (grade III).[1] Metaplastic meningiomas are very rare (accounting for 0.2%–1.6% of all meningiomas), slow-growing, grade I meningiomas that are characterised by the presence of mesenchymal components.[2] Here we present the case of an intra-cranial osseous metaplastic meningioma where a dramatic improvement in vision was achieved with postoperative stereotactic radiotherapy.
Case Report
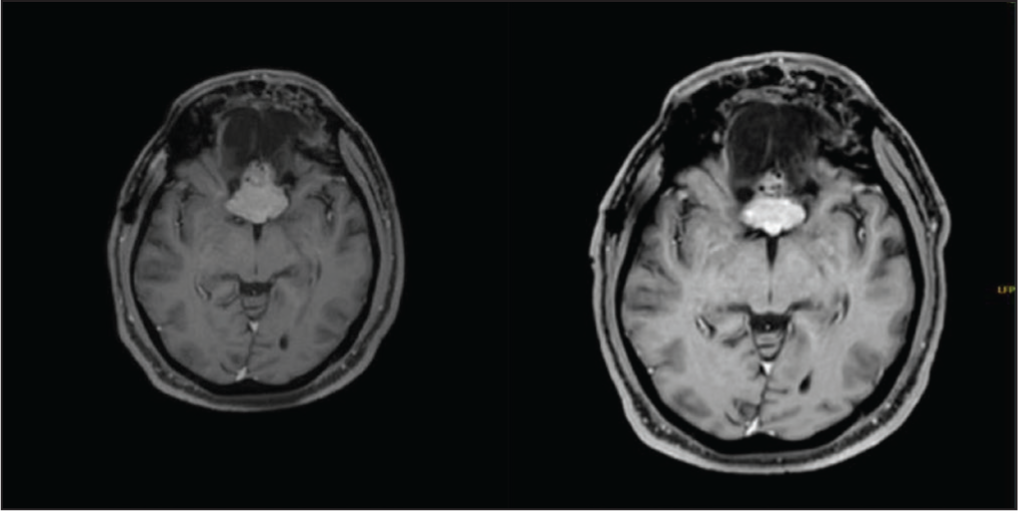
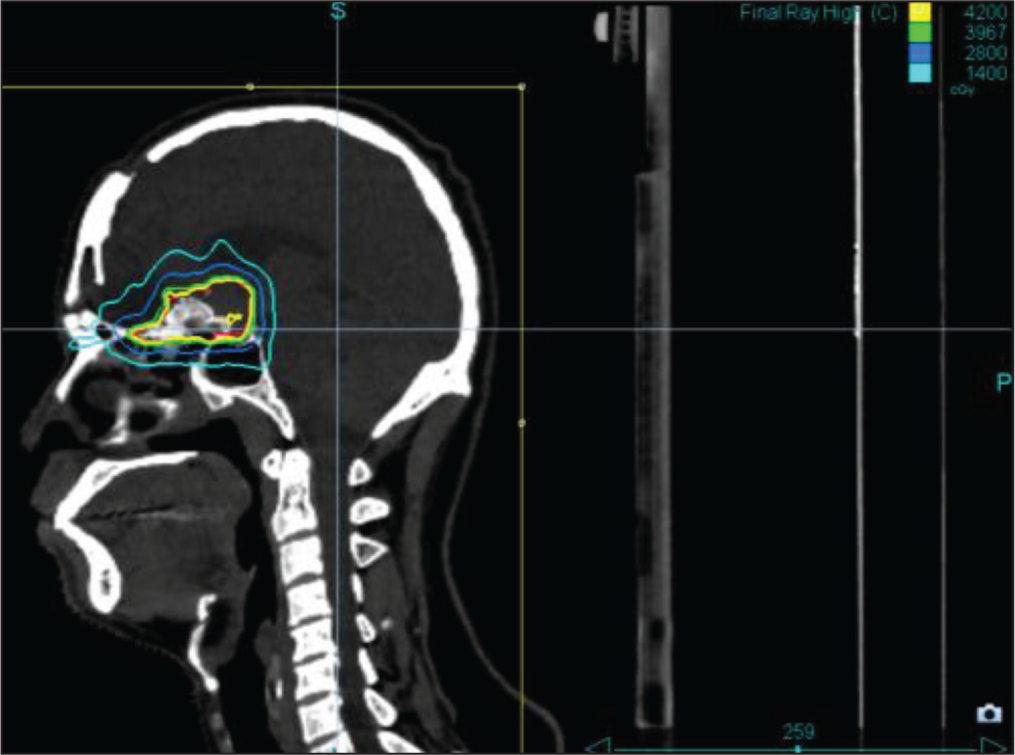
A 44-year-old gentleman presented with complaints of visual disturbances, anosmia and sleeplessness. Magnetic resonance imaging (MRI) brain showed features suggestive of planum sphenoidale and falx meningioma, measuring 6.4 cm × 5.5 cm × 6.2 cm, in bilateral frontotemporal lobes, compressing the body, genu and rostrum of the corpus callosum, and the frontal horns of bilateral lateral ventricles, partly extending into the intra-sellar region [Figure 1a]. He underwent a bifrontal craniotomy and excision of the lesion, with histopathology showing a neoplasm composed of lobules, whorls and fascicles of meningothelial cells with round nuclei, and fine chromatin in a vascularised stroma with areas of osseous metaplasia, confirming the diagnosis of olfactory groove metaplastic osseous meningioma, World Health Organisation (WHO) grade 1. Following the finding of a residual lesion measuring 2.81 cm × 2.93 cm × 2.37 cm in a follow-up computed tomography (CT) scan, he received stereotactic radiotherapy in a CyberKnife machine to a total dose of 42 Gy in 15 fractions [Table 1, Figure 2, Appendix 1 & 2 in supplementary data]. Radiotherapy course was uneventful. At one-year post-treatment, patient had dramatic improvement in the field of vision and quality of life [Figure 3]. Review MRI brain [Figure 1b] showed regression in the lesion size (3.0 cm × 1.6 cm × 1.8 cm) and volume analysis has shown 6cc volume reduction.
Stereotactic radiotherapy using CyberKnife

(a). MRI brain (contrast) T1 contrast image showing the lesion (pre-radiation). (b). Post-treatment imaging
Sagittal image. Multiple plans were created and the one with adequate target coverage and acceptable normal tissue doses was finalised for treatment. Conformity index was 1.19 and Homogeneity index was 1.12
Visual field examination—(a) prior to radiotherapy showing severe visual field deficit in the left eye, post-radiotherapy improvement in the visual field of (b) left eye, (c) right eye

Discussion
Meningiomas are classified into various histological subtypes according to the 2021 WHO classification, and metaplastic meningioma is one rare entity that is WHO grade I.[2,3] The transformed neoplastic cells show the histological features of the cells they mimic, hence the name ‘metaplastic’. Metaplastic meningioma can be osseous, cartilaginous, myxoid, lipomatous or xanthomatous based on the histology.[2,4]
The most common presenting symptoms are headache and seizures.[5] Tumours commonly localise to the convexity of the brain, with patients often presenting with symptoms reflecting the tumour location.[5,6] In our case, patient presented with anosmia and visual disturbances which correspond to the location of the tumour in the olfactory groove.
MRI is the standard modality of imaging in meningioma, which typically reveals a dural-based homogeneously enhancing well-circumscribed lesion. Intralesional calcifications and bony changes can be seen in CT. Although not a pathognomonic feature of meningioma, ‘dural tail’ is still useful for distinguishing it from other lesions.[1] The osseous changes in meningioma are better defined with CT.[5,7] Kim et al. failed to identify any unusual radiological characteristic that can distinguish metaplastic meningioma from others.[4] Xu et al. reported that the final diagnosis should be histological since imaging cannot differentiate between ossification and calcification.[8] In our case, MRI showed a T1 and T2 isointense broad-based lesion, and MR Spectroscopy showed an elevated NAA peak.
Histologically metaplastic meningioma contains focal or more widespread mesenchymal components like bone, cartilage, fat, myxoid or xanthomatous tissue.[9,10] In the osseous type, histological characteristics encompass the existence of bone tissue and tumour cells within areas devoid of psammoma bodies.[4]
The standard treatment for all meningiomas is surgery. Radiotherapy is an alternative option for tumours at eloquent locations, such as the skull base where the lesion encases vascular-nervous structures. Radiosurgery, fractionated and hypofractionated stereotactic radiotherapy often provides a tumour control rate of over 85% at five years.[11-13] Adjuvant radiotherapy is indicated for grade II and III meningiomas due to their higher recurrence rates even after a gross total resection.[11]
Following a sub-total resection (STR) of a grade I meningioma, the five- and ten-year local progression rates can be up to 62% and 100% respectively.[14] Adjuvant radiotherapy following STR is not without any controversy, with some advocating for observation owing to higher five-year PFS in the range of 81%.[15] Hence, there is a tendency to omit radiotherapy for patients with advanced age, multiple comorbidities and small residual tumours.[14] Other studies have shown excellent results with adjuvant radiotherapy following STR, with local control rates of up to 100% at extended follow-up.[14] In addition, some of the studies have also observed a cause-specific survival benefit with radiation after STR.[16] Fractionated stereotactic radiotherapy or SRS after STR also offers higher local control in the range of 92%–100% at five years.[14] SRS is usually reserved for tumours <3 cm or <10 cm3 in volume. Smaller tumours have higher five-year disease-free survival (91.9% for tumour volume <10 cm3) and lesser complication rates (4.8% for tumours <3.2 cm3).[14,17,18] Fractionated radiotherapy is preferred for larger tumours. Although Unger et al. reported that post-treatment symptomatic oedema was higher with single-session radiosurgery than with multisession, Conti et al. reported that treatment modality (single vs. multiple sessions) was not a significant factor causing post-treatment oedema.[19,20]
Pollock et al. observed complications like cranial nerve deficits, headaches, hemiparesis, new/worsened seizure, cyst formation and stroke, following radiotherapy, especially for larger tumours.[17] As many patients will achieve long-term tumour control, late effects should be addressed. Neurocognitive decline, hypopituitarism, brain necrosis and rarely malignant transformation are the other possible long-term effects.[14]
In view of critical location of the tumour, we have delivered the radiation in over 15 fractions respecting the tolerances of nearby structures (refer to Appendix 2 in supplementary data). We have observed a significant reduction in the residual tumour volume (~6 cc) one year after radiotherapy, with significant improvement in the presenting symptoms including the visual disturbances, and no clinically relevant adverse effects. To the best of our knowledge, this is the first case report describing the use of stereotactic radiotherapy specifically in metaplastic meningioma. With the sophistication of the technology, radiation can be delivered very safely limiting the normal tissue dose spillage to obtain excellent clinical outcomes and long-term control.
Conclusion
Metaplastic meningiomas, though uncommon, can have devastating symptoms due to their location affecting severely the quality of life of the patient. A gross total resection is often not possible when tumours occur in critical regions due to the close proximity to nearby normal structures. Local control of the residual lesion can be effectively achieved by stereotactic radiotherapy. Proximity of the critical structures and their dose tolerances should be a key consideration while deciding on the fractionation.
Supplemental material
Supplemental material for this article available online.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Not applicable.
Informed consent
The authors certify that they have obtained informed consent from the patient. The patient has given his consent for the usage of images and other clinical information for publication in the journal. The patient understands that his name or initials will not be published, and while all attempts will be made to conceal his identity, complete anonymity cannot be guaranteed.
Credit author statement
Kesav Ajith was involved in data collection and compilation of the article. R. Rathnadevi, B. Subathira and Mohammed Sathik Ali contributed to guidance in compilation and critical review of the article.
This manuscript has been read and approved by all the authors. All the authors believe that the manuscript represents an honest work, that contributes to the knowledge pool in the field of radiotherapy.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
Use of artificial intelligence
None.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
