Abstract
Introduction:
Pediatric Acute Liver Failure (PALF) incidence has been increasing in recent times. It is an emergency and needs appropriate management in a specialized center with availability for liver transplant.
Methods:
In this article, we shall provide an overview of etiologies and approach to the management of PALF.
Conclusions:
Various known etiologies for pediatric acute liver failure involve infections, immunologic disorders, and metabolic and drug/toxin-mediated illnesses. However, a known etiology for liver failure may be lacking in approximately 30–50% of cases. In developing countries like India, infections still dominate as the leading cause of pediatric ALF, with the hepatitis A virus (HAV) being the most identified etiology. Metabolic disorders account for approximately 23–30 percent of the PALF in infancy or early childhood (less than 5 years). Around 9–52% of children with PALF have indeterminate etiology. Management requires a multidisciplinary approach involving hepatologists, pediatric intensive care specialists, nephrologists, and a liver transplant surgery team. Cerebral edema is the most frightful complication of acute liver failure, which may progress rapidly, leading to irreversible brain damage, so every possible measure should be taken to identify as well as prevent the progression of cerebral edema and raised intracranial pressure. Elective intubation and mechanical ventilation should be started in patients with significant encephalopathy (Grade II). Identification of the need for liver transplantation and early referral for the same before irreversible brain damage sets in. Extracorporeal liver assist devices have shown promising results in bridging these children to native liver survival or liver transplantation, but continuous careful monitoring should be done to assess for treatment failure and the need for liver transplantation. Liver transplantation is the only definite treatment that improves survival in PALF, and with several advances in surgical techniques and good intensive care, the overall survival after LT for ALF has improved significantly.
Introduction
Paediatric acute liver failure (PALF) is a distinct clinical syndrome characterised by massive hepatocellular necrosis leading to synthetic dysfunction and hepatic encephalopathy (HE). It progresses rapidly and leads to devastating consequences if not managed appropriately. As per the Paediatric Acute Liver Failure Study Group (PALFSG), PALF is defined as hepatic-based coagulopathy defined as (a) prothrombin time (PT) 15 seconds or international normalised ratio (INR 1.5) not corrected by vitamin K in the presence of clinical HE or a PT 20 seconds or INR 2.0 regardless of the presence or absence of clinical HE, (b) biochemical evidence of acute liver injury and (c) no known evidence of chronic liver disease.[1] There is a wide spectrum of aetiologies causing ALF in children which differ not only within several age groups but also among various geographical locations. Management requires a multidisciplinary approach involving hepatologists, paediatric intensive care specialists, nephrologists and a liver transplant (LT) surgery team.
Aetiology
Various known aetiologies for PALF involve infections, immunologic disorders and metabolic and drug/toxin-mediated illnesses. However, a known aetiology for liver failure may be lacking in approximately 30%–50% of cases.[2] In developing countries like India, infections still dominate as the leading cause of PALF with hepatitis A virus (HAV) being the most commonly identified aetiology.[3] Metabolic disorders account for approximately 23%–30%of the PALF in infancy or early childhood (less than 5 years).[4] Around 9%–52% of children with PALF have indeterminate aetiology.[5]
Diagnostic Approach
An age-based diagnostic approach is followed to improve diagnostic yield. If a specific diagnosis with a curative treatment can be identified, it can alter the natural history of the disease and prevent complications. A detailed history of prodrome, onset and duration of jaundice, change in mental status, drug or alternative medicine intake and past and significant family history may help in planning the relevant investigations and reaching a diagnosis and thus plan management. Antenatal and birth history, history of developmental delay or seizures may play an important role in infants with ALF which may be due to metabolic disorders in a significant proportion. History of growth failure, jaundice in past, pruritus and ascites may point towards underlying chronic liver disease.
Physical examination should be oriented towards identification of the severity of illness, complications already present, underlying cause and presence of any signs suggestive of underlying chronic liver disease. Haemodynamic instability is commonly seen in advanced stages of ALF, so all these children should be nursed in a well-equipped intensive care unit and continuous and close monitoring of haemodynamic as well as neurological status should be practised. Children with significant encephalopathy (altered sleep cycle followed by excessive irritability, inconsolable crying, aggressive behaviour, confusion or stupor) should be identified carefully and appropriately intervene. Other parameters include evaluation of growth and development as well as stigmata of chronic liver disease (clubbing, anaemia, palmar erythema, spider angioma, xanthomas, pruritus marks, prominent abdominal vessels, left lobe hypertrophy, splenomegaly, ascites and growth failure). Some specific examination findings depending upon the age and presenting complaints may point towards specific aetiologies. KF ring is seen in Wilson’s disease, cataract in galactosemia and retinopathy in congenital infections which can all present with ALF. Other specific examination findings include dysmorphism, multisystemic involvement (metabolic disorders), arthritis and skin rash (autoimmune hepatitis), signs of haemolysis like cola-coloured urine and severe pallor (Wilson’s disease or autoimmune hepatitis), renal failure suggested by oliguria, anuria (Wilson’s disease, Leptospira or hepatitis A infection).
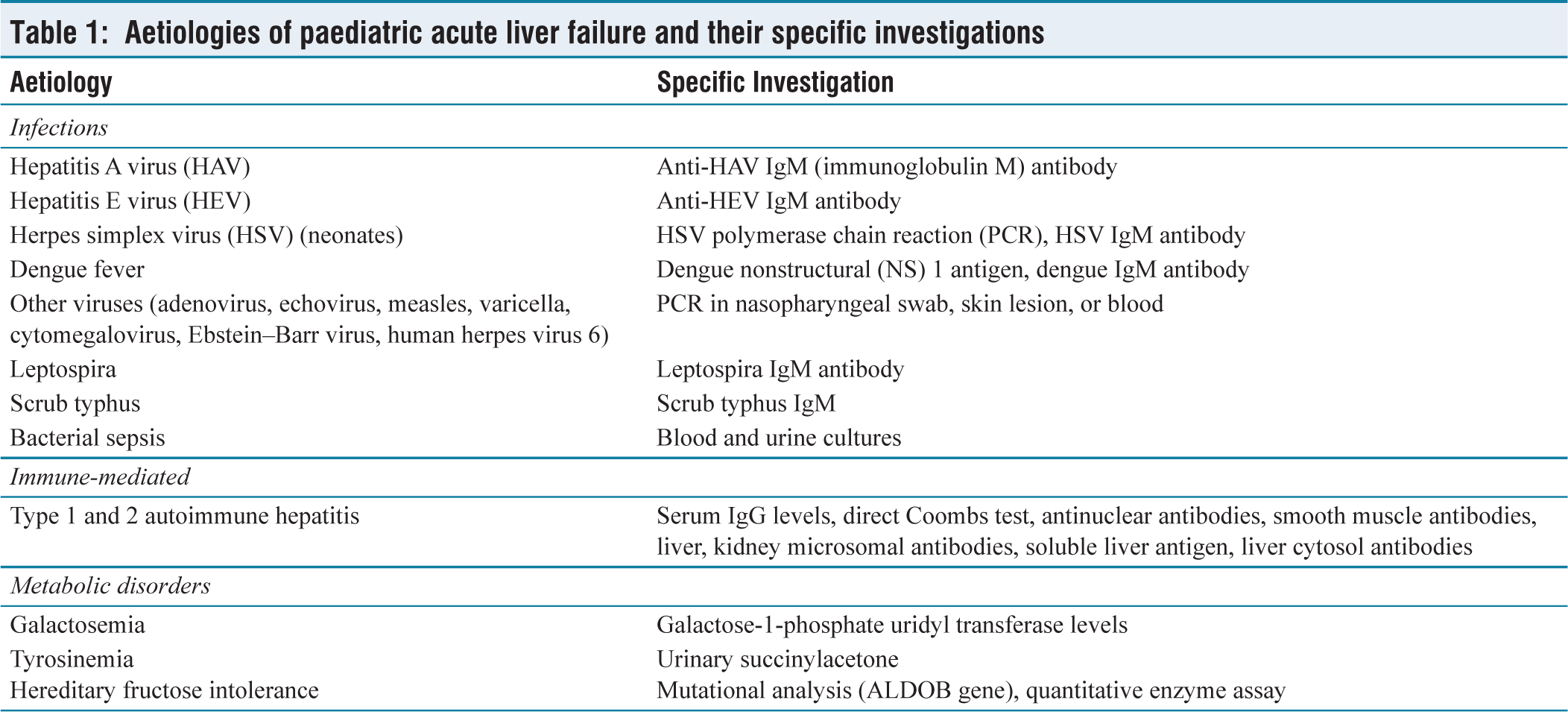
Lab investigations should be tailored to monitor the progression of liver failure and to identify treatable aetiologies. Liver function tests, coagulation profiles and ammonia trends are regularly trended to monitor the progression. Complete hemogram and renal function tests, electrolytes, blood glucose and lactate are other parameters needing regular monitoring. Ultrasound abdomen and Doppler may help identify children with acute chronic liver failure. Liver biopsy via transjugular route may be attempted in certain children suspected to be suffering from autoimmune hepatitis, lymphoreticular malignancies or other multisystem aetiologies that may render these children unsuitable for LT. It is usually safe when performed by experts with appropriate measures. However, it is costly, requires specialised equipment, has an inherent risk of bleeding and may not be possible in children younger than two to three years.[6] Aetiology-specific investigations can be planned depending on the age, clinical presentation and prevalent causes in the particular geographical area [Table 1].
Aetiologies of paediatric acute liver failure and their specific investigations

Management
General Management Principles
Children with ALF should be nursed in a quiet environment; vital parameters like heart rate, mean arterial blood pressure, respiratory rate, oxygen saturation, Glasgow coma scale and pupillary reaction should be regularly monitored using appropriate equipment. Elective intubation and mechanical ventilation should be started in patients with significant encephalopathy (Grade II).
Intravenous fluids should be started depending on the clinical status to maintain euvolemia as both positive and negative fluid balances may be detrimental. Vomiting and poor oral intake may lead to dehydration and few children may be hypotensive or in shock due to systemic inflammatory response requiring fluid resuscitation or vasopressors. Crystalloids should be used with careful avoidance of hypovolemia and hypervolemia. As hypoglycaemia is a common complication of liver cell failure, and hypoglycaemia may be detrimental to the already affected brain, blood glucose levels should be regularly monitored and appropriate glucose infusion rates should be continued to keep blood glucose in normal range (100–150 mg/dL). ALF is a state of immune dysregulation and these children are at an increased risk of infections and sepsis is an important cause of mortality. Bacterial infections may account for 60%–100% and fungal for 40% with average time to bacteraemia as short as two days for bacterial infections and four days for fungal infections.[7] There should be a low threshold for starting prophylactic antibiotics in children with ALF. Broad-spectrum antibiotics covering both gram-positive and gram-negative bacteria and antifungals should be started. Timely upgradation of antibiotics is warranted depending upon the clinical course, culture sensitivities and prevalent flora in the respective institution. The idea of protein restriction to limit the possibility of HE has now been disregarded and adequate calories should be provided. A plant-based diet which contains more branched-chain amino acids is preferred over animal protein that contains more aromatic amino acids. Proton pump inhibitors should be started in all children, especially those who are on mechanical ventilation as stress ulcerations may lead to bleeding. N acetylcysteine (NAC) has a proven role in children with acetaminophen poisoning but recent studies on the use of NAC in children with non-acetaminophen ALF failed to show any significant benefit when compared to placebo.[8]
Monitoring and Management of Cerebral Oedema
Cerebral oedema is the most frightful complication of ALF which may progress rapidly leading to irreversible brain damage; so, every possible measure should be taken to identify as well as prevent the progression of cerebral oedema and raised intracranial pressure (ICP). Factors associated with raised ICP include hyperacute ALF, haemodynamic instability requiring vasopressors, high serum ammonia, Grade III or IV encephalopathy and sepsis.[9] West Haven criteria and modified HE assessment scale are used for the assessment of HE in children more than and younger than three years respectively. Symptoms and signs of cerebral oedema include altered sleep cycle, excessive irritability, inconsolable crying, aggressive behaviour, confusion or stupor.[10] Examination findings suggestive of significantly raised ICP include 6th nerve palsy, Cushing’s triad (bradycardia, hypertension and irregular breathing), spasticity and decerebrate posturing. In addition to signs and symptoms of HE, pupillary size and reaction should be carefully monitored in all children with ALF.
Several noninvasive measures have gained importance in monitoring ICP. The commonly used noninvasive measures for monitoring raised ICP and reduced cerebral perfusion include optic nerve sheath diameter (ONSD), transcranial Doppler (TCD), near-infrared spectroscopy (NIRS) and reverse jugular venous oxygen saturation (SjvO2). ONSD is most commonly used due to the easy availability of ultrasound in most of the intensive care units managing these sick patients. ONSD values more than 4.6 mm signify raised ICP in children with ALF and persistent elevation for more than 24 hours is suggestive of poor prognosis.[11] ONSD can be used to monitor the progression or response to treatment. Using TCD to assess the middle cerebral artery dynamics is another important noninvasive tool to study elevated ICP. Raised ICP leads to increased peak systolic velocity, decreased end-diastolic flow velocity and decreased mean flow velocity, leading to a high pulsatility index.[12] SjvO2 refers to oxygen saturation measured in the jugular vein (venous drainage of the brain). Normal SjvO2 is 60%–80% and SjvO2 less than 60% signifies increased oxygen consumption in the brain suggestive of increased metabolic activity in the brain due to seizures, fever or raised ICP. On the other hand, SjvO2 of more than 80% signifies brain hyperaemia, thus guiding appropriate strategies for raised ICP management.[13] NIRS also provides an estimate of cerebral oxygenation and cerebral blood flow.[14] It is recommended to use two modalities simultaneously depending on expertise and availability. Children with ALF may have clinical or subclinical seizures. Subclinical seizures may easily be missed unless monitored by continuous electroencephalogram (EEG). It is important to monitor for subclinical seizures as they may further exaggerate the ICP. It is recommended to use nonhepatotoxic antiepileptics prophylactically to prevent seizure activity especially when continuous EEG is not being monitored.
To prevent the progression and detrimental consequences of raised ICP, all children should be nursed in a quiet room with minimal stimulation, adequate sedation (if intubated) and head elevation to 20–30 degrees to improve venous drainage and as a preventive measure for aspiration pneumonia. Oxygen saturation should be maintained above 95% to prevent hypoxemia of an already compromised brain. Hyperosmolar therapy using hypertonic saline and/or mannitol is the first line of therapy for the prevention as well as management of cerebral oedema. Three per cent saline to maintain serum sodium between 145 and 150 mEq/L is commonly used to create a more favourable osmotic gradient to extract water from the brain.[15] Mannitol is an osmotic diuretic; a rapid bolus of 0.5 gm/kg over 15 minutes is recommended in an acute rise in ICP and the dose can be repeated if the serum osmolarity is less than 320 mOsm/L.[16] In case of an acute rise in ICP, hyperventilation could be done for a brief period till there is clinical improvement. There is no consensus on the use of propofol or dexmedetomidine for sedation in PALF. Thiopentone infusion can be considered in refractory cases. Normocarbia and euthermia should be maintained in all children.
Coagulopathy
As INR is a dynamic indicator of disease progression, coagulopathy should be corrected only if the patient is bleeding actively or before a high-risk invasive procedure. Bleeding manifestations are uncommon in ALF as there is a proportional reduction in both procoagulants and anticoagulants.[17] This delicate balance can get disturbed by sepsis, disseminated intravascular coagulation, thrombocytopenia or hyperfibrinogenaemia. The most common site of bleeding is the gastrointestinal (GI) tract and major bleeding during low-risk procedures is also uncommon.[18] Thromboelastography (TEG) may act as an important tool in guiding about the risk of bleeding. To correct coagulopathy, fresh frozen plasma could be given at a dose of 10 mL per kg and cryoprecipitate at 5 mL per kg (if fibrinogen is < 1 gm/L). Platelet count should be maintained above 50,000 as it is an important cause of bleeding.
Specific Therapies
Some of the aetiologies of PALF may be amenable to specific treatment, thereby changing the fulminant course of illness. All attempts should be made to identify these treatable causes and appropriate therapies instituted as early as possible to prevent irreversible damage. NAC in paracetamol-induced ALF at a rate of 6.25 mg/kg/hr until normalisation of the INR has been shown to be efficacious.[19] Evidence of NAC in nonacetaminophen ALF has been inconclusive. Autoimmune hepatitis presenting as ALF may be considered for oral or intravenous steroids in patients with no or early encephalopathy. All children with high-grade encephalopathy or poor PELD scores should be prepared for LT before irreversible injury ensues.[20] Other aetiologies amenable to specific therapies include gestational alloimmune disease in the neonatal period (intravenous immunoglobulin coupled with exchange transfusion), herpes simplex virus-induced ALF (acyclovir), galactosemia (lactose-free diet), tyrosinemia (nitisinone), hereditary fructose intolerance (fructose free diet), HLH (immunosuppression, bone marrow transplant) or hepatitis B (entecavir). LT should be considered at appropriate times in all these aetiologies, if there is no improvement or as per standard indications based upon various prognostic scores as well as clinical features.
Bridging Therapies/Liver-assist Devices
ALF is associated with systemic inflammatory response syndrome in response to microbial or non-microbial factors, resulting in dysregulation of the immune system and accumulation of toxins and inflammatory cytokines which not only further perpetuates the liver injury but also leads to multisystemic dysfunction, and cerebral oedema and culminates in sepsis. Liver assist devices or extracorporeal liver support systems (ELSS) act by removing these toxins, and inflammatory mediators from the blood with simultaneous replacement with plasma or albumin. These may play an important role in bridging PALF to spontaneous recovery or LT. Various modalities evaluated for the paediatric population include therapeutic plasma exchange (TPE), continuous renal replacement therapy (CRRT) and molecular adsorption and recirculation system (MARS). TPE acts by removing pro-inflammatory cytokines, inflammatory macromolecules and toxins, resulting in the amelioration of the systemic inflammatory response system (SIRS) and the improvement of phagocytic function.[21] The volume of plasma exchanged varies from 0.5 times (low volume), 1.5–2 times (standard volume) to 2–3 times (high volume), depending upon the underlying aetiology and haemodynamic stability of the patient. As it is intermittent and ALF is a rapidly progressive disease, its effect may be short-lived. It has been shown to play an important role in PALF due to Wilson’s disease or due to other toxic ingestion such as yellow phosphorus.[22] CRRT (continuous venovenous haemofiltration) is the most commonly available and applied ELSS for PALF. It is highly efficacious in removing water-soluble toxins such as ammonia, lactate, urea and creatinine. Indications for starting CRRT in PALF include acute kidney injury suggested by oliguria and anuria resulting in fluid overload, metabolic acidosis (pH < 7.1), persistent hyperlactatemia, hyperammonaemia (>150 µmol/L) and dyselectrolytaemia.[23] CRRT may be combined with TPE for better outcomes.[24] MARS is an efficacious ELSS that removes both water and protein-bound toxins without the need for replacement with exogenous blood products.[25] Its high expenses and need for specialised equipment limit its use. As ELSS can artificially alter crucial prognostic parameters, it may be difficult to know the trajectory of the disease and the need for further intervention like LT. In children on CRRT, INR and bilirubin can be used for prognostication while in those on TPE, rebound INR after TPE (50% increase in 12 hours or 100% increase in 24 hours) may be used for guiding further treatment. While on ELSS, one has to carefully assess the need for LT based upon the age of the child, aetiology of ALF, overall clinical progression of disease and response to treatment.
Prognostic Scores
LT is life-saving when a condition without specific therapy is irreversible or fails to respond to treatment. Also, LT has profound lifelong consequences on both the recipient and the family with its associated morbidity, risks and need for lifelong immunosuppression. Immediate complications and long-term outcomes following LT for ALF are inferior to those for chronic liver diseases. Hence, a suitable prognostic model is needed to readily and effectively differentiate the patients unlikely to respond to medical and supportive management, from the ones who would survive without LT. The liver injury in PALF is rapidly evolving, so it is also important to identify when is the appropriate time to go ahead with LT and when it would be futile to proceed with LT. Widely used prognostic criteria include King’s College Hospital (KCH) criteria, paediatric end-stage liver disease score (PELD score), model for end stage liver disease (MELD) score, liver injury unit and Children’s Hospital of Los Angeles-Acute Liver Failure Score.[26] KCH criteria should be used for LT listing and in children fulfilling KCH criteria, an INR > 4 carries a high mortality needing close monitoring and preparation for LT [Table 2].[27,28] As aetiology is one of the most important determinants of outcome in ALF, several aetiology-specific prognostic scores have been discussed and validated. Peds HAV model for hepatitis A virus-induced ALF, New Wilson Index for Wilson’s disease-associated ALF, Kochi criteria for Yellow Phosphorus poisoning-induced ALF have been found to offer better prognostic-accuracy-as compared to non-aetiology specific prognostic models.[29,30] Wherever possible aetiology-specific prognostic models should be used for determining need for and listing for LT.
Kings College Hospital criteria

Liver Transplantation
All children with PALF should be managed at a centre equipped with paediatric LT facilities. LT has significantly improved the outcome of PALF in the last decade. With the advent of newer surgical techniques, specialised equipment as well as ever-improving paediatric intensive care, both the short- and long-term survival rates post-LT for PALF, has improved. Split liver grafts, reduced grafts and blood group incompatible transplants have increased the number of patients who can use this life-saving modality when nothing else is helping.
Careful patient selection needs to be done, and several prognostic indices (general and aetiology specific) as discussed above may be used to identify patients unlikely to improve with medical management. Absolute contraindications for LT include fixed and dilated pupils suggestive of irreversible brain injury, uncontrolled sepsis and multisystemic mitochondrial or other metabolic disorders. Also, attempts should be made to rapidly determine the aetiology of PALF to identify conditions amenable to specific treatment as well as those posing contraindications to the same.
To conclude, ALF in children is a medical emergency as it may progress rapidly leading to cerebral oedema, irreversible brain damage and multi-organ dysfunction. All children with coagulopathy or encephalopathy should be referred to tertiary care centres equipped with taking care of such children and LT services. All attempts should be made to identify aetiologies to administer specific treatment in the early course of the disease and to prognosticate when specific treatment is not available. Extracorporeal liver assist devices have shown promising results in bridging these children to native liver survival or LT, but continuous careful monitoring should be done to assess for the treatment failure and need for LT. LT is the only definite treatment that improves survival in PALF and with several advances in surgical techniques and good intensive care, the overall survival after LT for ALF has improved significantly.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Not applicable.
Credit author statement
Jaswinder Kaur: Writing: Original draft.
Smita Malhotra: Conceptualization, Methodology, Writing-review and editing, Supervision.
Karunesh Kumar: Writing-review and editing.
Anupam Sibal: Supervision.
Data availability
Not applicable.
Use of artificial intelligence
No use of AI.
