Abstract
Background:
Cerebral venous thrombosis (CVT) is a rare form of stroke in infants and young children, with multifactorial causation and a poor prognosis. Bilateral haemorrhagic thalamic infarction secondary to deep CVT is an extremely rare presentation.
Case report:
We report the case of a 12-month-old infant with bilateral haemorrhagic thalamic infarction secondary to deep CVT, presenting with fever and seizures. The infant was initially diagnosed with acute meningoencephalitis of viral aetiology and treated accordingly. However, the patient had a sudden clinical deterioration. Neuroimaging revealed deep CVT with bilateral haemorrhagic thalamic infarction. Anticoagulant therapy in conjunction with physiotherapy was initiated and the infant gradually improved and was discharged.
Conclusion:
CVT is a rare form of stroke in infants and young children, with multifactorial causation and poor prognosis. Our case highlights the importance of careful evaluation of cerebral venous drainage on routine neuroimaging in infants with fever and seizures, especially in those who do not respond to standard treatment for meningoencephalitis.
Synopsis:
Here we report a unique and challenging case of deep cerebral venous thrombosis with haemorrhagic infarction of bilateral thalami mimicking viral encephalitis in an infant with acute febrile encephalopathy.
Background
Cerebral venous thrombosis (CVT) is an uncommon cause of stroke in infants, with a reported incidence of 0.6-1.3 per 100,000 children per year.[1] CVT is common in neonates and is rare in infants and older children. In infants, the risk factors include dehydration, fever, anaemia, sepsis, haemolytic anaemias, congenital heart disease, chronic disorders like inflammatory bowel disease, nephrotic syndrome and autoimmune disorders.[2] Deep CVT in children is an extremely rare form of venous thromboembolism and shows multifactorial causation with poor prognosis.
Case Presentation
A 12-month-old infant was brought with complaints of unresponsive fever for 7 days. The infant was symptomatically treated in a local secondary health care facility and was referred to our hospital in view of recurrent episodes of seizures and projectile vomiting over the past few hours. On general examination, the infant was undernourished, febrile and stuporous with a GCS of E1 V1 M3. Examination of the central nervous system revealed dystonia with signs of meningeal irritation. Provisionally, a diagnosis of acute meningoencephalitis was considered. Mechanical ventilation was initiated. Fundoscopy showed papilledema. Hemogram with peripheral blood smear examination revealed severe microcytic hypochromic anaemia leucocytosis. CSF evaluation showed elevated level of proteins (232 mg/dl), normal sugar level (85 mg/dl) and leucocyte count of 5 cells/cu.mm with all lymphocytes. CSF culture revealed no growth. A clinical diagnosis of acute meningoencephalitis of viral aetiology was considered. The infant was treated with intravenous fluid replacement, blood transfusion, antiepileptics and cerebral decongestants (parenteral hypertonic saline and mannitol). Initially, there was partial resolution of the signs of intracranial hypertension following which sudden clinical deterioration was observed with reappearance of the features of intracranial hypertension and meningism. Neuroimaging revealed deep CVT with haemorrhagic infarction of bilateral thalami. Anticoagulant therapy in conjunction with physiotherapy was initiated. Child gradually improved and was discharged.
Imaging Findings
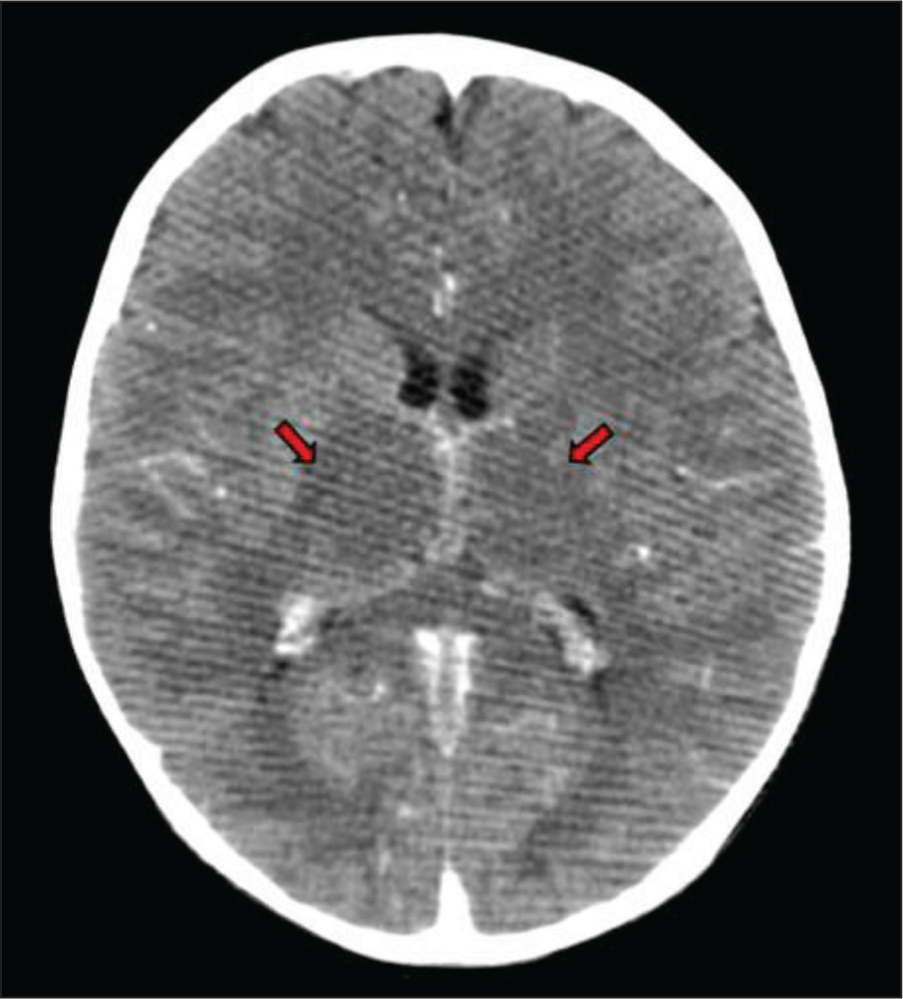
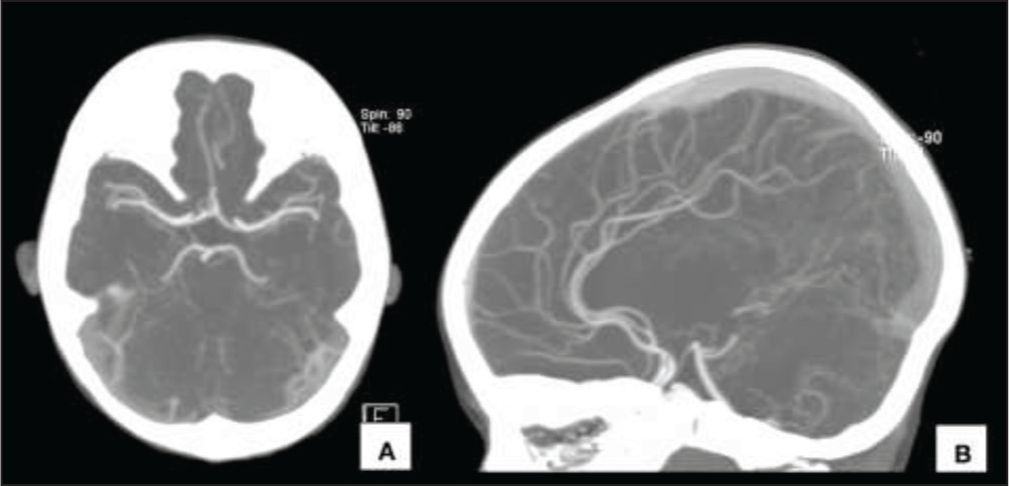
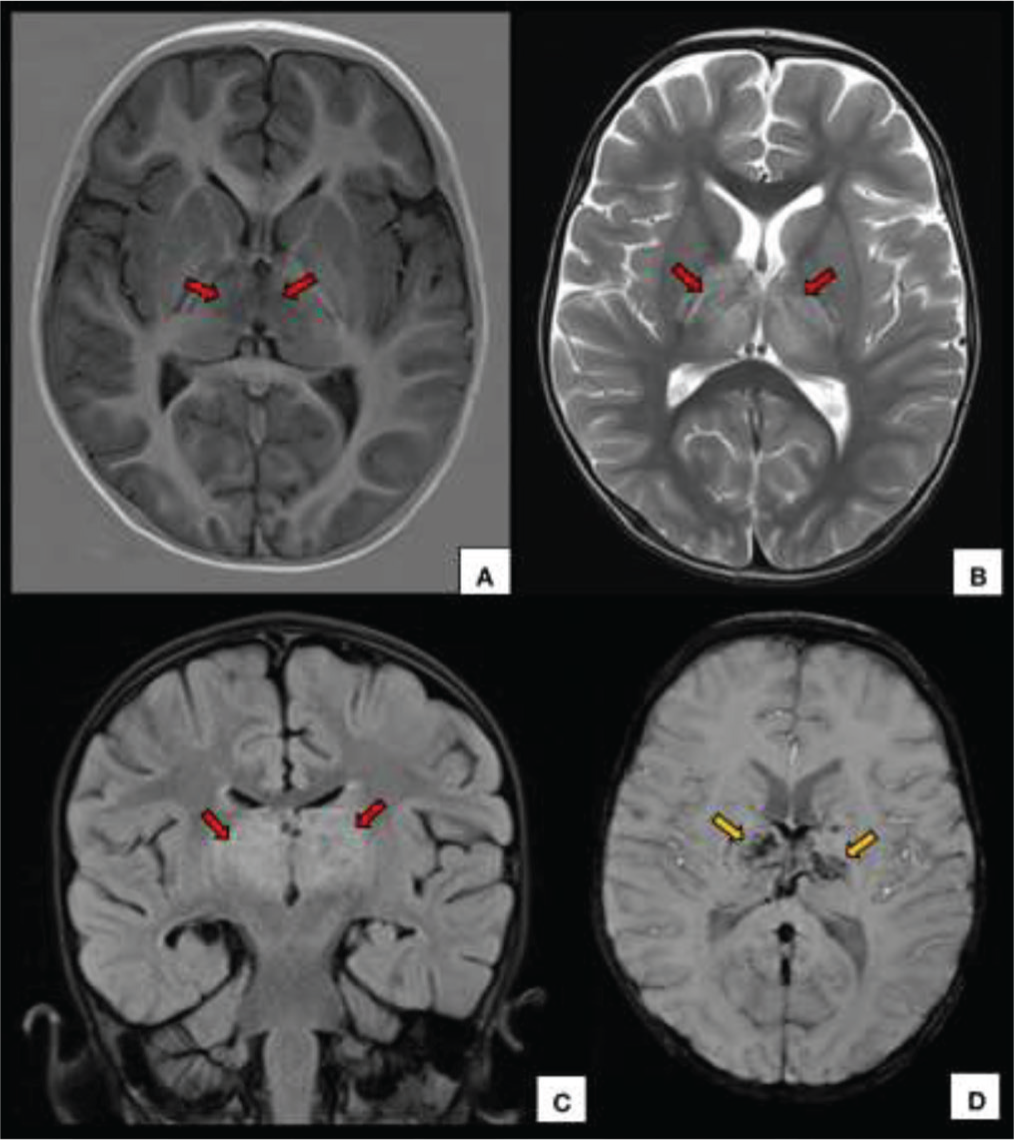
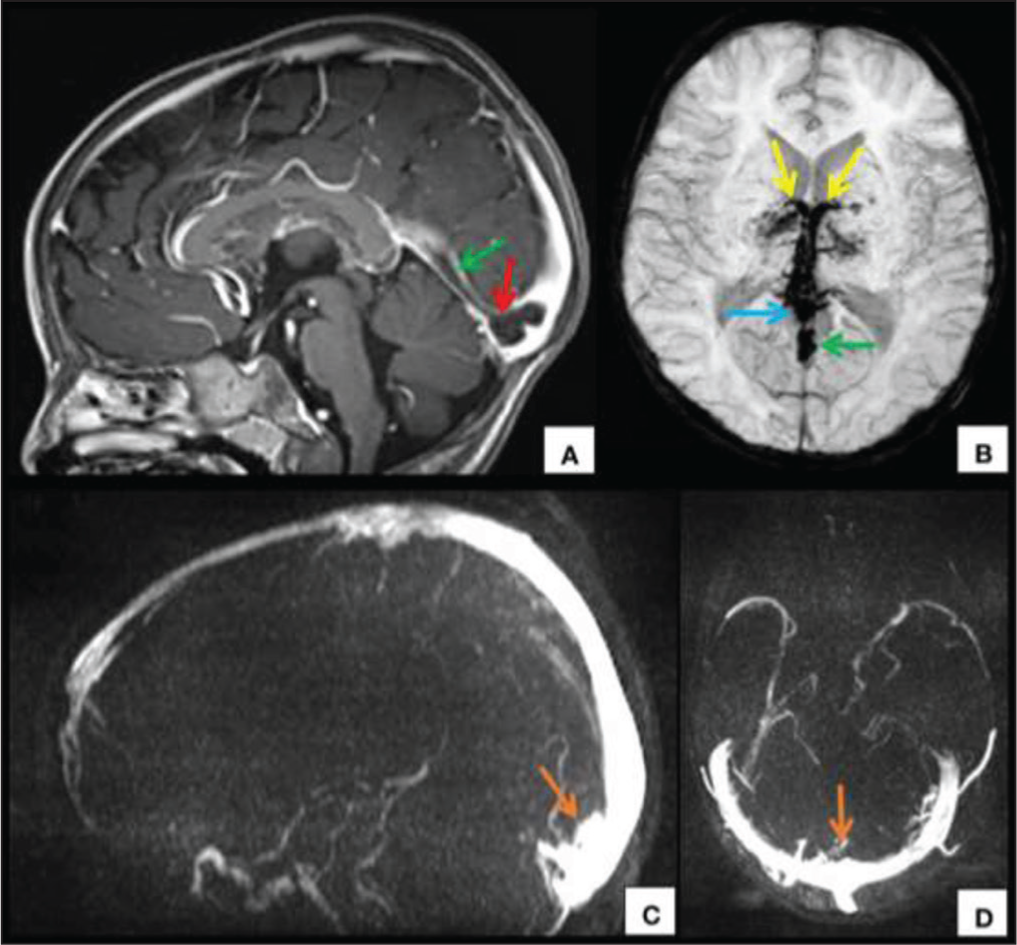
CECT of the brain showed symmetrical hypodensity involving the bilateral thalami [Figure 1] and normal angiography [Figure 2]. MRI revealed symmetric enlargement of bilateral thalami with altered signal intensity appearing hyperintense on T2W/FLAIR images and hypointense on T1W images with multiple foci of haemorrhage and no evidence of restricted diffusion [Figure 3]. Adjacent posterior limbs of bilateral internal capsules were also involved. Magnetic susceptibility was seen along the bilateral internal cerebral veins, vein of Galen and straight sinus. MR venography revealed non-visualisation of the straight sinus and the Galenic venous system. Post-contrast images showed a hypointense filling defect in the torcular herophili extending into the straight sinus, vein of Galen and bilateral internal cerebral veins [Figure 4]. A radiological diagnosis of deep CVT with bilateral haemorrhagic thalamic infarction was made.
Discussion
CVT comprises thrombosis of dural venous sinuses, cortical veins and deep veins of which thrombosis involving the deep venous system is the rarest form and carries a poor prognosis. Deep cerebral venous system drains the deep white matter and central grey nuclei and includes internal cerebral veins, basal vein of Rosenthal, and vein of Galen. It is centred at the vein of Galen (also known as the Great cerebral vein) which drains into the straight sinus. Bilateral thalamic infarction is seen in thrombosis involving the straight sinus and vein of Galen and is often associated with haemorrhage secondary to venous congestion. Few cases of unilateral thalamic involvement have also been reported if diagnosed earlier during the course.[3,4]
Differential diagnoses include infective, vascular and metabolic causes.[5] Infective causes include viral encephalitis (Japanese encephalitis, Influenza and West Nile encephalitis) which is associated with meningeal enhancement on imaging. Meningeal enhancement can also be seen following lumbar puncture for CSF examination. Vascular causes comprise of hypoxic ischaemic encephalopathy (HIE) and artery of Percheron infarct. HIE secondary to hypoperfusion is common in infants with dehydration and sepsis. Diffusion-weighted imaging is a most sensitive sequence. Imaging findings include T2W/FLAIR hyperintensity involving basal ganglia and thalami with restricted diffusion. Artery of Percheron infarction is seen as symmetric involvement of bilateral paramedian thalami and rostral brainstem. Metabolic causes include extrapontine osmotic myelinolysis, Leigh’s disease and Wilson’s disease. Extrapontine osmotic myelinolysis is associated with rapid correction of hyponatraemia and might show restricted diffusion. Patients with Leigh’s disease typically exhibit elevated lactate levels in both serum and CSF, abnormal involvement of the basal ganglia on imaging, and a distinct lactate peak at 1.3 ppm on MR spectroscopy. Wilson’s disease presents with hepatomegaly, Kayser Fleischer ring and basal ganglia involvement.
The management is complex, and treatment options include anticoagulation, thrombolysis, and surgical intervention. Anticoagulation is the mainstay of therapy, and the recommended duration of treatment is usually 3 to 6 months.[6] Thrombolysis is reserved for cases where anticoagulation is contraindicated or ineffective. Surgical intervention is considered in cases of massive intracranial haemorrhage, deteriorating consciousness, or progressive neurological deficits. Complications include haemorrhagic infarcts, intracranial haemorrhage, hydrocephalus, seizures, and cerebral oedema. Long-term complications are cognitive deficits, behavioural abnormalities, and epilepsy.
Conclusions
Deep CVT in children is an extremely rare form of venous thromboembolism and shows multifactorial causation with poor prognosis. It is a rarely recognised cause of infantile stroke and can radiologically mimic features of acute viral encephalitis and metabolic encephalopathies. Possibility of deep CVT should be excluded on imaging in febrile infants with deep grey nuclei involvement as early diagnosis is warranting. The necessity for careful evaluation of cerebral venous drainage on routine neuroimaging cannot be overemphasised.
List of abbreviations
GCS- Glasgow coma scale
CSF- Cerebrospinal fluid
CECT- Contrast-enhanced computed tomography
MRI- Magnetic resonance imaging
FLAIR- Fluid- attenuated inversion recovery
T2W- T2 weighted
SWI- Susceptibility weighted imaging
MPRAGE- Magnetisation- prepared rapid gradient- echo
CVT- Cerebral venous thrombosis
HIE- Hypoxic ischaemic encephalopathy
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
The case was a part of a research project and a blanket institutional ethical committee approval will be applicable (dated 14/01/2022 with ref. no. 82/10/2020/200). All procedures performed in this study involving a human participant were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from the parents of the patient to publish the case and use her radiological involved in the preparation of this manuscript.
Credit author statement
EA: guarantor of integrity of the entire study, study of concept and design, literature research, manuscript preparation, manuscript editing.
AA: guarantor of integrity of the entire study, study concepts and design, literature research, manuscript preparation, manuscript editing.
GK: study concepts and design, literature research, manuscript editing.
GSP: study concept and design, manuscript editing.
SS: study concept and design, manuscript editing.
ASS: study concept and design, manuscript editing.
Data availability
Data sharing is not applicable to this research article as no new data were created or analysed in this study.
Use of artificial intelligence
Authors would like to acknowledge the use of a generative AI tool (ChatGPT v. 3.5) for language improvement only.
