Abstract
Background:
Calcific myonecrosis (CMN) is a rare condition, often considered to be a late sequela of antecedent trauma.
Case Report:
We describe here our experience in managing a 56-year-old male referred with a painless swelling on the lateral aspect of the left lower leg with a suspicion of a tumour.
Results:
Clinical and imaging features were classic of CMN.
Conclusion:
Clinical assessment, role of ultrasound scan in reaching a diagnosis and the management are highlighted.
Keywords
Introduction
Calcific myonecrosis (CMN) is a rare entity occurring latent to historic trauma and may mimic a soft tissue tumour on an ultrasound scan (USS). It was first reported in 1960 by Galie and Thompson as a subsequent consequence of compartment syndrome of the leg.[1] Clinically, it is presented as a slowly enlarging, firm and painless mass which most frequently occurs in the anterior compartment of the lower leg and less commonly in other lower limb, thigh or forearm compartments. It can present a diagnostic challenge given its overlapping features with other benign and malignant soft tissue tumours or mimics, with a few case reports describing its sonographic appearances.[2-5] Given that it is most commonly managed conservatively, accurate diagnosis is essential to reduce patient morbidity from unnecessary biopsy and surgery.[6]
Case Report
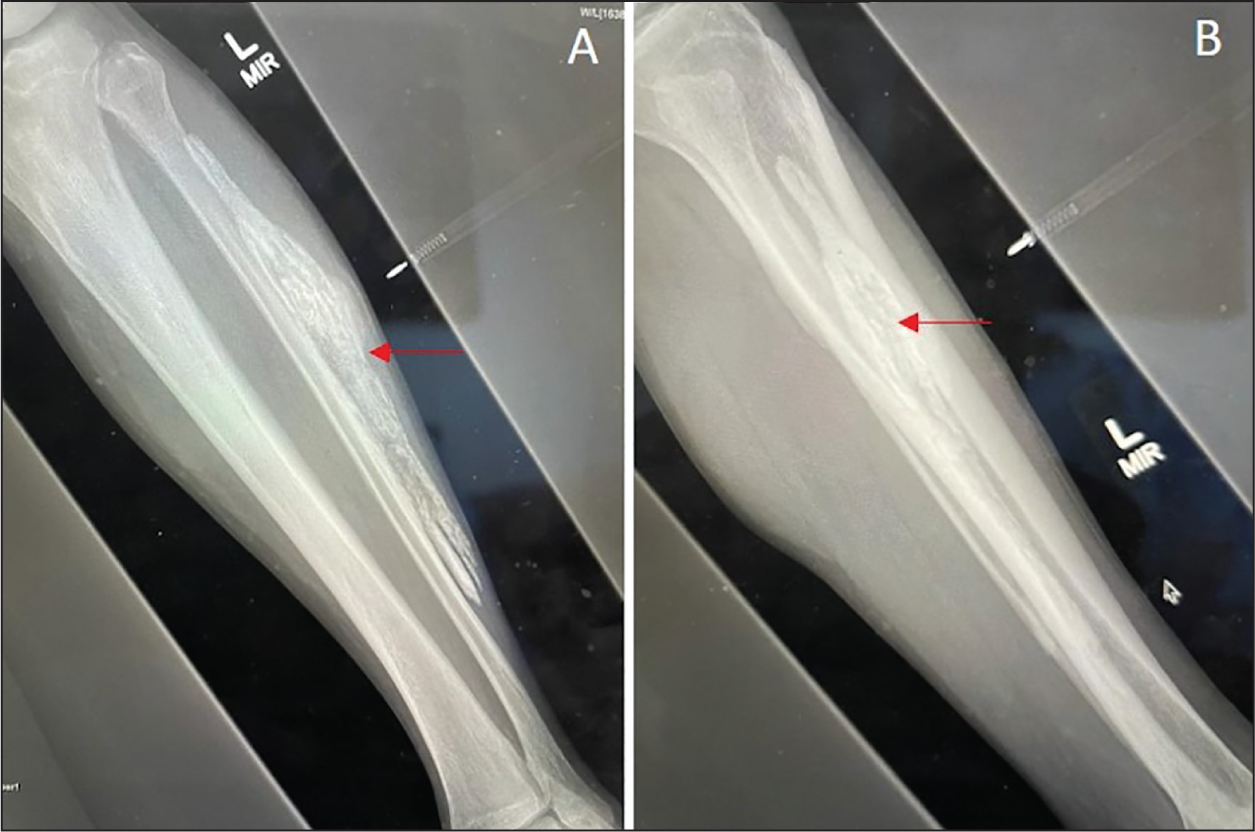
A 56-year-old male was referred to our Tertiary Oncology unit, with swelling on the lateral aspect of the left lower leg growing slowly. The patient reported that the painless mass had been slowly enlarging for many years without significant impact on daily activities. His biochemical and inflammatory markers were within normal limits. On examination, there was a 15 cm × 4 cm linear and ovoid mass within the lateral compartment of the lower leg. There were no overlying skin changes, scars or sinus tracts. The mass was of firm consistency, non-tender and fixed to underlying structures. The patient was neurovascularly intact with normal movements at the knee and ankle joints. A standing radiograph was performed which demonstrated a fusiform mass with sheet-like calcifications, within the lateral compartment of the lower leg [Figure 1].
Ultrasound of calcific myonecrosis within the lower limb lateral compartment. (A) Axial and (B) longitudinal images demonstrate a cystic lesion with internal mobile debris. There is a hyperechoic periphery and internal echogenic foci corresponding to low-density calcification with minimal posterior acoustic shadowing (red arrows). There is also Irregularity of the underlying fibula (yellow arrows). Fib = fibula
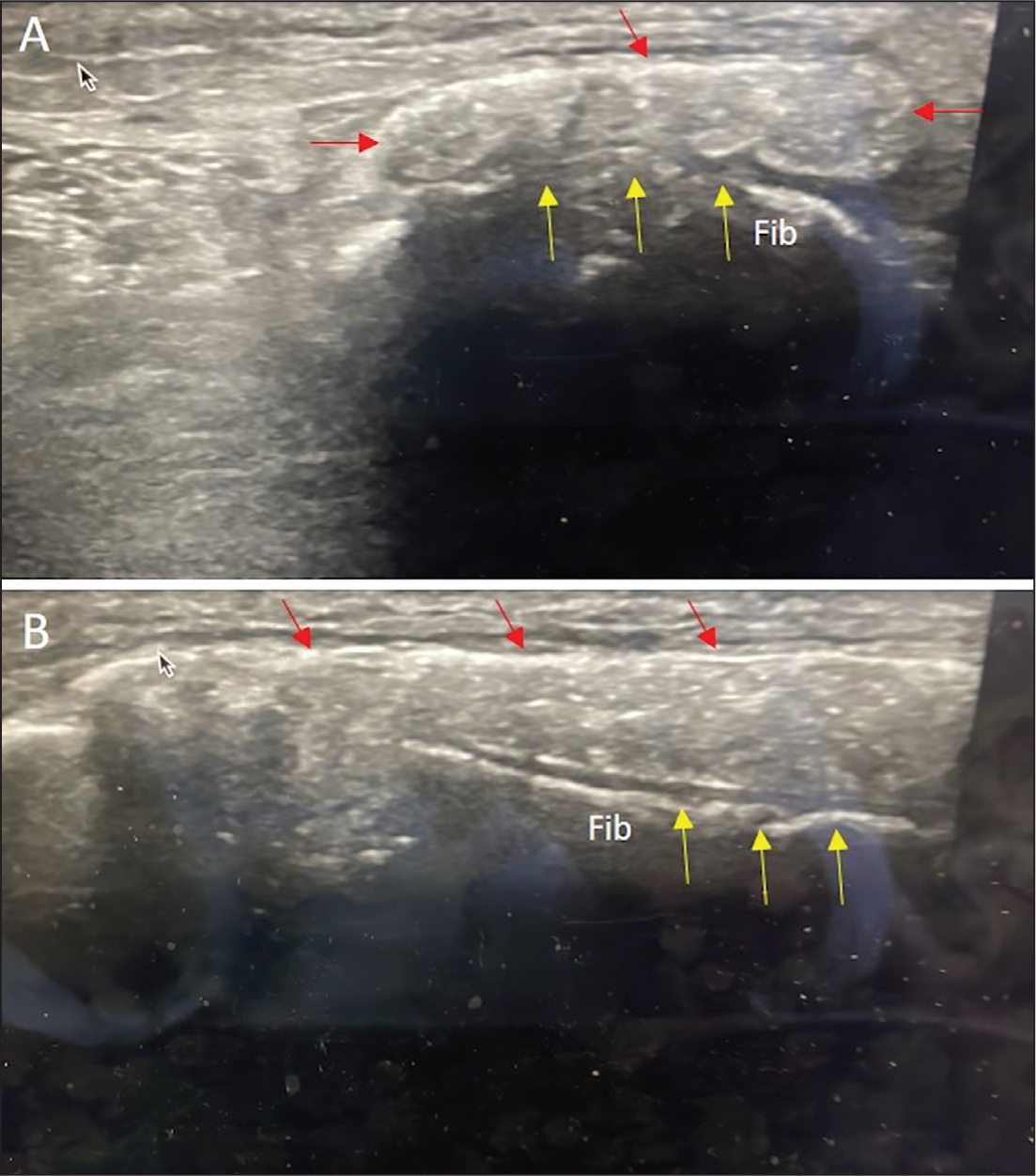
On USS, the mass appeared as a large predominantly cystic lesion with internal mobile debris, confined to the lateral muscle compartment. It demonstrated a linear hyperechoic rim and internal hyperechoic foci, suggestive of low-density calcification with deeper cortical irregularity of fibula. It demonstrated no internal vascularity on doppler studies [Figure 2]. Magnetic resonance imaging (MRI) demonstrated diffuse predominantly low-signal abnormality on T1 and fluid-sensitive sequences involving the entire peroneal.
Radiographs of calcific myonecrosis within the lower limb lateral compartment. (A) and (B) demonstrate a mass in the lateral compartment with sheet-like calcifications
The patient reported a historic trauma to the lower leg 40 years prior to his clinic attendance. The clinical and radiological findings were diagnostic of CMN. The patient was informed, reassured about the benign nature and was counselled about the high rate of complications that can be associated with attempted surgical intervention. Following a shared decision-making process, he opted for conservative management. In the interim, he was advised to continue with his normal daily activities, allowing full weight-bearing mobilisation and monitor the mass for increasing size or new or concerning symptoms such as a sudden increase in size or pain or feeling acutely unwell.
Discussion
It is currently understood that CMN occurs as a long-term sequel of post-traumatic compartment syndrome complicated by necrosis, fibrosis and repeat intralesional haemorrhage which subsequently forms a slowly growing mass.[7] The underlying trauma, which may be a high-impact or repeat low-impact injuries, is reported to occur between 10 and 64 years before clinical presentation.[7,8] It most frequently occurs in the lower leg anterior compartment followed by the lateral and deep posterior compartments, with few reports occurring in the forearm.[6,9-12]
Ultrasound is an effective tool for the initial investigation of soft tissue tumours owing to its relatively low cost, wide availability and ease of access to clinicians. The reported sonographic appearance of CMN is of a peripherally calcified mass with central liquefication and no internal vascularity. If the calcification is sufficient to allow acoustic penetration (low concentration calcification),[13] internal matrix and underlying cortical irregularity may be observed, such as in our case.[5,14] Other authors have reported dense calcification with acoustic shadowing, owing to a higher concentration of calcium, despite a central liquefied component being demonstrated on cross-sectional imaging.[3,4,15-17] CMN may demonstrate overlapping features with other benign entities and can be misdiagnosed as an aggressive lesion. Key differential diagnoses are highlighted in Table 1.
Differential diagnosis of calcific myonecrosis on ultrasound (USS) imaging

Myositis ossificans is a benign entity of extraosseous heterotopic ossification within the muscle and soft tissues. Sonographic appearances vary but may be differentiated by its dense calcification with posterior acoustic shadowing, lack of a cystic component and relatively short history after injury. Chronic haematoma may also demonstrate calcification in the chronic phase. It is typically associated with a history of antecedent trauma, relatively short history after known trauma (weeks to months) and demonstrates reticular or ‘lace-like’ appearance in the subacute phase.[18] Post-traumatic pseudoaneurysm demonstrates internal vascular flow on Doppler studies (yin-yang sign), close proximity to a vessel and relatively less pronounced calcifications.[19] Tumoral calcinosis demonstrates similar calcification and cystic components but can be distinguished by its lobulated appearance and distribution in peri-articular regions of large joints. In addition, biochemical markers may reveal co-existing renal failure.[20] Dermatomyositis and polymyositis generally have a more widespread pattern of involvement involving multiple areas of the appendicular skeleton.[3] Together with diabetic myonecrosis, which may develop calcification, a suggestive history is essential to clinch the diagnosis.
Aggressive lesions that demonstrate calcification, soft tissue component and cystic spaces include synovial sarcoma, epithelioid sarcoma and paraosteal osteosarcoma. These may be distinguished by mineralisation throughout the lesion rather than predominantly peripheral area.[2,21]
Although early reports have advocated surgical excision to treat CMN, this is associated with adverse complications such as large-volume bleeding, infection, chronic cutaneous fistulation and then need for subsequent below-knee amputation.[10,22-25] CMN is considered a ‘don’t touch lesion’; an awareness and recognition of its imaging appearances, thorough history taking avoid unnecessary interventions and potentially adverse complications.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors Rajesh Botchu and Karthikeyan P Iyengar are members of the Editorial Board of Apollo Medicine. The authors did not take part in the peer review or decision-making process for this submission and have no further conflicts to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional ethical approval number
Not required for this study.
Informed consent
The patient provided written informed consent to the authors for the publication of this article.
Credit author statement
All authors contributed significantly to the project.
Data availability
No.
Use of artificial intelligence
No.
