Abstract
Background and Aims:
This research aimed to determine which of three spinal anaesthetic medication combinations—clonidine–bupivacaine, buprenorphine–bupivacaine and bupivacaine alone—is the most effective for total abdominal hysterectomy.
Material and Methods:
In this prospective study, approved by the Institutional Ethical Committee, 120 patients aged 35–60 years scheduled for elective total abdominal hysterectomy under spinal anaesthesia were included, following informed consent. Comprehensive pre-anaesthetic assessments, including detailed medical histories, clinical examinations and routine surgical investigations, were conducted. The study, spanning 24 months, utilised purposive sampling and a cross-sectional design. The inclusion criteria encompassed ASA I and II patients aged 35–60 years, undergoing elective total abdominal hysterectomy with a weight exceeding 45 kg, height over 150 cm, and valid informed consent. Exclusion criteria involved ASA Class III or higher, patient unwillingness for spinal anaesthesia, gross spinal deformities, bleeding diathesis, known allergy to the test drug, history of cardiac, respiratory, or central nervous system disease, hepatic or renal dysfunction and elevated intracranial pressure.
Results:
The onset time of sensory blockade was faster in the Bupivacaine + Buprenorphine group than in the Clonidine + Bupivacaine group, with the Bupivacaine group exhibiting the slowest onset. The Bupivacaine + Buprenorphine group had a significantly faster onset of motor block than the Clonidine + Bupivacaine and Bupivacaine groups. The total duration of motor block was shorter in both the Bupivacaine + Buprenorphine and Bupivacaine groups than in the Clonidine + Bupivacaine group. The Clonidine + Bupivacaine group showed higher incidences of bradycardia and hypotension than the other groups. The Bupivacaine + Buprenorphine group reported more side effects, including chest pain, cough, nausea and vomiting, than the other groups.
Conclusion:
This study found that spinal clonidine, in comparison to bupivacaine alone, gives better post-operative analgesia and sensory and motor blockage over a longer period of time.
Introduction
Total abdominal hysterectomy (TAH) necessitates effective perioperative pain management to optimise patient recovery and satisfaction.[1] Neuraxial anaesthesia using local anaesthetics with adjuvants such as buprenorphine and clonidine is pivotal for superior pain control.[2] clonidine, a selective beta 2-adrenoceptor agonist with a high selectivity ratio (220:1) for beta 2 over beta 1 adrenoceptors, exhibits analgesic properties by inhibiting substance P release and reducing nociceptive neuron firing.[3,4] Acting centrally and peripherally, it decreases peripheral vascular resistance, heart rate, blood pressure and cardiac output, making it useful in labour analgesia, gynaecological and orthopaedic surgeries either alone or with other analgesics. Buprenorphine, a partial mu-opioid receptor agonist derived from thebaine, offers potent and prolonged analgesia owing to its high lipophilicity and strong affinity for beta opioid receptors, minimising the risk of late respiratory depression compared to morphine. It functions effectively at lower intrathecal doses, enhancing post-operative analgesia quality and duration when combined with bupivacaine.[5-9] The integration of buprenorphine and clonidine with local anaesthetics like bupivacaine in neuraxial anaesthesia has emerged as an effective strategy for enhancing perioperative analgesia while minimising adverse effects in TAH. This multimodal approach aims to achieve optimal pain management, facilitating early recovery and patient satisfaction. This study aims to assess and compare the efficacy of two drug combinations with a local anaesthetic agent in spinal anaesthesia for TAH: clonidine–bupivacaine, buprenorphine–bupivacaine and bupivacaine alone.
Material and Methods
In this prospective study, approved by the Institutional Ethical Committee, 120 patients aged 35–60 years, scheduled for elective TAH under spinal anaesthesia, were included, following informed consent. Comprehensive pre-anaesthetic assessments, including detailed medical histories, clinical examinations and routine surgical investigations, were conducted. The study, spanning 24 months, utilised purposive sampling and a cross-sectional design. The inclusion criteria encompassed ASA I and II patients aged 35–60 years, undergoing elective TAH with a weight exceeding 45 kg, height over 150 cm and valid informed consent. Exclusion criteria involved ASA Class III or higher, patient’s unwillingness for spinal anaesthesia, gross spinal deformities, bleeding diathesis, known allergy to the test drug, history of cardiac, respiratory or central nervous system disease, hepatic or renal dysfunction and elevated intracranial pressure.
Data were collected using multiparameter monitoring tools, a 25G spinal needle, 0.5% bupivacaine injection, 150 μg clonidine injection, 300 μg buprenorphine injection and an anaesthesia workstation. After ensuring a free flow of cerebrospinal fluid, Group A received injection bupivacaine 0.5% (heavy) 2.5 mL + saline 0.5 mL. Group B received injection bupivacaine 0.5% (heavy) 2.5 mL + 50 µg buprenorphine (in 0.5 mL of normal saline [NS]). Group C received injection bupivacaine 0.5% (heavy) 2.5 mL + preservative free injection clonidine 50 µg (in 0.5 mL of NS).
Methodology involved patient preparation, overnight fasting, administration of pre-operative medications and citing an intravenous line. Patients were monitored for various parameters before induction, followed by spinal block procedures based on group assignments. They were divided into three groups of 40 each, using the chit-in-a-box technique.
Parameters measured during the study included heart rate, systolic and diastolic blood pressure, mean arterial blood pressure and end-tidal CO2. Patient preparation included overnight fasting and pre-operative medications. Spinal blocks were performed using 25-gauge Quincke spinal needles, and patients were closely monitored for vital signs and other relevant parameters.
Statistical analysis involved descriptive statistics for ‘quantitative and categorical variables, chi-square tests for associations between categorical variables’, independent sample t-tests for treatment effects between groups and repeated measure analysis of variance (ANOVA) for differences in parameters at various time intervals. ‘The analysis was carried out using statistical software SPSS, version 26 (IBM Corp., Chicago, IL, USA)’.
Results
There were no significant differences in age among the three study groups (P = .67), indicating that the age distribution was similar across the groups. Similarly, there were no significant differences in weight among the groups (P = .93), suggesting that the weight distribution was comparable. The duration of surgery among the groups was comparable. Significant differences were observed in the onset of analgesia among the groups (P < .001). The bupivacaine + Buprenorphine group had a significantly shorter onset of analgesia than both the bupivacaine group and the clonidine + bupivacaine group. There was a significant difference in the spread of analgesia among the groups (P = .04). Specifically, the bupivacaine + Buprenorphine group had a slightly greater spread of analgesia than the other two groups. Significant differences were observed in the onset of motor block among the groups (P < .001). The bupivacaine + Buprenorphine group and the clonidine + bupivacaine group had significantly shorter onset times than the bupivacaine group. There were significant differences in the total duration of analgesia among the groups (P < .001). The clonidine + bupivacaine group had a significantly longer duration of analgesia than both the bupivacaine group and the bupivacaine + Buprenorphine group. Significant differences were observed in the total duration of motor block among the groups (P < .001). However, the differences were relatively small, and all groups had a similar duration of motor block. There were significant differences in the visual analogue scale (VAS) score among the groups (P = .002). Specifically, the bupivacaine + Buprenorphine group and the clonidine + bupivacaine group had lower VAS scores than the bupivacaine group, indicating better pain relief in these groups [Table 1].
Distribution of study variables among the study participants (N = 120)
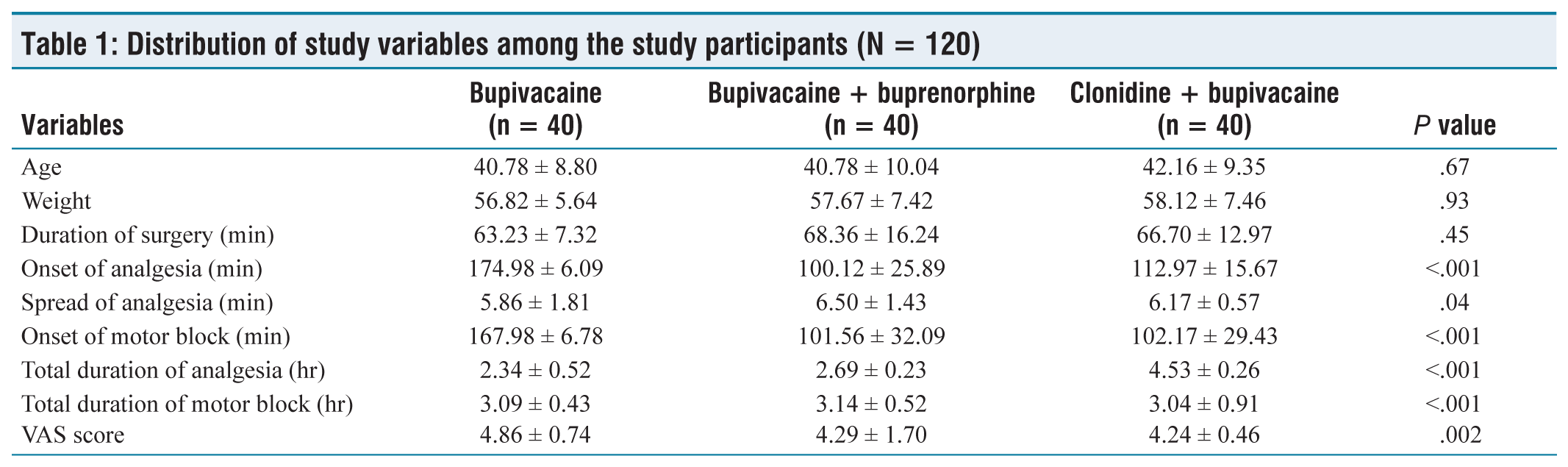
Overall, there was a noticeable trend of decreasing systolic blood pressure across all groups as time progressed from pre-operative baseline measurements to 70 minutes post-spinal block (SBP 70). The P values, which represent the significance of the observed differences, consistently indicated highly significant variations among the groups at each time interval (P < .001). Comparing the three study groups, it is evident that clonidine + bupivacaine consistently resulted in the lowest mean SBP values across all time points, followed by bupivacaine + Buprenorphine. The bupivacaine group generally exhibited the highest mean SBP values among the three groups. At the earliest post-spinal block time point (SBP 2), significant differences were observed between the groups (P = .019), indicating that the treatments had immediate effects on systolic blood pressure. As time progressed, these differences became more pronounced, with clonidine + bupivacaine consistently producing more significant reductions in SBP than the other groups [Figure 1].
Distribution of systolic blood pressure among the study participants (N = 120)
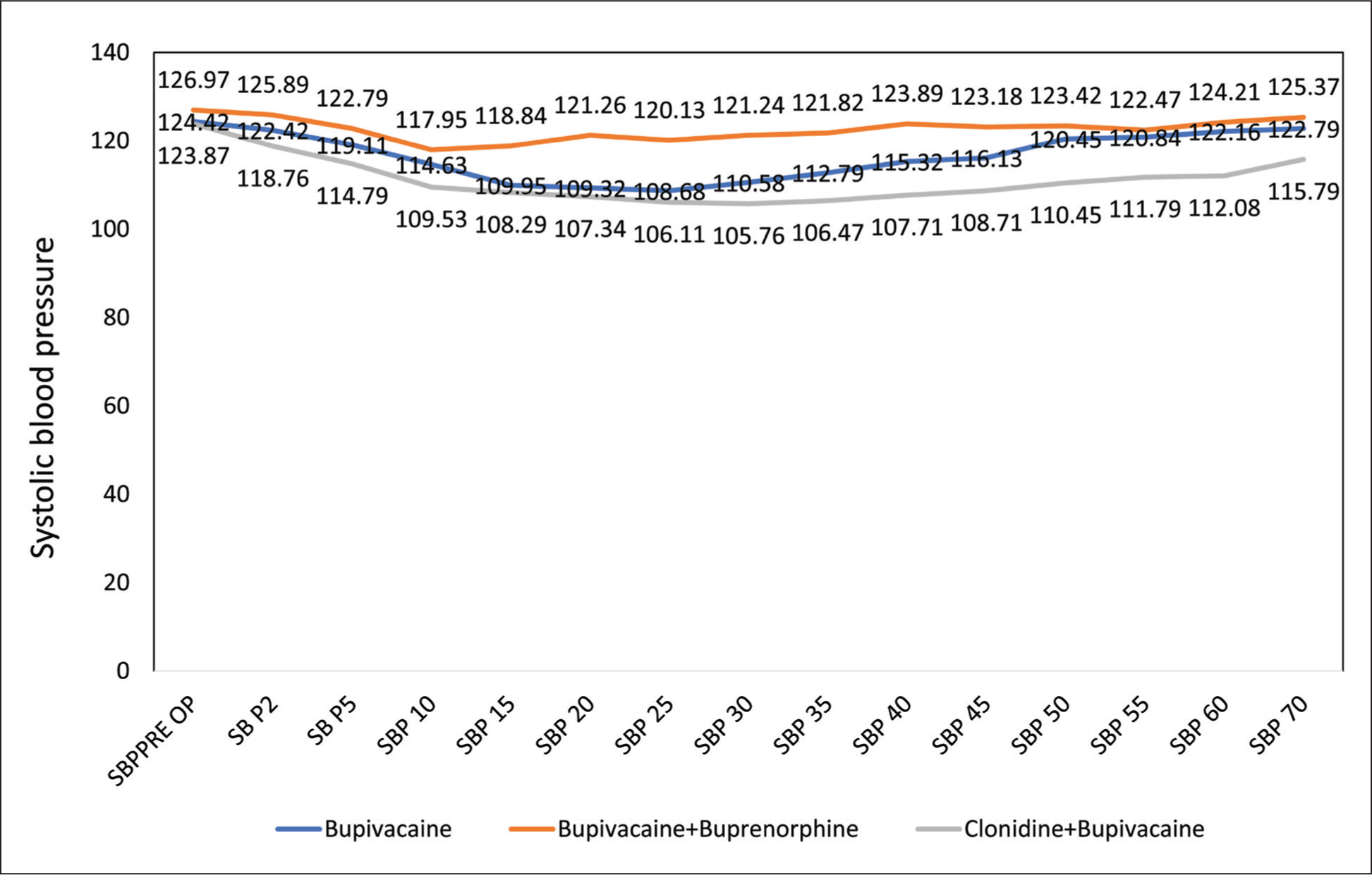
There was a noticeable trend of decreasing diastolic blood pressure across all groups as time progressed from the pre-operative baseline measurements to 70 minutes post-spinal block (DBP 70). The P values, which represent the significance of the observed differences, consistently indicated highly significant variations among the groups at each time interval (P < .001). Comparing the three study groups, it is evident that clonidine + bupivacaine consistently resulted in the lowest mean DBP values across all time points, followed by the bupivacaine + Buprenorphine group. The bupivacaine group generally exhibited the highest mean DBP values among the three groups. At the earliest post-spinal block time point (DBP 2), there were no significant differences observed between the groups (P > .05). However, as time progressed, significant differences became apparent, with clonidine + bupivacaine consistently producing the most significant reductions in DBP compared to the other groups [Figure 2].
Distribution of diastolic blood pressure among the study participants (N = 120)
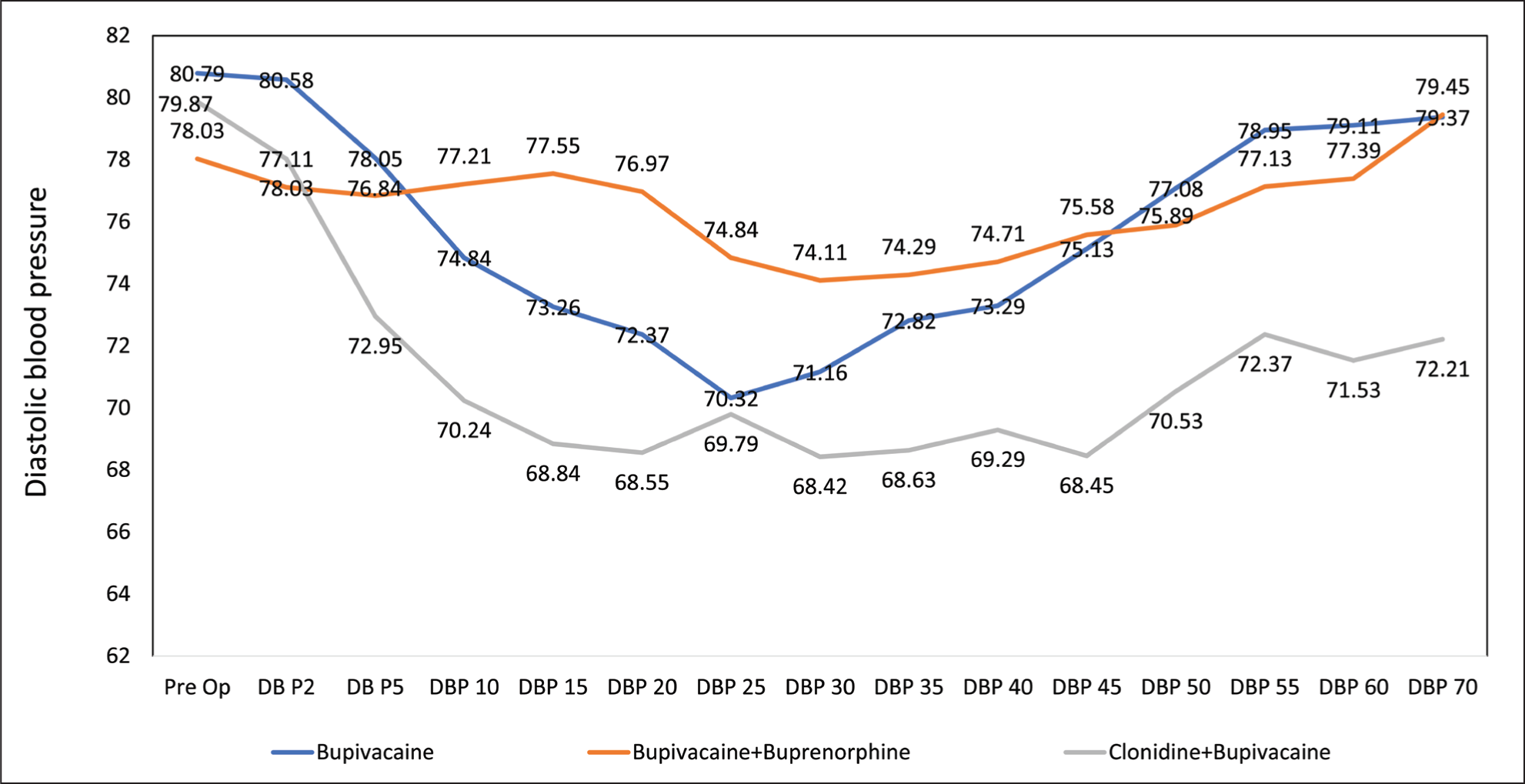
There was a slight variation in oxygen saturation across the groups, with some fluctuations observed over time. The P values suggest that there were significant differences in oxygen saturation among the groups at certain time points. At the pre-operative baseline and at the earliest post-spinal block time point (SPO2 2), there were no significant differences observed in oxygen saturation between the groups (P > .05). However, as time progressed, significant differences became apparent. Between the groups, there was no consistent pattern in terms of which group exhibited higher or lower oxygen saturation values across all time points. The differences in oxygen saturation among the groups were generally minimal and might not be clinically significant. The most notable differences in oxygen saturation among the groups were observed at SPO2 5, SPO2 20, SPO2 35 and SPO2 50, where the P values indicated significant variations (P < .05). However, these differences, while statistically significant, were relatively small in magnitude and might not have a significant clinical impact [Figure 3].
Distribution of SPO2 among the study participants (N = 120)
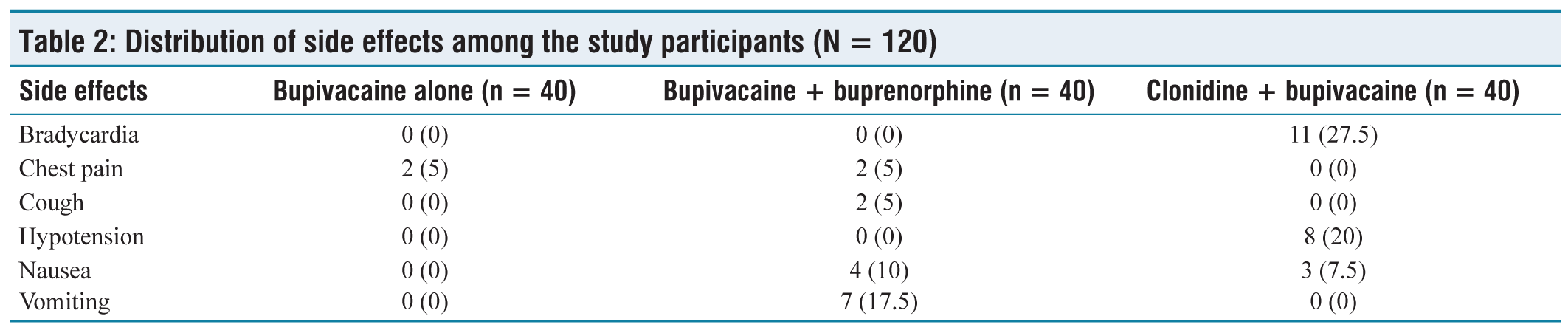
None of the patients in the bupivacaine and bupivacaine + Buprenorphine groups experienced bradycardia, whereas 11 out of 40 patients (27.5%) in the clonidine + bupivacaine group reported bradycardia as an adverse event. Two patients (5%) in both the bupivacaine group and the bupivacaine + Buprenorphine group reported chest pain, whereas no patients in the clonidine + bupivacaine group experienced this adverse event. None of the patients in the bupivacaine group experienced cough, whereas 2 out of 40 patients (5%) in the bupivacaine + Buprenorphine group reported cough as an adverse event. None of the patients in the bupivacaine group and the bupivacaine + Buprenorphine group experienced hypotension, whereas 8 out of 40 patients (20%) in the clonidine + bupivacaine group reported hypotension as an adverse event. None of the patients in the bupivacaine group experienced nausea, whereas 4 out of 40 patients (10%) in the bupivacaine + Buprenorphine group and 3 out of 40 patients (7.5%) in the clonidine + bupivacaine group reported nausea as an adverse event. None of the patients in the bupivacaine group experienced vomiting, whereas 7 out of 40 patients (17.5%) in the bupivacaine + Buprenorphine group reported vomiting as an adverse event [Table 2].
Distribution of side effects among the study participants (N = 120)
Discussion
In the anaesthesia field, effective management of post-operative pain and provision of adequate muscular relaxation and analgesia during surgery are recognised as fundamental practices. Ensuring optimal pain control following surgery is critical for improving patient outcomes and reducing the risk of morbidity and mortality. Spinal anaesthesia is commonly preferred for lower abdominal procedures because of its simplicity and ease of administration compared with epidural and general anaesthesia. However, a notable limitation of spinal anaesthesia is its limited duration of post-operative analgesia.
This study aimed to investigate the impact of clonidine with bupivacaine, bupivacaine + Buprenorphine and bupivacaine alone on the duration of post-operative analgesia and intraoperative hemodynamic stability in patients undergoing TAH. A total of 120 ASA I and II patients of both sexes and aged 35–60 years, scheduled for TAH, were included after pre-anaesthetic assessment and obtaining informed consent. They were randomly assigned to three groups, each comprising 38 patients.
The mean age and weight of participants were similar across all groups, ensuring a balanced distribution of these demographic factors. The onset of analgesia was significantly faster in the bupivacaine + Buprenorphine group and the bupivacaine group than in the clonidine + bupivacaine group. This observation is consistent with findings from previous studies.
For instance, in a study conducted by Pan et al.[10] in 1998, the analgesic efficacy of intrathecally administered neostigmine and clonidine combined with bupivacaine in spinal anaesthesia for patients undergoing caesarean section was compared. The study[10] found that the onset of sensory block was quicker in groups receiving neostigmine than those receiving clonidine. Similarly, Yoganarasimha et al.[11] in 2014 compared intrathecal clonidine 75 mg with intrathecal neostigmine 50 mg as adjuvant medications for spinal anaesthesia using 0.5% hyperbaric bupivacaine 12.5 mg. They reported that the onset of sensory and motor blocks was quicker with neostigmine than with clonidine and plain bupivacaine.[11]
In our investigation, the extent of analgesic spread was significantly reduced in both the bupivacaine + Buprenorphine group and the bupivacaine group, with measurements of 5.68 ± 1.18 and 6.05 ± 1.33, respectively, compared with the clonidine + bupivacaine group which had a spread of 6.37 ± 0.75. In a study by Elia et al.,[12] involving 1,445 patients, intrathecal clonidine was utilised as an adjunct to 0.5% hyperbaric bupivacaine. They observed a significant prolongation in the time to two-segment regression (mean range: 14–75 minutes), a delay in the regression time to L2, and an extension in the duration of analgesia and time to the first rescue analgesic, with no dose-related relationship identified.[12] Furthermore, Andrieu et al.[13] investigated patients undergoing radical retropubic prostatectomy, who received intrathecal morphine at a dosage of 4 μg/kg with or without clonidine at a dosage of 4 μg/kg. They found that adding clonidine to spinal morphine increased the overall duration of analgesia and decreased the intraoperative requirement for sufentanil.
In our investigation, the onset of motor block was significantly earlier in the bupivacaine + Buprenorphine group. Specifically, the onset times were as follows: 165.89 ± 7.80 min for the bupivacaine group, 100.66 ± 33.90 min for the bupivacaine + Buprenorphine group and 102.71 ± 30.34 min for the clonidine + bupivacaine group. In a study conducted by Kanazi et al.[14] in 2006, the effects of spinal anaesthesia were analysed. They compared the administration of clonidine 30 and dexmedetomidine 3 added to 12 mg of hyperbaric bupivacaine at 0.5% with spinal anaesthesia using 12 mg of hyperbaric bupivacaine at 0.5% alone in 60 transurethral resections of the prostate. The patients who received the alpha-2 agonist experienced a quicker onset of motor block, although both sensory and motor regression were significantly prolonged.
In our investigation, the total duration of motor block was significantly shorter in both the bupivacaine + Buprenorphine group and the bupivacaine group. Specifically, the durations were as follows: 3.10 ± 0.34 hours for the bupivacaine group, 3.41 ± 0.25 hours for the bupivacaine + Buprenorphine group and 3.40 ± 0.19 hours for the clonidine + bupivacaine group. In a study conducted by Merivirta et al.,[15] ambulatory individuals undergoing surgery were administered either 6 mg of spinal hyperbaric bupivacaine or 6 mg of bupivacaine alone. It was demonstrated that administering clonidine resulted in a prolonged motor block in the patients. Another study by Tuijl et al.[16] in 2010 investigated the effect of different doses of clonidine combined with hyperbaric bupivacaine on motor block duration, voiding ability and analgesia following knee arthroscopy. The study findings revealed that clonidine significantly increased the duration of motor block by 25 and 34 minutes, respectively. Additionally, Elia et al.[12] conducted a study involving 1,445 patients, which found that administering intrathecal clonidine as an adjunct to bupivacaine increased the incidence of arterial hypotension and bradycardia, independent of the dose. Similarly, in a study by Merrivirta et al.[15] in 2010, a comparison was made between adding 75 μg of clonidine to 6 mg of spinal hyperbaric bupivacaine and administering 6 mg of bupivacaine alone to 60 ambulatory individuals undergoing surgery. The findings of this study indicated a higher requirement for vasopressors and significantly less post-operative pain.
In our study, the bupivacaine + Buprenorphine group exhibited more pronounced side effects than the clonidine + bupivacaine group, and this difference was statistically significant. Within the bupivacaine group, two cases of chest pain were reported. In the bupivacaine + Buprenorphine group, there were two reports of chest pain, two cases of cough, four instances of nausea and seven episodes of vomiting. Among those who received clonidine + bupivacaine, 11 cases of bradycardia, 8 reports of hypotension and 3 cases of nausea were documented. In a study conducted by Pan et al.,[10] the focus was on patients undergoing caesarean sections. The study randomly assigned 80 patients scheduled for lower segment caesarean section under spinal anaesthesia to one of four groups: the bupivacaine group received 10 mg of bupivacaine alone; the bupivacaine + neostigmine group received 10 mg of bupivacaine plus 50 mg of neostigmine; the bupivacaine + clonidine group received 10 mg of bupivacaine plus 150 mg of clonidine. According to the findings of this study, participants in both the bupivacaine group and the groups receiving bupivacaine with adjuvants experienced significantly higher rates of adverse effects such as nausea, vomiting and dizziness.
Conclusion
The results of this study indicate that spinal clonidine provides a longer duration of sensory and motor blockages than bupivacaine alone, along with effective post-operative analgesia. These findings suggest that the inclusion of clonidine with bupivacaine in TAH offers several advantages over using bupivacaine alone. These advantages include longer duration of sensory and motor blockages, quicker onset time of sensory block and effective post-operative pain relief. This combined approach also facilitates early ambulation, reduces the duration of hospital stay and ultimately lowers post-operative morbidity. Moreover, it mitigates the risk associated with simultaneous exposure to multiple drugs and their potential adverse effects.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional ethical committee approval number
Institutional Ethical Committee Approval Number is 22M066 (dated 10 November 2022).
Informed consent
Informed consent was obtained from all the participants in the study.
Credit author statement
All authors contributed equally to the manuscript.
Data availability
Data are included in the manuscript.
Use of artificial intelligence
Artificial intelligence was not used.
