Abstract
Background and Aims:
Antipsychotic drugs are critical in managing psychosis but they also carry risks when misused, leading to toxicity.
Case Presentation:
A patient overdosed on olanzapine, resulting in anticholinergic toxidrome with symptoms like tachycardia and altered mental status. Immediate recognition and management of antipsychotic toxicity-induced toxidromes in emergency settings are crucial. Treatment strategy includes maintaining airway, breathing and circulation along with decontamination. There is no specific antidote.
Conclusion:
This case underscores the need for emergency physicians to remain vigilant and proactive in identifying and addressing such toxicity by identification of toxidromes to prevent complications and missed diagnosis in emergency department.
Introduction
Antipsychotic, including olanzapine, are widely used for psychiatric disorders, such as schizophrenia and bipolar disorder. Despite their efficacy, they carry the risk of adverse effects, including toxicity in overdose scenarios.[1] Olanzapine, a second-generation antipsychotic, has been associated with a range of adverse effects, but reports of anticholinergic toxidrome following its toxicity are limited.
Every year, roughly 5,800 patients visit to emergency department (ED) in United States, because of symptoms linked to typical antipsychotic drugs. That’s about 26 ED visits for every 10,000 patients. Haloperidol is the most prominent among them which attributes to 43.3 visits per 10,000 visits.[2] In 2018, American poison control centres received reports of around 4,100 cases involving antipsychotics poisoning with around two deaths directly associated with it.[3]
Anticholinergic toxidrome is a constellation of symptoms resulting from the blockade of muscarinic acetylcholine receptors in the central and peripheral nervous systems. These symptoms include dry mouth, blurred vision, urinary retention, tachycardia, hyperthermia and altered mental status, among others. While anticholinergic toxidrome is commonly associated with medications possessing anticholinergic properties, its occurrence following olanzapine toxicity remains underreported and warrants further investigation.[4]
This case report delves into the anticholinergic toxidrome secondary to olanzapine overdose. Its aim is to illuminate the clinical presentation, management and potential outcomes of this rare adverse effect of this class of drugs. Understanding the mechanisms underlying anticholinergic toxidrome in the context of olanzapine toxicity is crucial for prompt recognition and appropriate management in clinical practice and it significantly impacts early detection, timely intervention and, ultimately, improved outcomes for affected individuals.
Case Presentation
A 47-year-old male was brought to our ED at around 3:00 PM, who was unresponsiveness for the last six hours prior to arrival to ED. The patient’s mother reported finding empty strips of olanzapine tablets under his pillow, indicating possible acute drug intoxication. The patient has a history of psychotic disorder and has been irregular with his medication. Additionally, he has developed an irregular sleep pattern over the past few days.
On primary survey there were signs of airway obstruction, with the patient unresponsive and having snoring sounds; thus, an oropharyngeal airway was inserted. He was breathing with a respiratory rate of 30 cycles per minute and had saturation of 80% on room air, improving to 90% with oxygen by simple face mask with 8 L/min. Bilateral equal air entry was noted with no additional sounds. He had warm peripheries, tachycardia (160 bpm) and hypertension (160/100 mm Hg). Glasgow Coma Scale (GCS) was 3/15 (E1 V1 M1), with pinpoint pupils and absent deep tendon reflexes. The patient’s temperature was elevated at 101°F, with dry mucosa and absence of secretions. On using adjuncts, his arterial blood gas showed a mild respiratory acidosis (pH 7.35, partial pressure of carbon dioxide [pCO2] 44 mm Hg and bicarbonate of 24.2 mEq) and electrocardiogram showed sinus tachycardia [Figure 1]. He was electively intubated for threatened airway by rapid sequence intubation after preoxygenation with bag and mask for three minutes and with inj. midazolam 4 mg intravenous (IV) as premedication and inj. propofol 60 mg IV as induction. Endotracheal intubation with a size 7.5 tube was successfully performed under direct laryngoscope. No complications were encountered during the procedure. Primary survey was reassessed. A drug history obtained from patients’ relatives revealed that he was on tab. olanzapine 5 mg and tab clonazepam 0.5 mg which he had regularly taking as over-the-counter medications from a nearby private pharmacy.
ECG showing sinus tachycardia
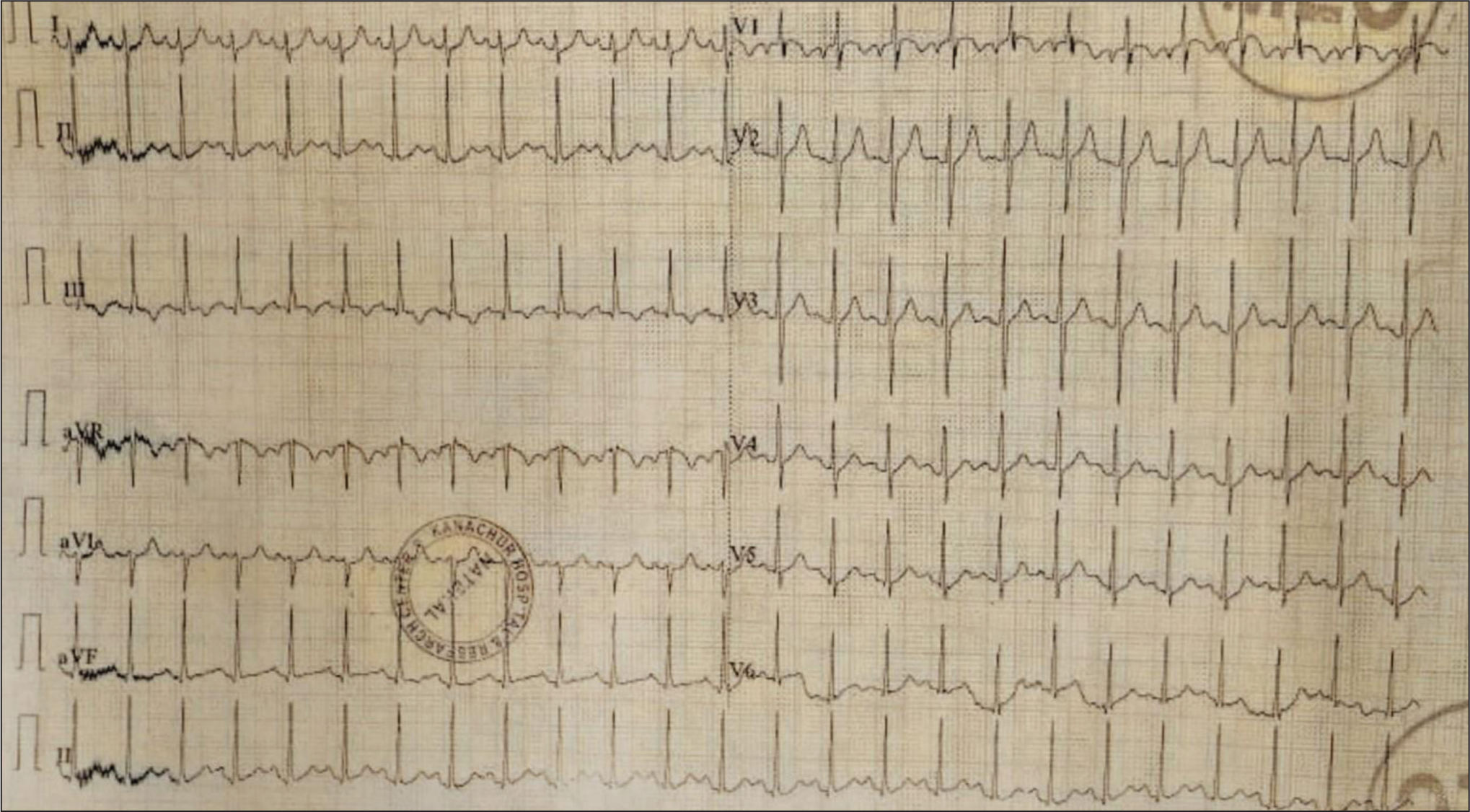
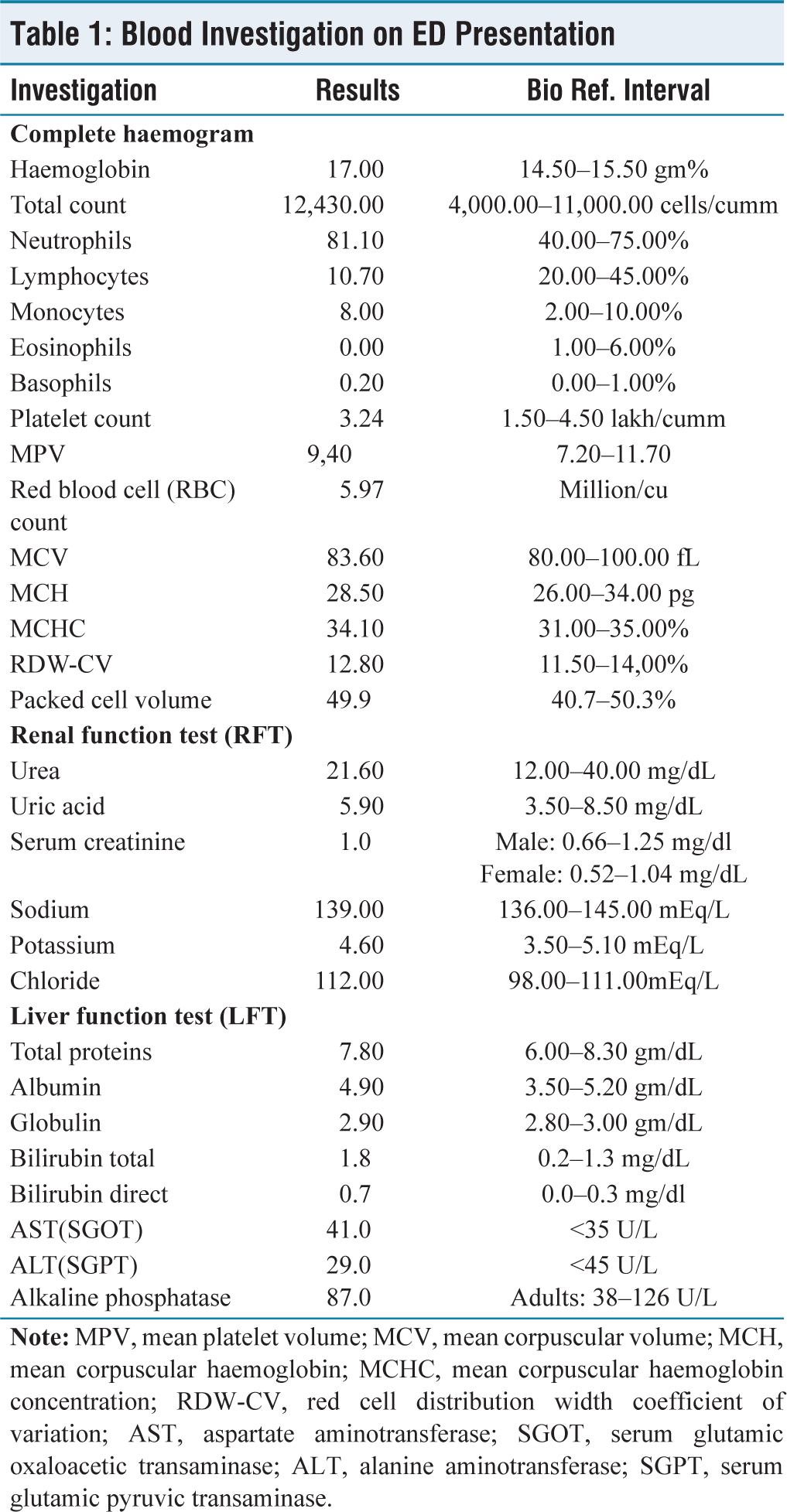
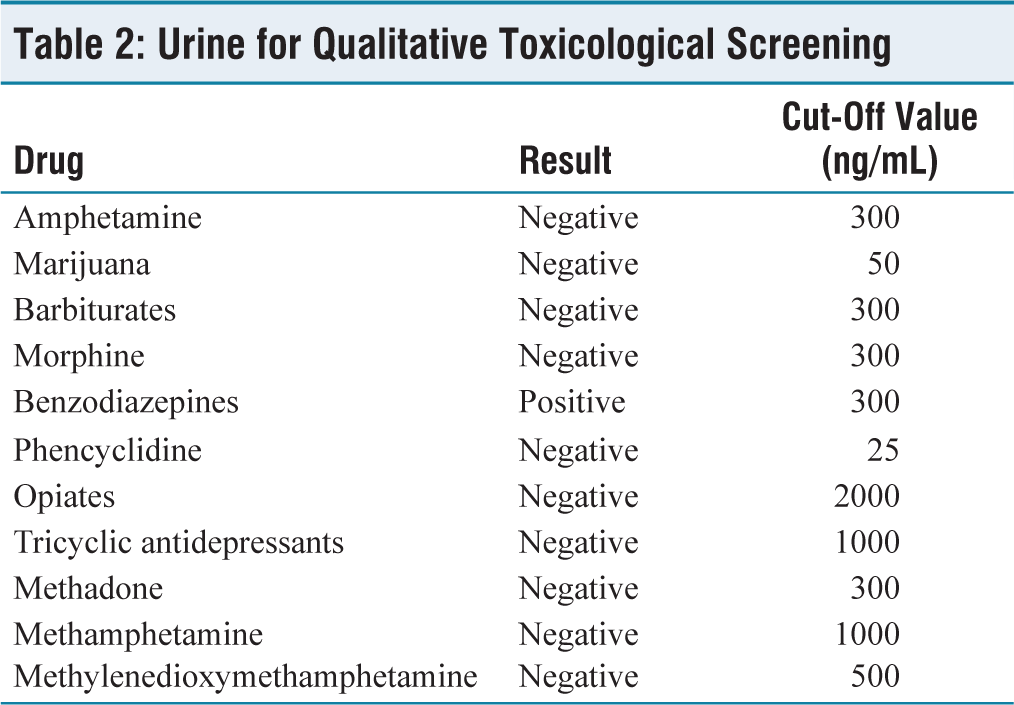
As patient had tachycardia, hypertension with a dry mucosa, a provisional diagnosis of acute drug intoxication with anticholinergic toxidromes secondary to atypical antipsychotic olanzapine was made. Decontamination procedures including gastric lavage, 100g of activated charcoal instillation, and urinary catheterisation for toxicology testing were initiated. Other measures like antipyretics were also included. Blood and urine samples were sent for investigations including qualitative urine toxicology test. Leucocytosis (12,430 leucocytes /mcL) and mild elevation in liver enzymes (aspartate aminotransferase 69 U/L) were noted, along with positive urine benzodiazepines in toxicology testing [Tables 1 and 2]. He was shifted to critical care unit for further management and ventilator support. In the intensive care unit (ICU), the patient’s vital signs gradually improved after day 2. Over the following days, the patient’s condition stabilised with supportive care. He was weaned from mechanical ventilator and successfully extubated on day 4. Psychiatry counselling revealed self-diagnosis of mental disorder and ingestion of 20 tablets of olanzapine 5 mg the previous night. A final diagnosis of acute drug intoxication with anticholinergic toxidromes with olanzapine toxicity along with severe depression without psychosis was established. He was taken off all the antipsychotic drug and tab clonazepam 0.5 mg as anxiolytic was started with a regular follow-up. After two weeks of follow-up in psychiatric outpatient department, the patient had experienced improved sleep and had returned to his routine work.
Blood Investigation on ED Presentation
Urine for Qualitative Toxicological Screening
Discussion
The extent of anticholinergic abuse in people with psychiatric conditions varies widely, with rates ranging from 6.5% to as high as 34%. However, there’s a challenge of underdiagnosis because symptoms of anticholinergic intoxication are often confused with signs of primary psychiatric disorders or other health issues.[3]
A French surgeon’s chance discovery of chlorpromazine’s calming effects in the 1950s transformed psychiatric care for psychosis and it was approved in next four years by US Food and Drug Administration (FDA). It replaced sedatives like barbiturates which were previously used for psychosis, by the 1960s. Newer drugs followed, with ‘atypical antipsychotics’ in 1989 offering fewer side effects and becoming first-line treatments for schizophrenia.[1] The atypical antipsychotics do have lesser side effects of extrapyramidal symptoms but are anticholinergic. Thus, such drugs were abused for its calming effect and for anticholinergic properties. Abuse of anticholinergic drugs was first reported in late 1960s with the description of patient who increased trihexyphenidyl to achieve antidepressant and euphoriant affect. Since then, reports of over 100 patients have been known to have abused anticholinergic drugs.[4]
Although clinicians are always vigilant about patients misuse of psychoactive agents, recent case reports have described abuse of atypical antipsychotics mainly, which have higher potency to produce anticholinergic toxidrome. According to anticholinergic exposures and outcomes reported to US poison centres in 2014, out of 35,822 anticholinergic exposures with clinical effects 11,816 patients had consumed atypical antipsychotics which is graded as second highest drug causing anticholinergic toxicity after antihistamines.[5]
These compounds competitively inhibit binding of acetylcholine to muscarinic acetylcholine receptors which are found on peripheral postganglionic cholinergic nerves in smooth muscle, the ciliary body of the eye, the central nervous system (CNS) and secretory glands. Symptoms of an anticholinergic poisoning, therefore, include both central and peripheral effects. Central effects manifest as agitation, psychosis and seizures. Peripheral effects manifest as tachycardia, cutaneous vasodilatation, leading to red skin, mydriasis, anhidrosis, hyperthermia, urinary retention and gastrointestinal dysmotility.[6,7] These symptoms are collectively described as ‘Blind as a bat, mad as a hatter, red as a beet, hot as Hades, dry as a bone, the bowel and bladder lose their tone, and the heart runs alone’.[8]
Treatment of antipsychotic overdose is mainly supportive and consists of IV fluids and urinary catheterisation, and in some cases, gastrointestinal decontamination and administration of activated charcoal in combination with a whole bowel wash, both of which are beneficial when used within two hours of ingestion. Severe agitation and seizures should be treated with benzodiazepines. Physostigmine, a short-acting anticholinesterase with a half-life of approximately 15 minutes, can be considered in case of a central anticholinergic syndrome.[9]
The patient reported here with severe CNS depression with bilateral pinpoint pupils, tachycardia, increased blood pressure, febrile, with mucosa being dry and maintaining a GCS of 3/15 arrived to our ED was in consistent with anticholinergic poisoning, and upon further questioning about drug history it was found to be secondary to antipsychotics. The urine for toxicology does not usually test for antipsychotics until specifically asked for. He was managed by supportive measures.
The case mentioned underscores the significance of clinically diagnosing patients and verifying it through thorough history-taking. Routine toxicology screenings often miss detecting antipsychotics, commonly abused drugs with potentially life-threatening anticholinergic effects. ED physicians often face challenges as quantifying serum olanzapine levels, for instance, requires expensive methods like high-performance liquid chromatography with ultraviolet (UV) detection, typically unavailable in many care centres.[10]
Conclusion
This case sheds light on the importance of recognising and managing anticholinergic toxidrome secondary to olanzapine overdose. While olanzapine and other atypical antipsychotics are effective for psychosis, they also carry the risk of adverse effects, including toxicity. Despite being less commonly reported, anticholinergic toxidrome can manifest following olanzapine overdose, presenting clinicians with diagnostic challenges. Prompt recognition through detailed history-taking and clinical evaluation is crucial for initiating prompt and appropriate management thus improving patient outcomes. Additionally, this case underscores the limitations of routine toxicology screenings in detecting antipsychotic levels, highlighting the need for more accessible methods for quantification in clinical settings. Overall, enhancing awareness among healthcare providers about the potential for anticholinergic toxidrome in olanzapine overdose is essential for ensuring timely intervention and optimising patient care.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this.
Declaration of patient consent
The authors confirm that they have obtained all necessary patient consent forms. The patient has consented to the publication of her images and clinical information in the journal. She understands that her name and initials will not be published, and while efforts will be made to conceal her identity, complete anonymity cannot be guaranteed.
Credit author statement
We, at this moment, state that the manuscript has been read and approved by all the authors. Afeefa Hakeem contributed to conceptualisation and data collection.
Vijay Kumar S. S. was involved in original draft preparation and editing.
Ananth Prasad Rao helped in supervision and editing.
Data Availibility
Not applicable.
Use of Artificial Intelligence
No.
