Abstract
Background and Aims:
68Ga-PSMA PET represents a reasonable biomarker for presence of cancer of prostate (CaP). 68Ga-PSMA PET combined with simultaneous ‘multiparametric MRI’ (mpMRI) has the potential to be used for the detection and localisation of primary prostate cancer. It is therefore essential to correlate the histopathological findings of robotic-assisted radical prostatectomy (RARP) specimens with pre-operative PSMA PET/MRI. In addition, imaging with PSMA could be useful in estimating the aggressiveness of CaP. The current study aims at the correlation of histopathologic findings of RARP specimens with pre-operative combined 68Ga-PSMA PET/MRI imaging.
Materials and Methods:
Specimens retrieved after RARP in 38 patients of localised CaP were synchronised with pre-operative images obtained with simultaneous 68Ga-PSMA PET/MRI. Maximum scores of individual parameters such as PI-RADS, Gleason’s score and PSMA uptake (in SUV) were taken for each sector and used for statistical analysis.
Results:
The positive predictive value (PPV) and negative predictive value (NPV) of simultaneous 68Ga-PSMA PET and MRI in detecting malignancy were found to be 84.23% (95% CI: 80.74%–87.18%) and 63.92% (95% CI: 57.77%–69.65%), respectively. Combining 68Ga-PSMA PET and mpMRI improves the sensitivity to 81.48% (95% CI: 76.70%–85.67%), but the specificity was found to be 68.24% (95% CI: 60.09%–75.64%). It was found that SUV max correlated with the severity of GS (P value < .001).
Conclusion:
Prediction and localisation of histological malignancy in the prostate could be improved pre-operatively by adding 68Ga-PSMA PET to mpMRI imaging. The SUV of abnormal uptake of 68Ga-PSMA radiotracer is predictive of aggressiveness of tumour as reflected in the Gleason score.
Introduction
Early detection of adenocarcinoma prostate (cancer of prostate [CaP]) can possibly lead to an increased cure rate.[1,2] MRI plays a significant role in comprehensive imaging of prostate suspicious for malignancy. Endorectal coil and multiparametric MRI (mpMRI) have reportedly shown good accuracy in local staging[3] of prostate cancer and are more accurate than TRUS in tumour detection and accurately guiding biopsy sites[4] and can even reclassify low-risk men.[5] However, MRI has been consistently reported to lack specificity in benign conditions such as prostatitis, post-biopsy bleeding, or scarring which may mimic cancer[6] owing to which several researchers have concluded that use of mpMRI for diagnosing significant CaP may result in a substantial number of unnecessary biopsies while missing a minimum number of significant CaP. Novel PET tracers like PSMA, a type 2 trans-membrane integral membrane protein frequently over expressed in primary CaP cells, has a large extra-cellular domain which potentially renders it as an advanced tracer compared to choline-based tracers.[7,8] Unlike choline, cytoplasmic immunoreactivity studies have shown significant differences in PSMA cellular expression among normal benign epithelium and primary malignant prostatic tissue.[9] Further, PSMA expression has been shown to be highly homogeneous in primary malignant tissue.[10] A study revealed that PSMA elevation is inversely correlated to time to prostate-specific antigen progression and disease-free survival.[11] Thus PSMA represents a potential biomarker estimating the aggressiveness of CaP. Positron emission tomography (PET) using 68Ga-PSMA has now become a widespread tool for re-staging recurrent prostate cancer and metastatic follow-up or guiding PSMA-based radionuclide therapies. However, its potential role in primary cancer detection and spread is still unexplored.
With the recent advent of fully integrated simultaneous PET-MRI systems, it has become possible to perform primary prostatic cancer detection using mpMRI and simultaneously evaluate PSMA expression in suspicious prostate.[12]
The aim of this study was to correlate the histopathological extent and characteristics of malignancy in the robotic-assisted radical prostatectomy (RARP) specimen, with the pre-operative 68Ga-PSMA PET/MRI. The secondary aim was to correlate the SUV max of PSMA PET with the Gleason score.
Material and Methods
This is a prospective, analytical, single-centre study of 38 cases where all patients with histological diagnosis of adenocarcinoma of prostate, who opted for RARP were included. Patients who were not suitable for radical prostatectomy or who refused radical prostatectomy and those with contraindications to MRI were excluded. The study protocol was approved by the institute’s ‘Ethic committee’ after following due procedure.
Methodology
Prospective data collection included clinical examination and baseline evaluation along with serum total PSA, TRUS-guided prostatic biopsy with Gleason’s score, 68Ga-PSMA PET with simultaneous MRI. Clinical staging was done as per TNM classification. Patients who were suitable for RARP and opted for it underwent radical prostatectomy.
Simultaneous 68Ga-PSMA PET/MRI was performed on ‘Biograph mMR’ (Siemens Healthcare) consisting of a seamlessly integrated 3-T MRI scanner with a PET insert operating an avalanche photodiode that allows whole-body imaging with Total Imaging Matrix (Siemens Healthcare) coil technology.
Image analysis was performed using a Syngo via workstation and software (Siemens Healthcare). Two experienced nuclear medicine specialists and two radiologists, with more than 15 years of PET/CT and 5 years of PET/MRI experience, who were blinded to the histopathologic report independently, read all images. Maximum lesion size was noted either on T2-weighted or apparent diffusion coefficient images. Care was taken in drawing the region of interest (ROI) to avoid a partial-volume effect.
Qualitative evaluation of multiparametric MR images was performed to assign a prostate imaging reporting and data system (PI-RADS) category to a lesion over a scale of 1–5, as per PI-RADS version 2 (PI-RADSv2). MRI-defined suspicious lesions (on the basis of PI-RADS category) were correlated with the PSMA uptake (SUV max) and histologic findings.
All radical prostatectomies were performed using the da Vinci Si Robotic system by experienced surgeons of our institute. Specimens were sent for histopathological examination in 10% formalin. The prostatectomy specimens were fixed in formalin and processed using standard techniques followed in the institution. During the grossing of the specimen, alignment with the radiological reference points was done by aligning the ‘axis’ of prostate, which was determined by a straight line joining the midpoint of the prostatic urethra at vesical end and the midpoint of urethral exit from prostate distally. Ready slides were reported according to ISUP protocol. Tumour location, Gleason’s score in each sector with tumour volume (percentage of entire prostate volume) and other significant findings like extraprostatic spread were reported. Section from the dissected seminal vesicles and vas were prepared separately and assessed for infiltration of tumour. Histopathologic examination was reported independently by one of the pathologists without any bias, who did not have access to imaging findings.
Correlation Between Imaging and Histopathology Data
Mapping tumour localisation: Axial sections of the MR image were cut perpendicular to the ‘axis’ of prostate gland starting from the neck of the bladder to correspond to the axial sections taken in the grossing of pathological specimen of the prostate for histopathological examination. On each MR slice, four sectors were drawn with an anteroposterior (AP) and transverse (TR) line at the midpoint of the section and this was followed in all consecutive slices across the whole of the prostate gland. Any focal area in a sector of a slice appearing suspicious on T2 or ADC images was taken as harbouring a prospective lesion and ROI drawn on that sector was copy-pasted on corresponding T2W, DWI, ADC and DCE images of MRI. Individual scores for these parameters were assigned for the lesion and final PI-RADS classification was arrived at and copy-pasted on delayed PSMA PET image at ~60 minutes to get PET uptake for that lesion as SUV max.
During histopathological examination, individual sections (slices) were divided into four sectors and corresponding Gleason’s scores were assigned to that sector. Once Gleason’s score assignment was completed, slices in MRI and PET were carefully grouped so as to match with histopathology findings taken as final. Maximum score of individual parameters, maximum grade of PI-RADS, highest Gleason’s score and maximum PSMA uptake (in SUV) amongst sectors across slices in a group were taken as characteristics of the lesion and used for statistical analysis.
Statistical analysis was performed using SPSS Statistical Software version 22.0. Kappa Statistics was calculated to assess the agreement between mpMRI and HPE, as well as, with 68Ga-PSMA PET and HPE. Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and accuracy were calculated by classifying patients into benign and malignant by mpMRI and 68Ga-PSMA PET.
Observations and Results
A total of 38 men who underwent RARP with pre-operative assessment by 68Ga-PSMA PET with simultaneous mpMRI as per the protocol were included in the study. A total of 456 sectors were prepared adhering to the principles outlined above for histological studies.
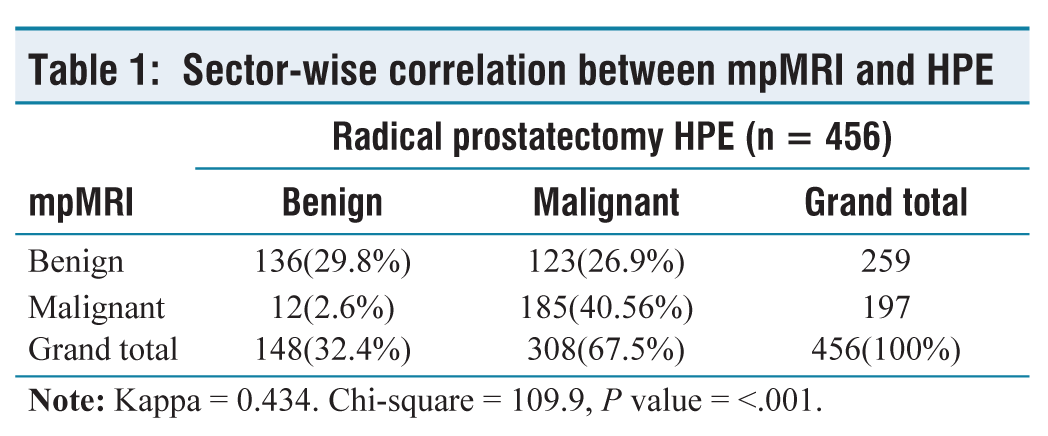
Out of 456 sectors, 308 were found to be malignant on HPE while 148 sectors were benign. mpMRI was found to be true positive in 185 sectors out of 308 sectors, while 123 sectors were detected as false negative by mpMRI. The sensitivity and specificity to detect malignancy sector-wise were 60.06% (95% CI: 54.36%–65.58%) and 91.89% (95% CI: 86.27%–95.74%), respectively. Twelve out of 148 sectors were detected as false positives by mpMRI. Out of 259 sectors negative on mpMRI, 123 were found to be positive for tumour on HPE. The PPV, NPV and accuracy of mpMRI in detecting malignancy were found to be 93.91% (95% CI: 89.89%–96.39%), 52.51% (95% CI: 48.88%–56.11%) and 70.39% (95% CI: 65.97%–74.55%), respectively (Table 2).
Sector-wise correlation between mpMRI and HPE
Sector-wise correlation between PET and HPE

In comparison to mpMRI, 68Ga-PSMA PET is more sensitive for detecting sector-wise malignancy. Out of 308 lesions detected by HPE, 241 sectors were found to be positive by 68Ga-PSMA PET with a sensitivity of 78.25% (95% CI: 73.22%–82.73%). However, the specificity was found to be less than that of mpMRI, that is, 72.97% (95% CI: 65.06%–79.94%). Out of 148 sectors diagnosed to be benign on HPE, 40 sectors were detected as false positives in 68Ga-PSMA PET. The PPV, NPV and accuracy of 68Ga-PSMA PET in detecting malignancy were found to be 82.12% (95% CI: 82.12%–88.77%), 61.71% (95% CI: 56.04%–67.06%) and 76.54% (95% CI: 72.37%–80.35%), respectively (Table 3).
Sector-wise correlation of simultaneous 68Ga-PSMA PET/MRI with HPE
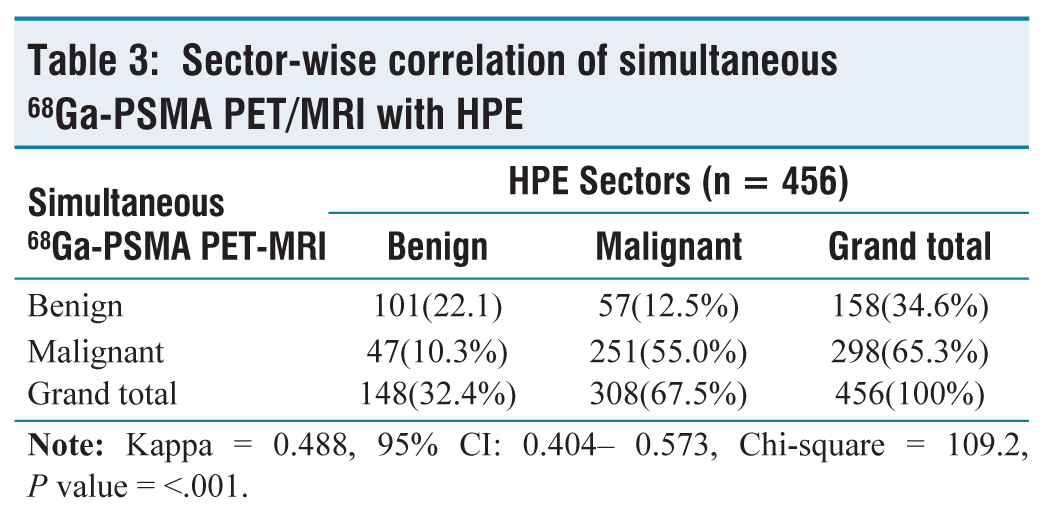
Combining 68Ga-PSMA PET and mpMRI improves the sensitivity to 81.48% (95% CI: 76.70%–85.67%), but the specificity was found to reduce, being 68.24% (95% CI: 60.09%–75.64%). The PPV and NPV of simultaneous 68Ga-PSMA PET and MRI in detecting malignancy were found to be 84.23% (95% CI: 80.74%–87.18%) and 63.92% (95% CI: 57.77%–69.65%), respectively. The strength of the agreement was considered to be ‘good’ (Kappa = 0.488, 95% CI: 0.404–0.573) (Table 4).
Overall detection of malignancy with different imaging modalities

When 68Ga-PSMA PET was added to mpMRI, it detected four more cases as positive on imaging and further increased the sensitivity of imaging modalities for diagnosing malignancy of prostate.
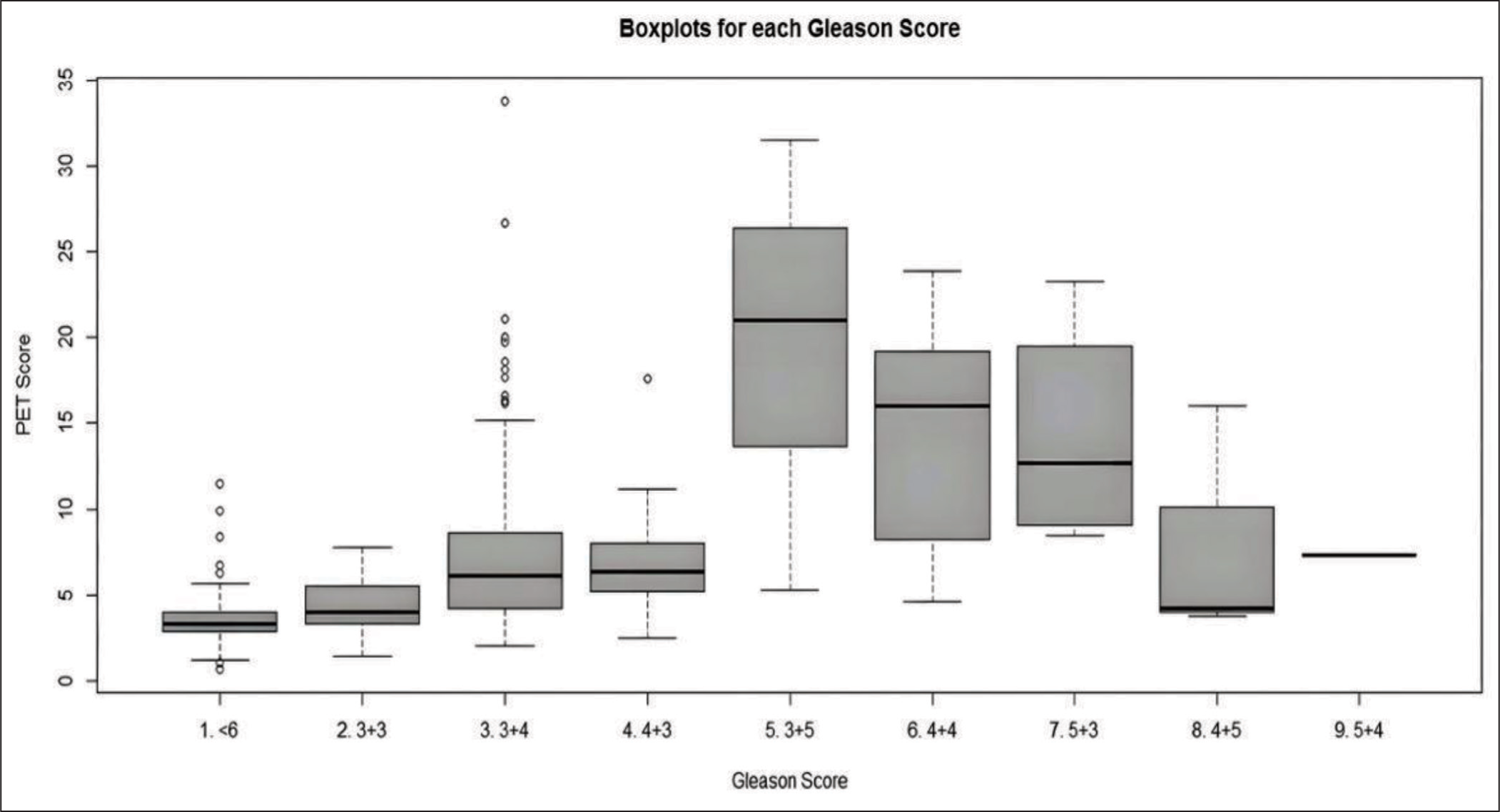
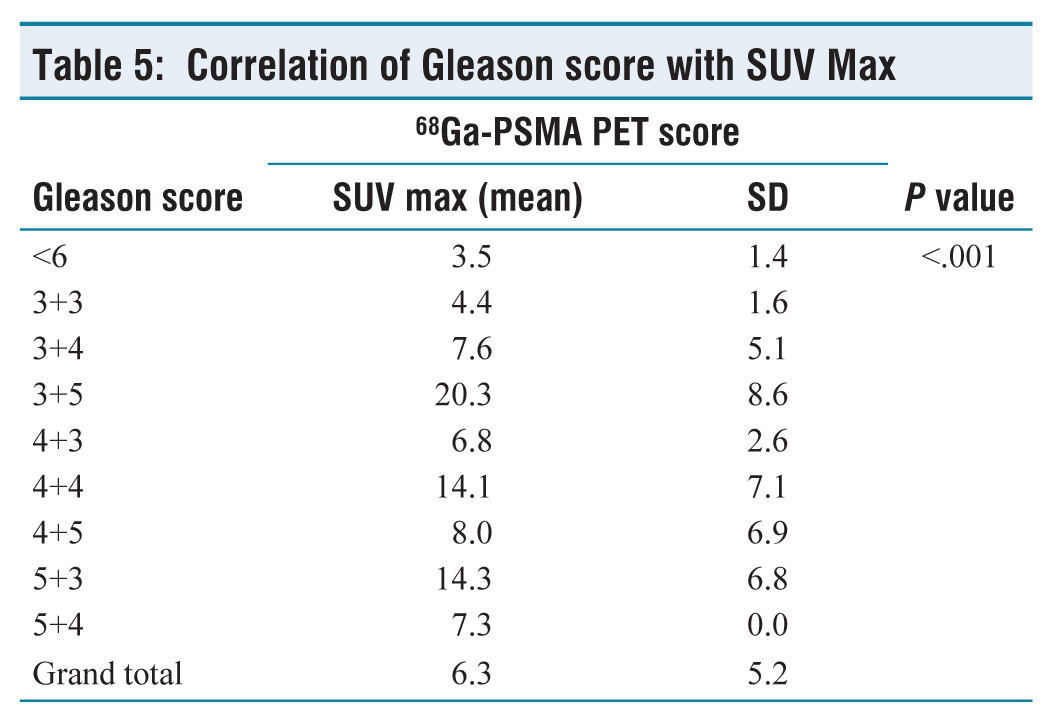
Pair-wise comparisons using the Wilcoxon rank sum test were used to find correlation between SUV max of 68Ga-PSMA PET with the Gleason score of individual sectors. It was found that SUV max correlated with severity of GS (P value < .001) (Figure 1, Tables 1 and 5).
Correlation of Gleason score with SUV Max
Correlation of Gleason score with SUV Max
Discussion
The combined anatomic and functional imaging provided by PET suggests that a PET radiotracer for a specific target may dramatically improve imaging of prostate cancer. PSMA is a promising, well-characterised biomarker specific for prostate cancer. 68Ga-PSMA ligand is theoretically a promising tracer for imaging prostate cancer in various disease stages.
According to mpMRI, 7.8% (3 of 38) and 5.2% (2 of 38) of the cases were considered equivocal (PI-RADS 3) and negative (PI-RADS 1/2), respectively. However, four out of these five patients were positive on 68Ga-PSMA PET. Hence this is to understand that combining 68Ga-PSMA PET scan to mpMRI increases the sensitivity of picking the malignancy in prostate, however, despite this advantage of combining these two modalities, 1 out of 38 cases (2.6%) of carcinoma prostate was missed on imaging in the present study [Table 6].
Profile of patients reported benign/equivocal on mpMRI imaging
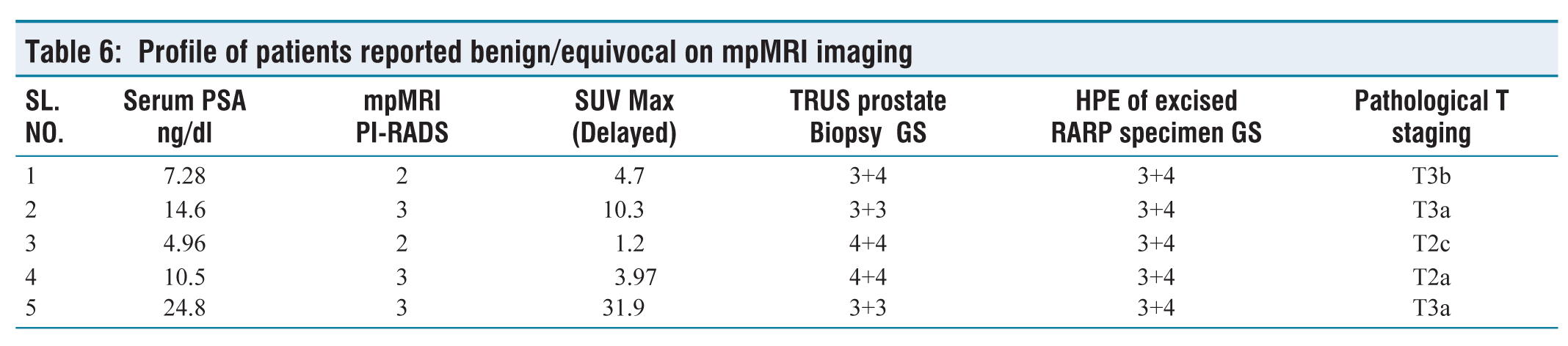
With regard to evaluation of sensitivity of imaging modalities, in the present study, out of 38 patients, two patients had no 68Ga-PSMA uptake on PET scan and missed on PET scan. On patient-wise analysis, in our study, we found that mpMRI, 68Ga-PSMA PET and simultaneous 68Ga-PSMA PET/MRI detected 86.84%, 94.73% and 97.36% patients, respectively.
Similarly, in a study done by Eiber et al.[13] on 53 patients to analyse the localisation of intraprostatic tumour by simultaneous 68Ga-PSMA PET/MRI, on patient-wise analysis, they found the sensitivity of mpMRI, PET and PET/MRI to be 66% (35 of 53 patients, 95% CI: 52%–78%), 92% (49 of 53 patients, 95% CI: 82%–98%) and 98% (52 of 53 patients, 95% CI: 89%–100%), respectively.
In another study by Robert et al.,[14] overall 68Ga-PSMA PET/MRI depicted prostate cancer in 97% of patients (31 of 32 men), compared with 79% of patients in whom prostate cancer was detected with mpMRI.
The findings of present study are consistent with the above-mentioned studies by Eiber et al.[13] and Robert et al.[14] Sector-wise correlation of HPE of RARP specimen with 68Ga-PSMA PET/MRI imaging.
The specificity of simultaneous 68Ga-PSMA PET/MRI is found to be less than either 68Ga-PSMA PET alone or mpMRI alone in the present study, that is, 68.24% (95% CI: 60.09%–75.64%), 72.97% (95% CI: 65.06%–74.94%) and 91.89% (95% CI: 86.27%–95.74%), respectively (Table 7).
Comparison of different studies for detection of sector-wise malignancy
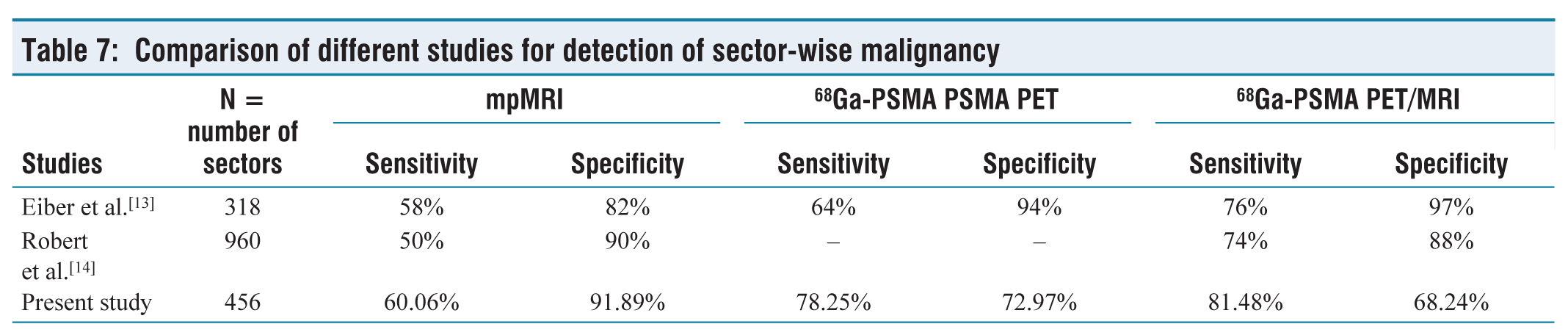
On reviewing the literature we found that Robert et al.[14] calculated the region-specific sensitivity and specificity for PET/MRI and mpMRI by using raw stringent and alternate neighbouring approach, they found region-specific sensitivities of PET/MRI and mpMRI to be 74% (95% CI: 70%–77%) and 50% (95% CI: 45%–54%), respectively, with the alternative neighbouring approach (P = .001 for both) and 73% (95% CI: -68% to 79%) and 69% (95% CI: 62%–75%), respectively, with the population-averaged generalised estimating equation (P = .04). Region-specific specificity of 68Ga PET/MRI was similar to that of mpMRI with the alternative neighbouring approach (88% [95% CI: 85%, 91%] vs. 90% [95% CI: 87%, 92%], P = .99) and in population-averaged estimates (70% [95% CI: 64%–76%] vs. 70% [95% CI: 64%–75%], P = .99).
In the study by Eiber et al.,[13] they divided the prostate into sextants and found that the specificity of mpMRI, PET and simultaneous 68Ga-PSMA PET/MRI to be 82% (95% CI: 69%–90%), 94% (95% CI: 86%–98%) and 97% (95% CI: 90%–99%), respectively. Out of 53 patients included in their study, mpMRI, PET, and simultaneous 68Ga-PSMA PET/MRI detected cancer in 66% (35 of 53), 92% (49 of 53), and 98% (52 of 53) of the patients, respectively.
Even though the sensitivity and specificity both were slightly better in our study when mpMRI alone was used, addition of 68Ga-PSMA PET increased the sensitivity. The specificity of combined modality in localising the cancer was less than the other contemporary studies. This may be due to the difference in the methodology, as in the study by Eiber et al.,[13] the mounted specimen was studied in six sectors while in the present study, we divided the prostate into 12 sectors.
In the present study, a positive correlation was found between SUV max and the histological parameters of aggressiveness of cancer. Pair-wise comparisons using the Wilcoxon rank sum test were used to find correlation between SUV max of 68Ga-PSMA PET with Gleason score of individual sectors. It was found that SUV max correlated with the severity of GS (P value < .001).
Similarly, Robert et al.[14] in their study used a generalised linear model and demonstrated an association between SUV max and Gleason score of 7 or greater (odds ratio = 2.22 [95% confidence interval: 1.46, 3.35], P value <.001). They concluded that abnormal radiotracer uptake accumulation is predictive of a tumour Gleason score of 7 and higher.
However, in another study, Eiber et al.[13] not find any correlation between SUV max and Gleason score on a patient basis (r = 0.096) or on sextant basis (r = 0.116). This difference may be explained on the basis of the difference in the methodology used.
Limitation of the Study
This study is limited by a small number of enrolments. Refinement in spatial correlation of radiological image and anatomical specimen needs to be evolved further for more accurate assessment of the utility of the newer imaging modalities.
Conclusion
In the present study, based on overall patient-wise analysis, prediction of histological malignancy in prostate is increased pre-operatively by adding 68Ga-PSMA PET to mpMRI imaging. On sector-wise analysis of the RARP specimen, the pre-operative sensitivity of simultaneous 68Ga-PSMA PET/MRI is increased while the specificity decreases in comparison with either mpMRI or 68Ga-PSMA PET alone.
Abbreviations
CaP, Cancer prostate; CI, Confidence interval; 68Ga-PSMA PET, Ga-PSMA PET; mpMRI, Multiplanar MRI; NPV, Negative predictive value; PET, Positron emission tomography; PI-RADS, Prostate imaging reporting and data system; PPV, Positive predictive value; PSMA, Prostate-specific membrane antigen; RARP, Robot assister radical prostatectomy; SUV, standardised uptake value.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
This study was approved by the Ethics Committee of the Hospital (Ethical No ECR/5/Inst/DI/2013 dated 28 June 2017.
Credit author statement
Sharwan Kumar: Correlation of gross specimen and alignment with images. Collection of data and writing the first draft.
Amarnath Jena: Reporting and correlation of MRI images with Histopathology.
Sangeeta Taneja: Examination, Reporting and correlation of PSMA PET images with Histopathology.
Vikas Kashyap: Reporting of Histopathological slides and correlation with Radiological imaging.
Ashutosh Kumar Singh: Collection, Compilation and Tabulation of raw data.
Ankur Sharma: Assisted in grossing of specimen and correlation of gross specimen with imaging and writing first draft.
Nilesh Taneja: Writing the final manuscript.
Apeksha Raheja: Assorting references and final proof reading.
Rajesh Taneja: Original idea of study, continual guidance, supervision and troubleshooting. Compiling final manuscript.
Data availability
The data is available with the first author, Sharwan Kumar, and can be accessed by a reasonable application procedure.
Use of artificial intelligence
No artificial intelligence has been used during the study period or preparation of manuscript.
