Abstract
Introduction:
Milan System for Reporting Salivary Gland Cytopathology (MSRGC) was introduced in the year 2015 to facilitate a standardised reporting format and to improve the overall effectiveness of fine-needle aspiration cytology (FNAC) in the preoperative evaluation of salivary gland lesions. Risk of malignancy (ROM) provided in each category guides the physician for appropriate clinical management. This study was performed prospectively for one year to evaluate the diagnostic utility and validate MSRSGC.
Results:
The study included 62 cases of salivary gland FNAC of which 35.5% of the cases had surgical resection specimens. Category IVA formed the major group, and only 1.6% was given category III. The ROM and RON were calculated for each category and were statistically significant and compared with other studies.
Conclusion:
MSRSGC is the recent classification system, and it suggests less than 10% of cases under category III to avoid it’s over utilisation. MSRSGC is a reliable classification in our population, and it was best with category IVA lesions in our study.
Introduction
Fine-needle aspiration cytology (FNAC) is an effective tool for preoperative evaluation of salivary gland lesions.[1] Other methods of evaluation, such as incisional or core needle biopsy, are less popular because of the complications, including the risk of infection and surgical plane contamination.[2] FNAC helps to distinguish non-neoplastic lesions, most common benign tumours and low- and high-grade malignant tumours.[3] The impact of FNAC in the management of salivary gland lesions and the institutional experience leads to the development of a risk-based classification scheme.[4] There are some limitations to the procedure, such as the FNA technique, cellularity, heterogeneity of the lesion and experience of the cytopathologist.[5] In salivary gland cytology, morphological overlaps and descriptive reports addle the treating clinician/ surgeon in patient management.[2,6] Earlier, various classification systems were used in the diagnosis of salivary gland cytology.[7] To provide standardisation in reporting salivary gland FNAC and to provide relevant information for patient management, an evidence-based tiered classification system: ‘The Milan System for Reporting Salivary Gland Cytopathology’ (MSRSGC) was introduced. The reporting system was proposed in 2015 at the European Congress of Cytology in Milan, Italy, by the American Society of Cytopathology (ASC) and the International Academy of Cytology (IAC).[7–9] MSRSGC contains six diagnostic categories with suggested risk of malignancy (ROM) for each category and clinical management algorithm. The categories comprise non-diagnostic (category I), non-neoplastic (category II), atypia of undetermined significance (category III), benign neoplasm (category IVA), salivary gland neoplasm of uncertain malignant potential (SUMP) (category IVB), suspicious for malignancy (category V) and malignant (category VI).[8] MSRSGC provides definitions, morphologic criteria, diagnostic category explanations and sample reports, which facilitate the cytopathologist to give a specific diagnosis wherever possible.[11] The diagnostic efficacy of the procedure can be further increased by rapid onsite evaluation (ROSE).[18] This study was undertaken to evaluate the diagnostic utility and to validate MSRSGC. The ROM was calculated wherever possible.
Materials and Methods
A prospective observational study was conducted in the Department of Histopathology for a period of one year from June 2022 to May 2023. The hospital’s ethical committee approval was obtained in May 2022. All the swellings in the salivary gland area (parotid and submandibular region), intraparotid lymph nodal samples without any ductal or acinar cells, both direct and ultrasonography (USG)-guided FNAC preparations and unstained smears for opinion were included in the study for analysis. Cell block preparations were made wherever possible. Samples with nodal tissue and without salivary epithelial cells in the submandibular region were excluded.
Relevant clinical history, physical examination details and imaging findings were recorded. Informed written consent was obtained for the procedure. ROSE was performed using field’s stain for the procedure done in our department. The rest of the smears were stained with Leishman and May Grunwald Giemsa (MGG), and smears in alcohol fixative were stained with Papanicolaou stain. The slides were re-evaluated and classified under the six categories of the Milan system. Histology follow-up specimens, wherever available, were reviewed and correlated with the cytology. ROM and RON were calculated for each category.
Regarding data management and statistical analysis, data entry was done in Microsoft Excel 2007. Data analysis was carried out by IBM SPSS for Windows Version 25.0, Armonk, NY: IBM Corp. Continuous variables were tested for normality using Shapiro–Wilk test. Normally distributed continuous variables were represented as mean + standard deviation. Non-normally distributed variables were represented by the median (interquartile range [IQR]). Categorical variables were represented as percentage. The chi-squared test, or Fisher’s exact test, was used to compare categorical variables. Comparison of continuous variables was done by an independent sample t-test. All ‘p’ values less than 0.05 were regarded as statistically significant.
Risk of neoplasm (RON) was calculated by dividing the neoplastic cases confirmed through histopathological examination by the total number of resection specimens in that particular category. (Neoplastic cases included both benign and malignant cases). Similarly, ROM was calculated by dividing the malignant cases (excluding benign cases) confirmed through histopathological examination in the particular category by the total number of resection specimens in that particular category.[4–11]
Overall, RON was calculated by dividing the number of neoplastic cases confirmed by histopathology by the number of cases in cytology for that particular category. Similarly, overall ROM was calculated by dividing the number of malignant cases confirmed by histopathology by the number of cases in cytology for that particular category.[4]
Results
A total of 62 cases were available for examination in this study. About 72.6% (
A total of 312 slides from 62 cases were screened blindfolded by three pathologists separately with clinical history, imaging and physical examination findings. Special stains like acid-fast bacilli (AFB), periodic acid-Schiff (PAS) and periodic acid-Schiff with diastase (PAS-D) were utilised whenever required. All the cases were categorised into the six categories of MSRGC detailed in Table 1.
FNAC, histopathological examination, ROM, RON, overall ROM and overall RON in this study
Possible differentials were provided apart from the mentioned category. Among 62 cases, 55 cases had the same MSRSGC category assigned by all three pathologists. For the rest of the seven cases, the slides were reviewed again, and the opinion of the most experienced pathologist was taken as the final decision. About 35.5% (
Discussion
FNAC is a sensitive and specific test to diagnose salivary gland lesions.[1] Accuracy ranges from 87.5% to 95.59% in various studies.[4,7,8,12] By using this technique, non-neoplastic lesions can be accurately detected before surgery, sparing the patient needless invasive procedures.[4] Morphological overlaps, the presence of basaloid component, squamous metaplasia, oncocytic change and cystic change make the FNAC even more challenging to interpret. Morphological patterns-based classification systems and their risk stratification had wide variation; hence, it was difficult to follow as it lacked consensus for the management plan.[7]
In this study duration, there were 62 cases of salivary gland cytology for analysis, of which 22 cases had histology follow-up. The patient’s age ranged from 13 to 70 years, with a mean age of 44, which was similar to the previous published studies.[4] The male-to-female ratio in this study was quite similar to the study by Gaikwad et al. (1:1.26).[8] Predominance of parotid gland involvement followed by submandibular gland was noted as in other studies.[6–13] No sublingual or minor salivary gland lesions were observed in this study duration. Similar to Tommola et al., there were three cases of intraparotid and periparotid lymph nodes cytology.[12]
About 35.4% of the cases in this study had histology follow-up, while the percentage ranged from 34.93% to 57.5% in other studies.[6-11]
The percentage of non-diagnostic cases assigned to category I in this study was 16.1%, while in other studies it ranged from 0% to 30%.[4–15] Paucicellular smears, haemorrhagic fluid, smear with cyst macrophages and cystic contents devoid of ductal epithelial cells or salivary acini were the findings. One discordant case in this group, the FNAC, showed predominantly haemorrhage and occasional acinar cell clusters. Metachromatic stromal elements or epithelial/ myoepithelial cells were absent. It was a USG-guided FNAC, and hence, ROSE could not be performed. The resection specimen of this case showed features of pleomorphic adenoma.
This study had 19.3% of the cases in category II, which was close to the results of the study by Singh et al. (21.1%) and Manucha et al. (18.7%).[4,11] However, in the study by Mishra et al., 55.4% were classified as non-neoplastic. As in other studies, there were no lymphoepithelial lesions or lymphoepithelial cysts in this category.[2,4,5–7] This study included 4.8% cases with lymph node hyperplasia causing parotid enlargement, while Kumari et al. found 2.4% of lymphoid tissue with reactive hyperplasia and Tommola et al. had 0.72% of parotid lymph nodes sampled.[12,16] Difference in the sample size explains the difference in the proportion of cases.
This study had 1.6% cases categorised as atypia of undetermined significance, while in other studies, it ranged from 0% to 8.5%.[2,4–15] This case had rare duct epithelial cells with oncocytic change and a background showing cellular debris and inflammatory cells but could not be categorised into an oncocytic neoplasm. This group should contain no more than 10% of cases, according to MSRSGC, in order to prevent overutilisation of this category. This category included cases that lacked both quantitative and qualitative cytomorphology findings to differentiate benign from neoplastic processes with confidence. The inherent nature of the lesions, in combination with preanalytical factors including smearing technique and fixation, are some of the reasons for the utilisation of this category.
This study had pleomorphic adenoma and Warthin tumour in the category IVA. At the same time, Singh et al. also reported cases of Warthin tumour, basal cell adenoma, oncocytoma and lipoma in addition to pleomorphic adenoma, and Felicelli et al. had Warthin tumour, basal cell adenoma and oncocytoma in this category.[4,15] About 72.72% of the total resection specimens were occupied by this category, with 100% in concordance with FNA diagnosis.
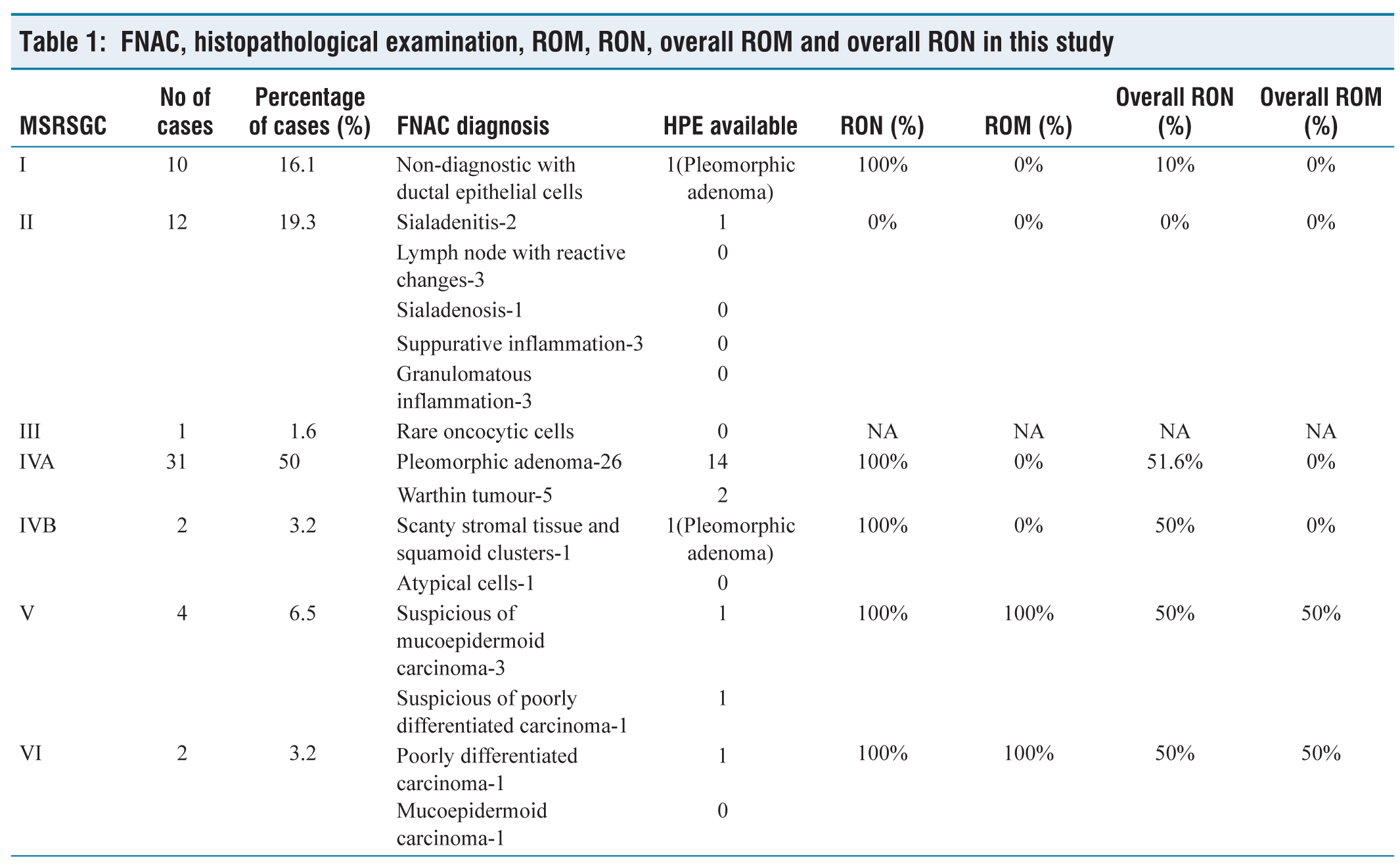
Category IVB was applied to neoplastic cases; however, a low-grade malignancy could not be completely ruled out. MSRSGC gave a detailed explanation of the differentials in this category, including basaloid neoplasms and oncocytic neoplasms and their malignant mimics. Category IVB accounts for 3.2%. However, other studies stated that 0%–13.1% of the cases were in this group.[4-21] The case with resection specimen had a biphasic population of large epithelial fragments and clusters, singly scattered spindle and epithelioid myoepithelial cells in a haemorrhagic background in the smear. A very scanty matrix was seen. The cell block section showed squamoid clusters with mild karyomegaly. The follow-up specimen was diagnosed as a pleomorphic adenoma with no atypical features [Figure 1].
Category IVB—(A) field stain (10×), (B) pap stain (40×), (C) cell block (40×), (D) pleomorphic adenoma–histopathological examination (HPE) (10×)
Legaspi et al. had 5.2% cases in category V where they had the diagnosis of acinic cell carcinoma, oncocytic variant turned out to be oncocytoma in resection specimen and suspicious of adenoid cystic carcinoma turned out to be pleomorphic adenoma in the histopathological examination.[17]. Our study had mucoepidermoid carcinoma and poorly differentiated carcinoma in the follow-up. Immunohistochemistry was not performed in the latter, though it was suggested. Interobserver variation was observed in our study in seven cases. The majority included categorising the lesions with either category IVB or V and category V or VI.
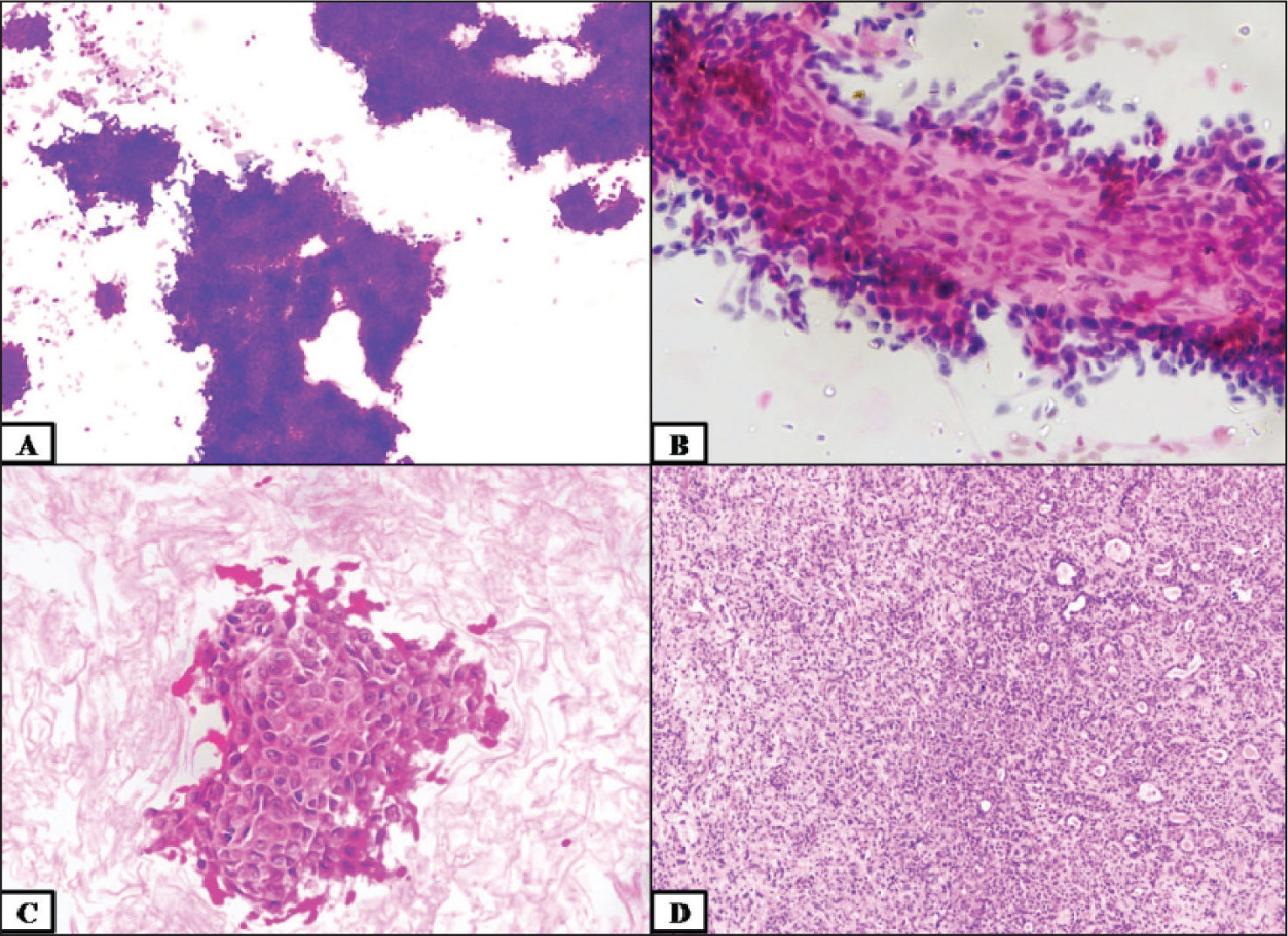
Of the two cases in category VI in our study, one case with follow-up showed similar morphology [Figure 2]. The other case did not have any follow-up specimen to compare. Mishra et al. had 12.6% cases in this category, which includes predominantly mucoepidermoid carcinoma, followed by other cases like adenoid cystic carcinoma, squamous cell carcinoma and acinic cell carcinoma.[2] Pal had 2.4% in this category, with one case with the diagnosis of carcinoma ex-pleomorphic adenoma on FNAC turned out to be pleomorphic adenoma with atypical features on histopathological examination.[20]
(A and B) Chronic sialadenitis (A: Leishman stain FNAC 40×, B: HPE 10×), (C) granulomatous inflammation 40× (PAP), (D and E) pleomorphic adenoma (D: field stain FNAC 40×, E: HPE 10×) (F and G) Warthin tumour 40× (F: MGG FNAC, G: HPE), (H and I) poorly differentiated carcinoma 40× (H: PAP FNAC, I: HPE)
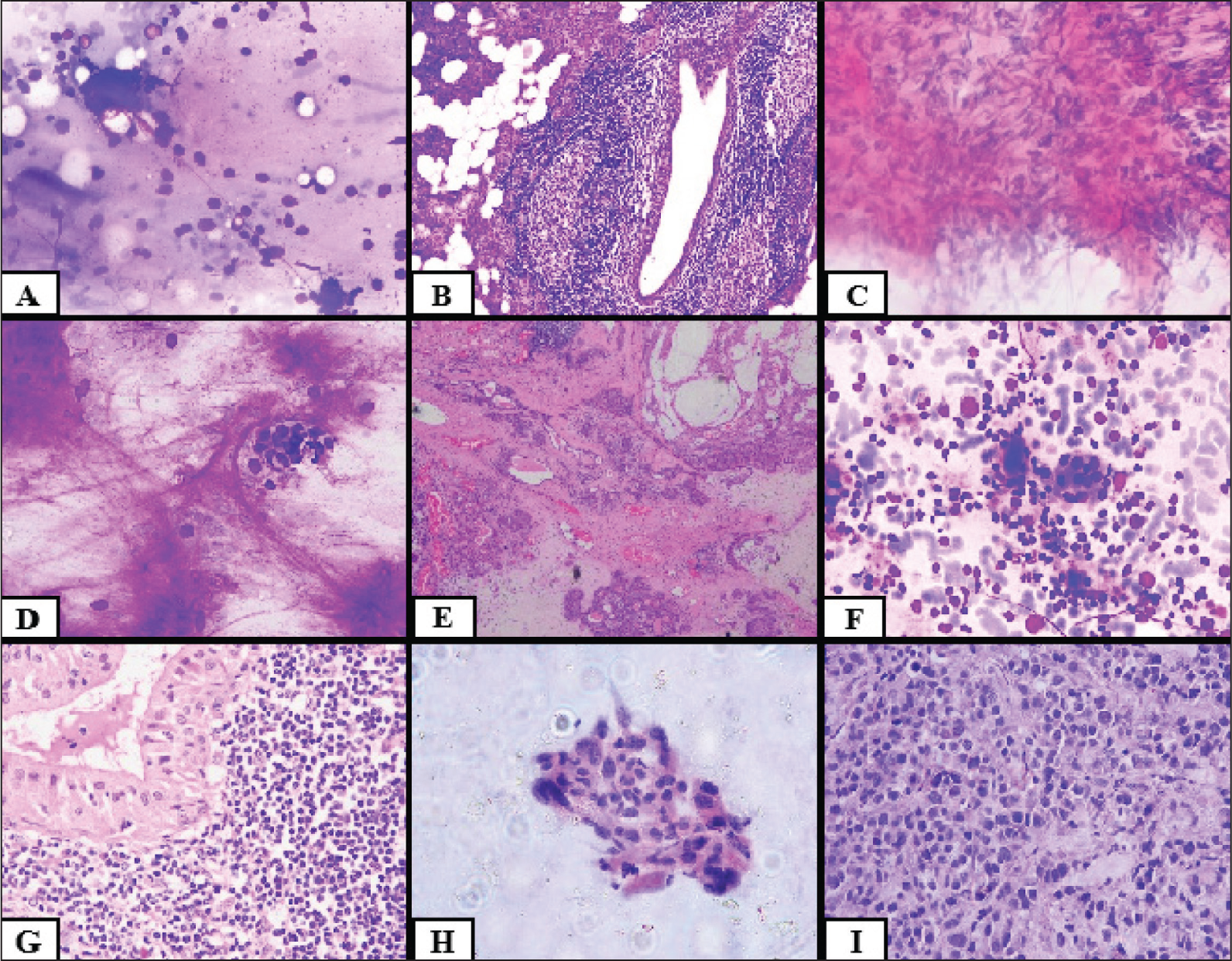
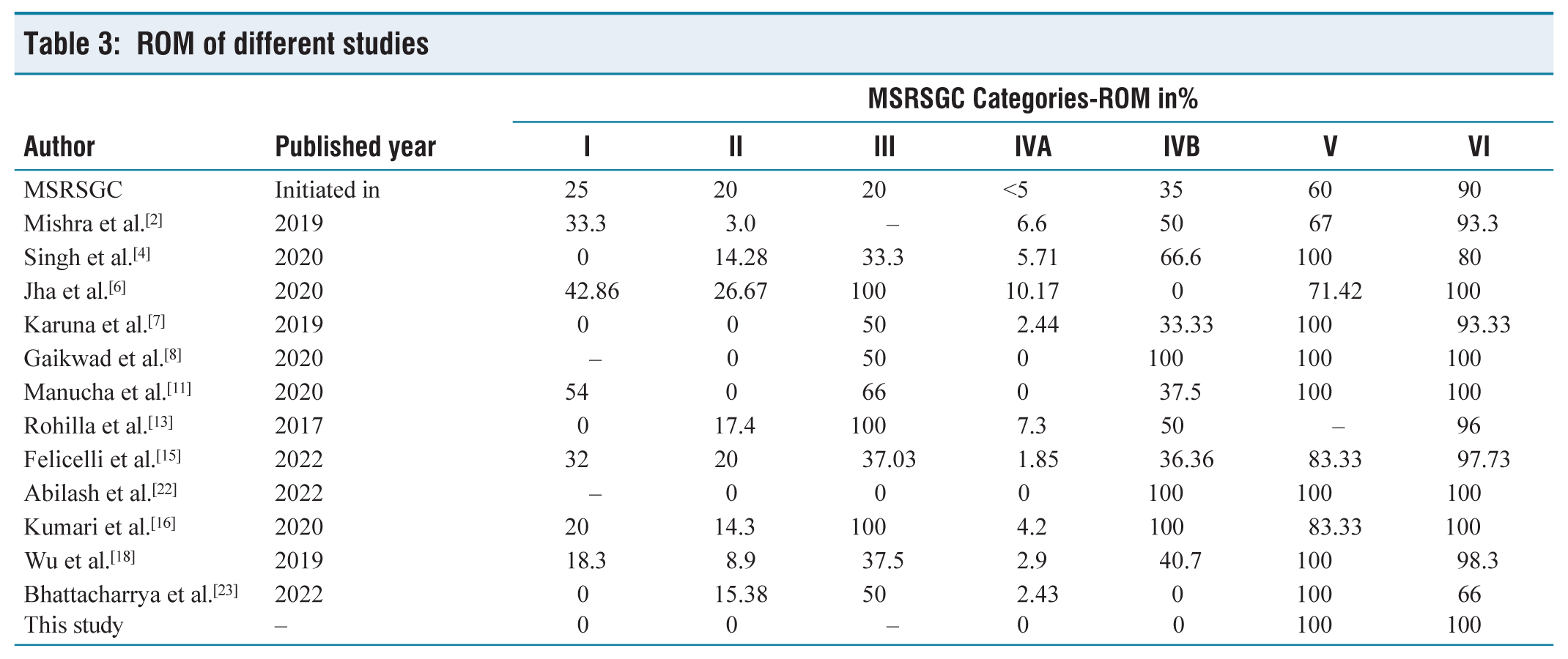
We calculated the RON and compared it with the study by Singh et al. and Rohilla et al., which were depicted in Table 2.[4,13] Despite the fact that MSRSGC has established the anticipated or estimated ROM for each category, differences in the institutional ROM were observed.[1] This study’s ROM included either 0% or 100%. This may be due to the small sample size, prospective study of short duration, the predominance of pleomorphic adenoma followed by Warthin tumour and non-neoplastic lesions and finally, an insufficient number of follow-up specimens. However, there were no major discrepancies in our study, in view of good clinical and imaging correlation, use of ROSE, well-preserved and stained smears and experienced pathologists. The ROM of various studies is depicted in Table 3.
RON of different studies
ROM of different studies
Conclusion
Salivary gland FNAC is a cost-effective, safe and sensitive diagnostic method for salivary lesions. MSRSGC is a relatively recent classification system in use for a uniform reporting format with a ROM for each category. MSRSGC suggests less than 10% of cases under category III, and this study had 1.6% of cases under category III. The most common salivary lesion in our study population was pleomorphic adenoma, which had a good histology correlation. Mucoepidermoid carcinoma was the predominant malignancy in this study, followed by poorly differentiated carcinoma. The association between the MSRSGC categories and the RON and ROM was statistically significant. Reliability in our study was good with category IVA lesions. Further prospective and retrospective study with the application of MSRSGC is needed to compare the results with other cohorts and evaluate/utilise the management guidelines. In order to obtain a better RON and ROM, future research should include a greater sample size of both FNAC and resection specimens.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional ethical committee approval number
Institutional Ethical Committee Approval Number is AMH-DNB-011/05-22.
Informed consent
Waiver of consent was obtained.
Credit author statement
Elamathi M contributed to collection of data, acquisition of data computing, interpretation of data, analysis of the data, drafting of the article and accountability for all the aspects of the study.
Suganthi K was involved in conception of the idea, interpretation of data, analysis of the data, correction of the chart and accountability for all the aspects of the study.
Maya Menon contributed to conception of idea, interpretation of data, reviewing the draft and final approval for the manuscript to be published and accountable for all the aspects of the study.
Data availability
Data supporting the findings of this study are available from the corresponding author upon request.
Use of artificial intelligence
Artificial Intelligence has not been used anywhere in the manuscript.
