Abstract
Background:
In India, 25%–65% of neonatal deaths are due to sepsis. Since it produces nonspecific symptoms, it is difficult to diagnose and manage. The present study assessed the value of brain natriuretic peptide (BNP) and C-reactive protein (CRP) to predict sepsis along with blood culture.
Introduction
Neonates are more prone to sepsis leading to high morbidity and mortality. Worldwide, the incidence of three million neonatal sepsis cases (2202/100,000 live births) has been recorded annually, whereas the highest incidence of clinical sepsis is recorded in India (17,000/100,000 live births). In India, 25%–65% of neonatal deaths are due to sepsis.[1,2] The mortality rate among neonates in Tamil Nadu is 17/1000 live births.[3]
Neonatal sepsis can manifest as early-onset neonatal sepsis (EONS) or late-onset neonatal sepsis (LONS) sepsis. EONS occurs within 72 h while LONS after 72 h of life.[4]
Neonatal sepsis can be diagnosed depending on the clinical presentation which is usually nonspecific, because similar presentation can be noticed with other noninfective conditions too. In this background, the present study was conducted to assess the value of brain natriuretic peptide (BNP) and C-reactive protein (CRP) in predicting sepsis along with the gold standard blood culture method and to detect the bacteriological profile with its sensitivity pattern.
Materials and Methods
The study includes 90 neonates (0–28 days) admitted to a tertiary care teaching hospital with clinical presentation suggestive of sepsis. Written informed consent was obtained from reliable informants of neonates. Institutional ethics committee clearance (TIREC-Ref. No.: 1602/MICRO/2019) was obtained before starting the study. Blood sample was collected under aseptic precaution before the initiation of antibiotics. Samples were inoculated into two conventional blood culture bottles, incubated at 37°C for 7 days, and observed for any growth. Identification was done by standard microbiological techniques, and antimicrobial susceptibility testing was done as per the Clinical and Laboratory Standards Institute guidelines.[5] Serum was separated and tested for CRP by latex agglutination kit (Pathozyme Diagnostics, Kagal, Kolhapur). Distinct agglutination denotes positive with a CRP value higher than 6 mg/L in undiluted serum. Remaining serum was stored at 2°C–8°C and tested for BNP by ELISA (Abbkine, Wuhan, China). Statistical analysis was executed using SPSS version 20 software. (Armonk, NY: IBM Corp).
Definition of sepsis
Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection.
Definition of neonatal sepsis
Neonatal sepsis is defined as a clinical syndrome of bacteremia with systemic signs and symptoms of infection in the first 4 weeks of life.
Inclusion criteria
Signs and symptoms suggestive of sepsis include pallor, grayish skin color, hypothermia, fever, poor cry, lethargy, refusal to suck, poor perfusion, prolonged capillary refill, muscular hypotonia, muscular hypertonia, absent neonatal reflexes, hyperexcitability, irritability on being touched, neck stiffness, bradycardia, or tachycardia. Respiratory signs include tachypnea (>60 respirations/min); dyspnea (grunting, nasal flaring, and retractions); apnea; FiO2 >0.21 (or rising FiO2 in previously stable infants); and respiratory insufficiency. Other metabolic manifestations are hypoglycemia or hyperglycemia, metabolic acidosis, and arterial hypotension (mean arterial pressure in mmHg <gestational age in weeks on day 1).[6]
Exclusion criteria
Neonates on antibiotic therapy
Neonates with birth asphyxia and aspiration syndrome
Neonates diagnosed with congenital anomalies and inborn errors of metabolism were excluded from the study.
Results
Of the 90 neonates, 49 (54%) and 41 (46%) were male and female, respectively. Based on neonatal risk factors, 42 (47%) were full-term babies (>37 weeks) and 48 were preterm babies (<37 weeks). Sixty-one (68%) had low birth weight (<2500 g). Forty-eight (53%) babies were born by spontaneous vaginal delivery, and 42 (47%) were delivered by lower-segment cesarean section. Maternal risk factors such as premature rupture of membrane (12%) and prolonged rupture of membrane (11%) were noted. Fetal distress was noted in 7 (8%) babies.
Blood culture was positive among 25 (28%) cases. Klebsiella pneumoniae (52%), Escherichia coli (16%), Pseudomonas aeruginosa (12%), Klebsiella oxytoca (4%), methicillin-sensitive Staphylococcus aureus (12%), and methicillin-resistant S. aureus (4%) are the common organisms isolated in the present study. Sixty-five percent of the isolates were sensitive to all routinely used antibiotics.
EONS was noted in 46 (51%) cases and LONS 44 (49%) cases. Of the 90 samples, blood culture was positive in 25 (28%) cases, BNP in 45 (50%) cases, and CRP in 36 (40%) cases. BNP was positive among 25 (100%) blood culture-positive cases. CRP, BNP, and blood culture were positive in 16 (18%) cases. CRP and BNP were positive in 13 blood culture-negative cases [Table 1].
Distribution of sepsis markers
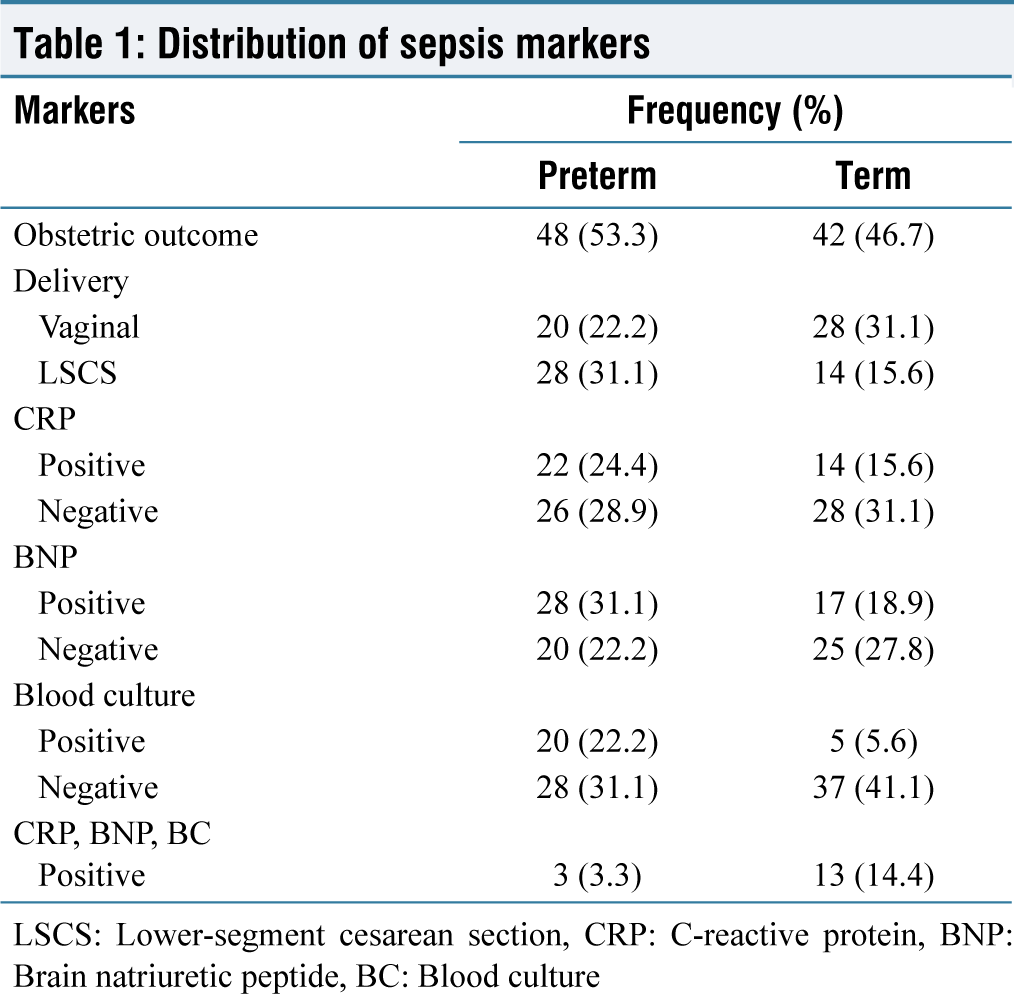
LSCS: Lower‑segment cesarean section, CRP: C‑reactive protein, BNP: Brain natriuretic peptide, BC: Blood culture
Of the 90 neonates, 87 (97%) recovered while 3 (3%) succumbed to sepsis. All the study participants were started with empirical antibiotics such as cefotaxime and amikacin after the sample collection.
Even with longer turnaround time, blood culture stands as the gold standard method in diagnosing sepsis, while the validity of CRP by latex agglutination method in predicting sepsis is 64% sensitive, 69% specific, 44.5% positive predictive value (PPV), and 83.3% negative predictive value (NPV). For BNP based on receiver operator characteristics curve (ROC) calculation, had 96% sensitivity, 69% specificity, 54.5% PPV, and 99% NPV, and the best cutoff was fixed at 75 pg/mL.
Discussion
Preterm infants who have sepsis show bradycardia, cyanosis, and apnea while full-term infants frequently have respiratory distress, which might resemble other illnesses. Diagnosis necessitates careful consideration. Neonatal sepsis may be caused by maternal risk factors such as amniotic infection, inadequate tocolysis, prelabor membrane rupture, raised maternal fever, foul-smelling amniotic fluid, and fetal tachycardia.[6,7]
The choice of antibiotics, transmission mode, and causative organisms in neonatal sepsis depend on its onset. Early-onset sepsis (EONS) is typically due to maternal transmission of Group B Streptococcus, E. coli, and Listeria monocytogenes. Late-onset sepsis (LONS) is predominantly caused by coagulase-negative Staphylococcus. High neonatal mortality is associated with organisms such as E. coli, P. aeruginosa, Serratia marcescens, and Candida albicans. Our study found a similar pattern, with Klebsiella (56%), E. coli (16%), P. aeruginosa (12%), and S. aureus (16%) as common isolates.[8]
The diagnosis of culture-proved sepsis includes the presence of at least one clinical sign suggestive of sepsis and a positive blood culture. The presence of at least one clinical sign suggestive of sepsis and a CRP >10 mg/L at 12–60 h afterthe first blood sample are designated as clinical sepsis. Patients who do not fulfill these criteria can be considered uninfected.[6]
Total leukocyte, differential leukocyte, and platelet counts are nonspecific in sepsis. Leukocytosis, driven by cytokines such as granulocyte-colony-stimulating factor (CSF), granulocyte-macrophage CSF, interleukin (IL)-3, and IL-6, is common, but leukopenia is a better predictor, especially in culture-negative cases. White blood cell (WBC) count’s reliability is hindered by age, gestational age, delivery method, and altitude variations. Viral infections (e.g., herpes simplex 1 and 2 and enteroviruses) often yield normal or slightly low WBC counts, posing challenges in neonatal sepsis diagnosis.[7,8]
Blood culture, the gold standard for sepsis diagnosis, exhibits a sensitivity below 50%.[9] In our study, 28% of probable sepsis cases tested positive for blood culture. Previous Indian studies reported sepsis rates ranging from 16% to 54%.[10] In addition, fungal sepsis incidence has surged in recent years.[11]
False negatives in blood cultures can result from inadequate sample volumes, intermittent bacteremia, antibiotic suppression, or due to other anaerobic, viral, or fungal pathogens.[12,13] Collecting 0.5–1 mL of blood is ideal for neonates, yet even with careful collection, contamination rates of 1%–3% can lead to inconclusive results and treatment uncertainty.[14]
Laboratory values in neonates fluctuate due to evolving physiology and metabolism in their early days, impacting parameters such as hormones, immunologic products, and cytokines. Careful interpretation of early neonatal reference values is crucial as they differ from later stages.[12]
In our study, BNP tested positive in all 25 (100%) blood culture-positive cases, proving its efficacy as a sepsis predictor. BNP, a cardiac hormone with diuretic and vasorelaxing properties, is released by the ventricular myocardium. Among clinically suspected cases, 45 (50%) showed BNP positivity. Elevated BNP in sepsis is attributed to factors such as proinflammatory cytokines, cardiac dysfunction, altered BNP clearance, renal issues, and sepsis-related lung problems. Medical interventions such as excessive fluid resuscitation, high positive end-expiratory pressure levels, and catecholamine infusion can also induce BNP release.[15]
CRP, an acute-phase protein produced by the liver, used to detect neonatal sepsis, takes 10–12 h to change after infection onset, with 94.8% specificity and 67.1% sensitivity. In our study, it was only positive in 16 blood culture-positive cases, indicating it as a late biomarker. Elevated CRP can occur in various conditions such as traumatic or ischemic tissue injuries, meconium aspiration syndrome, and hemolysis. Reference values, birth weight, and gestational age affect CRP kinetics. CRP rises in healthy term newborns typically from 1 mg/L at 12 h to 4 mg/L at 48 h. Infants born preterm exhibit a less significant CRP response.[16] The 95th percentile for healthy newborns at 48 h is 12–14 mg/L, despite a suggested maximum limit of 10 mg/L.[17] Contrary to this, in the current study, 46% of preterm infants and 33% of term infants were CRP positive, depending on sampling time and clinical symptom presentation. Furthermore, CRP and BNP were positive in 13 blood culture-negative cases, suggesting that CRP is almost similar to BNP in predicting sepsis.
Serial monitoring of CRP I at the time of presentation, CRP II at 24 h, and CRP III at 48 h was performed in a study by Sundarapandian et al. Of the 100 cases, CRP I was positive in 30% CRP II in 59% and CRP III in 72%. Hence, serial monitoring helps in early diagnosis, initiation, and management of antibiotic therapy.[18]
Other biomarkers include procalcitonin (PCT), serum amyloid A, lipopolysaccharide-binding protein, α-1 antitrypsin, lactoferrin, haptoglobulin, fibronectin, and neopterin are also used recently in diagnosing sepsis.[8]
PCT could distinguish bacterial infection reliably from other inflammatory conditions. However, elevated PCT values can be observed on day 1 of life in healthy neonates (mean, 3.8 μg/L; range, up to 15 μg/L). It is raised in infants with respiratory distress syndrome, heart failure, and perinatal hypoxia.[6]
Cytokines such as IL-6, IL-1β, SIL2R, tumor necrosis factor-alpha, and IL-8 are often present during infections, even before clinical symptoms appear. These biomarkers cannot cross the placental barrier, making them valuable for sepsis diagnosis. As each biomarker has limited sensitivity and specificity, it may be necessary to combine several biomarkers.
In recent times, highly accurate genetic biomarkers for sepsis diagnosis have emerged, including the hlyA, cnf1, sat1, focG, cfb, rov 5, and neuA genes.[8]
Although blood culture is the gold standard test in the diagnosis of sepsis, culture positive is being noted only in few cases which jeopardize the trustworthiness of the “gold” standard method. During the course of sepsis, the level of biomarkers may be altered; hence, it can be used as an indicator in early infection and also helps in monitoring and efficient management. In recent years, the use of molecular approaches has become common for etiological diagnosis.
Conclusion
As the clinical signs of sepsis are nonspecific, it should be anticipated, especially in high-risk neonates. Although BNP is an equally good marker in predicting sepsis, it is not widely available. Hence, screening by serial CRP along with blood culture alone reinforces the diagnosis in probable sepsis cases. Hence, early diagnosis and aggressive management with appropriate antibiotics are critical to rescue most cases of neonatal sepsis.
Limitation
Serial estimation of CRP is not done.
Footnotes
Acknowledgements
All persons who have made substantial contributions to the preparation of the manuscript, but who are not authors, are named in the Acknowledgment section and have agreed in writing to be named. No contributor has been omitted.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional Ethical Committee Approval Number
TIREC-Ref. No.: 1602/MICRO/2019.
CRediT Author Statement
All the authors have substantial contributions to each of the three components mentioned below:
Concept and design of study or acquisition of data or analysis and interpretation of data; Drafting the article or revising it critically for important intellectual content; and Final approval of the version to be published. The prominent roles of each also included the following.
RAG: Concept and design of study, literature review, and drafting the manuscript;
PSK: Literature review, clinical analysis, data analysis and interpretation, and drafting/editing and finalizing the manuscript.
Data Availability
Data supporting the study will be available with the Corresponding author and is not published or presented anywhere else.
Use of Artificial Intelligence
Nil.
