Abstract
Introduction:
Limited data exist on interphase fluorescent in situ hybridization (iFISH)-based survival outcomes in newly diagnosed multiple myeloma (NDMM) from India.
Objectives:
To study the demographics and iFISH-based survival outcomes in NDMM patients treated with proteasome inhibitors from a community-based cancer setup.
Materials and Methods:
We reviewed the records of 25 patients treated with proteasome inhibitors between June 2017 and April 2023 using five high-risk (HR) iFISH markers based on mSMART 3.0.
Results:
The median age was 60 years (range 34–87). HR iFISH was detected in 12 (48%) patients. With a median follow-up of 27 months, the overall response at the last follow-up was 80% (very good partial response - 52%, complete remission - 20%, and partial response - 8%), with 8 (32%) relapses. Twenty (80%) patients remain alive, with five deaths in HR (sepsis [n = 3]). The 2.5-year overall survival in HR and standard risk was 55.6% ± 15.2% and 100% (P = 0.01), and event-free survival was 32.4% ± 16.5% and 77.8% ± 13.8% (P = 0.02), respectively.
Conclusions:
Using limited iFISH HR markers helps in the early and effective stratification of NDMM in the real world. Sepsis remains an important cause of mortality in an Indian setup.
Introduction
Risk stratification in newly diagnosed multiple myeloma (NDMM) has evolved significantly over the last two decades, with interphase fluorescent in situ hybridization (iFISH) being the current practical standard of care for its prognostication.[1,2] Most retrospective studies from India are limited to large tertiary care government-run hospitals, with an overall paucity of iFISH-based survival outcomes.[3] Yanamandra et al. reported the largest study demonstrating a 5-year overall survival (O.S) of 83.1% among 696 patients with NDMM, but its iFISH positivity rate was 7.47% (n = 48/647).[3] Kadam Amare et al. did describe the most extensive cytogenetic data on 1104 patients with NDMM from India from a private diagnostic laboratory, but it needs more survival data.[2]
More information is, thus, needed from India. Our study describes the 2.5-year long-term clinical outcomes of NDMM treated at a community tertiary cancer setup, which constitute a significant proportion of cancer care facilities in India.
Materials and Methods
Study population
Inclusion criteria
All consecutive patients with an NDMM diagnosed[4] and treated between July 1, 2017, and April 30, 2023, were included in our study.
Exclusion criteria
All other plasma cell disorders were excluded from the study.
Stratification
Five high-risk (HR) iFISH markers based on mSMART 3.0 were tested in all 25 patients (del17p-TP53, IGH-FGFR3, IGH-MAF, IGH-MAFB, and 1q gain) and were reported according to the International System for Human Cytogenetic Nomenclature 2020.[5]
Treatment strategy
Induction
The following protocols were used:
Four induction cycles were administered before consolidation with either an autologous transplant (ASCT) or continuation of 4 more cycles as in induction. All drugs used were generics, except daratumumab. Subcutaneous (S.C) bortezomib was preferred.
Transplant
Eligible patients were offered an ASCT with injection melphalan 200 mg/m2 after completing induction therapy and achieving at least a very good partial response (VGPR) based on the International Myeloma Working Group (IMWG) guidelines.[10] Transplant-ineligible patients continued with a total of eight cycles of induction followed by maintenance indefinitely.
Maintenance
Both the transplant and nontransplant groups received oral lenalidomide 10 mg (3 weeks every month) until the last available follow-up (L.F.U), along with oral Ecosprin.[11] Supportive care was as per the guidelines.[1]
Time points for response assessment
This was based on the established IMWG criteria.[4,12] Bone marrow examination as part of response assessment was limited to ASCT recipients due to patient choice.
Statistical analysis
Descriptive statistics were calculated for all variables. O.S and event-free survival (E.F.S) were computed using the Kaplan–Meier method. The log-rank test was used to compare the outcomes of the standard risk (SR) and HR cohorts. Statistical Product and Software Solutions (SPSS) (version 23.0) was used for all statistical analysis.
Ethics
Our institutional review board approved this retrospective single-center study (AHM-ACD-067/07-22). All procedures performed were in accordance with the ethical standards of institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was waived due to the study’s retrospective nature.
Results
Baseline characteristics
Over the study period, 25 adults were treated for NDMM at our institution. The median age was 60 years (range 34–87), with 12 patients (48%) ≤60 years old. Eight patients (38%) each were identified in I.S.S II and III (n = 16/25), and five (20%) in stage I (n = 5/25). Beta 2 microglobulin was unavailable in 4 patients [Table 1].
Baseline demographics and treatment outcomes of entire cohort
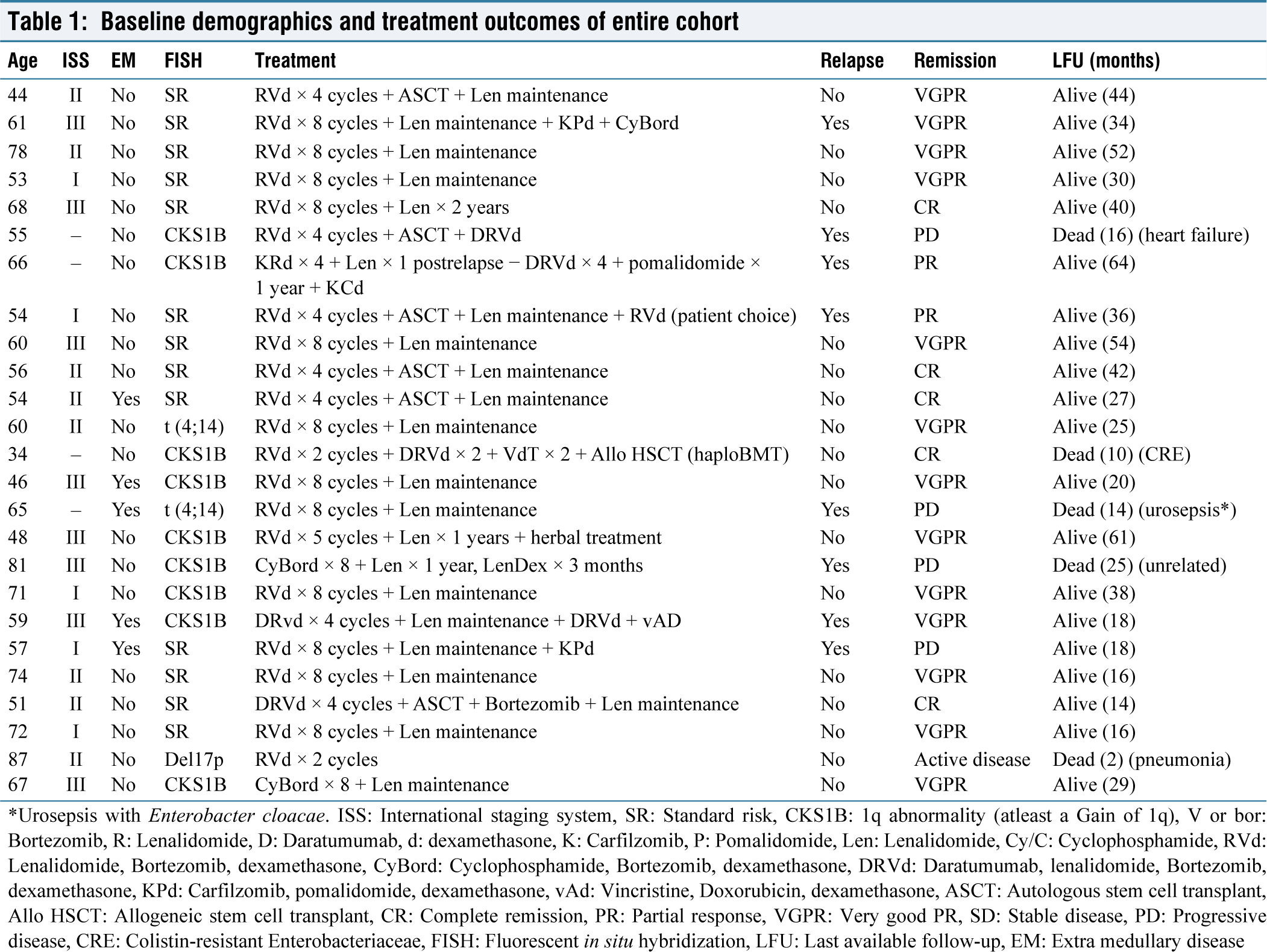
*Urosepsis with Enterobacter cloacae. ISS: International staging system, SR: Standard risk, CKS1B: 1q abnormality (atleast a Gain of 1q), V or bor: Bortezomib, R: Lenalidomide, D: Daratumumab, d: dexamethasone, K: Carfilzomib, P: Pomalidomide, Len: Lenalidomide, Cy/C: Cyclophosphamide, RVd: Lenalidomide, Bortezomib, dexamethasone, CyBord: Cyclophosphamide, Bortezomib, dexamethasone, DRVd: Daratumumab, lenalidomide, Bortezomib, dexamethasone, KPd: Carfilzomib, pomalidomide, dexamethasone, vAd: Vincristine, Doxorubicin, dexamethasone, ASCT: Autologous stem cell transplant, Allo HSCT: Allogeneic stem cell transplant, CR: Complete remission, PR: Partial response, VGPR: Very good PR, SD: Stable disease, PD: Progressive disease, CRE: Colistin‑resistant Enterobacteriaceae, FISH: Fluorescent in situ hybridization, LFU: Last available follow‑up, EM: Extra medullary disease
The most common presentation was anemia in 18 (72%) patients, followed by back pain in 17 (68%) and kidney dysfunction with lower extremity edema/oliguria in 6 (24%) patients. Twelve (48%) of the 17 patients with back pain had a vertebral collapse/fracture, with one having a paraplegia with complete urinary and bowel incontinence. Extramedullary presentation was seen in 2 (8%) patients, with one having left psoas muscle plasmacytoma and the other having multiple lung nodules and left-sided pleural effusion. Hypercalcemia was seen in 5 (20%) patients.
Cytogenetics in multiple myeloma
Twelve (48%) patients were detected to have HR on iFISH, with 9 (32%) positive for CKS1B, 2 (8%) IGT-FGFR3, and 1 (4%) with a del17p mutation [Table 1].
Treatment received
A total of 20 (80%) patients received RVd (SR - 12 and HR - 8), 1 (4%) patient received KRd (HR - 1), 2 (8%) patients received Cybord (HR - 2) considering renal dysfunction, and 2 (8%) received DRVd (SR - 1 and HR - 1) based on patient choice and clinical profile [Table 1]. Three (12%) patients underwent a decompressive laminectomy for impending or existing neurological deficits. Seven (28%) patients underwent transplants after achieving at least a VGPR, including 6 ASCT and one haploidentical transplant for refractory young myeloma with 1q gain. S.C injection of bortezomib 1.3 mg/m2 once in 2 weeks was added to lenalidomide in two HR patients.[11] Maintenance (post-ASCT or post 8 cycles of induction therapy) with lenalidomide was offered in 22 patients (SR - 13 and HR - 9). Subcutaneous bortezomib was added to lenalidomide in 2 HR patients.
Toxicity
Four patients developed Grade II anemia and neutropenia, and another three patients developed Grade II thrombocytopenia, all predominantly attributed to lenalidomide during cycle 3 of RVD regimens. A lower dose of lenalidomide at 15 mg was initiated in that cycle. Full-dose lenalidomide was restarted in subsequent cycles. One patient also developed Grade 3 sensorimotor peripheral neuropathy in bilateral lower limbs, attributed to bortezomib (after four cycles of RVd). She recovered completely within 6 months after bortezomib discontinuation. During maintenance, autonomic diarrhea was seen in two HR patients with S.C bortezomib maintenance.
Outcomes
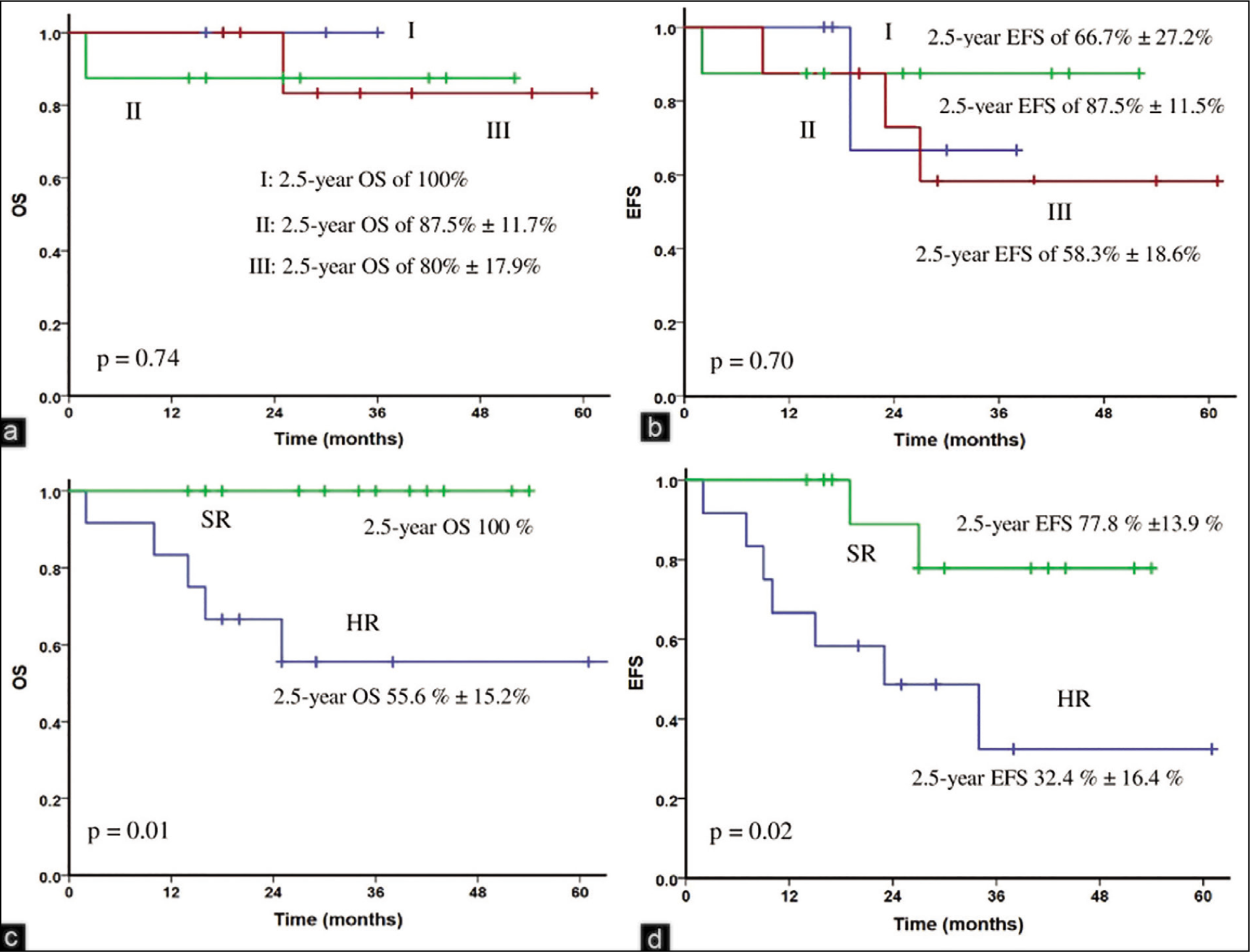
At the end of four induction cycles, the overall response was 92% (VGPR - 76% and CR - 16%) (refractory - 4% and early death - 4%) (bone marrow assessment limited to seven patients). The 2.5-year O.S in HR and SR cohorts was 55.6% ± 15.2% and 100% (p=0.01), (median follow-up 27 months, range 2-64), respectively [Figure 1c]. The 2.5-year E.F.S was 32.4% ± 16.5% and 77.8% ± 13.8% (p=0.02), (median follow-up 25 months, range 2-61), respectively [Figure 1d]. The 2.5-year O.S and E.F.S in 1q gain cohort was 60.3 ± 18.2% and 40% ± 20.3 % [Supplementary Figure 1a and b].
(a and b) 2.5-year overall survival (O.S) and event-free survival (E.F.S) of the I.S.S stages I, II, III; (c and d) 2.5-year O.S and E.F.S of standard risk and high-risk multiple myeloma based on interphase fluorescent in situ hybridization. OS: Overall survival, EFS: Event-free survival, SR: Standard risk, HR: High-risk
At the L.F.U as on April 30, 2023, 20 (80%) patients remain alive. Eight (32%) patients (HR, n = 5 [62.5%]) developed a relapse. One patient had an isolated extramedullary relapse with atypical plasma cells in the pleural fluid, and six had a biochemical relapse. All five deaths were from the HR cohort, with 4 having an underlying relapse and 1 NDMM [Table 1]. Three (12%) deaths were attributed to severe sepsis (colistin-resistant Klebsiella post haplotransplant, urosepsis with Enterobacteria cloacae, and pneumonia).
Discussion
This single-center retrospective study over 2.5 years demonstrates the demographics, cytogenetic profile, and responses to proteasome inhibitor-based therapy in a cohort of 25 patients with NDMM.
Our study included a large group of patients with anemia (A), with only few presenting with hypercalcemia (C), renal dysfunction (R), or bone disease (B). This is in contrast to the study by Yanamandra et al., in which a significant group of patients presented with substantial end-organ damage other than anemia, particularly CRB.[3] This could be attributed to regional differences, with our patients being referred earlier.
Using I.S.S as a risk stratification criterion could not reflect the true nature of the disease profile in our setup [Figure 1a and b].
In comparison, stratification using iFISH [Figure 1c and d] helped identify the HR cohort early. In addition, the high incidence of 1q abnormalities (gain of 1q) in our cohort was consistent with previously reported data.[13,14] Our outcomes in the SR cohort [Figure 1c and d] were also comparable with those of previous studies from our subcontinent.[3,15] Our HR cohort had an inferior outcome due to high relapse rates and a higher life-threatening sepsis due to more immunosuppression. Using generics and S.C bortezomib in outpatient departments did help in reducing admission costs.
Cost analysis
Single iFISH probe ₹3500 ($ 42),
HR iFISH panel (5 markers) ₹13,000 ($ 158).
Limitations
We had some limitations in our study. First, all deaths were in HR subgroup and largely attributed to sepsis, a significant challenge in India [Table 1]. The lack of uniform antibacterial prophylaxis and limited use of immunoglobulins added to the risk. Another limitation was the lower number of ASCTs, related partly to funding restrictions due to self-pay and added familial apprehensions. Our study partly reflects the trend in our country’s diversity in accessing quality health care, only with approximately 2500 transplants being performed annually (source - Indian Society of Stem Cell Transplant Registry) all over India. Limited use of bortezomib as additional maintenance therapy in HR also contributed to higher relapse.
Conclusions
Despite these limitations, our study reflects the unique challenges in treating NDMM in India. Using the five-HR iFISH panel helps in early risk stratification and limits overall cost. Sepsis remains a significant challenge in patients who relapse, and a comprehensive model with antibiotic stewardship is needed to improve outcomes.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
We acknowledge all our team members from the Department of Pathology and Microbiology at Apollo Hospital, Navi Mumbai (AHNM), and Helix Genetic and Pathology Laboratory, Powai, for performing this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional Ethical Committee Approval Number
Institutional Ethical Committee Approval Number is AHM-ACD-067/07-22.
CRediT Author Statement
Punit Jain performed the research and its design and wrote the manuscript.
Poonam Jain performed the research and its design and wrote the manuscript.
Agnivesh Tikoo performed the research and its design and wrote the manuscript.
Tejinder Singh provided concept and contributed in editing of the manuscript.
Salil Patkar contributed in editing of the manuscript.
Vaishali Lokhande contributed in editing of the manuscript.
Anand Mishra contributed in editing of the manuscript.
Bharat Agarwal contributed in editing of the manuscript.
Ashwathy Haridas contributed in editing of the manuscript.
Kanika Khandelwal contributed in editing of the manuscript.
Data Availability
The data that support the findings of this study are openly available.
Use of Artificial Intelligence
Artificial Intelligence has not been used anywhere in the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
