Abstract

Original research articles communicate new findings. Akin to the rigor of conducting original research, this requires considerable discipline. Reporting guidelines are available from the EQUATOR network for various types of original research articles, including the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) checklist for observational studies and the Consolidated Standards of Reporting Trials (CONSORT) checklist for randomized controlled trials. 1 Such checklists serve as ready reckoners of the minimum information required to assure quality. Irrespective of the type of study, most original research articles follow the Introduction, Methods, Results, And Discussion (IMRAD) format. 2 This editorial serves as a primer to the IMRAD format. These principles are also applicable to the Introduction and Methods sections of a study protocol, written in future tense as opposed to the past tense used while reporting original research. It is preferable the text of an original article is written in full sentences rather than bulleted or numbered lists.
Introduction
The introduction helps the reader understand why the research was conducted. As a rule of thumb, this section comprises a tenth of the manuscript. If targeted to a general audience, then a detailed background of the disease or condition that forms the study population might be required. This is not needed when such a paper is submitted to a specialty journal. For example, the readership of this journal would not require a detailed introduction to rheumatoid arthritis.
Thereafter, the introduction should try to bring into context the research being reported within the wider framework of what is already known. The authors should identify knowledge gaps or unmet needs, and pinpoint how their study addresses such lacunae. It is useful to visualize the introduction as a funnel, bringing into focus the relevance of the present research. 3 While narrowing down to the research question, it is good practice to state your hypothesis, even if your results eventually disprove the said hypothesis! The introduction should conclude with the aims (viz., the broader goals of the research question) and specific objectives of the study. Primary (generally, up to two) and secondary objectives should be defined here. 4
Methods
The methods section is the backbone of an original research paper, and carries the greatest weight while decisioning manuscripts. This section determines the reproducibility of a study. A good question to ask yourself is whether it would be possible for a reader to replicate the study after reading the methods. The methods comprise approximately a fourth of the manuscript. Consider using subheadings for the ease of the reader in the methods and results sections. The methods must contain information about the Patient population, Intervention(s), Comparator(s), Outcome(s) and Time period (T) of the study (abbreviated as PICOT).
The methods should begin with the study design (cross-sectional, cohort – prospective or retrospective, single arm or randomized controlled trial), followed by a description of the study population: which disease, with what complication or phenotype, source of subjects – community or hospital, duration of enrolment/ study period, whether they were part of an existing cohort (if so, reference relevant prior publications from the cohort). Thereafter, clear inclusion and exclusion criteria for the purpose of the study should be detailed. In the inclusion criteria, please specify which classification criteria the study subjects fulfilled, which ensures uniformity of study cohorts across geographic locations to preserve external validity. Please also define whether there was a control group of diseased or healthy individuals, with inclusion and exclusion criteria for the same.
At this stage, it is imperative to mention whether written informed consent was obtained from study participants (for a prospective study) or such a requirement for consent was waived by the relevant ethics committee for a retrospective chart review. For studies involving human participants or animals, authors should report the full name of the approving ethics committee, the document submission number to the said committee, and the date of approval. The latter is important, as prevalent ethical standards require studies to be initiated only after ethics approval. Ethics approval is not needed for studies lacking direct participation of subjects, such as systematic reviews or meta- analyses of published literature. Clinical trials mandatorily require pre-registration in clinical trial registries before enrolment of the first patient. Here, it is also important to mention about the funding for the study. In a clinical trial, it is essential to know if the funder was the commercial entity that manufactured the trial product, and what role they had in the design of the study. 5 In this era of large language models and artificial intelligence, authors must transparently declare whether and to what extent such tools were used to aid the writing of their manuscript. Please note that most publishers today do not allow the use of artificial intelligence to generate content for publication. However, this might be used ethically to improve the language of papers with transparent declaration. 6
Thereafter, please detail the study variables and how they were collected (case record form, clinic files, prospective interviews). If questionnaires were filled out by the study participants, they should be validated ones. Please validate any new questionnaires (at a minimum, face validity, content validity, test-retest reliability) before reporting them. It is the obligation of the authors to seek due permission from copyright holders of existing questionnaires or scales before using them in the study or reporting them. An inability to do so might result in legal complications or form grounds for errata or retractions of published studies. It is essential to reference any criteria or disease assessment tools used in the study to their source. If the use of laboratory equipment or reagents is being reported, then their make and country of origin should be mentioned within brackets.
The methods should conclude with a section detailing the statistical analysis to be reported in the results. Please specify which statistical software was used, including its version, company, and country of origin. It is essential to justify the sample size and detail the assumptions made for this to ensure adequate study power. If a sample size of convenience is being used, the reasons for this must be made explicit. It is not recommended to estimate post hoc study power if a sample size was not calculated a priori. 7 Instead, mention this as a limitation. Due attention to the use of appropriate statistical tests, depending on the nature of data points (paired or unpaired), is essential. Regression models should be used wherever appropriate; if multivariable-adjusted models are used, then these should be adequately powered. 8
Results
The results should correspond to the methods. Detail the characteristics of the study participants (including controls) in as much detail as required to understand the relevant research question. Thereafter, please report the analyses detailed in the methods in a logical order. A flow between the different analyses being reported and the judicious use of figures and tables helps keep the reader engaged. Keep in mind that the text of the results should complement rather than simply repeat the information presented in figures or tables. It is preferable that the results should focus on effect sizes and 95% confidence intervals rather than just p-values.
Discussion (and Conclusion)
The discussion brings into light the findings of the study in the context of what was already known. It is useful to visualize the discussion as an inverted funnel (whereas the introduction is better visualized as a funnel). The discussion should start with a paragraph summarizing the findings of the study without repeating numbers or figures from the results. Here, it is useful to state whether your study hypothesis was proven or disproven by the results obtained from your study. Remember to highlight the new and important findings from your study.3,9
Thereafter, each paragraph should discuss one key finding of the study in the context of the existing knowledge on the subject. Try to mention what the study adds to what is known. If your results are disparate from the existing dogma on the topic, try to explain why this might be the case, utilizing references from the literature or hypothesizing the reasons for this. Similarly, discuss contradictory findings from your study. The penultimate paragraph of the discussion should critically analyze the limitations and strengths of the study. In this section, it is crucial to justify why the study’s findings are relevant despite these limitations. The concluding paragraph of the discussion (often, a separate section, the Conclusion) should briefly highlight the key findings of a study while identifying a few (up to three) avenues for future research in the area. 9
The discussion should use references liberally to provide an appropriate context. It is imperative to avoid overstatements in the discussion, whilst noting the novelty or importance of the findings of a study with humility. Please do not draw conclusions that your study was not designed to answer.
Tips and tricks
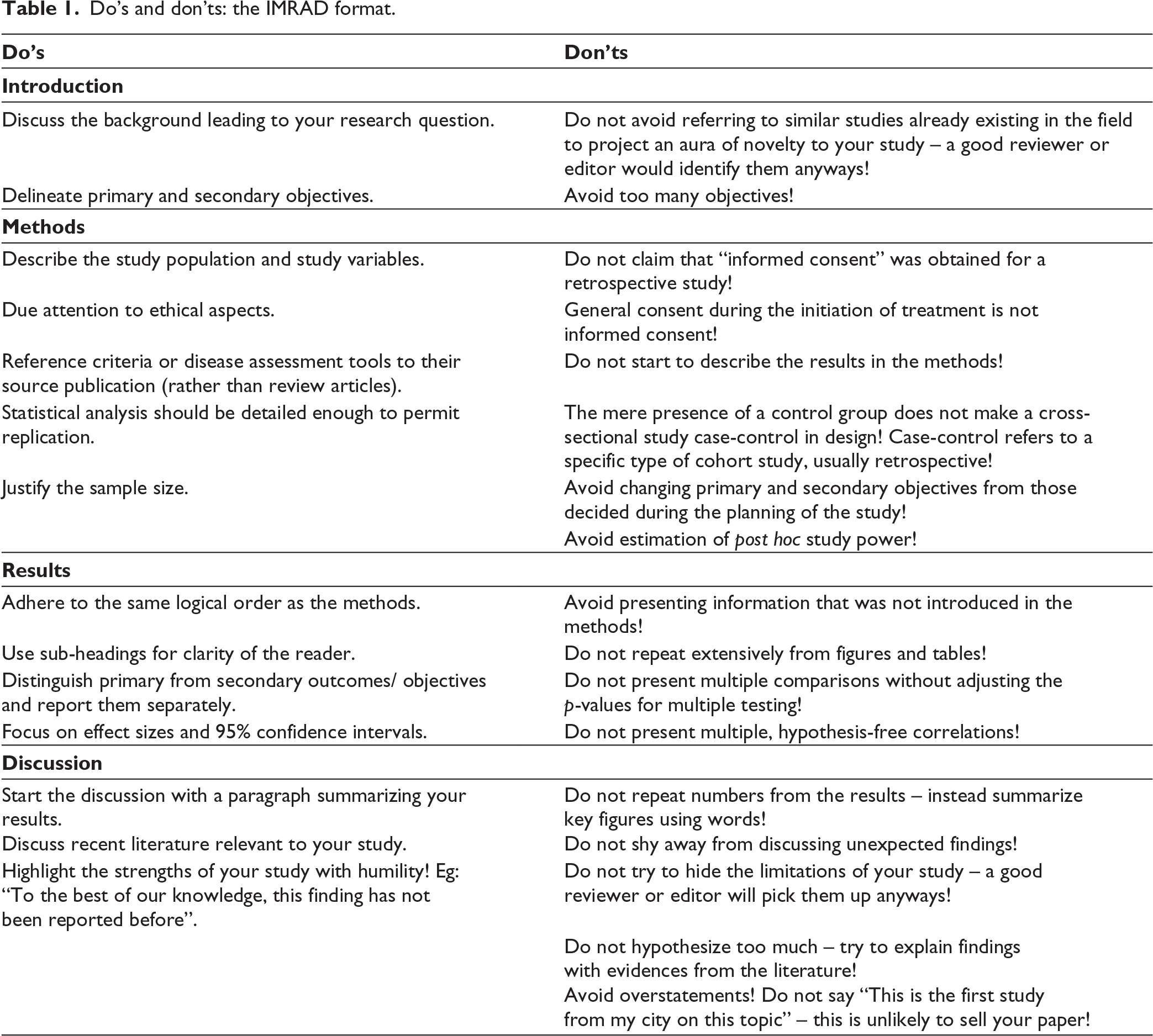
Table 1 lists do’s and don’t’s for original research articles. Clarity, reproducibility, transparency and due attention to ethical aspects are key to effective scientific publication through original research articles.
Do’s and don’ts: the IMRAD format.
Footnotes
Author’s Contribution
Substantial contributions to the - conception or design of the work, the acquisition, analysis, or interpretation of data for the work - DPM Drafting the work – DPM Revising it critically for important intellectual content – DPM Final approval of the version to be published - DPM Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved - DPM.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data Availability Statement
All the analyses performed for this article have been reported in the main text or in the supplementary files. Data pertaining to the article shall be shared on reasonable request to the corresponding author (Durga Prasanna Misra, durgapmisra@gmail.com).
Ethical Approval
Not applicable.
Funding
The author disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Use of AI Tools
The author used Grammarly to assist with the correction of grammar during the preparation of the manuscript. The author takes full responsibility for the content.
