Abstract
Introduction:
Systemic lupus erythematosus (SLE) is a chronic inflammatory autoimmune disease influencing numerus tissues in body. Ubiquitin carboxyl-terminal hydrolase 27 (USP27X) is a deubiquitinating enzyme involved in the type I interferons (IFNs) generation, apoptosis and production of proinflammatory mediators. DEAD-box helicase 3 X-linked (DDX3X) is a ribonucleic acid (RNA) helicases and contributes to the generation of type I IFNs, and proinflammatory mediators. Considering the involvement of IFN type I, apoptotic cell death and proinflammatory mediators in the pathogenesis of SLE, the expression levels of USP27X and DDX3X genes were assessed in SLE patients.
Methods:
Two groups, including SLE patients and healthy subjects, were included in the study and the expression levels of USP27X and DDX3X genes in their peripheral blood mononuclear cells (PBMC) were quantified by the real-time polymerase chain reaction (PCR).
Results:
The results demonstrated the expression level of DDX3X gene was 5.9-fold higher in the SLE patients compared to the healthy subjects (P < .01). No significant difference in the expression level of USP27X gene was detected between both groups (P > .05).
Conclusion:
A significant elevation in the level of DDX3X gene in SLE patients prepossess the potential involvement of DDX3X in SLE pathogenesis possibly through the induction of proinflammatory mediators. In spite of the potential involvement of USP27X in SLE through the induction of type 1 IFNs, and apoptotic cell death, our finding did not provide evidence in the contribution of USP27X in SLE pathogenesis.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease resulting in inflammation, and injury in the tissues and organs. Various organs and tissues, including skin, brain, skin kidneys, lungs and blood vessels are affected. 1 Signs and symptoms such as fatigue, skin rashes, fever are recognised in the disease. Two periods, including flare, and remission are recognised in the disease. In the flare, the signs and symptoms are present, while they are silent in the remission. 1 The causes of SLE are hidden; however, it may be correlated with the genetic, environmental and hormonal backgrounds. The incidence is different in various countries, ranging from 0.007% to 0.0017%. The disease is more abundant in women compared to men. 2 Immunosuppressive drugs aid to the disease control, and include hydroxychloroquine and corticosteroids that control the immune response. The first Food and Drug Administration (FDA)-approved belimumab tin 2011 composed of human monoclonal antibodies against B-cell activating factor (BAFF), and is the first biologic consumed in SLE therapy. 3
Ubiquitin carboxyl-terminal hydrolase 27 (USP27X) is a deubiquitinating enzyme that eliminates ubiquitin from the proteins. 4 Ubiquitin is a small protein composed of 76 amino acids, and is added to a protein in a process called ubiquitination highlighting the protein for the degradation in the proteasome. 5 In addition, USP27X generates type 1 interferons (IFNs), inflammation and apoptotic cell death.6,7 Given the elevated level of type 1 IFNs, including IFN-α, and IFN-β, inflammatory cytokines and apoptotic death in SLE, USP27X may be involved in the SLE pathogenesis.8-11 Therefore, we evaluated the expression level of USP27X gene in SLE patients, and compared with the healthy subjects.
DEAD-box helicase 3 X-linked (DDX3X) is a ribonucleic acid (RNA) helicase with DEAD motif. The family members contribute to the RNA metabolisms, including RNA transcription, splicing and translation. 12 They have been recognised as the detectors of the innate immunity that contributes to the generation of type 1 IFNs, and inflammatory mediators. 13 Considering the elevated level of type 1 IFNs, and inflammatory mediators in SLE patients, DDX3X may contribute to the SLE pathogenesis. Therefore, we quantified the expression level of DDX3X gene in SLE patients, and compared with the healthy subjects.
Materials and Methods
Sample Collection
In this study, a case–control study was carried out. Blood samples from the SLE patients, and healthy subjects were acquired in Kashan, Iran. The blood samples of entire subjects in both groups were collected after acquiring satisfaction. Peripheral blood mononuclear cells (PBMCs) were isolated from the blood samples (Lymphodex, Innotrain, Germany) based on the manufactures՜ protocol, and stored in –20°C.
SLE patients were included into the study according to the criteria from systemic lupus international collaborating clinics (SLICC, 2012). 14 Criteria included malar rash, photosensitive rash, acute cutaneous lupus, discoid, oral ulcers, alopecia, arthritis, serositis, renal, neurologic, haemolytic anaemia, leukopenia, lymphopenia, thrombocytopenia, antinuclear antibody (ANA), anti-double-stranded deoxyribonucleic acid (anti-dsDNA) antibody, anti-Sm antibody, antiphospholipid antibody and low complement. 14
SLE patients with active disease were distinguished from inactive form according to the systemic lupus erythematosus disease activity index (SLEDAI). Only subjects with the active disease were entered into the study (36 patients).15,16 Patients with SLEDAI score ≥ 6 were regarded to have an active disease, while those with score < 6 regarded to have an inactive disease. The SLEDAI criteria included seizure (8 score), organic brain syndrome (8 score), visual disturbance (8 score), cranial nerve disorder (8 score), lupus headache (8 score), cerebral vascular accident (CVA) (8 score), vasculitis (8 score), arthritis (4 score), myositis (4 score), urinary casts (4 score), haematuria (4 score), proteinuria (4score), pyuria (4 score), new rash (2 score), alopecia (2 score), mucosal ulcers (2 score), pleurisy (2 score), pericarditis (2 score), low complement (2 score), increased DNA binding (2 score), fever (2 score), thrombocytopenia (1 score) and leukopenia (1 score). 16
The samples of the healthy subjects were also collected (40 samples), and included into the study based on negative results for C-reactive protein (CRP) experiment.
Measurement of the Expression Level of USP27X and DDX3X genes
To quantify the expression levels of USP27X and DDX3X genes, a real-time polymerase chain reaction (PCR) was carried out. Initially, RNA was extracted from the PBMCs according to the manufactures՜ protocol (Parstous, Iran). Briefly, 750 μL of RL solution was added to PBMCs pellet, and homogenised. After 5 minutes of incubation in the room temperature, 150 μL of chloroform was added and shaked fully for 15 seconds, and incubated for 3 minutes at room temperature. Thereafter, microtube, containing mixture, spined for 12 minutes at 13000 rpm at 4°C. After centrifugation, 400 μL of upper phase transferred to a new microtube, and 400 μL of 70% ethanol was added to the microtube, and mixed thoroughly. Afterwards, the mixture transmitted to the spin column and spined for 1 minute at 13000 rpm. The flow-through of the collection tube was discarded, and 700 μL of PW solution was added to the spin column and spined for 1 minute at 13000 rpm. The flow-through of the collection tube was discarded. Finally, 30 μL of diethyl pyrocarbonate (DEPC)-treated water was added to the spin column, and after 3 minutes incubation at room temperature, the solution was spined for 1 minute at 13000 rpm. The eluted RNA solution was stored at –70°C.
Thereafter, complementary DNA (cDNA) synthesis was performed from the RNA according to the manufactures՜ protocol (Parstous, Iran). Briefly, cDNA synthesis was performed in a 20 µL volume consisting of 10 µL of buffer mix (2X), 2 µL of enzyme mix and 8 µL of the RNA (2.5 μg RNA). Afterwards, the mixture was incubated in 25°C for 10 minutes, followed by incubation in 47°C for 60 minutes. Finally, the reaction was finished by warming the mixture at 85°C for 5 minutes, and chilling at 4°C.
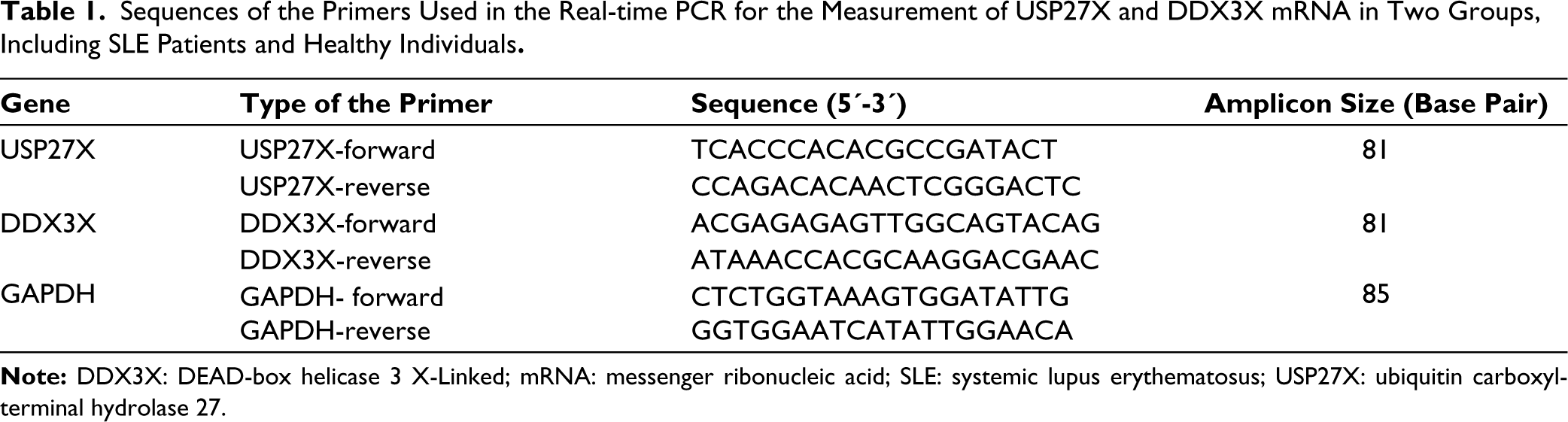
The real-time PCR reaction was carried out in a 20 µL volume composed of 2X green buffer (10 µL), 0.4 µL for forward primer (10 μM), 0.4 μL for reverse primer (10 μM), cDNA template (1 μL) and DEPC treated water (8.2 μL). Primer sequences are demonstrated in Table 1. Cycling real-time PCR conditions included an initialisation step of 10 min at 95°C, continued by 40 cycles, including a denaturation step at 95°C for 15 seconds, an annealing step at 60°C for 30 seconds, and an elongation step at 72°C for 1 minute, continued by a final elongation step at 72°C for 5 minutes. Relative expression levels of genes were calculated using 2-∆∆ct method. 17
Sequences of the Primers Used in the Real-time PCR for the Measurement of USP27X and DDX3X mRNA in Two Groups, Including SLE Patients and Healthy Individuals.
Statistical Analysis
Statistical analyses were performed using statistical package for the social sciences (SPSS) programme (v. 28; Chicago, USA). Comparison between groups was performed based on the Student’s t test (in data with normal distribution) or Mann–Whitney U test (in data with non-normal distribution) tests. P value < .05 was statistically significant.
Results
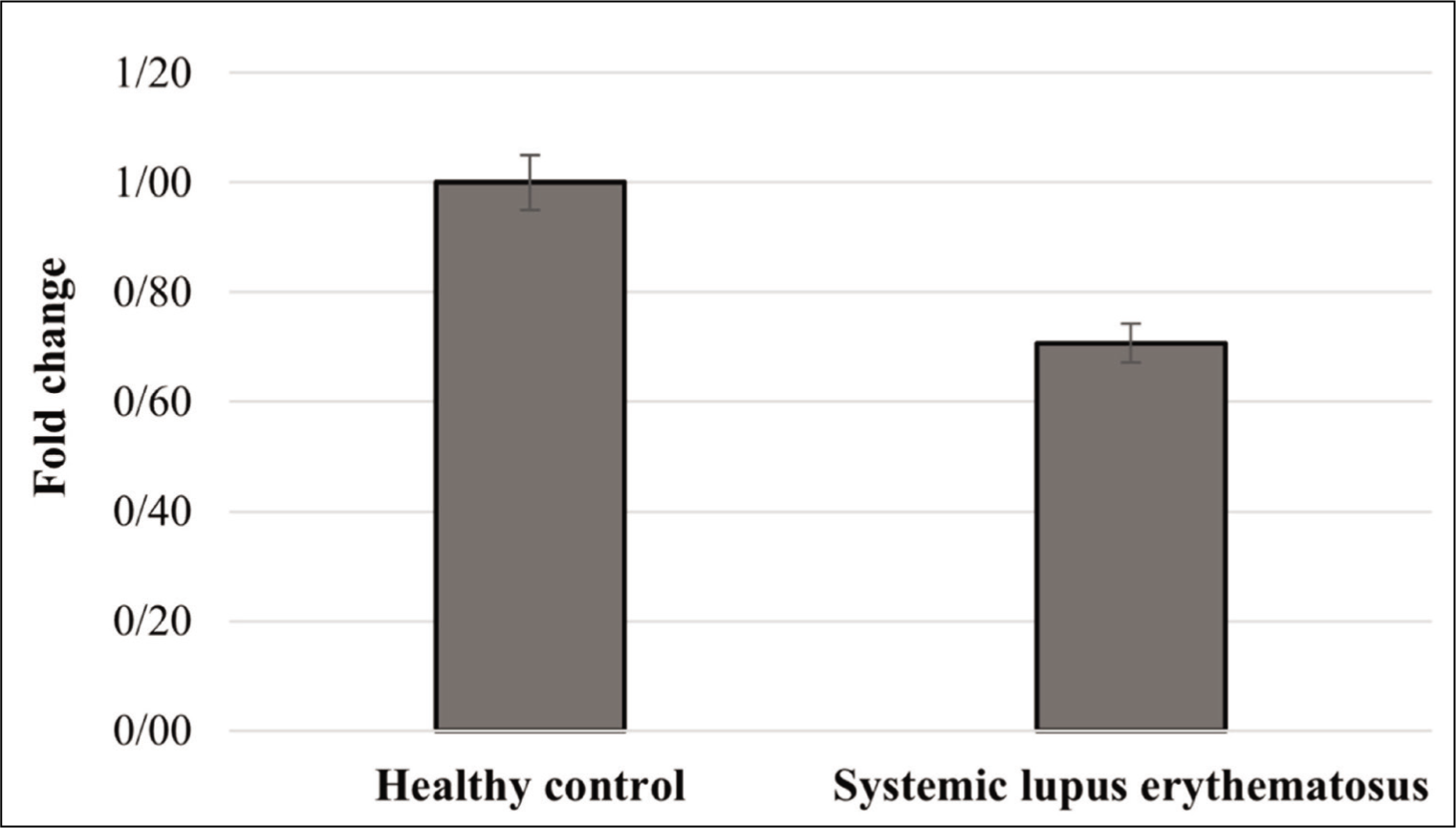
The Expression Level of USP27X Gene in SLE
According to the results, no statistically significant difference in the expression level of USP27X gene was recognised between two groups (P > .05; Figure 1). No significant difference was found in the expression level of USP27X between SLE patients and healthy subjects suggesting USP27X may not be involved in SLE pathogenesis. However, more studies including analysis of protein level and functional analysis are necessary to uncover potential contribution of USP27X in SLE pathogenesis.
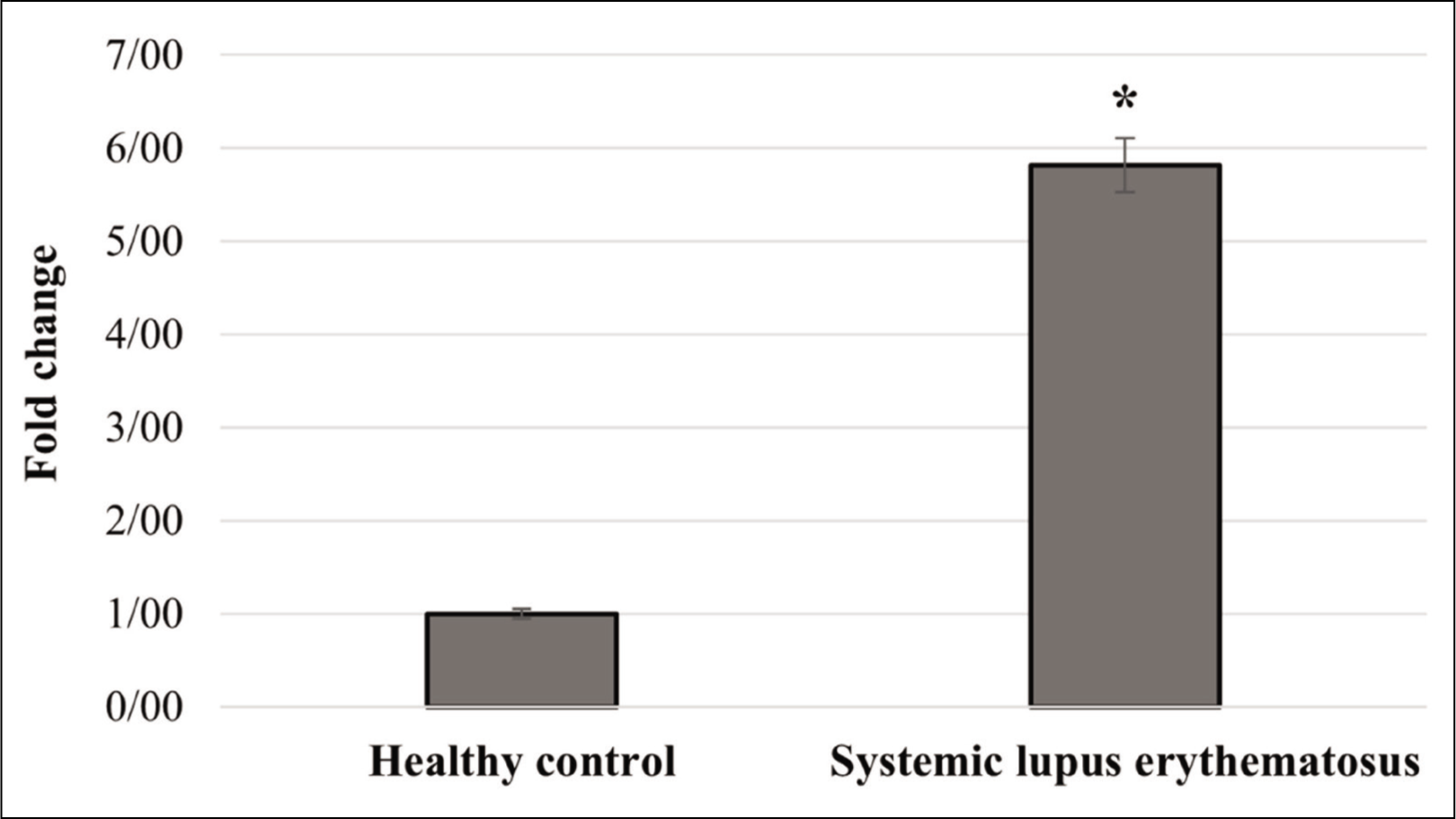
The Expression Level of DDX3X Gene in SLE
The results demonstrated that the expression level of DDX3X gene was 5.9-folds higher in the SLE patients in comparison to healthy subjects, which was significant (P < .01; Figure 2). Altogether, this may be a significant finding, providing new views into the contribution of DDX3X in SLE pathogenesis.
Discussion
In the current survey, the mRNA levels of USP27X and DDX3X genes were investigated in SLE patients. According to the results, the expression level of DDX3X gene was significantly higher in the SLE patients compared to healthy subjects. No significant difference in the level of USP27X gene was recognised between two groups.
SLE is an autoimmune disease in which the immune system attacks to the self-tissues. The results are inflammation, and tissue injury. 1 Cytokines, including proinflammatory cytokines (tumour necrosis factor-alpha (TNF-α), interleukin (IL)-1β, IL-6), chemokines and IFNs type І, contribute to the disease pathogenesis. 18
USP27X is a deubiquitinating enzyme that removes ubiquitin from cyclic guanosine monophosphate (cGMP)-adenosine monophosphate (AMP) synthase (cGAS) leading to its activation. Activated cGAS detects cytosolic DNA and exerts inflammation. Activation of cGASby self-DNA mediates the pathology of a number of autoimmune diseases. 19 In addition, cGAS activates phosphorylated TANK-binding kinase 1 (TBK1) that in turn initiates activation of interferon regulatory factor 3 (IRF3). IRF3 is a transcription factor that triggers the transcription of inflammatory genes, especially type I IFNs. Altogether, USP27X initiates inflammatory response and generates IFNs type I via the cGAS-TBK1-IRF3- IFN-β axis.4,20 Accordingly, USP27X-deficient macrophages that were acquired from the USP27X-deficient mice demonstrated the disturbed innate antiviral response against herpes simplex virus type 1 (HSV-1) infection accompanied by a reduction in the production of type I IFN. The decreased level of type I IFNs was along with a reduction in cGAS level, reduction in TBK1 phosphorylation and the reduced activation of IRF3. 4 The pathological function of type I IFNs, including IFN-α, and IFN-β, is greatly known in the SLE pathogenesis. 21
USP27X may also be involved in autoimmunity by increasing apoptotic death. Increased apoptosis contributes to the pathogenesis of a number of autoimmune diseases, especially SLE. 22 USP27X removes ubiquitin from the phosphorylated and ubiquitinated Bim 7 that is a pro-apoptotic protein inducing apoptosis after its activation. 23 USP27X stabilises Bim proteins through the deletion of the ubiquitin from this protein. 7 Correspondingly, overexpression of USP27X in tumour cell lines, including melanoma, and non-small cell lung cancer cells induces a pro-apoptotic activity. 7
Notably, USP27X may mediate the pathogenesis of autoimmune fibrotic diseases such as scleroderma, 24 because USP27X activates fibroblasts through deubiquitination, and stabilisation of SNAIL1 (snail family transcriptional repressor 1) which is a transcription factor that mediates fibroblasts activation. 25 Activated fibroblasts increase angiogenesis and fibrosis through the production that has of transforming growth factor β (TGF-β), vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF). 26
In this study, possible involvement of USP27X in SLE pathogenesis was investigated by quantification of the expression level of USP27X gene. Our result provided no evidence of the USP27X contribution to the SLE pathogenesis, as no significant difference in the expression level of USP27X gene was detected between SLE patients and healthy subjects.8-11 However, more studies are essential to answer the issue if there is a significant difference in the protein level of USP27X between SLE patients and healthy subjects. As no full concordance exists between expression level of a gene and its protein level.27,28 Moreover, functional analysis is necessary to investigate activation state of USP27X (deubiquitination of the proteins) in SLE patients and healthy subjects. 4
DDX3X is from RNA helicases family that has been recognised as an immune detector in innate immunity, especially in the antiviral immune responses by the induction of type I IFNs. For example, chicken DDX3X produced IFN-β through STING-TBK1-IRF7-IFN-β pathway. In addition, DDX3X generated the proinflammatory mediators, such as IL-1β, IL-6, IL-8, myxovirus resistance 1 (MX1) and protein kinase R (PKR). 29 Accordingly, porcine DDX3X increased generation of IFN-β. 30 Fish DDX3X also induced an antiviral response mediated by the transcription factors, including IRF3 and IRF7 and consequently IFN-β production. 31
Samir et al. observed that the DDX3X-deficient macrophages obtained from bone marrow have the lower activation of the NLR family pyrin domain containing 3 (NLRP3) inflammasome suggesting that DDX3X promotes the inflammatory response by the generation of proinflammatory cytokines, including IL-1β and IL-18. 32
DDX3X-deficient macrophages infected with Listeria monocytogenes were impaired in toll-like receptors (TLRs) signalling, after their activations with TLRs agonist, including TLR3 agonist (poly (I:C)) and TLR4 agonist (lipopolysaccharide (LPS)). Notably, TLRs are a main family of pattern recognition receptors (PRRs) that render the first line of immune system against microbes by the detection of invading microbes. In addition to a disturbance in TLRs signalling, DDX3X-deficient infected-macrophages produced a lower amount of the proinflammatory cytokines, including IL-1β, IL-6, IL-12, TNF-α and IFN-β. Another line of experiments demonstrated that three transcription factors, including IRF3, IRF7 and nuclear factor kappa B (NF-κB), were responsible for the cytokine production. 33
In this study, possible involvement of DDX3X in SLE pathogenesis was investigated by quantifying the expression level of DDX3X gene. Our result provided preliminary confirmation in the contribution of DDX3X gene in SLE pathogenesis, as a significant elevation in expression level of DDX3X gene was detected between SLE patients and healthy subjects (a 5.9-fold elevation in the expression level of DDX3X gene in SLE patients compared to healthy subjects). Altogether, this study is the first one to investigate the level of DDX3X in SLE patients, providing a new view into the function of DDX3X in SLE pathogenesis.
Conclusion
Our result provided initial confirmation in the contribution of DDX3X gene in the SLE pathogenesis. However, no significant difference in the expression level of USP27X gene was recognised between SLE patients and healthy subjects. More experiments are needed to evaluate the issue if there is a significant difference in the protein level of DDX3X and USP27X between SLE patients and healthy subjects, as no entire concordance exists between the expression level of a gene and its protein level.
Footnotes
Acknowledgements
The authors thank the individuals who participated in this study. Kashan University of Medical Sciences financially supported this study.
Author Contributions
All persons who meet authorship criteria are listed as authors, and all authors certify that they have participated sufficiently in the work to take public responsibility for the content. Supervision, conception, and design of the study: S.G and B.Z, Investigation, Methodology, Resources: A.R.O and H.M, Writing – review & editing: Z.M, Data analysis: M.J.M.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The research related to human use complied with all the relevant national regulations and institutional policies was performed in accordance with the tenets of the Helsinki Declaration, and has been approved by the Research Review Board of Kashan University of Medical Sciences, Kashan, Iran. Before sampling, the study procedure was completely explained to all participants, and informed consent was obtained from all individuals included in this study.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
Before sampling, the study procedure was completely explained to all participants, and informed consent was obtained from all individuals included in this study.
