Abstract
Background:
In this study, we aimed to investigate subclinical ultrasonographic change suggestive of gouty arthritis in the hyaline cartilage, joints and tendons from asymptomatic hyperuricemia (AH) individuals with hyperuricemia.
Methods:
We conducted a cross-sectional, observational study including US examinations of the knees and first MTPJs as well as of the tendons and enthesis. The normal distribution of different parameters was tested by the Shapiro–Wilk normality test. Quantitative data were expressed in mean ± standard deviation and the difference between means of two groups was tested by Student’s t-test while qualitative data were expressed in frequency and the difference between proportions was tested by the chi-square test or Fisher exact test.
Results:
Fifty-nine AH individuals and 52 normouricemic subjects were included. A total of 118 knees, ankles and first MTPJs in patients with AH and 104 in the normouricemic subjects were studied. The most common abnormality found on USG was power Doppler signal in 1st MTPJ (37% vs. 0%; P < .001) followed by joint cavity widening (31% vs. 0%; P < .001). Double contour sign was found in 11% of the first MTPJs from AH individuals versus none in the control group (P < .001). Posterior tibialis tenosynovitis (6% vs. 0%; P < .01) and Achilles tendon enthesopathy (8% vs. 0%; P < .001).
Conclusions:
These data demonstrate that subclinical changes detected in the US suggestive of gouty arthritis induced by chronic hyperuricemia frequently occur in both intra- and extra-articular structures of clinically AH individuals.
Keywords
Introduction
The serum uric acid level reflects the stability between the degradation of purine compounds and the speed at which monosodium urate is excreted by the kidneys. When the urate level exceeds >7 mg/dl, there is an increased chance of MSU crystal tissue deposition. 1 Urate-lowering treatment (ULT) use is well known in gouty arthritis or nephropathy, but its use in asymptomatic hyperuricemia (AH) individuals is controversial.2,3 Ultrasound (US) has been demonstrated to be an effective imaging technique for identifying musculoskeletal involvement in gout sufferers.4–11 US findings include double contour sign (DCS), hyperechoic spots (tendons), tophi and bone erosions.6,12,13 Enhanced blood perfusion around the MSU deposits observed through power Doppler US (PDUS) indicates inflammatory activity.5,14 This observation aimed to take a look at the role of US/PDUS in figuring out latent musculoskeletal manifestations in the tendons, soft tissues and lower extremity joints (knee, ankle and first MTPJ) in patients with subclinical hyperuricemia.
Materials and Methods
We executed this study at the departments (hidden for anonymity) over a period of two years (August 2019–August 2021). Approval from the Ethical Committee was granted to conduct it. All participants provided informed consent. It included 59 individuals with serum uric acid levels of ≥7.0 mg/dl. The sample comprised individuals diagnosed with AH who visited the outpatient department and those admitted to the department of general medicine. Fifty-two healthy controls, each with uric acid levels less than 7 mg/dl, were selected from relatives of patients. Complete medical history and physical examination were done and a uniform standard procedure was used to check for joint examination. Investigations relevant to hyperuricemia patients were done. Serum uric acid was measured in a fasting state on two occasions at least one week apart by enzymatic method (URICASE).
Ultrasound Examination
All patients subjected to blinded PDUS and grey scale ultrasound (GSUS) examination within 48 hours of clinical assessment. US examinations were conducted using an ALOKA PRO SOUND 3500 with linear high-resolution 10–12 MHz transducers. PDUS examinations of the knee, ankle and first MTPJ, as well as GSUS examinations for effusion/hypertrophy (E/H), were done. The US examiner conducted assessments without being informed of the clinical findings. During PD examination, the Doppler settings were fine-tuned for detecting low flow, utilising a medium wall filter to reduce flash artefacts. The pulse repetition frequency (PRF) was optimised to maximise sensitivity at the lowest feasible level for each joint, resulting in a PRF of 500–750 Hz. Colour gain was adjusted just below the noise floor. Joint effusion was documented upon observing widening of the joint cavity characterised by anechoic or hypoechoic features. At the same time, synovitis was determined through detection of tissue within the joint that exhibits either reduced or increased echogenicity. Hyperechoic enhancement along the outer edge of hyaline cartilage suggested MSU crystal deposition, referred to as DCS. Enthesopathy or tendinopathy was distinguished by uneven thickening of tendons or entheseal areas and intratendinous hyperechoic bands. Erosion was defined by a distinct interruption in cortical structure displaying a step-down change in contour visible from both longitudinal and transverse views. An enthesophyte was identified by an elevated prominence where the bone profile normally terminates. GSUS E/H was assessed qualitatively to determine absence (0) or presence (1) in the specified joints: first MTPJ, ankle joint and knee joint.
Data Collection and Statistical Methods
The collected data underwent variable transformation, coding and were entered into Microsoft Excel. Statistical analysis was conducted using SPSS-PC-25. The normality of different parameters was assessed using the Shapiro–Wilk test. Mean ± standard deviation was used to present quantitative data. Group mean differences were analysed using Student’s t-test. Frequency distributions represented qualitative data, and differences in proportions were assessed using either the chi-square test or Fisher’s exact test. Statistical significance was defined as a P value less than .05.
Findings
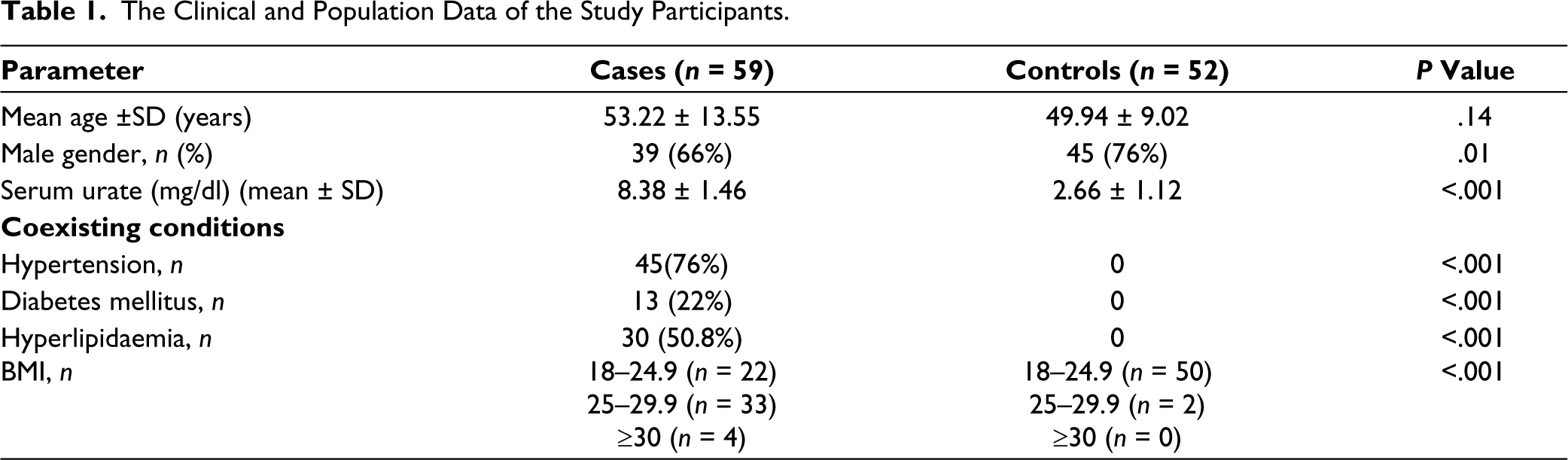
The study included 59 individuals with hyperuricemia (mean age 53.22 years) and 52 controls (mean age 49.94 years). Table 1 presents detailed demographic and clinical characteristics of the participants. Importantly, hyperuricemic patients showed a higher prevalence of diseases linked to metabolic syndrome compared to those with normal uric acid individuals.
The Clinical and Population Data of the Study Participants.
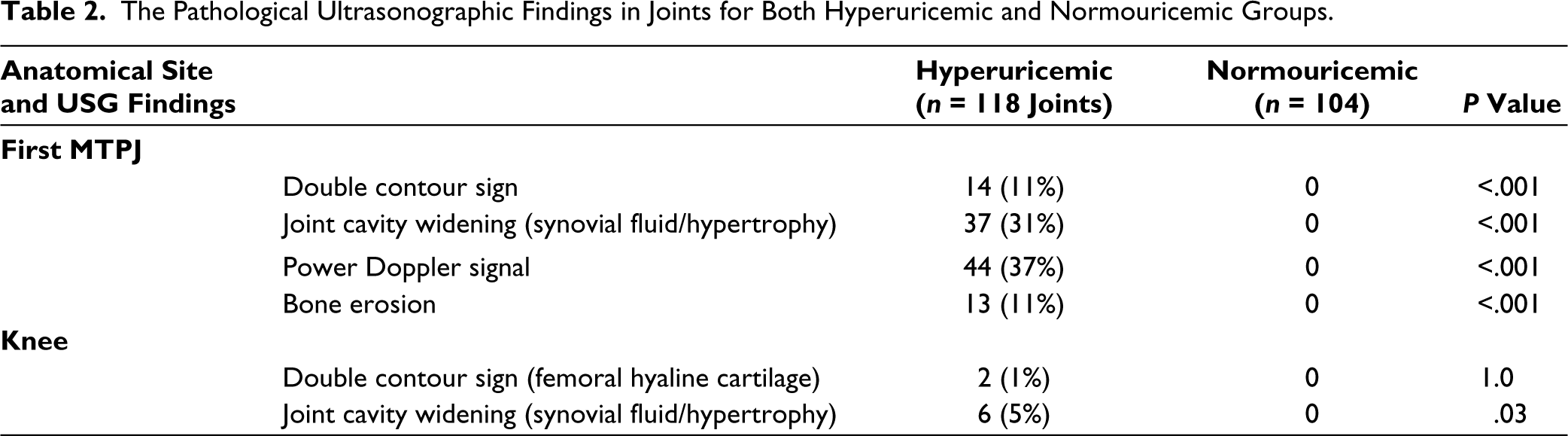
The study examined a total of 118 knees, 118 ankles and 118 first MTPJs in hyperuricemic patients and 104 (knee, ankle and first MTPJ) in the comparison group. Joint cavity widening was observed in 6 (5%) of 118 knees from hyperuricemic patients, but none (0; P = 0.03) in healthy controls. In hyperuricemic patients, articular space widening in the first MTPJ was observed in 31%, whereas none of the comparison group showed this condition (P < .001). The DCS was detected on femoral hyaline cartilage in 2 (1%) of 118 knees from hyperuricemic patients but was absent in the control group (P = 1.0). Hyperuricemic patients exhibited a higher prevalence of the double-line appearance (DCS) in the first MTPJs (11% vs. 0%; P < .001). Power Doppler signal was present in the first MTPJ of 37% of hyperuricemic individuals, whereas no such activity was found in the reference group (P < .001) (Table 2). Tendon examinations revealed that posterior tibialis tenosynovitis (6% vs. 0; P < .01) and Achilles enthesopathy (8% vs. 0; P < .001) were more prevalent among hyperuricemic patients compared to comparison group (Table 3).
The Pathological Ultrasonographic Findings in Joints for Both Hyperuricemic and Normouricemic Groups.
The Pathological Tendon Abnormalities Identified Through Ultrasound for both Groups.

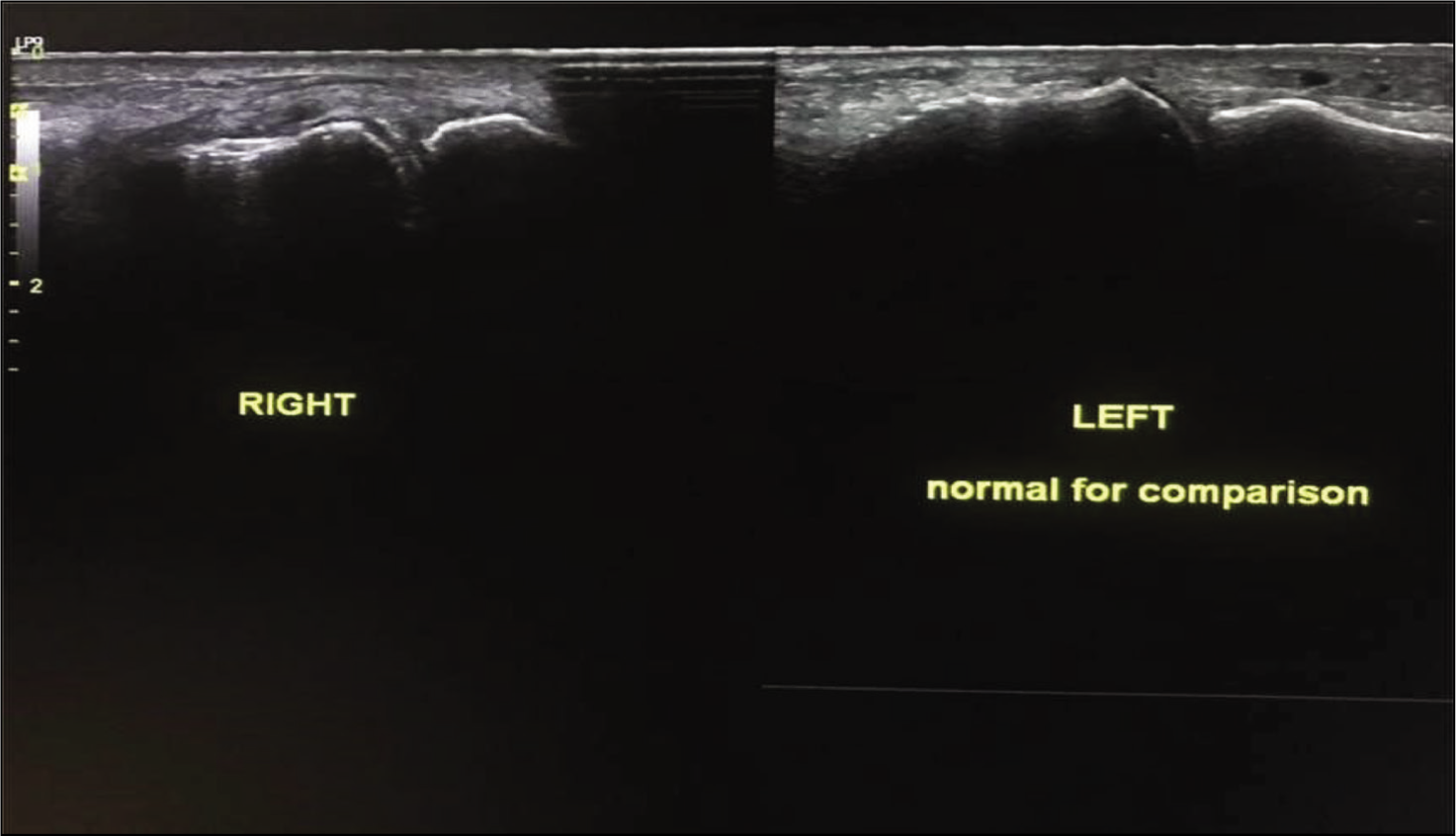
Grey Scale Ultrasound Showing Synovial Thickening with Tiny Specks of Refractile Calcifications Around First MTPJ. An Echogenic Line Parallel to the Articular Surface is also Noted Suggesting a Double Contour Sign.
Discussion
This study aimed to illustrate the broad range of subclinical structural changes in asymptomatic hyperuricemic individuals. Our findings provide evidence that hyperuricemia can cause morphostructural changes indicative of gouty arthritis in both joints and surrounding tissues, even in those without symptoms.
Musculoskeletal US is a highly effective imaging technique for confirming early morphostructural damage. US has found to be effective in detecting subtle inflammation in joints and tendons among patients with inflammatory conditions such as rheumatoid arthritis,15,16 psoriasis
17
and Sjögren’s syndrome.
18
This prompted us to investigate its capacity to detect involvement of the hyaline cartilage, tendons and joints in individuals with AH who show no signs of inflammation or musculoskeletal complaints. In our study, the prevalence of subclinical joint activity in the US in AH patients was 61%. The prevalence of subclinical joint activity in AH patients varies between 34% and 64% in studies conducted earlier.14,15,19,20–22 The variation may be due to various geographical, ethnic and other factors like US assessing parameters utilised for diagnosing the same. The prevalence of subclinical joint activity was
Conclusion
The findings endorse musculoskeletal US as a valuable, non-invasive method for detecting structural damage in the hyaline cartilage, synovial tissue and tendons of individuals with hyperuricemia who do not exhibit symptoms. There is limited evidence concerning the outcomes of patients with clinical AH who have MSU crystal deposition identified through imaging. ULT is not currently recommended for these conditions and remains a subject of ongoing debate. Further research is required to document resolution of crystal deposition in joints following ULT, which would support the formulation of guidelines emphasising the treatment necessary for AH.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The ethical approval was obtained from the Institutional Ethics Committee, SKIMS Medical College, Bemina, Srinagar, India.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
