Abstract
Vasculitis is a group of heterogeneous conditions characterised by inflammation of blood vessels. Based on the predominant size of the affected vessel, vasculitis is classified into small, medium, and large vessel vasculitis (LVV). Giant cell arteritis (GCA) and Takayasu’s arteritis (TAK) are the two major types of LVV. GCA was thought to affect only the external carotid artery and its branches, whereas, TAK was thought to affect the aorta and its major branches. Advances in imaging techniques have led to a better understanding of these conditions. It has become increasingly clear that GCA can involve the aorta and its branches. GCA and TAK have overlapping features like propensity to affect the aorta and its branches, similar histopathology, and similar clinical features. However, there are differences in age, geographic location, genetics, and predominant vascular territory involvement. There are subtle differences in clinical features. Are these conditions different ends of the same spectrum or different conditions with similar features? This review compares and contrasts these two conditions based on epidemiology, genetics, pathology, predominant vascular territory involvement, and any differential response to treatment. Based on current evidence, GCA and TAK seem to be distinct conditions and not different ends of the same spectrum.
Keywords
Introduction
Vasculitis comprises a heterogeneous group of disorders. Vasculitis is classified into small, medium, or large vessel vasculitis (LVV) on the basis of the size of the predominant vessel affected. 1 LVV is defined as ‘vasculitis that affects large arteries more often than do other vasculitides’. 1 In adults, giant cell arteritis (GCA) and Takayasu’s arteritis (TAK) are the predominant types of LVVs. 1 GCA and TAK are granulomatous vasculitis with similarities and differences in their geographic location, ethnicity, clinical features, histopathology, and imaging features. One of the most important differentiating features between these two conditions is the age of onset. For example, if a person presents with LVV predominantly involving the aorta and its branches, will the diagnosis differ based on age, ethnicity, and geographic location? Would an age <50 years and an Asian ethnicity make a diagnosis of TAK more likely? Conversely, would an age > 60 years and a European ethnicity change the diagnosis to GCA with predominant large vessel involvement? Are these two conditions different or the same but at different ends of the same spectrum?
In this review, based on the current evidence, we have tried to summarise the similarities and differences between GCA and TAK based on epidemiology, genetics, histopathology, predominant vascular territory involvement, and differential response in treatment, if any.
1. Classification of LVV
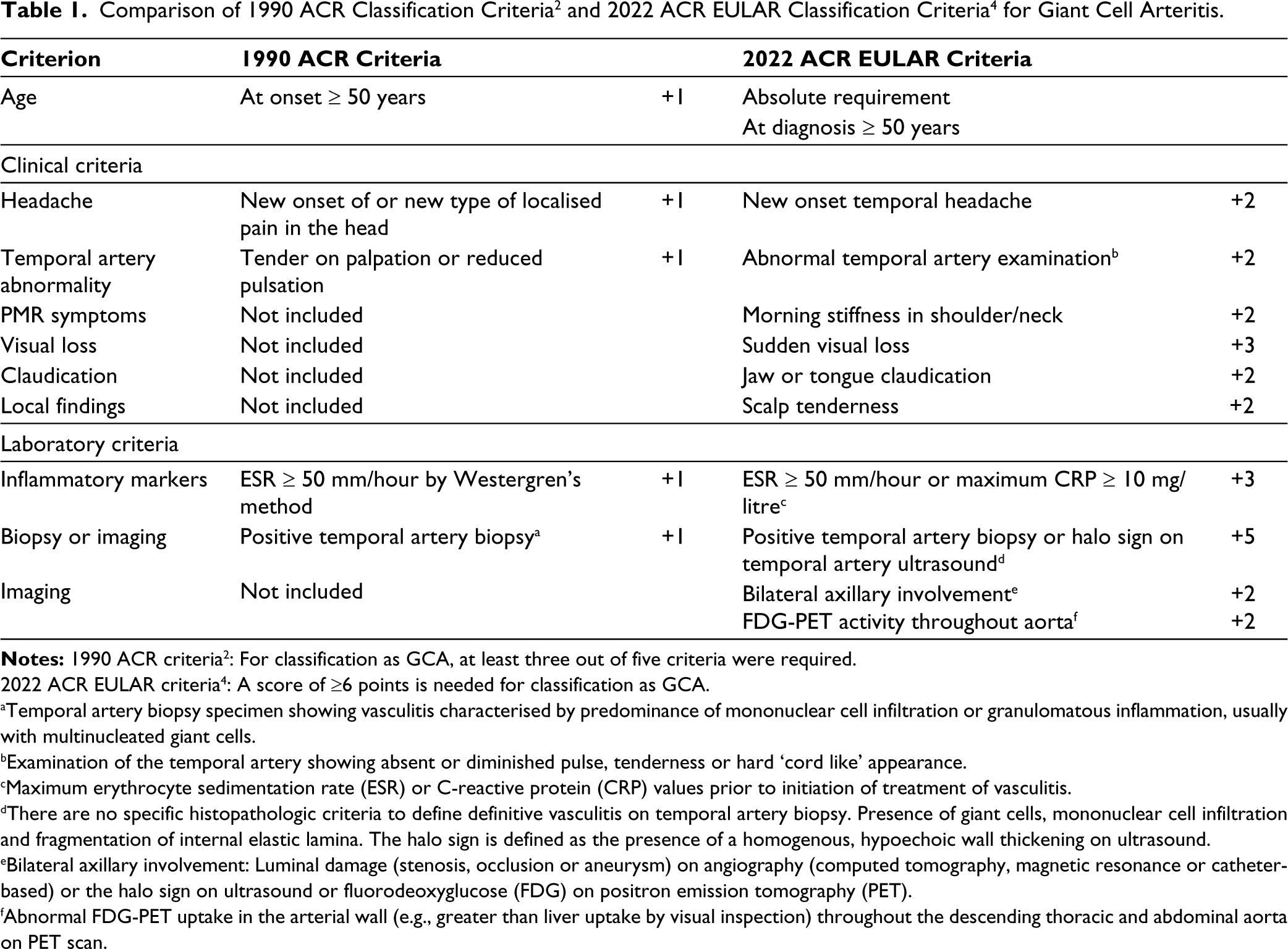
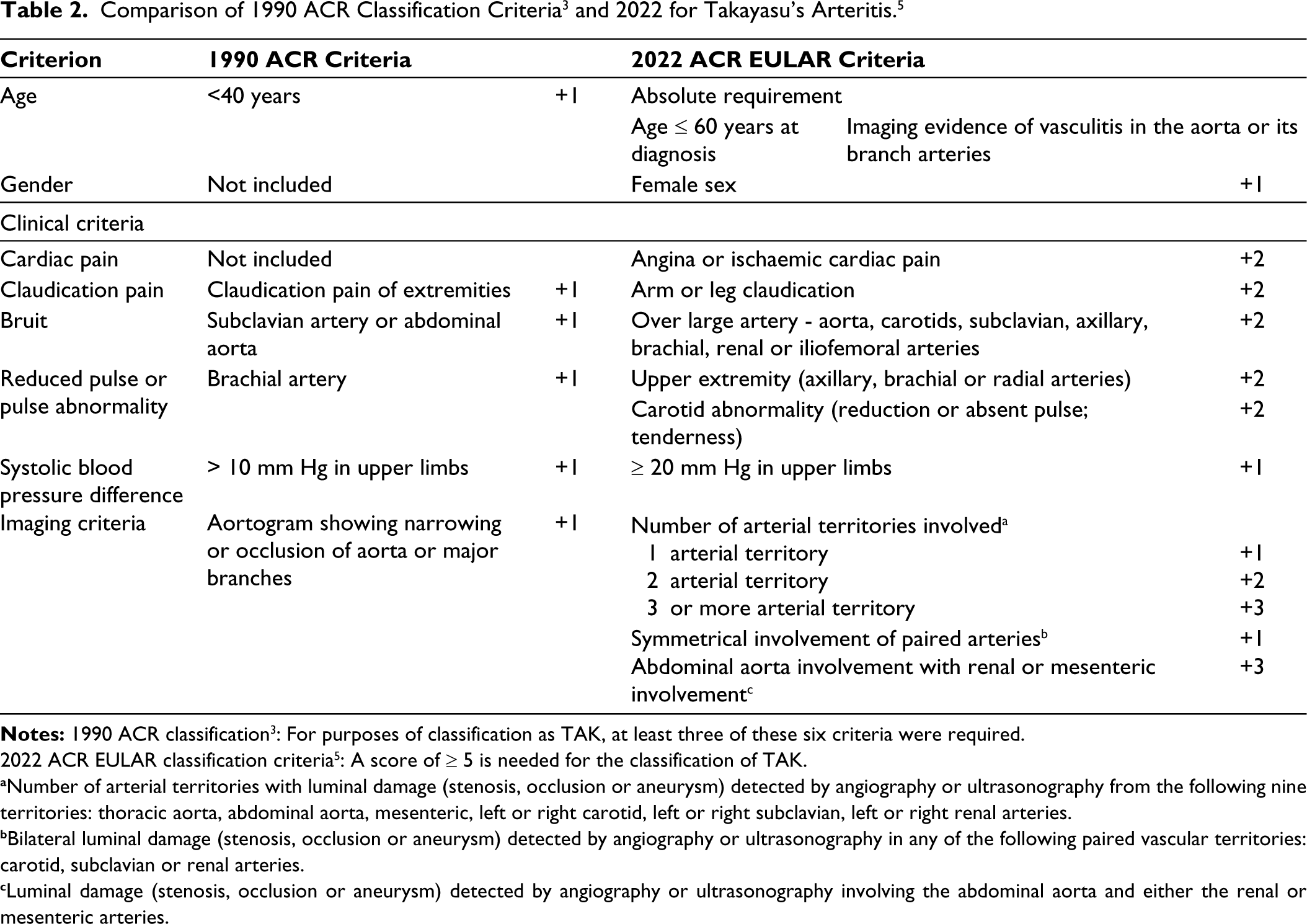
Classification of vasculitis is a challenging and evolving field. The 1990 American College of Rheumatology (ACR) classification criteria2,3 was one of the first systematic efforts in this direction. It had limitations like small numbers, limited geo-ethnic representation, and underrepresentation of some forms of vasculitis. The purpose of ‘Diagnostic and Classification Criteria for Vasculitis’ (DCVAS) project4,5 was to address the shortcomings associated with the 1990 ACR classification criteria. The recent 2022 ACR/European Alliance of Associations for Rheumatology (EULAR) classification criteria for GCA and TAK are the result of this collaborative effort (DCVAS).
Classification of GCA
The 1990 ACR classification criteria for GCA 2 were developed before the availability of advanced non-invasive imaging techniques. The older criteria were more focused on the cranial manifestation of GCA. Their performance was suboptimal in the identification of large vessel involvement. It followed the ‘number of criteria’ rule wherein each criterion was given equal importance. The recent classification criteria developed and validated by the multi-national DCVAS group have addressed limitations associated with the previous criteria. It is a weighted scoring system that reflects the importance of specific items. 4 One of the main features of the new criteria over the older one is the inclusion of advanced imaging features and additional clinical features. 4 These changes have improved the sensitivity of the new criteria for classification for GCA as a whole and more so especially for detection of large vessel GCA (Table 1). The sensitivity and specificity of the newer criteria are 87% and 94.8%, respectively, when compared to older criteria (80.3% and 92.5%). The sensitivity of newer criteria for classification of large-vessel GCA is 55.7% which is better than the older criteria (37.1%). 4 On comparison with the older criteria (1990 ACR criteria), the new criteria is more sensitive in identification and classification of patients with the large-vessel GCA subtype. 4
2022 ACR EULAR criteria 4 : A score of ≥6 points is needed for classification as GCA.
aTemporal artery biopsy specimen showing vasculitis characterised by predominance of mononuclear cell infiltration or granulomatous inflammation, usually with multinucleated giant cells.
bExamination of the temporal artery showing absent or diminished pulse, tenderness or hard ‘cord like’ appearance.
cMaximum erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP) values prior to initiation of treatment of vasculitis.
dThere are no specific histopathologic criteria to define definitive vasculitis on temporal artery biopsy. Presence of giant cells, mononuclear cell infiltration and fragmentation of internal elastic lamina. The halo sign is defined as the presence of a homogenous, hypoechoic wall thickening on ultrasound.
eBilateral axillary involvement: Luminal damage (stenosis, occlusion or aneurysm) on angiography (computed tomography, magnetic resonance or catheter-based) or the halo sign on ultrasound or fluorodeoxyglucose (FDG) on positron emission tomography (PET).
fAbnormal FDG-PET uptake in the arterial wall (e.g., greater than liver uptake by visual inspection) throughout the descending thoracic and abdominal aorta on PET scan.
Classification of TAK
The 1990 ACR classification criteria 3 for TAK had limitations due to its small patient sample (n = 63) drawn solely from North America. This restricted the generalisability of the criteria and limited their usage in diverse populations around the world. 5 Similar to GCA, these criteria were developed prior to the availability of advanced non-invasive techniques. The recent 2022 ACR/EULAR classification criteria 5 by the DCVAS project have been developed from a much larger and diverse patient pool. Imaging evidence of vasculitis (aorta or in its branch arteries) is an absolute requirement. 5 The age limit has been increased (≤60 years) and, along with imaging evidence of vasculitis, is an absolute requirement. 5 There are other changes like the inclusion of additional clinical features and more details about the arterial territory involvement (number of territory involvement; symmetric involvement) (Table 2). It is also a weighted score designed to reflect the relative importance of specific items (Table 2). When compared to earlier criteria 3 these changes have improved the sensitivity of the new classification criteria 5 (93.8% vs. 84%).
2022 ACR EULAR classification criteria 5 : A score of ≥ 5 is needed for the classification of TAK.
aNumber of arterial territories with luminal damage (stenosis, occlusion or aneurysm) detected by angiography or ultrasonography from the following nine territories: thoracic aorta, abdominal aorta, mesenteric, left or right carotid, left or right subclavian, left or right renal arteries.
bBilateral luminal damage (stenosis, occlusion or aneurysm) detected by angiography or ultrasonography in any of the following paired vascular territories: carotid, subclavian or renal arteries.
cLuminal damage (stenosis, occlusion or aneurysm) detected by angiography or ultrasonography involving the abdominal aorta and either the renal or mesenteric arteries.
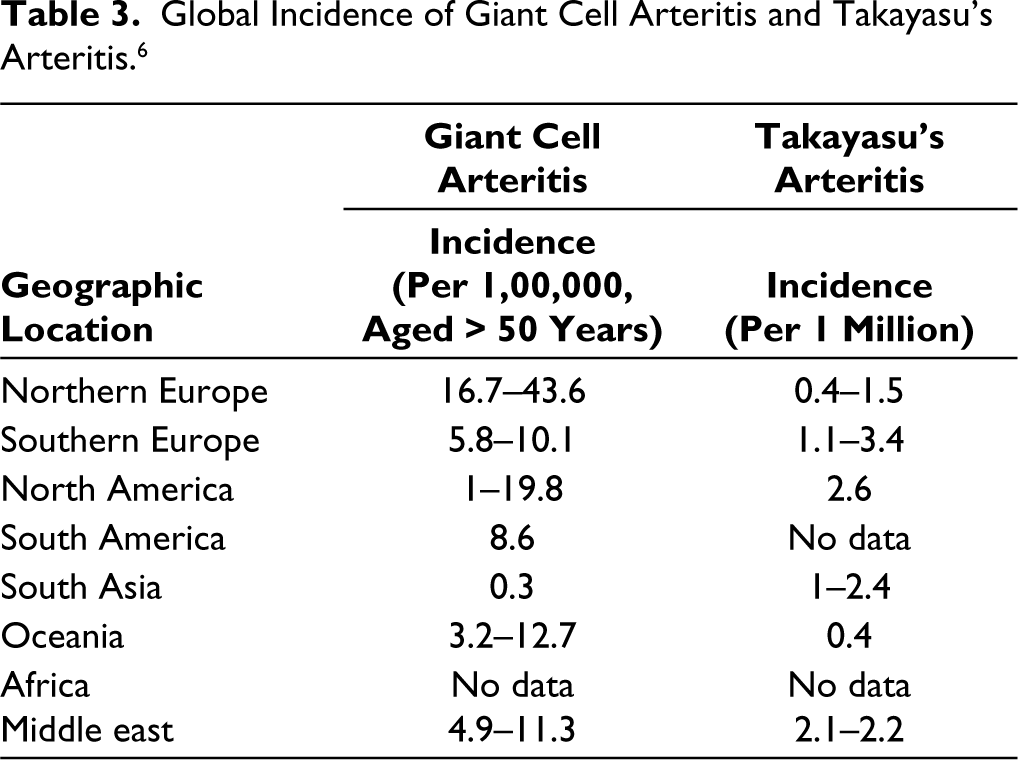
2. Epidemiology of LVV (Table 3)
Geographical location is an important risk factor for the occurrence of LVV, especially GCA.6,7 Latitude is known to influence the incidence of GCA. The incidence of GCA increases with an increase in latitude.6,7 Higher incidence rates are recorded in northern European countries and populations with Scandinavian ancestry.6,7 Lower incidence rates are noted in the Mediterranean and Asian countries.6,7 TAK is more common in the Asian population. Both are more common among females. The common age group for GCA is > 50 years and < 40 years for TAK.6,7 GCA is more common than TAK. However, this could be artefactual, and the epidemiological data should be interpreted with caution as most of the data are either from Europe or North America.6,7 There are inadequate data from Asia and Africa. Factors like differences in access to healthcare may also contribute to a lack of data, especially among low-income countries.6,7 The incidence rates for TAK may also be falsely low due to long asymptomatic periods resulting in diagnostic delay, which may make the incidence rates unreliable.6,7
Giant cell arteritis
In the DCVAS cohort, 4 the mean age was 72 years and <1% (7/942) had an age at diagnosis of <50 years. Large vessel GCA has a younger age at onset when compared to cranial GCA. However, even in these cases, the mean age at diagnosis is 70 years. 8 In a recent multicentre data9(S212) of 73 patients from India, the mean age at diagnosis was 68 years. Therefore, GCA seems to affect the older population and largely occurs in those aged > 50 years. With an increase in age, the incidence also increases. The peak incidence is observed in the 8th decade, who have a 44-fold increased risk of GCA.6,7 Females are more commonly affected when compared to males.6,7 The female-to-male ratio is 3:1. 7 There is marked variability in the global incidence of GCA, and it is more common in populations with northern European ancestry.6,7 Europe has higher incidence rates when compared to South Asia.6,7 Even in Europe, there is considerable variability with a north-south gradient, with higher incidence in Northern Europe.6,7 The annual incidence in ages >50 years per 100,000 in various populations is as follows: 44 cases in Northern Europe, <10 cases in the Mediterranean, and ~ 0.3 cases in Southern Asia (Table 3). 6 GCA is believed to be less common in Asia and Africa. However, the lack of epidemiological data from these populations is a confounding factor.
Takayasu’s arteritis
Age is a major differentiating factor between GCA and TAK. Age at onset is usually <40 years.6,7 However, a proportion of patients can have a late onset. In the DCVAS cohort, 5 approximately 24% had late onset (age at diagnosis > 40 years). It is a condition which is more common in females. The female-to-male ratio varies from 4:1 to 9:1.10,11 TAK is more common in Asia when compared to Europe or North America.6,7 A recent meta-analysis 12 estimated the pooled incidence of TAK to be 1.1 per million person-years. The data were predominantly from Europe, and there was only one study from Asia (South Korea). 12 There is a lack of epidemiological data from Asia and Africa (Table 3). Therefore, it is difficult to assess the incidence and prevalence rates accurately and compare them with those of GCA.
Global Incidence of Giant Cell Arteritis and Takayasu’s Arteritis. 6
3. Genetics
In the UK, GCA is less common in populations with Indian subcontinent ethnicity. 13 TAK is more common in Norwegian populations with African and Asian ancestry. 7 Populations in North America with Scandinavian ancestry have higher rates of GCA when compared to other populations (Hispanics, Asians and Afro-Americans).6,7 This geo-ethnic variability suggests a significant genetic contribution to the aetiology of LVV and also to differences between GCA and TAK.
GCA has an association with HLA class II and TAK has an association with HLA class I.6,7 The association of HLA class II with GCA has been known for a long time. However, the first large-scale genetic study in GCA was done in 2017. A multicentre genome-wide association study (GWAS) 14 which included >2000 individuals of European ancestry confirmed a strong HLA class II association. 14 This association may explain the presence of CD4+ T cells in the inflammatory lesions.7,15 The GWAS also identified risk polymorphisms in non-HLA regions: plasminogen (PLG) and an isoform of the α-subunit of collagen prolyl 4-hydroxylase (P4HA2). 14 Other genetic studies have described associations with other genes: tumour necrosis factor (TNF) and its receptors, IL17A, intercellular adhesion molecule 1 (ICAM1), vascular endothelial growth factor (VEGF), Toll-like receptor 4 (TLR4) and protein tyrosine phosphatase non-receptor type 22 (PTPN22). 6
TAK has been consistently associated with HLA class I compared to the class II association with GCA. 6 A multi-ancestral GWAS 16 confirmed the HLA class I association with TAK. The population was from different ethnic backgrounds and comprised individuals from five different populations (Turkish, Northern European descendant, Han Chinese, South Asian, and Italian inheritance). The strongest association was noted with HLA-B allele HLA-B*52:01. 16 This HLA class I association explains the predominant CD8+ T cell infiltrate in the inflammatory lesions of TAK.7,15 Four novel non-HLA loci associated with genetic susceptibility were also detected: SVEP1 (Sushi Von Willebrand factor type A, EGF and pentraxin domain containing 1), CFL2 (Cofilin 2), VPS8, and chr13q21. 16 It also confirmed the earlier known non-HLA associations: PTK2B, IL12B, and a locus on chr21q22. 16
The strong genetic difference between TAK and GCA was confirmed by an Immunochip genotyping multicentre study 17 which included patients from North America and Europe (Spain, Italy, and Turkey). The study included 1434 individuals affected by LVV (GCA = 997 and TAK = 437) and compared them to 3814 unaffected controls. The study confirmed the distinct HLA associations of GCA and TAK (GCA – HLA class II and TAK – HLA class I). 17 The only shared genetic risk was observed in the non-HLA region: IL12B. 17 Association of IL12B with TAK is well known. Along with its association with TAK, its association was described for the first time with GCA. 17 IL12B encodes the P40 subunit, which is shared between the interleukins IL-12 and IL-23. These cytokines are important in T cell differentiation into Th1 and Th17, respectively. Th-1 and Th-17 cells have important roles in the pathogenesis of LVV. 7 Ustekinumab is an IL12/23 blocking monoclonal antibody, and there are reports of its successful use in refractory LVV.18-21
4. Histopathologic Findings (GCA Aortitis vs. TAK Aortitis)
Though both are granulomatous vasculitis, there are subtle differences in the histopathology of aortitis in GCA and TAK (Figure 1).15,22,23
Comparison of Histopathology of Aortitis Between Giant Cell Arteritis (GCA) and Takayasu’s Arteritis (TAK).
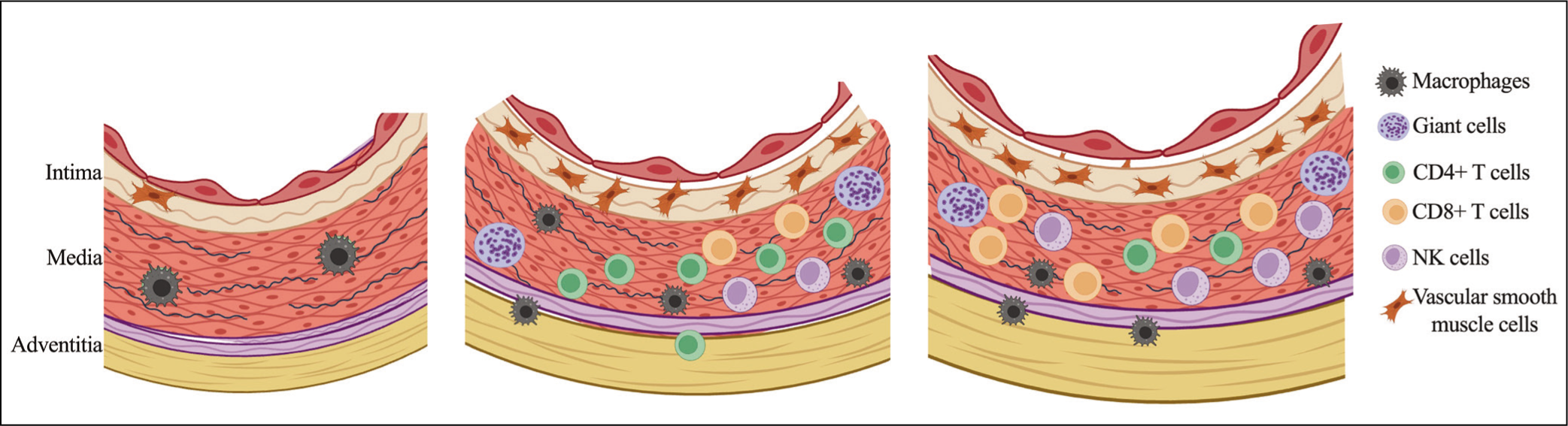
In both conditions, granulomatous inflammation is composed of inflammatory cell infiltrates and multinucleated giant cells. The primary location of this granulomatous inflammation is the media and the intima. 15 Loss of medial smooth muscle cells is one of the features which is common to both GCA and TAK. 15 This could be responsible for aneurysmal deformation and dilatation of the wall. 15
Features that differentiate TAK from GCA aortitis include the composition of the predominant T cell population of the inflammatory infiltrate, characteristics of the granuloma, and adventitial involvement. The predominant T cell population in GCA consists of CD4 T cells, and in TAK, CD8 T cells. 15 In TAK aortitis, the number of NK cells is higher. 15 These dissimilarities indicate different effector pathways. 15 The granulomas in TAK are compact and well-formed. 22 GCA primarily involves the intima and inner media more frequently. 15 TAK predominantly involves the outer media and adventitial tissue. 15 In TAK, there is marked adventitial thickening and, in late phases, severe adventitial scarring and resultant fibrosis.22,23 Adventitial thickening, fibrosis or scarring are usually not seen in GCA. 22
5. Clinical features
The clinical features of LVV depend on the predominant vessel or vascular territory involvement. In GCA, with a propensity for involvement of cranial branches of the external carotid artery, the predominant presenting feature is headache (70%). 10 Other clinical features include constitutional features (50%), PMR symptoms (40%), jaw claudication (33%), ocular symptoms (20%), and cerebrovascular accidents (stroke and TIA – 7%). 10 The common ocular features are amaurosis fugax, diplopia, and visual loss. 24 They are usually secondary to anterior ischaemic optic neuropathy. 24 Additional ocular features that can be observed are as follows: large peripapillary cotton wool spots, arterial occlusions (cilio-retinal artery or central artery), oculomotor cranial nerve palsy, and posterior ischaemic optic neuropathy. 24 Extracranial large vessel involvement is being increasingly observed in GCA and has been reported in 30%–83% of individuals. 8
In one of the earliest studies that looked at differences between GCA and TAK, GCA had higher levels of inflammatory markers. 25 At presentation, in GCA the mean erythrocyte sedimentation rate (ESR) and C-reactive protein level (CRP) were 83 mm/h and 10 mg/dL, respectively. In TAK, the ESR was 68 mm/h and CRP was 7 mg/dL. 25 Though both are inflammatory conditions, the levels of inflammatory markers are higher (ESR, CRP) in GCA when compared to TAK.25,26
The clinical features of TAK depend on the disease phase. In the pre-stenotic phase (inflammatory) the features include constitutional symptoms and vessel wall tenderness (carotidynia). 10 In the ischaemic/pulseless phase (mainly stenosis and aneurysms) the signs and symptoms include absent/unequal pulses, absent/unequal blood pressure, and features of ischaemia (depending on the affected arteries). 10 The clinical features include—constitutional (33%–65%), hypertension (40%–60%), carotidynia (4.4%–33%), and ocular (1%–8%). 10 Permanent visual loss is rare in TAK. 10 Ocular manifestations in TAK can be secondary to hypertensive arteriopathy, treatment-related cataracts, hypoperfusion secondary to cerebral ischaemia, and retinal microaneurysms. 10 Upper limb symptoms which include pulselessness, vascular bruits, blood pressure difference, fatigue, coldness, and numbness have been reported in 18%–94%. 10 Head and neck symptoms like dizziness, vertigo, syncope, headache, carotidynia, and jaw claudication have been reported in 6%–56%. 10
Features of arterial insufficiency like limb claudication, bruits, pulse anomalies, and blood pressure discrepancy are considered to be hallmarks of TAK. 27 Similarly, symptoms like headache are considered to be characteristic of GCA. 27 However, headaches have been reported in 9%–70% of individuals with TAK, and limb claudication has been noted in 15%–60% of individuals with GCA.11,25,26,28
Large vessel GCA
GCA has been classically described as arteritis affecting the branches of the external carotid artery (ECA). With advances in imaging techniques, it has been recognised that GCA is not just isolated involvement of branches of ECA; involvement of the aorta and its major branches has been increasingly described. This entity with the predominant involvement of the aorta and its branches has been labelled as large vessel GCA (LV-GCA). 29 The classical GCA with the predominant involvement of branches of ECA has been labelled as cranial GCA (C-GCA). 29 The frequency of LV-GCA was initially estimated to be 15%. 8 However, recent studies have reported rates of 29%–83%. 8 When compared to C-GCA, LV-GCA patients are younger and have lesser cardinal GCA manifestations. A recent meta-analysis 8 compared 3724 LV-GCA patients versus 2118 C-GCA patients. Patients with LV-GCA were younger (mean age – 69.8 years vs. 73.4 years). LV-GCA had significantly lesser classical cranial features of GCA like headache (49%), jaw claudication (23%), scalp tenderness (25%), and visual disturbance (14%). They were less likely to undergo temporal artery biopsy (TAB) and also less likely to have a positive result on TAB (OR 0.45). The CRP levels were similar. LV-GCA had similar rates of constitutional features like fever (34%) and PMR symptoms (37%). LV-GCA had higher rates of limb claudication (23%) and was 14.8 times more likely to have limb claudication symptoms. Distribution of vessel involvement showed higher rates of involvement of the thoracic aorta (73%), followed by the subclavian (58%), brachiocephalic trunk (58%), and axillary arteries (48%). LV-GCA also had a 3.6 times higher risk of developing an aortic aneurysm.
LV-GCA seems to be a distinct subset of GCA with a younger age at onset, lesser frequency of cranial GCA features, higher frequency of limb claudication symptoms, predilection to involve the aorta, and higher risk of aortic aneurysm. Though LV-GCA has a younger age at onset, some of the LV-GCA features that help to differentiate it from TAK are the higher age (69.8 years vs. usually <40 years), presence of PMR features (37%) and higher risk of aneurysms. 8
Late-onset TAK
In a proportion of patients with TAK, the age of onset is >40 years. The number of such patients varies from 13% to 43%. 27 How do we label these patients? Are these patients LV-GCA or late-onset TAK?
Literature on differences between late-onset (>40 years) and early-onset TAK (<40 years) is scarce. Studies from Japan, Italy and North America (multi-ethnic population) have specifically compared late-onset TAK with early-onset TAK.30-32 One of the largest studies to have looked at differences between late-onset and early-onset TAK is a nationwide registry 32 from Japan, and about 43.2% (N = 594/1372) patients had late-onset TAK. These studies have looked at differences between early-onset and late-onset TAK in terms of clinical features and predominant vascular territory involvement in angiography. The mean age at diagnosis in late-onset TAK varied from 42 to 60 years.30,31 There was no difference between the early-onset and late-onset TAK in clinical features or predominant vascular territory involvement in angiography.30-32 Symptoms of GCA, like cranial and PMR symptoms, were absent.30,31 Apart from age, early-onset and late-onset TAK seem to be similar to each other.30-32
The mean age at diagnosis in LV-GCA is approximately 70 years. 8 Even though TAK can have a late onset, these late-onset TAK patients are comparatively younger when compared to LV-GCA (42–60 years vs. 70 years). 32 Features differentiating late-onset TAK from LV-GCA include the absence of cardinal features of cranial GCA, the absence of PMR features, a younger age of onset, similar angiographic involvement as early-onset TAK, and predominantly angiographic findings of stenosis or occlusion, and lesser risk of aortic aneurysm formation.8,30-32 Therefore, late-onset TAK seems to be distinct from LV-GCA.
6. GCA versus TAK - Angiographic patterns and distribution
GCA was initially thought to affect only extracranial branches of the carotid artery and involvement of the aorta and its branches were considered to be classical of TAK. With improvements in imaging, the involvement of the aorta and its branches is being increasingly recognised in GCA. This brings into question whether there is any difference between TAK and GCA in terms of predominant vascular territory, inflammation versus damage, and type of lesion (stenosis/occlusion vs. aneurysm).
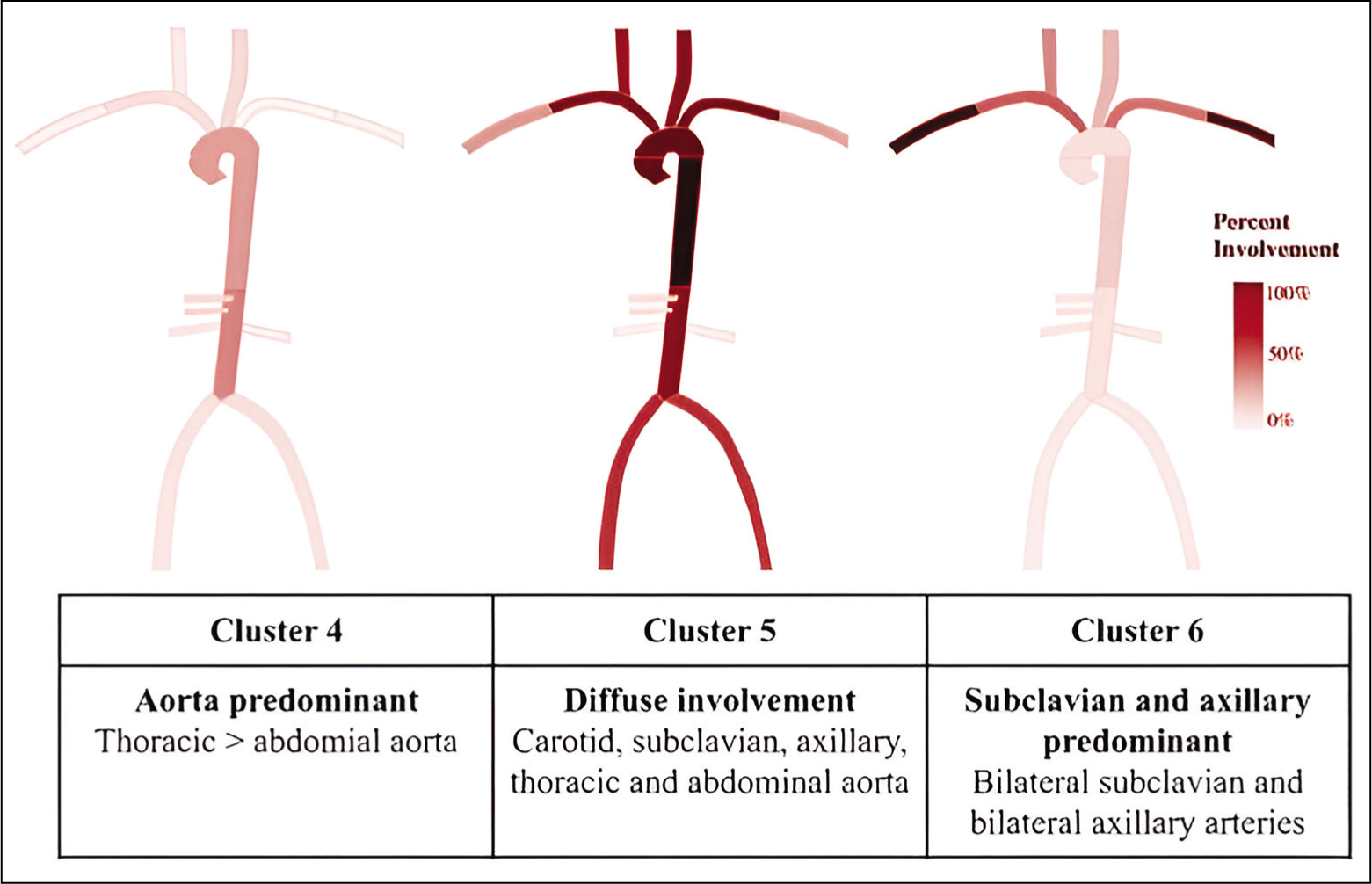
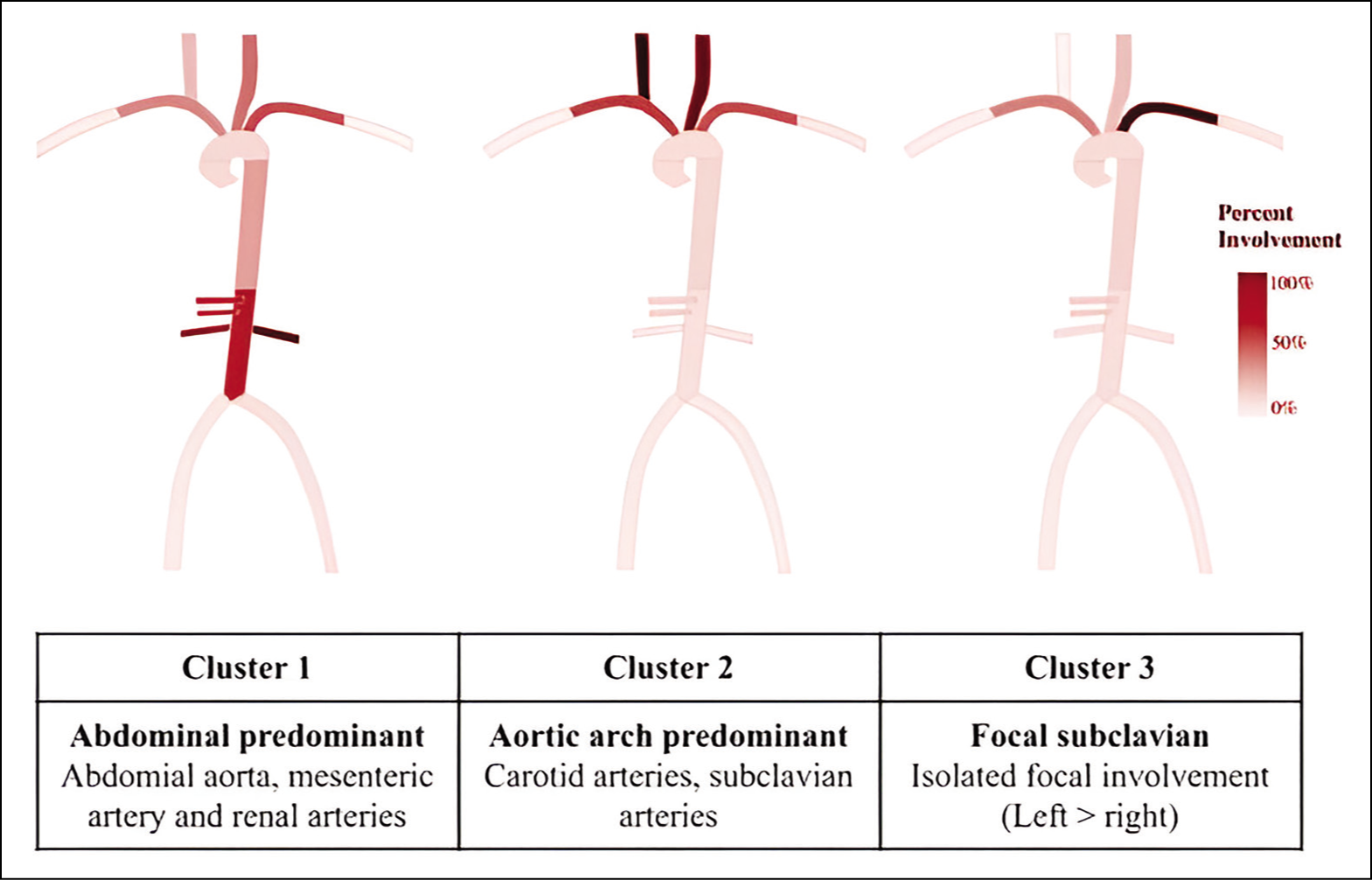
One of the earliest comparisons showed that TAK more commonly had carotid, subclavian, and iliac artery disease, while GCA had a greater frequency of axillary artery involvement. 26 Subsequently, based on predominant vascular territory involvement in imaging a cluster analysis revealed six distinct clusters. 33 The study group 33 included 1068 (TAK - 686; GCA - 382). The cluster analysis 33 included 11 arterial territories: right and left carotid, subclavian, axillary, and renal arteries; mesenteric arteries; and descending and abdominal aorta. Clusters 1, 2, and 3 were associated with TAK, whereas, Clusters 3, 4, and 5 were associated with GCA (Figures 2 and 3). One of the important outcomes of this cluster analysis is the identification of ‘diffuse involvement throughout the aorta and the aortic arch branch vessels’ in GCA (Cluster 4). Comprehensive information on imaging was available in 289 of these patients. When compared to GCA, TAK had more arterial damage without inflammation, whereas GCA had more vascular inflammation without arterial damage. In a large study, 34 that compared 581 Indian patients with 225 TAK patients from North America, similar results were replicated. Based on predominant angiographic patterns, TAK patients could be grouped under three clusters: abdominal predominant, aortic arch predominant, and focal disease. 34 These clusters were similar to Clusters 1, 2, and 3. 33
Length and shape of stenotic lesions especially in the subclavian and carotid arteries can also help to differentiate between GCA and TAK. A retrospective analysis 26 showed that TAK had more stenotic lesions (96% vs. 50%). They also looked at the length and shape of stenotic lesions in the subclavian and carotid arteries. The stenotic lesions were grouped as short (<5 cm), medium (5–10 cm), and long (>10 cm). 26 Depending of the characteristics of the shape of stenotic lesions, they were grouped as tapered stenosis, non-tapered stenosis, and complete occlusion. 26 Long tapered-type stenotic lesions were more common in GCA (GCA - 73% vs. TAK - 6%). 26 Whereas, short non-tapered-type stenotic lesions were common in TAK (TAK - 69% vs. GCA - 9%). 26
Though both GCA and TAK are LVV affecting the aortic and its branches there are differences in the pattern of predominant vascular territory involvement, morphology of stenotic lesions (in subclavian and carotid arteries), and the predominance of either vascular inflammation or damage.
A detailed review of imaging in LVVs is beyond the scope of this article. Readers can refer to recent guidelines to better understand the role of imaging in LVVs. 35
7. Treatment
In treatment, the main feature that highlights the differences between GCA and TAK is the differential response to biological therapies. This differential response has been reported with tumour necrosis factor inhibitors (TNFi) and Abatacept (ABT). Treatment with IL-6 receptor antagonist Tocilizumab (TCZ) is effective in GCA and likely to be beneficial in TAK. However, some of these results may need to be interpreted in the context that the evidence base for TAK is less robust than that of GCA.
There are no randomised control trials (RCTs) that have looked at the role of TNFi in TAK. There are approximately 19 observational studies that have looked at treatment with TNFi, and have been summarised in a recent meta-analysis. 36 Considerable heterogeneity was observed across studies. TNFi seems to be effective in the treatment of TAK. Treatment with TNFi was effective in attaining partial clinical response, stabilisation of angiographic findings, decrement in levels of inflammatory markers, betterment in PET CT scores, and reduction in the dose of glucocorticoids. 36 Relapses were noted in 32%. 36 In contrast to TAK, the use of TNFi in GCA is not effective. There have been three RCTs that have looked at the role of TNFi in GCA, and TNFi was found to be ineffective.37-39
ABT exerts its anti-inflammatory effect by blocking co-stimulatory signals to T lymphocytes. T cells have an important role in the pathogenesis of TAK and GCA. ABT was studied in a randomised, multicentre, placebo-controlled withdrawal study in patients with active TAK and GCA.40,41 It was not found to be effective in TAK. At 12 months there was no difference in the primary endpoint of relapse-free survival or in the median time to first relapse. 40 In contrast to TAK, the use of ABT in GCA has had promising results. At 12 months, ABT group had higher relapse-free survival and a significantly higher median duration of remission. 41
IL-6 is an important mediator in the T cell differentiation to the Th17 subset. Th17 cells are important in the pathogenesis of both of these conditions. In GCA, TCZ, an IL6 receptor inhibitor, is effective in inducing remission and in reducing the glucocorticoid dose. GiACTA was a phase 3 RCT 42 that compared TCZ with placebo in the treatment of GCA. At 52 weeks, TCZ was superior to placebo. When compared to placebo, the TCZ group had higher rates of sustained glucocorticoid-free remission and had an overall reduced cumulative dose of glucocorticoids. 42 In the open-label extension of this trial, at 3 years, a higher number of patients who received TCZ patients were in sustained treatment-free remission. 43 There is only one RCT 44 that has looked at the role of TCZ in TAK (TCZ 18 pts and Placebo 18 pts) and its open-label extension study 45 (36 pts). The RCT and about 19 observational studies have been summarised in a recent meta-analysis. 36 The RCT did not reach its primary endpoint but was favourable towards TCZ. 44 Patients on TCZ had a longer time to relapse and a higher relapse-free rate. 44 In the open-label extension, 45 at 96 weeks, TCZ was associated with a significant reduction in prednisolone dose and better quality of life. The pooled data from observational studies has shown treatment with TCZ was effective in attaining partial clinical response, stabilisation of angiographic findings, decrement in levels of inflammatory markers, betterment in PET CT scores, and reduction in the dose of glucocorticoids. 36 Relapses were observed in 26%. 36 However, considerable heterogeneity was observed across studies. Overall, these results are encouraging for the use of TCZ in TAK. However, there is a need for RCTs with a larger sample size which includes individuals across ethnicities and geographic locations.
A detailed review of the treatment or pharmacotherapy of LVV is beyond the scope of this article. Overall in LVVs, there is a paucity of high-quality literature regarding pharmacotherapy, especially regarding the use of non-biologics or conventional disease-modifying agents. There are only four RCTs in TAK, and out of these, two were for biological agents. Similarly, in GCA, there is a distinct lack of quality data with respect to the use of non-biologic agents. Readers can refer to recent reviews and treatment guidelines for more information regarding the pharmacotherapy of LVV.36,46-48
8. Unmet needs and future directions
Some of the unmet needs in LVV are as below:
GCA
Lack of data about genetics and epidemiology (incidence/prevalence studies) from Africa, Asia and South America. Lack of quality literature regarding the use of non-biologic agents. Challenges in the interpretation of temporal artery biopsy. Availability of experienced personnel for the interpretation of temporal artery doppler. TAK
Though TAK is more common in Asia compared to Europe or North America, there is a lack of data about genetics and epidemiology (incidence/prevalence studies) from Asia. Paucity of data from Africa. Better characterisation of late-onset TAK. Lack of quality literature regarding the use of non-biologic agents.
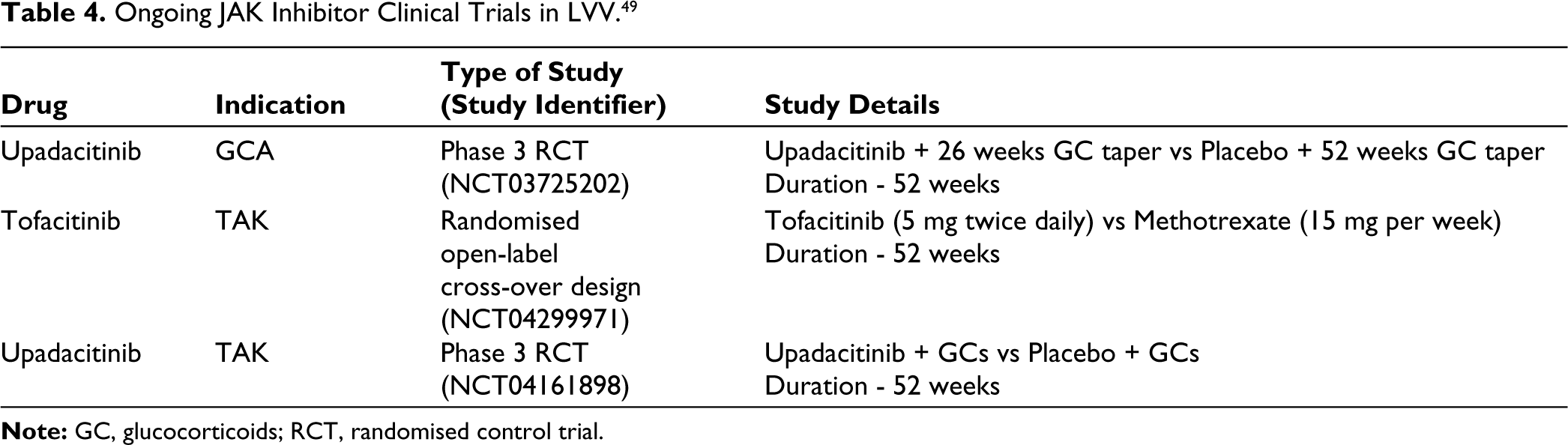
Other unmet needs common to both these conditions include the need for biomarkers and better outcome measures. Future directions should be aimed at addressing the unmet needs of LVVs. Treatment options with non-biologics are an area where ongoing work offers hope, especially in resource-poor settings. Table 4 lists ongoing trials which are looking at JAK inhibitors in LVV.
Conclusion
GCA and TAK have similarities and differences (Table 5). They have distinct age groups, genetic associations, epidemiology, clinical features, and differential responses to biological therapies. Challenges in classification may arise in individuals who present with LVV in the age group of 50–60 years. The distinct angiographic patterns may be helpful in such situations (GCA - Clusters 4, 5 or 6 and TAK - Clusters 1, 2 or 3). Apart from differences in the pattern of involvement of the arterial tree, patients with TAK are more likely to have vascular damage, and patients with GCA are more likely to have vascular inflammation without concomitant damage. Though both of them affect large vessels and share several similar features, based on current literature evidence, they are different conditions and not ends of the same spectrum.
Similarities and Differences Between Giant Cell Arteritis and Takayasu’s Arteritis.
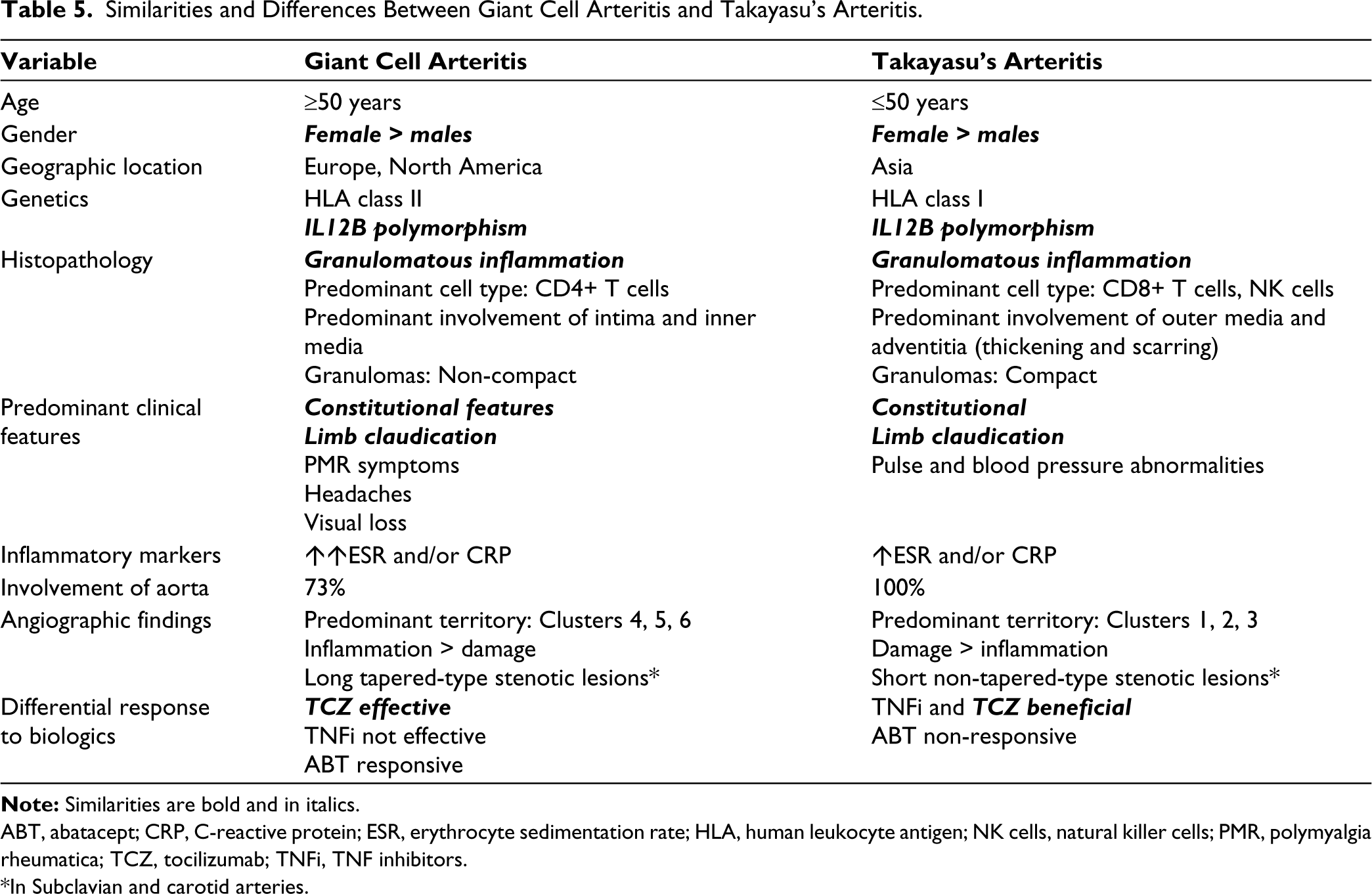
ABT, abatacept; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA, human leukocyte antigen; NK cells, natural killer cells; PMR, polymyalgia rheumatica; TCZ, tocilizumab; TNFi, TNF inhibitors.
*In Subclavian and carotid arteries.
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This review did not require ethical approval as it does not involve any new data collection from human or animal subjects.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
This review did not require patient consent as it does not involve any new data collection from human subjects.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.