Abstract
Introduction:
Majority of available protocols for isolation of chondrocytes from articular cartilage tissue rely on the enzymatic digestion of the tissue by collagenase type 2. The yield of chondrocytes in such protocols is low. Herein, we designed a novel indigenous sequential digestion by dual enzyme Pronase and Collagenase Type 1 for isolating human Chondrocytes from articular cartilage.
Methods:
Articular cartilage of Osteoarthritis (OA) patients undergoing total knee replacement were collected for the isolation of chondrocyte cells and subjected to sequential digestion by Pronase for three hours followed by Collagenase 1 overnight. Pellet of cells collected after digestion was plated on culture flask in 5% CO2 incubator.
Results:
From day three onwards, round to elongated cells adhered to the flask were visible which developed into elongated cell population of homogenous morphology, expressed Aggrecan (Agg), Collagen 2a (Col2a) and SRY-box transcription factor (Sox9) and had chondrogenic differentiation similar to a commercially available healthy chondrocyte. These cells were negative for Alizarin red stain, thus confirming the purity of chondrocytes.
Conclusion:
We have successfully established a sequential dual enzyme digestion-based culture technique for isolating the human chondrocytes from the articular cartilage biopsy derived from OA knee joints.
Introduction
Osteoarthritis (OA) is a degenerative joint condition that affects mobility and quality of life. This condition is primarily caused by cartilage injury. 1 Chondrocytes are the main type of cell that constitute the framework of cartilage and keeps it healthy. 2 Chondrocytes are the primary cells impacted during the pathophysiology of OA. Its shape and extracellular matrix (ECM) get altered during its pathogenesis, with Agg and Coll2a replaced by Col1a and ColX.3–7 For a better understanding of OA pathogenesis, behaviour and pathophysiologic alterations in chondrocytes biology need to be explored further. For this, one need primary chondrocyte cells cultured from OA cartilage.8, 9 Although a few protocols are already known for the isolation of chondrocytes from the cartilage, however, these are generally complex, require special culture media and growth factors, lengthy and are expansive.11–13 To overcome this problem and save time, commercially available chondrocyte cell lines are available which are widely utilised. Despite having a huge advantage of immense potential to grow, these cells loose polarity, key traits over time, are genetically unstable and their concordance with in vivo systems is low as compared to primary isolated chondrocyte cells.9, 14 Primary chondrocytes outperform commercial cell lines by being genetically stable, maintain normal healthy cell shape, and expression of surface markers as in in vivo situation.8, 9 Therefore, primary chondrocyte cells reflect in vivo situation more truly and act as better models for pathophysiology studies as compared to the commercially available cell lines. Consequently, there is a need to develop simple, reproducible, and cost-effective culture protocol of a primary chondrocytes that can be easily reproduced as growing number of in vitro, ex vivo and in vivo experiments are being performed in research labs worldwide.
Herein, we established the primary chondrocyte culture protocol, using two enzyme combinations, from OA cartilage biopsy tissue taken from the knee joint of OA patients who underwent total knee joint replacement.
Method
Preparation of Chondrocyte Dulbecco’s Modified Eagle Medium (DMEM) Media
Prepare the chondrocyte DMEM growth Medium by adding the following components to high-glucose DMEM (Sigma D1152-10X1L): 12% foetal bovine serum (FCS) (Heat inactivated, Cat No. 10270-106, Gibco), 1% non-essential amino-acid (Sigma, Cat No.M7145), 30–40 µg ascorbic acid (Sigma, A4403-100 G, SLCF6711, always add freshly prepared), 1% Anti-mycotic antibiotics (Sigma, 100×, Cat No. A5955), 2.0 g/L Sodium bicarbonate (Sigma, Cat No. S8875), and 0.11 g/L sodium pyruvate (Sigma, Cat No. P5280). All components were added in sterile conditions and filtered with 0.2 µm Syringe filters (Sartorius, cat no. 16534-k). The pH of the media was less than 7.5.
Preparation of Enzyme Solutions
The Pronase (derived from Streptomyces griseus CAS 9036-06-0, 33902221, merk) and collagenase Type I (prepared from CIostridium histolyticum, USA, 17100017, Gibco) were prepared in the incomplete chondrocyte DMEM with a concentration of 2 mg/ml each for Pronase and collagenase type I. Both the solutions were filtered through a 0.2 µm syringe filter. Note always use freshly prepared solutions.
Collection of Human Subjects Cartilage Biopsy
Cartilage tissue from 10 patients fulfilling the ACR Criteria for OA knee,15, 16 age ≥ 50 years, and undergoing total knee joint replacement at the Radius Hospital, Gomti Nagar, Lucknow were obtained after written informed consent. Patients with infection, other rheumatological disorders, spondylitis, bleeding diathesis, and trauma were excluded.
Culture of OA Derived Primary Chondrocytes Cells (OPCs)
The cartilage biopsy samples were transported to the culture room aseptically in the 50 ml falcon tube containing the sterile normal saline at 4°C, immediately. The tissue samples were washed thrice with the normal saline containing 1× antibiotics to avoid contamination. The cartilage tissue was mechanically minced to around 2 mm × 2 mm size. Minced tissue was subjected to enzymatic digestion with Pronase (2 mg/ml) for three hours followed by collagenase Type I digestion (2 mg/ml) overnight at 37°C incubator containing the 5% CO2 in the incomplete chondrocyte DMEM. After the enzymatic digestion, cell suspension was thoroughly mixed 5–10 times with 10 ml syringe and then centrifuged at 1000 rpm for 10 minutes. Supernatant was carefully discarded and the pellets were gently pipetted out in fresh media and cells containing suspension were seeded into the 12% chondrocyte growth medium in 5% CO2 incubator at 37°C. Media was changed every 72 hours. After seven days ellipsoidal to polygonal cells adhered to the floor of the flask were visible. The photomicrographs were taken in an inverted Phase contrast microscope (Nikon).
Characterisation of Human OA Derived Primary Chondrocyte Cells (OPCs)
The phenotypic conformation of OPCs was performed using Immunocytochemistry (ICC) using the specific markers Aggrecan [(Agg), cat no: sc-33695, Abcam] and Collagen 2a [(Col2a), Cat no: AB761, 3229338] and cartilage-specific Alcian Blue staining methods.
Monolayer chondrocytes culture of OPCs were trypsinized with Tryplee Gibco (1×, Cat no: 12605-010) and 25,000 cells were seeded into the eight-well chamber slides [LAB-TEK→ Permanox Chamber Slides (Nalgene Nunc International, Naperville, IL, USA)]. For ICC, the cells were fixed with 4% Paraformaldehyde for 20 minutes and washed thrice with the 1× PBST. The cells were permeabilised with 0.01% triton in 1× PBS for 10 minutes at RT. The antigen retrieval was done for the Agg antibody specifically with an ABC enzyme (cat no: C3667, Sigma) for 180 minutes at 37°C. Then 3% H2O2 was added to each well for 10 minutes followed by 3% bovine serum albumin (BSA) for one hour. This was followed by overnight incubation with primary antibodies Agg (1:200) and coll2a (1:200) at 4°C followed by HRP-conjugated secondary antibody (1:2000) for 90 minutes at RT. Treatment with DAB for five minutes for the antibodies and haematoxylin for 30–45 seconds for the nuclear staining was done. Image analysis was carried out by using the compound Light microscope.
Chondrogenic Differentiation Staining
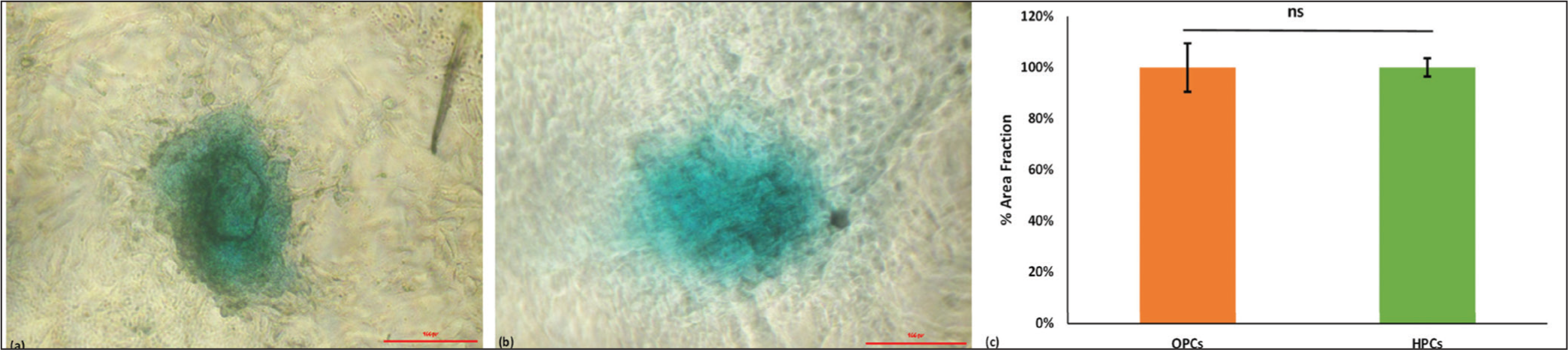
The cartilage-specific differentiation was confirmed using 1% Alcian blue staining in 96 well U-bottom plates as briefly described elsewhere.17–19 Approximately 2,00,000 cells were seeded per well in 96-U-Bottom plate and kept at 37°C incubator with a 5% CO2. The media was replaced every 3rd day until the spheroidal cells appeared at the bottom of the plate. Carefully, media was removed and washed with sterile 1× PBS. Thereafter, cells were fixed with 4% formaldehyde at RT for 30–60 minutes and washed with double distilled water (DDW). Freshly prepared 1% Alcian blue solution was added overnight, protected from light as described by the manufacturer’s manual. 18 After that solution was removed and washed with 0.1 N HCL thrice followed by DDW. The stained spheroid was visualised with a Carl Zeiss phase contrast microscope and the percentage of the relative intensity of stained area (area fraction) to the total area of the cells, using ImageJ, was determined.
Osteogenic Differentiation Staining
Osteogenic differentiation of cells was performed using the Alizarin Red-S staining. The Alizarin Red S Solution was prepared by dissolving the 2g Alizarin Red in 100 ml of DDH2O and pH adjusted to 4.1–4.3. 19 The solution was filtered through a 0.2 µm filter and stored at 4°C in the dark until use. Approximately, 50,000 cells were seeded onto the 12 well plates (Nunc). The cells were allowed to grow up to 80%–90% confluency. Media was aspirated and cells were fixed with ice-cold 70% ethanol for one hour at RT. The fixing solution was removed and rinsed with DDH2O thrice. The Alizarin Red solution (approx. 1000 µl) was added to each well and incubated at RT for 30 minutes. The cells were washed four times with DDH2O. The images of the stained cells were captured by a Carl Zeiss phase contrast microscope.
Expansion of Human Primary Chondrocytes Cell Line
HPC cell lines (Cat no: 12710) were purchased from promo cell and expanded in the chondrocyte’s growth medium (cat no: C-27101, Promo cell;) containing supplements with chondrocytes growth supplements (Cat no: C-39635 Promo cell). Both were mixed aseptically prior to use as described by the manufacturer.
Results
Patient Characteristics
Knee osteo-articular tissues were available from 10 OA patients (eight males and two females) undergoing total knee joint replacement. The mean age of patients was 63.1 years (range 57–71) and mean duration of OA of 12.4 years (range 7–19).
Morphological Characterisation of the OA-derived Chondrocytes
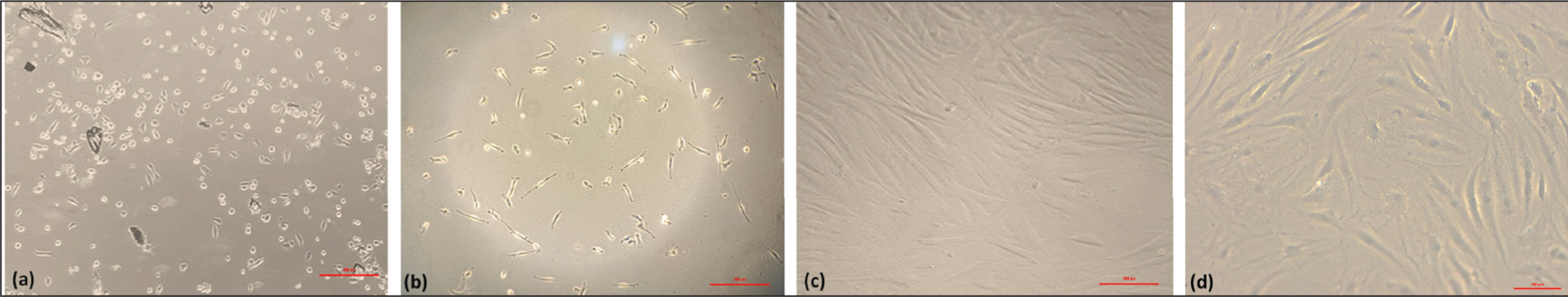
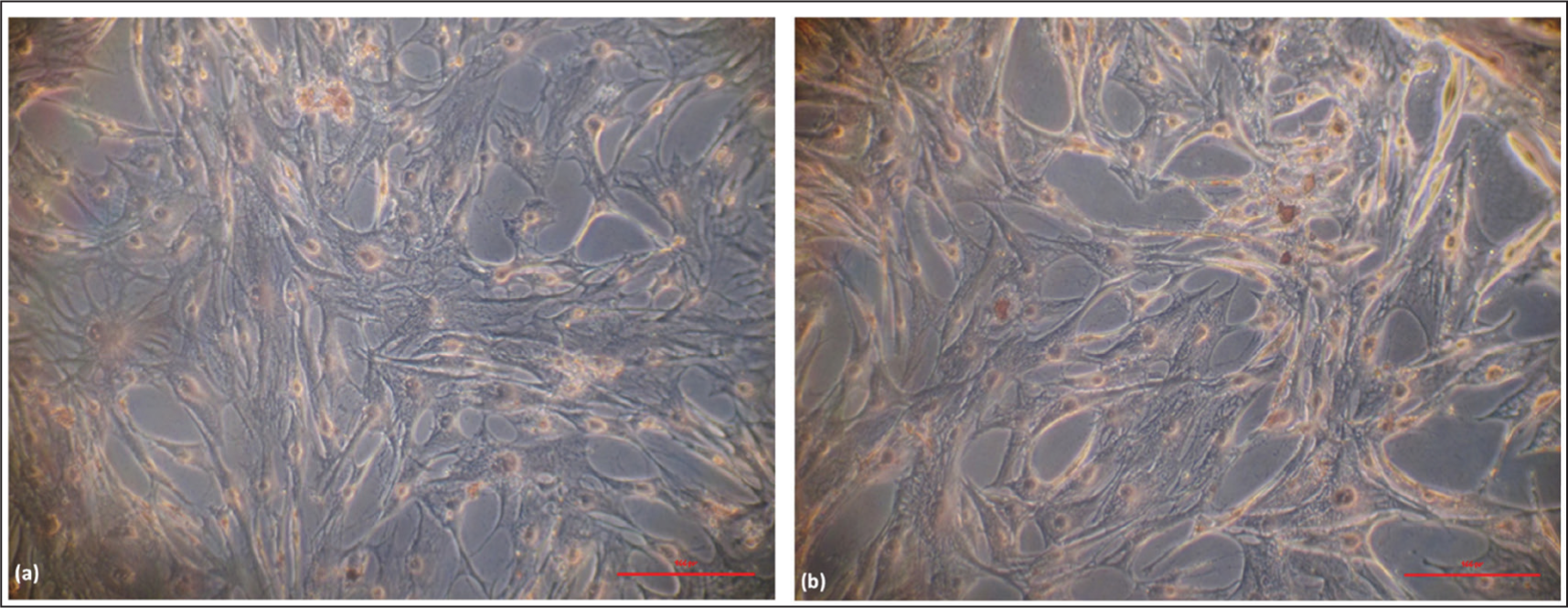
Cell viability of chondrocytes varied from 80% to >95% following isolation from the articular cartilage. The flasks were observed on the third day after the seeding, and a few cells started to take their own elongated shape (Figure 1a). After the media replacement, the number of cells increased, and cells started dividing (Figure 1b). The flasks become >90% confluent after 10–12 days of the seeding (Figure 1c). After the 1st passage, all grown chondrocytes exhibited a similar elongated morphology. The HPCs cell line was also expanded and compared with the OPCs cells, exhibiting a comparable morphology (Figure 1d).
Immunophenotypic Characterisation of the OA-derived Chondrocytes
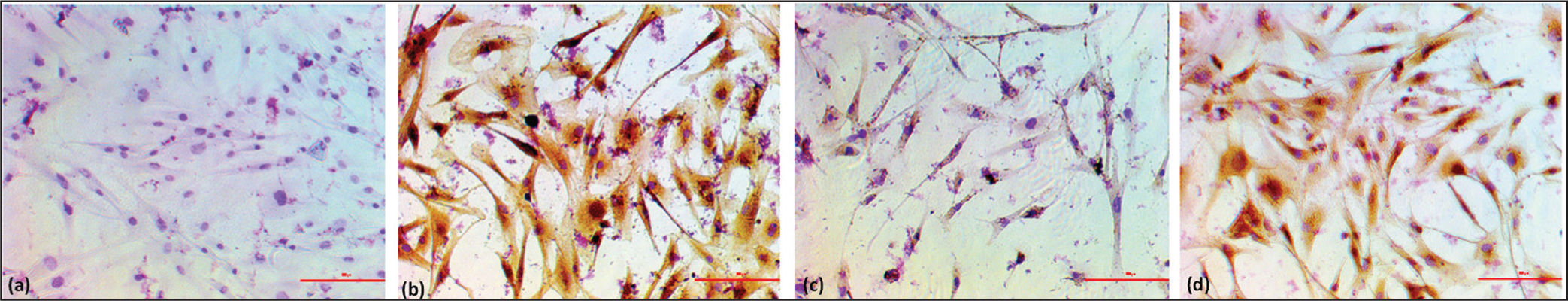
The Phenotypic marker characterisation via the ICC revealed that OPCs cells express Agg and Collagen2a, as shown in Figure 2a–c, confirmatory markers for the chondrocyte. The same expression we have observed with HPCs is shown in Figure 3a–c. The assessment of chondrogenic lineage differentiation through Alcian blue staining showed similar blue-stained positive cells and % of Area fraction (Figure 4a–c) in both types of cells, HPCs and OPCs.
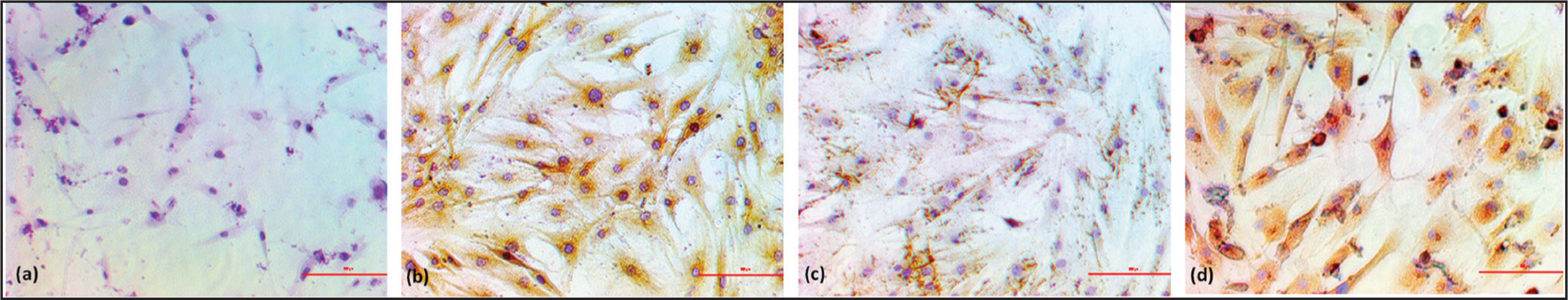
Sox9 is a key transcription factor (TF) expressed by the chondrocytes throughout their journey and acts as the safeguard for lineage fate.20–22 A high expression of Sox9 protein was observed in both HPCs and OPCs, thus confirming the healthy chondrocytes (Figure 2d).
Negative Alizarin Red staining (Figure 5a and b) ruled out osteogenic differentiation and confirmed these isolated cells to be purely chondrocyte cells.
Discussion
We have successfully established the novel indigenous technique, which is a feasible, reliable, and cost-effective and can be reproduced by any lab for research purposes. Our results indicate that cells isolated from cartilage tissue are chondrocytes as these cells have morphology like the commercially available HPCs, high expression of Agg, Collagen2 and Sox9 and negative staining for Alizarin Red.
Compared to available protocols for primary chondrocyte culture which utilises collagenase II,10–13 we have utilised two different enzymes for dissociation of cells from the osteochondral tissue. Articular cartilage is usually pauci-cellular, chondrocytes comprise 5%–10% of volume, and contains a large amount of matrix rich in collagen type II and sulphated proteoglycans. 24 Therefore, enzymatic digestion protocols that utilises digestion of ECM proteins by trypsin, Pronase, hyaluronidase or tosyllysylchloromethane are likely to yield a greater number of chondrocytes as compared to protocols not utilising these enzymes.23, 24 Jakob et al. studied various protocols (collagenase II, trypsin/EDTA, hyaluronidase, and tosyllysylchloromethane) for digestion of adult human articular cartilage and isolation of chondrocytes from them and reported that combination of collagenase II with trypsin or tosyllysylchloromethane accelerated the digestion process but did not significantly increase cell yield. 24 Digestion of articular cartilage with proteolytic enzymes may induce death of chondrocytes, affect cell growth, or alter the metabolic and functional properties of isolated chondrocytes, therefore, choice of proteolytic enzymes remains a critical step in isolation of chondrocytes from articular cartilage.24, 25 The rationale of Pronase used in the present study is its ability to dissolve ECM of proteoglycan rich glycosaminoglycans, found abundantly in the cartilage tissue. Pronase used in the present study is a mixture of 10 proteases derived from Streptomyces griseus. This loosens the chondrocytes and facilitates their isolation after further treatment with collagenase which digests the collagen II present in the ECM. Collagenase IA has more potent collagenase activity then collagenase II and has been reported to be more efficient in digestion of various human tissues including lung, brain and tumours. 26 Additionally, collagenase IA has been used for isolation of chondrocytes from bovine cartilage as well as ankle cartilage of a young patient. 27 In another study, Muhammad et al reported, chondrocyte yield to be higher with collagenase II as compared to Trypsin digestion of the articular cartilage. 28 Similar to the present study, Xiong and colleagues too have reported that sequential digestion of human articular cartilage tissue by Pronase followed by collagen IA yields better (increased number and better viability) chondrocytes population than the collagen II digestion alone. 23 In earlier studies, morphological phenotype of the chondrocytes has been reported to be well preserved till passage 2-3.23, 24 We monitored the morphology and phenotypic characteristics of isolated chondrocytes and did not observe any alteration in it until passage 5 (data not shown).
Our protocol for chondrocyte isolation was cost effective as it did not require addition of growth factors, cytokines, and insulin like growth factor-1 for expansion of the chondrocytes which would have escalated the cost of the protocol. 29 A two step sequential digestion protocol of articular cartilage followed by culture in a culture flask is easily reproducible in any basic culture lab and it did not require special equipment like orbital shaker.
We do not see any limitation to our protocol, however, correct concentration and duration of treatment of cartilage tissue with the digestion enzymes are crucial steps which need to be strictly adhered to otherwise yield of chondrocytes from the cartilage tissue may not be optimum. Both over and under treatment with digestion enzymes will affect both quantity, quality, and viability of chondrocytes. Since the present study was conducted on articular cartilage available during total knee joint replacement in elderly patients with long standing OA, we cannot generalise it to articular cartilage available from the either non-OA knee joints or cartilage tissue available from the younger individuals as both the cellular constituents and ECM contents are different.
Conclusion
We have established a sequential dual enzyme digestion-based simple, feasible, and cost-effective culture method for the isolation of chondrocytes from the articular cartilage biopsy derived from OA patients.
Footnotes
Acknowledgements
AKA acknowledges receipt of the DBT -JRF/SRF -programme fellowship from the Department of Biotechnology, Ministry of Science and Technology, Government of India. We acknowledge the support of Dr Sanjay Srivastava and staff of Radius Hospital, Gomti Nagar, Lucknow, for providing cartilage biopsy samples. We also acknowledge the support of Manali Jain from the Department of Haematology, SGPGIMS, Lucknow for providing the reagents. VA acknowledges Intramural Grant from SGPGIMS, Lucknow. Authors acknowledge the voluntary contribution of Akshat Agarwal and Harshita Pandey towards data acquisition, English language editing, and bibliography annotations in preparation of this manuscript.
Authors Contribution
The conception and design of the study: AKA, MKR, LG, VA. Acquisition of data, analysis, and interpretation of data: AKA, NN, LG, VA. Drafting the article: AKA, VA. Revising it critically for important intellectual content: VA, NN, LG, MKR. Final approval of the version to be submitted: AKA, MKR, NN, LG, VA.
Compliance with Ethical Standards
No part of this study, including graphics, tables and text, are copied or published elsewhere in any language. Study was approved by IEC, SGPGI. Approval number 2018-172-IMP-107, letter number PGI/BE/25/2019 dated 28/01/2019.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Disclaimer
No part of the research is copied or published elsewhere.
Funding
This study was funded by an Intramural grant SGPGIMS, Lucknow.
