Abstract
Background:
Many recent studies have suggested a changing paradigm of management of psoriatic disease. One of the main concerns is the lost opportunity of controlling ‘psoriatic disease’ during the early phase, especially when the disease is limited to the skin.
Objective:
The study’s main aim was to determine psoriasis (PsO) treatment before the patient’s presentation to the rheumatologist. We also studied the associated comorbidities in these patients.
Methods:
This was a cross-sectional study done at eight centres. Data was collected from 375 patients with psoriatic arthritis (PsA) about their demography, details of psoriatic lesions, treatment taken for them, additional co-morbidities and pattern of PsA.
Results:
Only 22.4% of patients had received oral methotrexate for PsO: the majority for less than a year. Plaque and scalp PsO were the most common, and 75% of patients had body surface area involvement greater than 5%. There was a significantly higher prevalence of type II diabetes mellitus (T2DM) (24% vs. 9.1%) and obesity as compared to the general population.
Conclusion:
Few patients received sustained systemic treatment for PsO prior to the development of arthritis. There was a significant association of co-morbidities such as T2DM and obesity with PsA.
Keywords
Introduction
Psoriatic arthritis (PsA) is a chronic, inflammatory arthritis associated with psoriasis (PsO). Estimates of PsA in patients with PsO vary from 20% to 30%, 1 with a relatively low prevalence (8.7%) reported among Indian patients presenting to dermatologists with PsO. 2 There is limited Indian data examining the factors affecting the progression of PsO to PsA.
Retrospective studies have suggested that early systemic treatment of PsO may provide a window of opportunity in preventing PsA. 3 In one such study, new onset PsA was seen in 12% of PsO patients treated with biologics, 9.6% of non-biologic systemically treated patients and 37.4% of topically or not treated patients (p < .001). In light of this, one of the main areas of this study was to examine the nature and duration of treatment received from dermatologists for PsO—local or systemic. We also studied the extent, site and type of PsO in these patients with PsA.
PsA is associated with many comorbidities. It is strongly associated with obesity. 4 Patients with PsA have a higher risk of developing type two diabetes mellitus (DM).5–7 The prevalence of metabolic syndrome in patients presenting with PsA at a tertiary care centre in Northern India was reported to be 58%. 8 A recent observational study has shown higher cardiovascular risk among patients with psoriatic spondyloarthropathy (SpA) compared to patients with non-psoriatic SpA. 9 A study among patients in a southern state of India has shown a higher prevalence of non-alcoholic fatty liver disease (NAFLD) among patients with PsO compared to controls. 10 The presence of NAFLD was significantly more in patients who had coexisting metabolic syndrome or DM.
With this background, we assessed the prevalence of obesity, DM, hypertension, dyslipidemia, coronary artery disease (CAD) and stroke in patients of PsA presenting to rheumatologists at multiple centres across Western India and its relation to the onset of PsO.
Methods and Design
Study Design
This was a cross-sectional, non-interventional study in which patients presenting with PsA to rheumatology outpatient departments were evaluated for past and current treatments of PsO, and cardiometabolic co-morbidities. Data collection was planned for 12 months.
Data Collection
Patients of age 18 years or more, presenting with inflammatory arthritis to multiple rheumatology centres in Mumbai and Nashik, Maharashtra were screened for PsA.
Patients who fulfilled the Classification Criteria for Psoriatic Arthritis. were included in the study. The diagnosis of PsO was based on the history of PsO previously diagnosed by a dermatologist or the presence of active psoriatic skin lesions confirmed by a rheumatologist or dermatologist. Patients with other forms of inflammatory arthritis were excluded from this study. Patients were enrolled from July 2021 up to March 2022. The target study sample size was achieved before the completion of a year. An ethics committee approval was taken at a tertiary care hospital in Mumbai (EC letter nos. 1389-20-₹, 1440-21-₹) for all centres. Informed consent was taken from all the patients included in the study.
The assessment of patients enrolled in the study was done according to a pre-determined ‘psoriasis proforma’. The proforma was discussed at length and agreed upon by all participating rheumatologists after several virtual meetings before the commencement of the study. The proforma is attached as a supplement.
Data was collected via online Google Forms, which ensured a standard collection of data across various centres. Data from Google Forms was captured in Excel sheets, which were accessible to the principal investigators and the statistician at this centre.
Demographic data, disease variables and treatment variables were recorded in the ‘psoriasis proforma’. This included age, gender, body mass index, disease duration of PsO and PsA, family history of PsO and PsA, site and maximum body surface area (BSA) ever involved, treatment received for PsO, type of PsA and history of smoking and alcohol consumption. Family history of up to second-degree relatives was collected. The presence and duration of type II DM (T2DM), hypertension, dyslipidemia, CAD, stroke and relevant treatments were determined. The American Diabetes Association criteria and the Japanese Atherosclerosis Association criteria were used respectively to diagnose previously undiagnosed diabetes and dyslipidemia.
Investigations were conducted as part of the standard of care by the treating rheumatologist, including a complete blood count, erythrocyte sedimentation rate, C-reactive protein, rheumatoid factor, liver function tests, serum creatinine, blood sugars (fasting and postprandial), glycated haemoglobin (HbA1c) and a lipid profile.
Study Size
The sample size was calculated based on the type of PsO, type of PsA, the extent of skin involvement, the prevalence of DM and the family history of Ps at a 95% confidence level and margin of error of 8%. Based on these, a minimum sample size of 140 patients was deemed necessary. Being a multi-centre study, it was planned to enrol 355 patients across all centres.
Statistical Analysis
The variables studied were type, site and extent of PsO, treatment received for PsO, type of PsA, DM, hypertension and dyslipidaemia. Data recording was done in MS Excel using Google Forms. Since the study is observational, descriptive statistics were used. Results for quantitative variables were expressed as mean (±SD), median (range) and IQR. Statistical analysis was done using the JASP statistics program.
Results
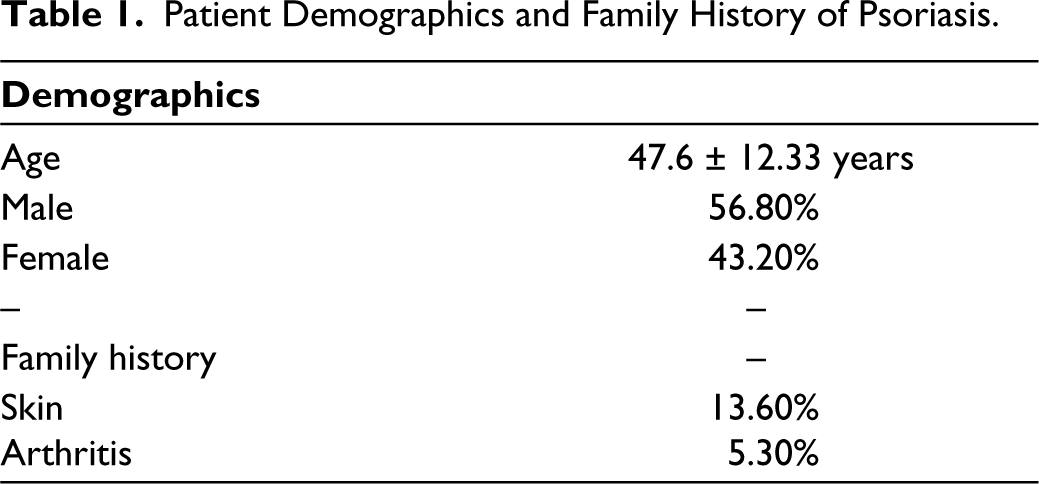
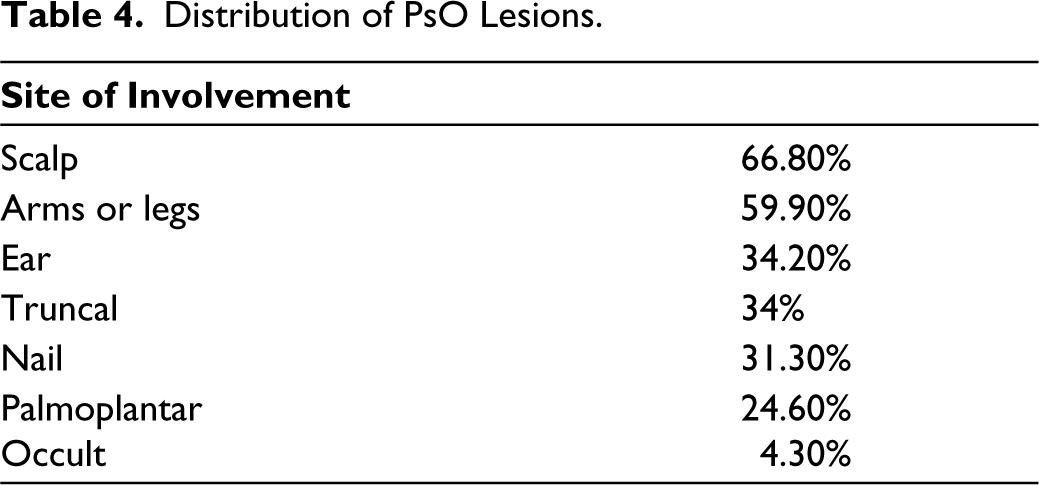
A total of 300 and 75 patients were enrolled in the study. Of these, 213 (56.8%) were males and the rest were females, with a ratio of 71:54. The mean age of patients was 47.6 ± 12.33 years (Table 1). The date of diagnosis of PsO by a dermatologist was considered as the onset of PsO. The duration of skin lesions ranged from 1 to 45 years (mean 10.25 years). Of the BSA involved, 32.1% of patients had >15% involvement, 25% had 5% and 43% had between 5% and 15% involvement (Table 2). The most common type of PsO observed was plaque PsO in 93.3% of the patients (n = 375), which was followed by guttate PsO in 5.6% (Table 3). Involvement of the scalp was seen in 66.8% of patients, followed by arms and legs in 59.9% and the trunk, ears and nails in 31.3% of patients (Table 4). Family history for PsO and PsA was negative in 86.4% of patients with PsO and in 94.7% of patients with PsA.
Patient Demographics and Family History of Psoriasis.
Maximum Body Surface Area Involved.

Type of Psoriasis.

Distribution of PsO Lesions.
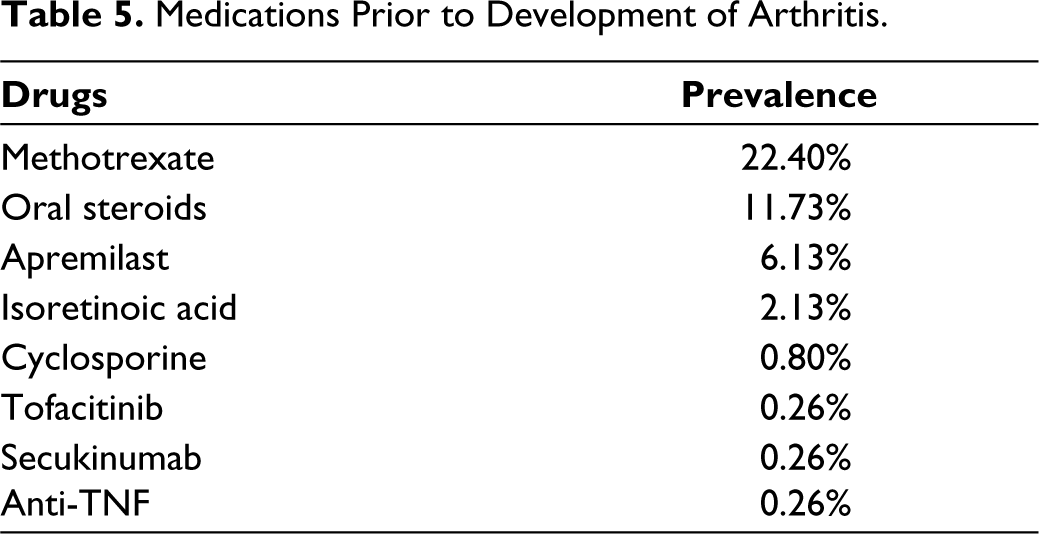
In this study, 77.6% of patients had never been prescribed DMARDs or biologics for skin manifestations before visiting a rheumatologist. A total of 22.4% of patients receiving methotrexate, received it for less than a year. Overall, 11.73% of the patients received corticosteroids, 6.13% got apremilast, and a handful of patients were given isoretinoic acid, cyclosporine, JAK inhibitors, IL-6 inhibitors and anti-TNF agents (Table 5).
Medications Prior to Development of Arthritis.
The date of diagnosis of arthritis was determined as the ‘Onset’ of PsA. Overall, 12.8% (n = 48) had the onset of arthritis before the onset of skin lesions, 76.3% (286) had it after developing skin lesions and 10.9% (41) had the simultaneous onset of both skin lesions and arthritis. The duration of PsA ranged from 6 months to 40 years, with a mean of 6.081 years. The mean duration between the onset of PsO and PsA was 6.4 years. Similar to other studies, PsO was considered to precede PsA if the interval between the two was more than a year or simultaneous if both occurred within a year of each other. 11
Among the patients, 4.3% were smokers; the majority had been smoking for four to five years. Overall, 8.3% of the patients had a history of alcohol consumption.
The prevalence of co-morbidities, namely, T2DM, dyslipidaemia, obesity, hypertension, cerebrovascular accidents (CVAs) and CAD was observed as mentioned in Table 6 and 7. Overall, 24% of the patients were diabetics. Overall, 3.2% of the patients who were previously undiagnosed were found to be diabetic as per ADA criteria. T2DM was diagnosed in 66.66% of people after the onset of PsO. The majority of them were diagnosed within five years of the onset of PsO. The diagnosis of T2DM prior to presentation to the rheumatologist was based on the patient’s history. Overall, 24.3% of the patients were hypertensive, of whom 71% were diagnosed after the onset of PsO, most commonly within the first five years of onset of PsO. Overall, 4.2% of the patients had CAD, of which 83.3% were diagnosed as CAD after the onset of PsO, most commonly within the first 15 years of onset of PsO. CAD was defined as the occurrence of myocardial infarction, unstable angina or need for revascularisation. Overall, 1.9% had a history of cerebral vascular accidents, of which 57.1% of patients developed cerebral vascular accidents after the onset of PsO. A CVA was defined as a transient ischemic attack or neurological deficit secondary to an ischemic or haemorrhagic event. Overall, 38.66% of the patients were diagnosed with dyslipidaemia at the time of enrolment, of whom 67.58% developed dyslipidaemia after PsO; 8.7% were previously undiagnosed.
Prevalence of Co-morbidities and Body Mass Index.


Overall, 36.8% of the patients had a body mass index of 25–29.9, followed by 33.1% who had a body mass index of 23–24.948; 16.3% of the patients had a body mass index of more than 30; and 13.9% of the patients had a body mass index of 18–22.9 (Table 4).
Discussion
Examining the relationship between the nature and extent of baseline skin lesions, initial therapy and the development of subsequent arthritis was one of the main aims of the study. In this study, PsO preceded PsA in most (76.3%) patients by an average of 6.4 years. This is similar to a recently reported review of 20 epidemiologic studies, the skin lesions were present on an average of up to 12 years before the development of joint problems in 84% of the patients. 1 Previous studies have shown that the severity of PsO is associated with the development of PsA.12–14 In our study, 78% of patients had >5% BSA involvement. However, the correlation between skin and joint severity may be low at a specific time. For example, it is not uncommon for a patient to have mild PsO and severe arthritis, and vice versa.
Most of our patients received only topical treatment before presenting with PsA. Very few patients received systemic therapy for PsO. In recent times, the PsA management paradigm has started to shift. Studies suggest that treating PsO effectively with systemic treatment provides a good window of opportunity to either prevent PsA or dilute its manifestations when they occur. PsO progressing to PsA is found less often in patients receiving biologic/systemic therapies than in patients receiving topical treatment or no therapy (12% vs. 37%, respectively). 3 A retrospective study of patients with PsA found no evidence of current or previous dactylitis in patients receiving biologic therapy for skin PsO. This is an important finding since dactylitis is suggested to be a marker of disease severity and progression, and its prevention is a manifestation of effective therapy.15, 16 Despite their availability and cost-effectiveness, conventional DMARDS and systemic therapies are used by only 0.5%–22.6% of the patients in the USA. 17 Our data also confirms the sparse use of systemic therapy for PsO. The majority of our patients received only topical treatment. Overall, 48.3% of patients had maximum BSA involvement of more than 10%, but only 22.4% received MTX, of which more than 50% reported an overall period on medication of less than one year. A reason reported in a survey for physicians for the underuse of systemic treatment in PsO was related to concerns about the long-term safety, tolerability and efficacy of the currently available oral systemic drugs. 16 Biologics are not widely used among Indian patients due to their high costs. 18 Personal communication with dermatologists also suggested that they might not consider systemic therapy in mild to moderate PsO for the fear of patient acceptability, the costs of therapy and the possibility of potential adverse events. In summary, our study suggests that we might be losing the window of opportunity for preventing PsA or reducing its severity. It is only recently that the Indian guidelines have been proposed for the early use of oral therapy and biologics in PsO. 19 It will be interesting to see the effect of these guidelines on further practice patterns.
Examining the prevalence of co-morbidities in PsA was another important aim of this study.
A remarkable finding in our study was the high prevalence of T2DM observed in our population of PsA patients, at 24%. This prevalence is much higher than the recently reported prevalence of 9.3% in the general Indian population. This study concurs with a meta-analysis of 44 observational studies that found a significant correlation between DM and PsA. Though the pathogenic link between PsA and DM is not fully understood, TNF- and adipokines may be important mediators of this correlation. TNF-alpha inhibits the autophosphorylation of the insulin receptors, thus inducing insulin resistance. Similarly, adiponectin is an adipokine with anti-inflammatory, insulin-sensitising and anti-atherogenic properties, whose secretion is decreased by proinflammatory cytokines.
Obesity is one of the strongest risk factors for PsA, both in patients with PsO as well as in the general population. It precedes its development by several years. Moreover, there is a dose effect, such that a higher body mass index is associated with an incrementally increased PsA risk.22–23 Using conventional BMI criteria, our prevalence of obesity and overweight in PsA patients is 16.3% and 36.8%, respectively, which is much higher than the data from the general Indian population. 20
In our population, hypertension was prevalent in one-fourth of the patients. This is not much different from the recently reported national prevalence of 25.8%. 21 Our data do not concur with the previous meta-analysis, which has shown that patients with PsA have an increased prevalence of hypertension (OR 2.07, 95% CI 1.41–3.04). 17 Numerous studies have shown that the prevalence rate of concomitant hypertension is up to 55%.22–23
In our study, 29.9% of the patients were being treated for dyslipidaemia, and an additional 8.8% were diagnosed at entry, based on the criteria mentioned. This prevalence is similar to the prevalence found in the general Indian population. A review of several epidemiological studies by Gupta et al. in 2017 suggested that high cholesterol of more than 200 is present in 25%–30% of urban and 15%–20% of rural subjects. 24 In studies on patients with PsA, dyslipidaemia has been found to be associated with high markers of inflammation (high-sensitivity C-reactive protein and platelet counts), suggesting a potential relationship with a higher level of inflammation. 25 There is also an association found between increased levels of total cholesterol and triglyceride levels and subclinical atherosclerosis in PsA.26, 27
Regarding the prevalence of CAD, only 4.8% of the study population developed CAD. The prevalence of CAD in Indians living in India is 21.4% for diabetics and 11% for nondiabetics. 28 A meta-analysis of 11 studies found a 43% increased risk of CAD in patients with PsA compared with the general population. 29 The high prevalence of metabolic syndrome and T2DM seems to be the main cause of the increased CAD events, acting through sustained and chronic systemic inflammation. It is possible that the higher levels of systemic inflammation emanating from joints as well as the skin could be contributing to sustained inflammatory changes in the cardiovascular system too, leading to a higher prevalence of CAD. Longer duration of psoriatic disease and disease severity has been associated with increased risk of cardiovascular disease: the risk of major atherosclerotic cardiovascular events (MACE) increased by 1.0% per additional year of PsO duration. It is difficult to reconcile the relatively low incidence of CAD in our population particularly in the light of a high prevalence of diabetes. However, we found a disproportionately higher prevalence of CVAs of 1.9 per 100 (5–20-fold) as compared to that in the general population. The estimated adjusted prevalence rate of stroke ranges in India are 84–262/100,000 in rural and 334–424/100,000 in urban areas 30 which is much lower than seen in our population. In one met analysis CVA risk was found to be increased by 22%in patients with PsA compared to the general population.
Since MACE by definition includes both coronary events and stroke, the overall cardiovascular risk in our population is high, although coronary events were relatively low.
There could be some limitations to this study. This being a cross-sectional study, limitations associated with proving causality will exist. Longitudinal studies from the onset of PsO will be needed to prove that PsO is responsible for the comorbidities that are present with it. These may be difficult, though not impossible. Though the study was multi-centric, all centres were situated in western India. As a result, generalising to all of India may be difficult. The maximum BSA involved was calculated from the patient’s recall. There could be some inaccuracies during the recall. Also, it was possible that the patient may have had undiagnosed PsO before presenting to the dermatologist. Our study determines the ‘Onset’ as the date of diagnosis by the dermatologist rather than the date of symptoms. The diagnosis of T2DM prior to presentation to the rheumatologist was based on the patient’s history and it is possible that some the patients diagnosed at presentation could have had T2DM beforehand. Lastly, the use of alternative medicine in detail could not be obtained and its impact on the progression from PsO to PsA could not be determined.
Conclusion
We found the use of sustained systemic therapy for PsO (before the onset of PsA) to be very low. We also found a much higher prevalence of obesity and diabetes in our patients with PsA compared to the general population, putting them at higher risk for cardiovascular events. Early diagnosis and better treatment of PsO may hold the key to better control of psoriatic disease.
Footnotes
Author Contributions
Drs Soham Kadam and Asna Shaikh were responsible for the collection, formulation of the dataset and writing of the article. Drs Varsha Bagul, Priti Nagnur-Mehta, Shubhada Kalke, C. Balakrishnan and Kaushik Bhojani were responsible for the collection of data. Drs Praveen Jadhav and Dr Sushant Shinde were responsible for the collection of data and writing the article. Dr Rohini Samant and Dr Jyotsna Oak were responsible for the conceptualisation of the project, collection of data and editing of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethics committee approval was taken at P. D. Hinduja Hospital in Mumbai (EC letter no. 1389-20-RS, 1440-21-RS). Informed consent was taken from all the patients included in the study.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
