Abstract
Background:
Methotrexate (MTX) at a dose of ≤25 mg/week is one of the most prescribed disease-modifying anti-rheumatic drugs (DMARDs) in a variety of rheumatic diseases. It can potentially cause life-threatening neutropenic sepsis, and acute renal and hepatotoxicity when taken inadvertently at high doses. We aim to analyse the clinical profile and risk factors of patients who presented with acute MTX toxicity.
Methods:
All patients presenting to the Rheumatology department with a history of inadvertent consumption of higher doses of MTX (>25 mg/week), from July 2021 to May 2023 were included. Additional data was extracted from hospital electronic medical health records. The clinical profile, risk factors, and outcome of patients with MTX toxicity were analysed.
Results:
The median age of the patients in our cohort was 52 IQR (40–62.5) years, with 80% females. The median cumulative dose of MTX was 120 mg (IQR 95–150). The reason for overdose in our cohort was medication error in comprehending once-weekly dosing. The most common major adverse event was neutropenia (80%). All our patients had stomatitis, with half of them having oral bleeding. Gastrointestinal adverse events like vomiting and diarrhoea were seen in 60% and 13% of the patients, respectively. Our cohort had two patients who succumbed to the complications due to neutropenic sepsis. The dose of MTX did not correlate with the severity of the disease or duration of hospital stay; however, the latter was significantly influenced by lower absolute neutrophil count (ANC).
Conclusion:
Acute MTX toxicity is one of the severe rheumatological emergencies and the toxicity profile includes haematological, gastrointestinal, hepatic, and renal adverse events. Severe neutropenia leading to sepsis can be fatal if not intervened early.
Introduction
Methotrexate (MTX) is one of the most widely prescribed disease-modifying anti-rheumatic drugs (DMARDs) in rheumatological practice. Rheumatoid arthritis (RA) is the most common indication owing to its high prevalence and the approval of MTX as first-line therapy. 1 The drug has emerged as the preferred line of treatment in various conditions like RA, systemic sclerosis with predominant skin involvement and arthritis, newly diagnosed non-organ or life threatening anti-neutrophil cytoplasmic antibody-associated vasculitis (ANCA), musculoskeletal manifestations of Sjogren’s syndrome, systemic lupus erythematosus, to name a few.1–4 Even when low-dose MTX came to be used in clinical rheumatology in the 1980s, the side effect profile of stomatitis and bone marrow suppression was reported; however, due to the risk-benefit ratio, MTX gained popularity. 5 It is administered as a single weekly dose for the treatment of these conditions and a medication error leading to fallacious intake of daily doses has the potential to cause life-threatening cytopenia, renal and hepatic dysfunction, mucositis and stomatitis. 6
Low dose MTX (<25mg/week) is mostly well tolerated, with the prevalence of stomatitis, bone marrow suppression causing cytopenia (anemia, neutropenia, thrombocytopenia) being as low as 3%. A recent meta-analysis analysing the side effect profile in nearly 4,000 patients showed that the incidence of neutropenia was ~1.77% and anaemia was 2.5%. 7 However, the data available for the adverse effects of the drug are predominantly in a stable-dose cohort, where these were attributed to chronic bone marrow suppression and not to an acute toxicity.
Though adverse events of MTX have been widely described in literature, data on acute MTX toxicity is from anecdotal reports and partly from an Indian series by Ajmani et al. describing pancytopenia with MTX, where among other cases, they also described 13 patients who had a dosing error.8–11 In this study, we aim to describe the clinical profile, outcome and possible factors associated with higher risk and poor prognosis in patients who presented with acute MTX toxicity due to unintentional fallacious consumption of a dose of >25 mg/week.
Methods
All patients admitted in the Department of Rheumatology from July 2021 to May 2023 with a history of medication error with MTX, resulting in an intake of >25 mg/week and presenting with symptoms of acute toxicity were included. We defined acute toxicity in patients who had consumed daily doses of the prescribed weekly dose, for two or more consecutive days, that resulted in one or more of the adverse events. The data regarding cumulative dose of MTX, prevalence and severity of neutropenia, renal and hepatic dysfunction, and presence of pre-existing poor prognostic factors like hypoalbuminemia, chronic kidney disease (CKD) or usage of another DMARD, were captured. Additional data was extracted from electronic medical health records and analysed. Haematological adverse events were further divided into anaemia, any leukopenia (<4000 cells/mm3), neutropenia (<1500 cells/mm3), and thrombocytopenia (<1 lakh cells/mm3).
All our patients were treated with the institutional protocol for acute MTX toxicity, which included adequate hydration, leucovorin rescue, granulocyte colony stimulating factor (G-CSF) in severe neutropenia, along with broad-spectrum antibiotics and add-on anti-fungal agents, that were decided on a case-to-case basis. In the patients who developed acute renal failure, additional step of alkalinisation of urine was done. Along with this, supportive measures, barrier nursing and strict hygiene for the maintenance of stomatitis and ulcers were followed.
Statistical analysis was carried out by using IBM SPSS version 25. Groups were compared by student’s t-test. The Karl Pearson correlation coefficient was calculated for lowest absolute neutrophil count (ANC) and duration of hospital stay with the total MTX dosage. The odds ratio was calculated for the covariate effects of mortality. The clinical and lab parameters were analysed as potential prognostic factors for survival/mortality. Univariate analysis regarding the risk factors for developing a serious neutropenia or a more severe course of illness was carried out. p Value <.05 was considered statistically significant. All median values are expressed with 25th to 75th interquartile range (IQR).
Results
Baseline Characteristics
Fifteen patients admitted with acute MTX toxicity from July 2021 to April 2023 were included (Table 1). Baseline characteristics are summarised in Table 1. The most common indication for prescribing MTX was RA (12/15); two patients had psoriatic arthritis and one was being treated for peripheral Spondyloarthritis. The cumulative median dose of MTX (taken together) that resulted in acute toxicity in our cohort was 120 mg (IQR 95–150).
Patient Characteristics.
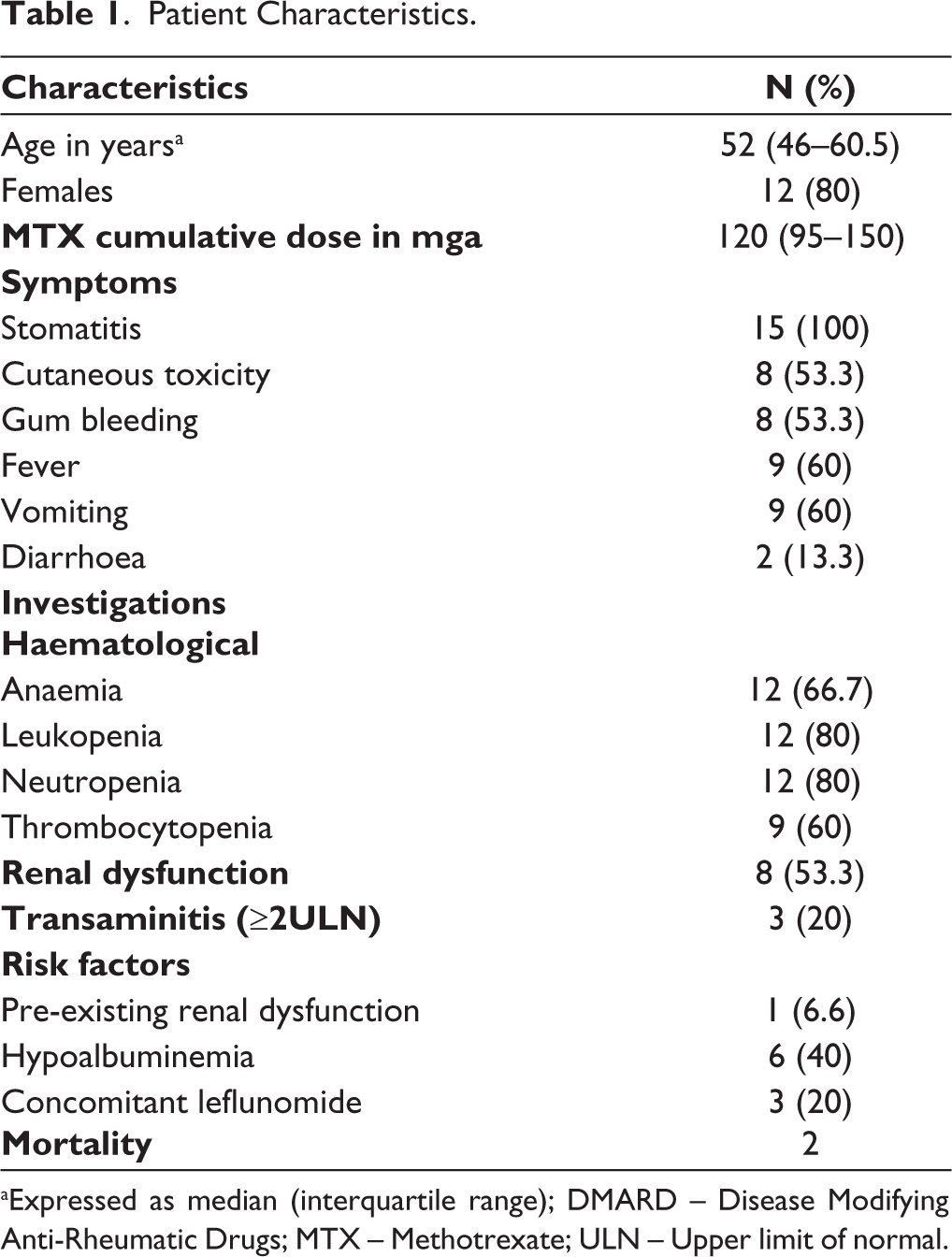
aExpressed as median (interquartile range); DMARD – Disease Modifying Anti-Rheumatic Drugs; MTX – Methotrexate; ULN – Upper limit of normal.
All the patients in our cohort had an accidental overdose. After a recall of prescriptions and thorough history taking, we determined that there were no prescription errors and there was no alleged intentional overdose. In 12/15 patients, toxicity occurred with the initial doses as they wrongly consumed the medication every day. Two patients were previously taking weekly MTX for six months and eight months for RA, then defaulted on treatment and presented with a flare. On restarting the medication, they unintentionally consumed it daily and alleged that they had forgotten that it was a weekly medication. The last patient took 15 mg on two consecutive days unintentionally and in the background of CKD, she developed acute toxicity with a dose of 30 mg taken within 48 hours.
Clinical and Laboratory Features of Acute MTX Toxicity
Neutropenia was the commonest adverse event with 80% of the patients demonstrating some degree of neutropenia. The lowest ANC in our cohort was 105 cells/mm 3 , and unfortunately this patient succumbed to sepsis. Severe neutropenia less than 500 cells/mm 3 was seen in eight patients. However, dose-dependency could not be established, possibly due to the limited sample size. Though the cumulative dose of MTX did not correlate with the ANC, or the duration of hospital stay, we observed a significant correlation between the severity of neutropenia (as quantified by ANC) and a longer duration of hospitals stay (p – .008).
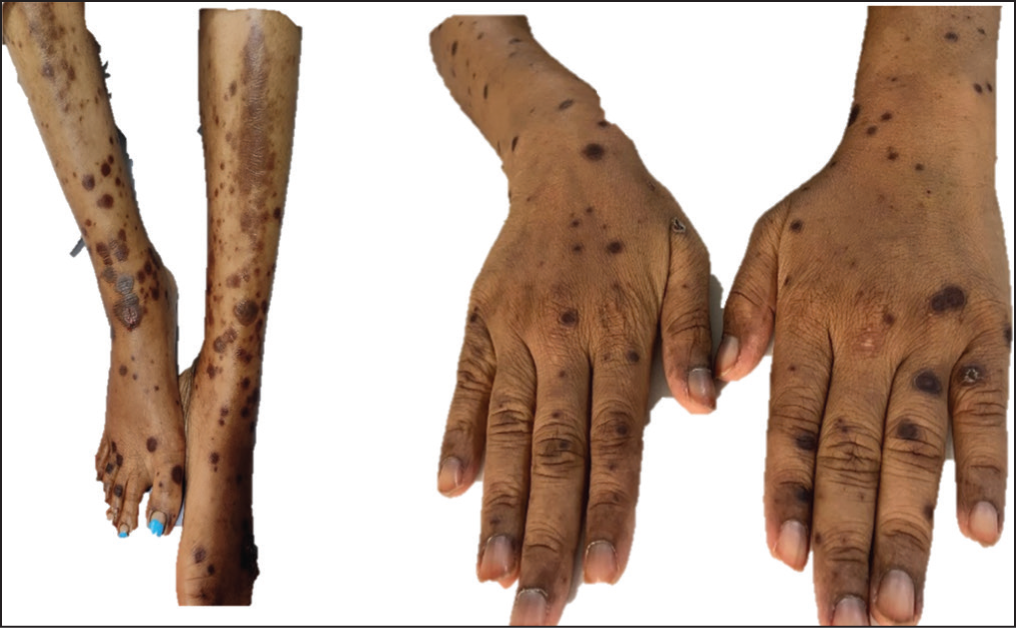
A and B: Cutaneous Eruptions due to Acute Methotrexate Toxicity.
Cutaneous Ulcer Secondary to Acute MTX Toxicity.
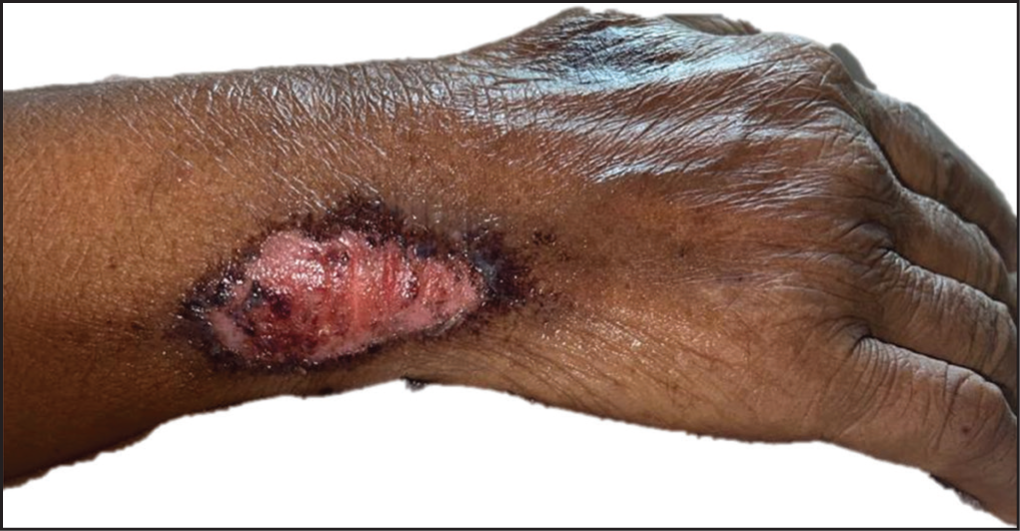
At presentation, all patients had stomatitis and over half of them developed gum bleeding and cutaneous toxicity (Figures 1–3).
Oral Mucositis and Stomatitis due to Acute Methotrexate Toxicity.
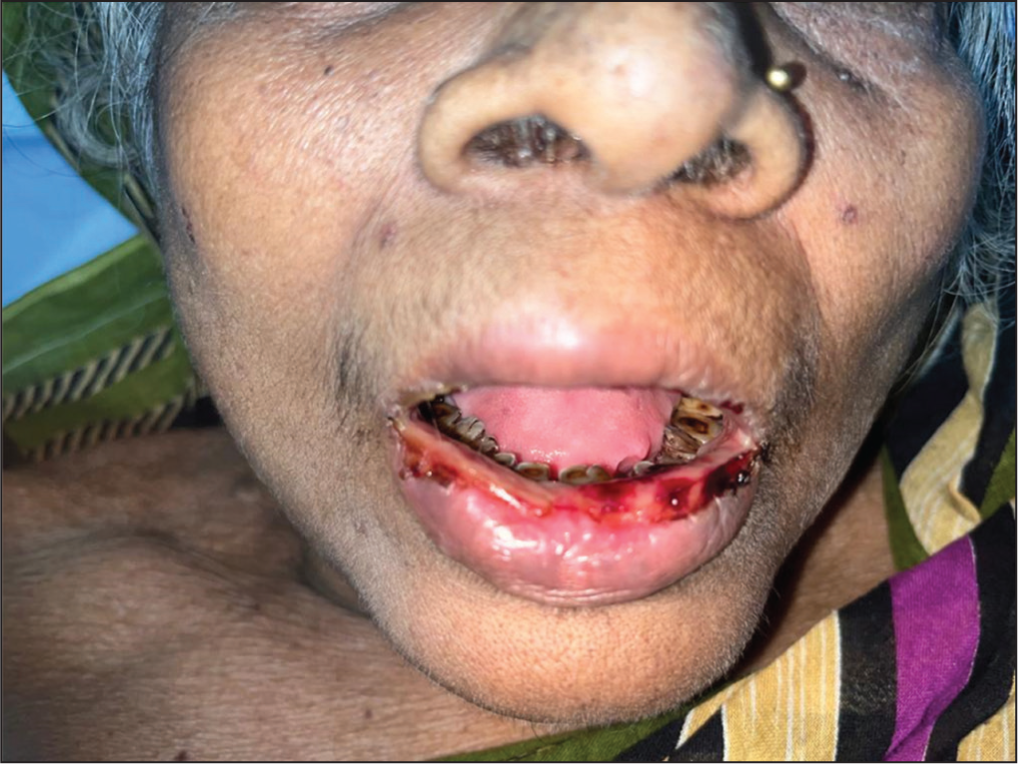
Transaminitis was seen in a minority of cases (n = 3). Fifty three percent of the patients had acute kidney injury, however, none of them were severe or progressive to warrant renal replacement therapy. Gastrointestinal adverse events like vomiting and diarrhoea were observed in 60% and 13.3% of the patients, respectively. None of the patients had hematemesis or rectal bleed.
Mortality
There were two deaths in our cohort, and they were both secondary to bacterial sepsis. One patient developed a Staphylococcus aureus septicemia while the other had a gram-negative sepsis with Escherichia coli, which was multi-drug resistant. A non-significant trend towards mortality was seen with total MTX dose with an odds ratio (OR) of 1.039 (95% CI of 0.95–1.12, p – .37). OR for other traditional risk factors of mortality like neutropenia, hypoalbuminemia and renal dysfunction was not significant.
Discussion
In our study, we analysed the clinical profile of acute MTX toxicity due to accidental erroneous intake of medication daily as opposed to a weekly dose. The commonest cause of acute MTX toxicity in literature was due to accidental drug overdose or due to erroneous dispensing of the drug.12, 13 Though unintentional overdose is commonly described, data from a poison control centre in the United States reported up to 20% consumptions that were intentional. 13 Bebarta et al., describing another retrospective cohort of acute MTX overdoses, reported no mortality in their series of 63 patients, with none of them developing any of the major adverse events like bone marrow suppression or death; however, this may have been due to the lower mean dose of MTX of 24 mg in this cohort, as opposed to a mean dose of 114.6 mg in our cohort. 14 A third retrospective series from the USA, described a small number of patients with acute MTX ingestion, where the maximum dose was ~13 mg and they reported no major adverse events. 15 An Iranian group of researchers reported a worrisome 37% of patients in their cohort who had an acute MTX poisoning due to suicidal intention. 12 Even among the patients who unintentionally overdosed, the most common reason was confusing the instruction of weekly dosing to a daily dose. 16 The stark difference in the incidence of major adverse events in our cohort, as compared to the series from USA is that our patients tend to receive a higher dose of MTX, usually starting at 15 mg weekly, while the commonly used oral dose described in the prior mentioned series were 2.5–5 mg. 15
Haematological complications are one of the most serious and potentially life-threatening. Vanni et al. in their meta-analysis about the cytopenia associated with MTX use in AIRDs, described an incidence of anaemia of ~2.5%, ~1.77% neutropenia and ~0.19% thrombocytopenia. 7 However, these observations cannot be extrapolated to acute MTX toxicity as the molecular mechanism causing an acute toxicity is different. However, in the isolated case reports and case series, all the patients had some degree of neutropenia and the ones who died, invariably succumbed to neutropenic sepsis. Contrary to the popular belief that says acute MTX toxicities do not cause cytopenia due to the saturation of the enzyme, we noted that 80% of our patients had neutropenia. 17
The cutaneous adverse events due to acute MTX toxicity can present as erythema, toxic skin eruptions (Figure 1A and B), ulcerations (Figure 2) or Steven-Johnson/Toxic Epidermal Necrolysis (SJS/TEN)-like lesions, however, the exact mechanism is not fully understood. It may result from hypersensitivity reactions or apoptosis due to the cytotoxic effects on keratinocytes and potentially endothelial cells, that triggers cellular apoptosis. 18
Patients who have pre-existing renal disease are at an increased risk of toxicity from MTX even at low doses and this risk increases when the eGFR <45 ml/min/1.75 m2.19, 20 In our cohort, only one patient had pre-existent CKD with an eGFR of 38 ml/min/1.75 m2, and she developed severe neutropenia with only 30 mg of MTX (15 mg taken on two consecutive days).
Effects on folate metabolism is one of the main mechanisms responsible for toxicity of MTX. This, along with polyglutamate accumulation were traditionally accepted as the major causes for adverse events, especially hepatic and gastrointestinal. Acute toxicities are described to result from intracellular accumulation of the drug, which has the potential to even form intrarenal MTX crystals which may result in acute kidney injury; but this mechanism has been described more commonly in the setting of toxicity resulting from high-dose MTX used in chemotherapy. 17 Recently many molecular mechanisms have forayed into the understanding of the safety profile of the drug, like the effect of oxidative stress on the cell, Sirt1/ Nrf2/γ-gcs, and γ-gcs/CaSR-TNF-α/NF-kB signalling pathways. 21
Among the factors predicting poor prognosis, hypoalbuminemia, preexisting renal failure and concurrent use of other DMARDs with similar toxicity profile are commonly quoted in literature.11, 22 However, in our cohort, we did not find a significant correlation between these poor prognostic factors and mortality, which is possibly due to our small sample size.
Two of our patients (13%) succumbed to sepsis secondary to severe neutropenia. A large series of 106 patients from the USA who reported medication errors with MTX, mortality was reported in a quarter of the cases. 16 Even in this cohort, the most common cause of overdose was mistaken consumption of weekly dose daily.
Though some authors recommend the estimation of serum MTX levels, there is no additional benefit of this value clinically, nor will it change line of treatment. However, most of this data is extrapolated from the treatment of toxicities resulting from high-dose MTX (>500 mg/m 2 ) used in cancer chemotherapy for malignancies like lymphoma, acute lymphoid leukaemia, osteosarcoma, etc.23, 24
Folic acid is supplemented at adequate doses with MTX to prevent toxicity. The FOLVARI trial that compared 10 mg/week of folic acid to 30 mg/week of folic acid did not report any significant benefits of a higher folate dose, nor were there any risks of supplementing it at a higher dose. 25 Though folic acid supplementation has proven benefits in preventing bone marrow suppression in chronic MTX use, data on it’s protective effects in acute toxicity may not be as robust. However, absence of folic acid supplementation is considered as a risk factor for developing toxicity. All the patients in our cohort were on regular 10 mg folic acid/week.
Though this is one of the largest series describing the clinical profile of acute MTX toxicity in rheumatic diseases, our study was limited by a single-centre and small sample size. Studies with a larger cohort may aid in confirming this hypothesis. Our study serves as an example for the need for repeated counselling regarding appropriate use of MTX. The physicians need to create better patient-information leaflets with pictorial presentation regarding MTX.
Conclusion
Acute MTX toxicity is not very commonly reported, and data is mostly derived from anecdotal evidence. Neutropenia is the commonest major adverse event associated with acute MTX toxicity. As medication error was the cause of all our acute overdoses, we need to re-evaluate the method of counselling and patient education regarding the drug.
Footnotes
Acknowledgements
We are grateful to Mr. Sourav Padhee for helping with the statistical analysis of the data.
Declaration of Conflicting Interests
SA has received honorarium as speaker from DrReddy’s, Cipla, Janssen, Pfizer unrelated to the current work. The other authors have no conflicts of interest to declare.
Ethics Approval and Patient Consent
A written informed consent was obtained from all patients/relatives for publication. Ethics clearance was exempted as this is a case series.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
