Abstract
Background:
Childhood cancer, specifically acute lymphoblastic leukemia (ALL), poses substantial psychological, physical, and emotional challenges, affecting the quality of life (QoL). Expressive art therapies integrate movement, art, and music and offer developmentally appropriate, nonverbal interventions that enhance resilience, coping, and emotional well-being.
Novelty:
Routine psychological interventions are not suitable for children and adolescents who may have difficulty formulating or expressing their experiences and feelings, particularly in our cultural context. In such clinical situations, creative therapies, such as expressive art therapies may be used.
Objectives:
The primary objective will be to evaluate the change in QoL with some secondary objectives to evaluate from baseline after 6 and 12 weeks of treatment with treatment as usual (TAU) plus expressive art therapy versus TAU amongst children with ALL.
Methods:
The proposed study is a prospective, randomized parallel design trial. Following the inclusion and exclusion criteria, children will be recruited and randomized into 2 treatment groups. One group will receive TAU plus expressive art therapy and another group will receive TAU for 12 weeks in a clinic-based setting. Clinical status and functioning will be checked by using Cancer Institute Pediatric Oncology Quality of Life Questionnaire-31 (CI‑Ped Onc QLQ 31) scale score, State-Trait Anxiety Inventory for Children (STAI-C) anxiety score, Rosenberg Self-Esteem Scale score, and perceived stress score at baseline, 6 and 12 weeks.
Conclusions:
Expressive art therapy may demonstrate greater reduction in anxiety levels, perceived stress, and improvement in self-esteem and QoL over the 12 week period in children with ALL. It may be used as add-on therapy in children with hematological cancer in Indian context.
Introduction
The annual incidence of cancer under 15 years of age is approximately 140 cases per million. An estimated 280,000 children were diagnosed with cancer in the year 2020 alone.1,2 Nearly 110,000 of them capitulated to the disease, which highlights the critical need for efficient treatment plans and supportive care strategies. 3 Childhood cancer is a cause of mortality in adolescents and children, and it is a major worldwide health burden, in which acute lymphoblastic leukemia (ALL) reports approximately 40%–50% of the total of pediatric cases of cancer in India. 4 It is the most common hematological malignancy in this population. In addition to its physical challenges, childhood cancer impairs the social, emotional, and psychological growth of patients. Physical deformities, chronic stress of managing the disease, isolation, and treatment-related discomfort eventually worsen the quality of life (QoL) in patients with pediatric cancer, along with their families. Conventional therapeutic modalities, such as cognitive behavioral therapy (CBT) have significantly reduced these impacts. 5 However, CBT heavily relies on verbal expressions and introspection, which can be difficult for adolescents and children as well, especially in those sociocultural settings where emotional articulation is restricted. 6 This emphasizes the need for different approaches that are attentive to the patient’s psychological needs, while still being developmentally appropriate. Expressive or creative art therapy offers patients various ways to express themselves in nonverbal ways, which eventually improves their QoL.
It has been demonstrated that creative art therapy improves resilience and emotional expression and reduces psychological distress. For instance, studies demonstrated the efficacy of painting-based interventions in eliminating fatigue, reducing anxiety, and elevating mood in children with cancer.7,8 Similarly, a recent systematic review highlighted the broad range of benefits of art-based therapies in reducing anxiety and depression and in fostering physical stability, resulting in improved QoL. 9 Beyond art-based therapies, integrative approaches that combine music, movement, and art have shown potential in addressing the comprehensive needs of children with cancer. It has been found that expressive art therapy helps children manage painful emotions and enables children to navigate difficult emotions through symbolic expressions. 10 Further, art therapy dramatically reduced depression, stress, and anxiety in children with ALL undergoing chemotherapy. 11 Study also revealed that group art-based therapy, creativity, and self-expression improved health-related QoL in pediatric cancer patients. 12 Movement and dance therapy paired with music provide children a kinesthetic outlet to process their sense of self and emotions and to enhance resilience. 13 Creative arts therapy improved QoL and coping in pediatric brain tumor patients, particularly by reducing anxiety and enhancing mood, which helped children to tolerate chemotherapy and other painful procedures. 14
Despite the proven benefits of creative art therapy in different cancer populations, there is a lack of comprehensive research in combining music, movement, and art known as expressive art therapy as a therapeutic framework for improving the QoL in children and adolescents with hematological malignancies. This study seeks to address this gap by evaluating the effect of expressive art therapy in conjunction with standard care. By leveraging expressive and creative interventions combined to the emotional and developmental needs of children with ALL, this study aims to enhance emotional well-being, healthy coping mechanisms, resilience, and general QoL in them.
Research Question
Does the change in QoL with TAU plus adjunctive expressive art therapy differ significantly from TAU alone in children diagnosed with ALL?
Hypothesis
Null: Change in QoL with TAU plus adjunctive expressive art therapy is similar to TAU alone in children diagnosed with ALL.
Alternate: Change in QoL with TAU plus adjunctive expressive art therapy is significantly different from TAU alone in children diagnosed with ALL.
Aim of the Study
To evaluate the efficacy of expressive art therapy on the QoL in children with ALL.
Study Objectives
The primary objective of the study is to compare the effect of expressive art therapy as an adjunct therapy with TAU on QoL amongst children with ALL at 12th week of treatment using Cancer Institute Pediatric Oncology Quality of Life Questionnaire-31 (CI-Ped Onc QoL-31). Secondary objectives are to compare the effect of expressive art therapy as an adjunct therapy with TAU on QoL from baseline to 6 weeks and from 6 weeks to 12 weeks as well as on anxiety, self-esteem, and stress amongst children with ALL using the State-Trait Anxiety Inventory (STAI) for Children (STAI-C), Rosenberg Self-Esteem Scale, and Perceived Stress Scale-10 (PSS-10) over a 12 week period.
Outcome Measures
The primary outcome measure is the change in QoL score on CI-Ped Onc QoL-31 from baseline to 12 weeks. The secondary outcome measures are (a) change in QoL score on CI-Ped Onc QoL-31 from baseline to 6 weeks and from 6 weeks to 12 weeks (b) changes in scores of STAI, Rosenberg Self-Esteem Scale and PSS-10 from baseline to 6 weeks, 6 weeks to 12 weeks, and baseline to 12 weeks, respectively.
Material and Methods
Ethics Review
The study strictly adheres to the ethical standards outlined by the Indian Council of Medical Research (ICMR) for biomedical research on human subjects (2006). The Institutional Ethics Committee approved the study prior to its commencement. After getting approval from the Institutional Ethics Committee, we intend to secure written consent, following all the predetermined exclusion and inclusion criteria. By coding the information, we plan to preserve the confidentiality of data. Participants have the authority to withdraw their consent at any stage, before, or during the study, without incurring any repercussions. The study is registered under the Clinical Trial Registry of India (CTRI/2023/11/059742).
Proposed Study Design, Participants, Setting
The proposed study is a prospective, open-label, randomized, parallel design trial and will be conducted at a single tertiary care center in the department of psychiatry and pediatric oncology. The inclusion criteria for the study comprise children diagnosed with ALL, regardless of their gender, ages ranging between 4 and 15 years. Additionally, the oncology team must anticipate a survival expectancy of more than a year for these participants. However, the exclusion criteria specify that children who are unable to communicate, or who are critically ill due to the severity of their illness will not be included in the study. Participants with comorbid diagnosed psychiatric conditions or significant cognitive impairments will be excluded from study participation. A detailed medical history will be obtained and clinical evaluations will be done using a semi-structured proforma at the baseline. The participants will be randomized by mixed block randomization into 2 treatment groups with an allocation ratio of 1:1. A random sequence will be generated through
Intervention (Expressive Art Therapy Protocol)
The expressive art therapy protocol will be based on Adaptive Response Theory: An Evolutionary Framework for Clinical Research in Art Therapy and work on dance/movement therapy in pediatric oncology.15,16 The expressive art therapy will consist of 2 sessions; the first will involve creative art therapy and storytelling, and the second will focus on dance, music, and movement therapy, which is novelty of our intervention. Therapy will be divided into 4 weeks apart into 3 stages for maximal therapeutic benefit for children with ALL.
In the first stage, consent will be taken from parents and participants will be assessed with parents for their current stress levels, anxiety, self-esteem, and QoL. The patients and parents will be psycho-educated on the stress, anxiety, fear of unknown, grief reactions, and developmental challenges that are faced by children due to long-term stays in ward. Each session will begin with 10 minutes of brief relaxation and positive affirmations, developed with the patient during the assessment session. In the first phase, it is important to understand how children of various ages think about their bodies. They will be shown an outline of the human body and asked to put up craft-made internal parts one by one to identify different body parts. This exercise helps the therapist to understand the child’s perceptions of the human body and provides the opportunity to offer information that may correct misunderstandings and allay fears. The next session will be about expressing difficult emotions, externalizing, and self-regulation through art. Free drawing allows children to externalize confusion and fears, talk about their emotions through drawing and colors.
The second stage will be developing a deeper therapeutic alliance and a nonjudgmental fun activity space for children through art. In this session, a clay activity will be introduced wherein the therapist will work with each patient to make clay figurines and objects. Working with clay may be rewarding for children who are hesitant about their creativity. The play therapy will be non-directive. Additionally, the important aspect of using clay which is often ignored in play therapy as we focus on the process rather than the product is the way it enables children to produce lasting pieces. Functional pieces will be produced (e.g., cups, bowls) children see themselves as capable of engaging in a truly purposeful activity.
The third stage of therapy will involve facilitating and modeling adaptive experiences, communicating (saying the unsayable), projecting inner thoughts and feelings, and symbolization of experience. Children will be asked to do structured drawing and will be shown various pictures and art to interpret in various ways. The final stage will be developing self-efficacy through creative decision making, encouraging resilience, and newly formed autonomy, rewriting the self-narrative through drawing stories of life and future ahead, herein in the last few sessions they will be helped to reimagine their stories with pictures or drawings and develop narrative (inner voices) for their character and imagine a positive outcome.
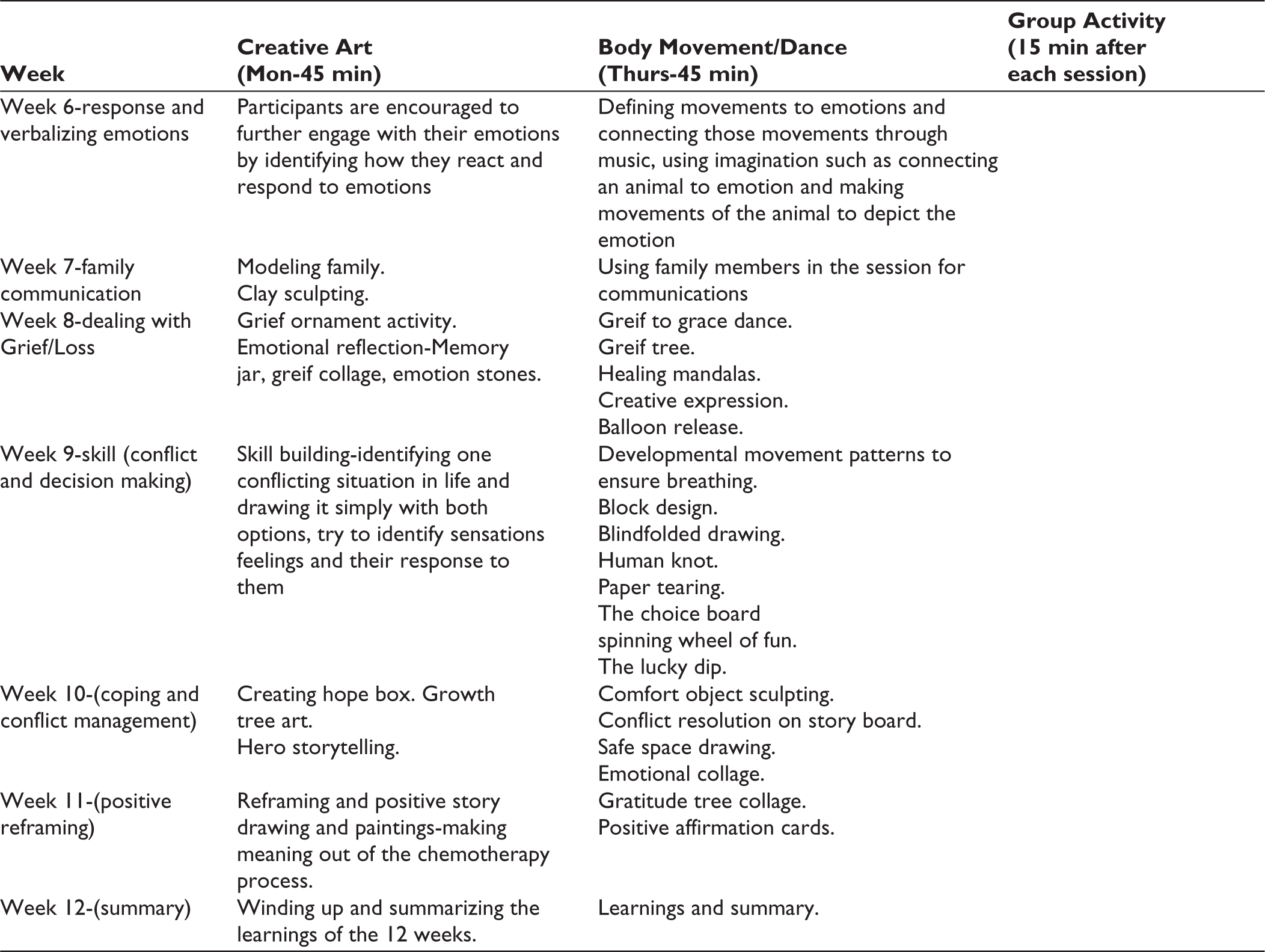
One session per week will be focused on creative art therapy (e.g., drawing and storytelling) and the second session in the week will be focused on dance, music, and movement therapy in which the therapist will collaboratively work with the patient and family to assess, conduct sessions, and strengthen skills. The assessment will be on the patient’s orientation to music and dance, the level they are able to perform with their body at the current stage of treatment, as well as their previous knowledge and liking of music and dance. The focus of these sessions will be resilience, coping, and improving confidence about their bodies. Patients will be asked about their musical choices. Their mood charting will be done and a playlist will be created for the patient based on each mood with the patient’s input. Each week the therapist will collaboratively work with the patient to devise movements that help adequately express the various moods the patient goes through during his cancer journey through the movement and music. The parent and caregiver will also be made part of this therapy to help the child and family reconnect with each other. Overall, the patients will be engaged in 24 sessions with 2 sessions per week of 45–60 minutes over a period of 3 months. Feedback will be taken from patients and parents at the end of the 3 months of therapeutic intervention. Table 1 shows detailed weekly session plan for creative art therapy and body movement/dance therapy sessions. There will be 2 concurrent individual sessions in a day. We plan to carry out 2 sessions per week per child. There will be around 5 participants in the group session, which will be carried out once a week.
Detailed Weekly Session Plan for Creative Art Therapy and Body Movement/Dance Therapy Sessions (= Expressive Art therapy).

Standard of Care/Treatment as Usual
Standard of care consists of medical standard care and education about the illness and prognosis to the parents. Medical standard of care for ALL includes an induction phase (prednisolone, vincristine, daunorubicin, L-asparaginase, methotrexate, cyclophosphamide, cytarabine, 6-mercaptopurine, methotrexate [IT]); consolidation phase (6-mercaptopurine, methotrexate, methotrexate [IT]); reduction phase (dexamethasone, vincristine, daunorubicin, L-asparaginase, methotrexate), maintenance phase (6-mercaptopurine (orally), methotrexate (orally), methotrexate [IT]).
Instruments for Measurement of Study Outcomes
The primary outcome of the study is QoL which will be measured using the CI-Ped Onc QoL-31. It is a 31-item questionnaire. 17 It has been validated and found to have a Cronbach’s alpha (internal consistency of the items) of 0.833. The questionnaire measures along 3 dimensions physical well-being, psychosocial well-being, and treatment-related concerns. For each item, the score is ranges from “3” (not at all) to “0” (always). Twelve items are negatively framed due to negative loadings, and are reverse scored. Scores below 47 will be interpreted as “low QoL,” those between 47 and 65 as “average QoL, and 66 and above as “high QoL.”
The secondary outcomes of the study are anxiety, self-esteem, and stress which will be measured by the following measurement tools with descriptions. All measurement tools are researcher-administered.
STAI-C: It is used to assess both state and trait anxiety for children and has 2 scales with 20 items each. The child responds to the STAI-C scale by choosing one of the alternative responses. In essence, each STAI-C item is a 3-point rating scale for which values of 1, 2, or 3 are assigned for each of the 3 choices. For items in which the key term indicates the presence of anxiety, “very” and “not” are assigned values of 3 and 1, respectively. The order of weighting is reversed for items in which the key terms indicate the absence of anxiety, that is, very = 1; not = 3. The score ranges from 20 to 60. The state part of the scale asks the participant how they feel from moment to moment in time. The trait scale asks questions that help understand how they generally feel. The Cronbach’s alpha for the scale is 0.88. 18
Rosenberg Self-Esteem Scale is a 10-item scale that measures global self-worth by measuring both positive and negative feelings toward oneself. Responses are on a 4-point Likert scale from strongly agree to strongly disagree. Higher sum scores indicate higher self-esteem. The scale is unidimensional. The Cronbach’s alpha is 0.709. 19
PSS-10: This is a popular measure of self-reported stress and the extent to which one’s daily life situations are perceived as stressful. We will use the 10-item version of the PSS which ranges from 0 to 40, which is a shorter version of the original PSS-14 and comprises 6 negative items and 4 positive items. The total score is calculated by summing scores across all items, after reversing the scores on the positive items, with higher scores indicating higher perceived stress. The Cronbach’s alpha ranges from 0.74 to 0.91. 20
Sample Size Estimation
As per the Cohen’s conventions for effect size (Ø = d/s) in case of difference of mean as outcome, variable in clinical trial, an effect size of 0.5 is considered medium effect: a = 5% 1-b = 80%, sample size = (1.96 + 0.84) 2 /d 2 ; where [d = 0.5]; where d = effect size = 7.86 = 31.44 ≈ 32; 10% attrition rate = 32/0.9 ≈ 36; d = 0.2 which means 36 children per arm.
Implementation Plan
Enrollment of children with ALL will be done after screening for eligibility and obtaining written informed consent following inclusion and exclusion criteria. The children’s sociodemographic details along with clinical variables such as diagnosis, duration of illness, age of onset, relevant history of medical diagnosis, and family history will be recorded using a semi-structured proforma. After recruitment, children will be randomized into 2 treatment groups. Group I-Patients (n = 36) will receive TAU plus expressive art therapy (EAT) and Group II-Patients (n = 36) will receive TAU. Both groups will be psycho-educated briefly in the beginning. In Groups I and II, TAU will involve antibiotics, analgesics, and nutritional support along with standard cancer care treatment. Subsequently, baseline assessment will be done using the CI‑Ped Onc QLQ 31 scale for QoL, STAI-C for anxiety, the Rosenberg Self-Esteem Scale for self-esteem, and PSS for stress. Group I children will be made to first listen to a music track, dance, and movement, or any other form of visual art session in a group session just before the personalized intervention in order to address their activation levels (baseline level of anxiety, stress, pain, and mood states) and familiarize with the expressive art sessions followed by the personalized sessions which were 45-60 minutes long twice a week. These sessions will be carried out over a period of 12 weeks (24 sessions) in the children admitted to the pediatric oncology ward for hematological cancer care. Clinical assessment will be done at baseline, after 6 and 12 weeks using the CI‑Ped Onc QLQ 31, STAI-C, PSS, and Rosenberg Self-Esteem Scale to assess the reduction in symptom severity and recovery process. The Standard Protocol items: Recommendations for Interventional trials (SPIRIT) guideline is followed for reporting this study protocol. 21 The implementation strategy is summarized in Figure 1.
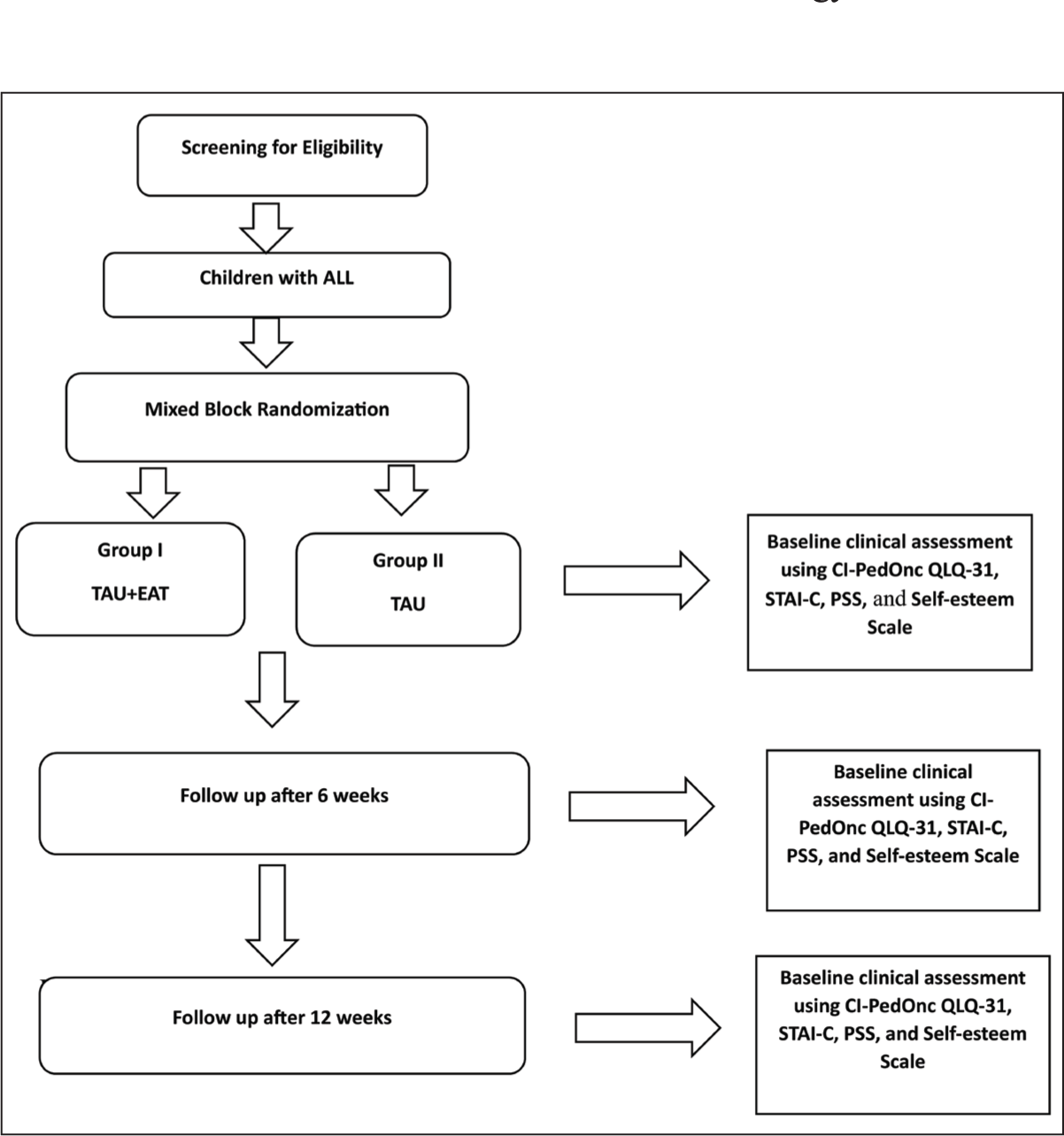
Statistical Analysis Plan
Descriptive statistics in the form of mean, median, range, frequency, percentages, and standard deviation will be used. Comparison of means of continuous variables within groups will be done using repeated measures Analysis of Variance (ANOVA) test and between groups will be done by 2-way ANOVA test. Fisher’s exact test and t-test will be used for categorical variables and continuous variables, respectively, for demographic data. Statistical analysis will be performed through an intention-o-treat approach using statistical software JAMOVI version 2.3.24, considering a significance level of <0.05 (2-tailed).
Timelines
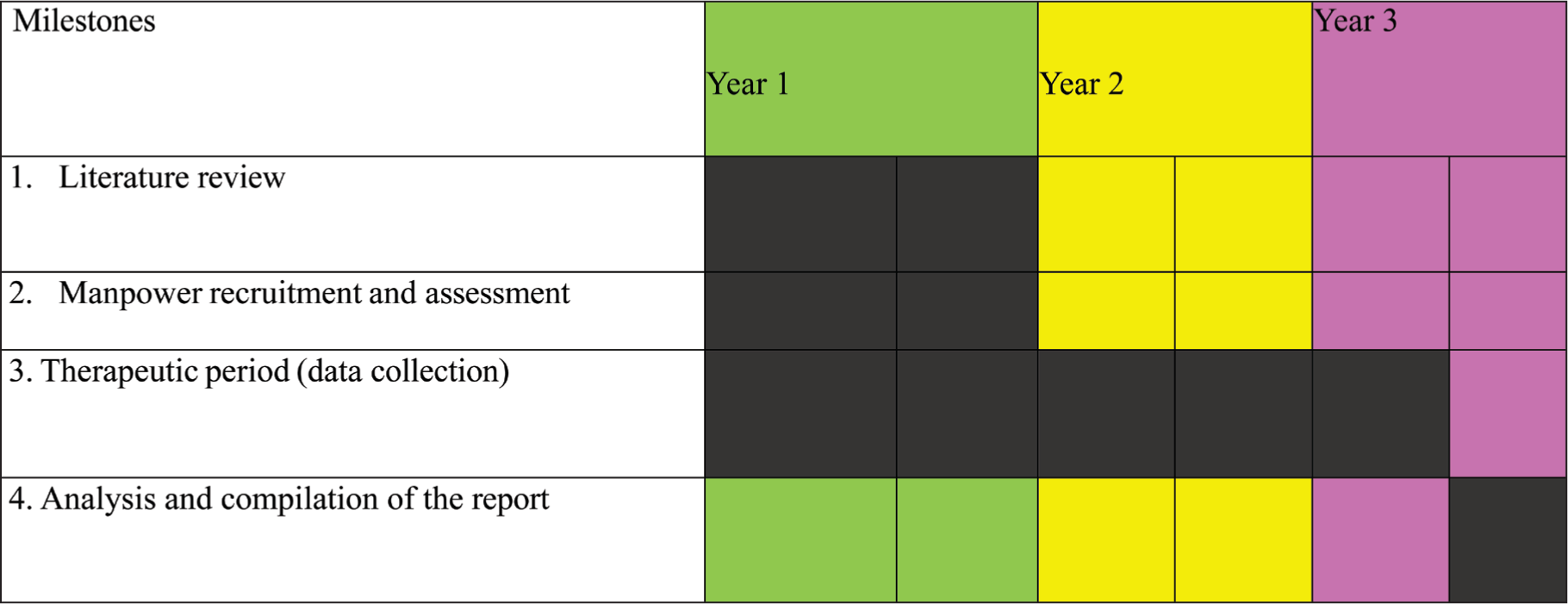
The phase-wise work to be done is illustrated in Figure 2. The work will be done phase-wise as in the following manner.
Timeline of the Study.
Literature Review (Year 1)
The thorough review of existing literature provides a better understanding of how creative art therapy is applicable in pediatric cancer care. This entails identifying research gaps, understanding specific emotional and psychological challenges faced by pediatric cancer patients, and examining how expressive art therapies (art, drama, music, movement, and dance) have addressed coping strategies and emotional expression. We followed the strategy to include the study population and intervention for our literature review. The following keywords or MeSH terms were used: [Neoplasms] OR [cancer] OR [childhood cancer] OR [pediatric cancer] AND [art therapy] OR [creative art therapy] OR [EAT]; filters: Child: birth-18 years, limited to English language only, from 1982/1/1-2024/12/28.
Manpower Recruitment and Assessment (Year 1)
For the smooth implementation of the therapeutic framework in first year, we will be hiring trained professionals such as psychologists and staff nurses who will be performing assessments and other support staff for data entry and other related tasks. We will be hiring an art therapist who has completed a certificate course in art therapy or a postgraduate diploma program in EAT or a child psychologist with experience in art therapy.
Therapeutic Period (Data Collection) (Year 1, 2, and 3)
During this phase, we will be focusing on data collection and intervention. Sessions of music-making, art, movement, dance, mindfulness, and painting will be tailored according to the specific developmental and emotional needs of children. Outcomes may include better resilience, enhanced social interaction, reduction in stress markers, and better emotional articulation.
Analysis and Compilation of Report (Year 3)
In the final 6 months, we will be analyzing the information gathered throughout the study and intervention phase to evaluate the efficacy of EAT in enhancing QoL amongst children with ALL.
Results
The study outlined here is merely a protocol, so no results to be presented at this stage. This protocol is intended to serve as a reference to illustrate the proposed investigation’s methods, design, and expected outcomes.
Discussion
The proposed study aims to evaluate the effect of EAT on psychological outcomes and overall QoL in children undergoing treatment for ALL. EAT can also be interchangeably called as creative art therapy. The underlying hypothesis is that when used in conjunction with standard medical care, EAT can enhance the QoL by improving anxiety symptoms and stress, boosting self-esteem and communication skills. Given the emotional toll and complications of cancer treatment, particularly in pediatric cancer, our study could provide important insights into alternative therapeutic approaches that complement conventional medical care in the pediatric population.
The use of EAT in cancer care is gaining popularity along with standard of care as cancer and its treatment interfere with a child’s normal development, activities, and social interactions through disfigurement, repeated school absences, losing contact with friends due to medical isolation from infections and other disruptions of daily activities. Children and their families see their daily lives disrupted and their overall QoL is widely affected. 22 Previous research has examined different forms of art, which include visual arts, music, and dance therapy in reducing psychological distress and improving QoL in cancer patients. However, the specific use of creative art therapy which combines multiple creative modalities (e.g., visual arts, dance, and music) in a personalized intervention, is a less explored topic in hematological cancer research. The next section highlights the distinctiveness of this approach by contrasting the current study with previous studies.
The General Impact of Art Therapy in Pediatric Oncology
Several studies have demonstrated that art therapy provides emotional and psychological advantages in pediatric oncology settings. For instance, a study was conducted (randomized controlled trial [RCT]) to assess the efficacy of group art therapy on QoL in pediatric cancer patients. They discovered that there are considerable impacts on social connections and psychological well-being. 12 Similarly, in an integrative literature review it was revealed that art therapy reduced depression and anxiety in children receiving treatment for cancer by fostering a positive environment. 13 The study demonstrated that creative arts therapy significantly improved the QoL in children with cancer by reducing anxiety, enhancing emotional expression, and fostering a sense of control during treatment. Participants reported increased joy and improved coping mechanisms. 23 All these findings are in line with the proposed hypothesis of the study, which implies that art therapy has the potential to help reduce psychological distress while simultaneously improving QoL. Earlier studies primarily focus on particular types of art forms, for example, visual arts and music therapy, on the other hand, our study uniquely investigates art therapy as a combination of multiple creative expressions, such as visual arts, dance, and music. This comprehensive approach is expected to offer a greater therapeutic effect, as it engages several emotional and cognitive pathways, allowing children to express a wider range of expressions and emotions. Previous studies did not investigate this multidimensional approach.
Psychological Effects of Art Therapy in Children with Cancer
A major psychological concern in pediatric cancer patients is anxiety. A systematic review of creative art therapies discovered that these programs helped lower stress and anxiety, which are prevalent in children receiving cancer treatment. 24 A study discovered that creative art therapies improved mood, and there was a reduction in anxiety in cancer patients. 25 These findings highlight the importance of art therapy in helping children cope with anxiety throughout the treatment process. While prior studies have largely concentrated on anxiety, the current study expands their focus beyond anxiety to include a wide range of psychological dimensions such as stress, depression, and self-esteem. The use of comprehensive assessment tools such as PSS, Rosenberg Self-Esteem Scale, and STAI in conjunction with the QoL, provides a more holistic understanding of the psychological consequences of art therapy. Furthermore, this study focuses on the long-term impact (6–12 weeks) of EAT, unlike previous studies, which focus on short-term impact.
Improvement in Quality of Life and Treatment Adherence
EAT has the potential to improve QoL and treatment adherence. A study showed that creative arts therapy improved QoL and coping in pediatric brain tumor patients, particularly by reducing anxiety and enhancing mood, which helped children tolerate chemotherapy and other painful procedures. 14 Similarly, a study highlighted that dance and movement therapy helped children with cancer develop better emotional regulation and coping mechanisms, which are crucial during long-term cancer treatment. 16 Dance and movement therapy along with music is another form of intervention in expressive arts that helps the child kinesthetically process their treatment and have an outlet for their emotions, thus helping with their sense of self and better QoL. 26 While these studies focused on specific forms of creative therapies, our study introduces EAT as a broad-based intervention, combining visual arts, dance, and music.
Long-term Follow-up and Implementation Strategy
Existing research in pediatric cancer care frequently assesses the short-term or immediate impact of art therapy. In this regard, studies demonstrated immediate enhancement in QoL and other psychological problems but fails to offer comprehensive follow-up data. 27 Our study takes an innovative approach by analyzing intervention outcomes every 6 and 12 weeks, which provides a detailed understanding of how sustained participation in EAT impacts the overall QoL in children with pediatric cancer. This extended examination allows a more subtle understanding of how therapy influences long-term emotional adjustments and QoL in children with ALL, which are frequently lacking in prior studies.
Innovative Methodological Approach
Most studies in this area have focused on longitudinal study designs with changes in anxiety or depression scores. However, the current study combines a rigorous study design (randomized clinical trial) with a 2-component therapeutic intervention that is a creative art and body movement/dance intervention, creating a robust framework for evaluating the effectiveness of EAT. The study’s use of a RCT design, along with comprehensive QoL and psychological assessments, further strengthens its methodological rigor compared to previous studies, which often lacked such robust designs. This study’s multimodal intervention, combining artistic expression with standard oncology care TAU, and its rigorous outcome measures (such as CI-Ped Onc QoL-31, STAI-C, and PSS-10) set it apart from prior studies. The novel inclusion of a 2-component therapeutic intervention that is, creative art and body movement/dance interventions tailored to the child’s emotional needs make this approach particularly innovative and comprehensive. Expressive art therapies have been found to reduce psychosocial distress thus improving QoL in children by allowing the expression of good and bad extreme experiences of cancer, producing, and sharing legacy, reducing stigma for both cancer and psychological intervention, ameliorating loneliness by expressing fear of sadness in the expressive art, allowing children in finding meaning of cancer experiences which increases strength and resilience. 28
Implication of the Study
This study aims to highlight the potential of EAT as an effective adjunct to standard treatment or TAU for children with ALL. Combining nonverbal therapies like music, movement, and art, it provides a culturally sensitive approach to improving resilience, self-esteem, stress, and anxiety in children. The findings could expand current psychosocial support methods in pediatric oncology, particularly in settings where conventional therapies such as CBT are less effective. Additionally, it also offers a model for applying innovative methods in resource-limited environments, boosting children’s self-esteem, reducing anxiety, and lowering stress thus improving the overall QoL in pediatric cancer patients. Previous studies demonstrate that creative art therapy has the potential to alleviate emotional barriers. This would aim to correlate our findings with previously available research, while possibly contributing new insights to pediatric cancer care. Thus, the outcome of our study could inform the broader adoption of such therapies across various pediatric cancer populations.
Challenges
The study may experience challenges in participant recruitment and retention, notably due to high attrition rates from illness and treatment adverse effects. Cultural variations could impact children’s willingness to participate in nonverbal therapies such as expressive art. Psychological assessments relying on self-reports may be inaccurate, particularly with younger children. Incorporating EAT with standard treatment schedules could be logistically challenging in the oncology ward. Parental acceptance of art therapy may vary, affecting children’s involvement. Last, sustaining long-term follow-up may be challenging due to health setbacks and complications in participants.
Expected Outcomes
The study aims to reduce anxiety and stress while boosting self-esteem and overall QoL. By integrating diverse art forms and considering cultural aspects, the study aims to provide useful insights into the effectiveness of EAT in pediatric oncology. The results will contribute to the expanding body of evidence supporting creative therapies in pediatric cancer treatment.
Footnotes
Acknowledgements
This study is supported by ICMR, Government of India (ICMR Grant-Sanction Letter No. EM/DEV/SG/ID-7139/2023). The authors also thank Project staff members and clinical and child psychologist of department of psychiatry, AIIMS Patna.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Patient Consent/Assent
The authors certify that they will obtain written informed consent from parents and verbal/written assent from children with ALL. The participants name and initials will not be published and due efforts will be made to conceal their identity.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support for the research from ICMR, Government of India (ICMR Grant-Sanction letter no. EM/DEV/SG/ID-7139/2023).
Statement of Informed Consent and Ethical Approval
The study is accorded Ethical Committee Approval vide Ethics Committee (Institutional Ethics Committee, All India Institute of Medical Sciences, Patna) No. AIIMS/Pat/IEC/2023/1145; dated 06.10.23. Written informed consent will be taken from all the participants. The study will be carried out in accordance with the principles as enunciated in the Declaration of Helsinki.
Clinical Trial Registration No.: CTRI/2023/11/059742 (registered with the clinical trials registry of India).
