Abstract
Introduction:
Treatment of childhood-onset schizophrenia (COS) and related psychotic disorders is challenging due to limited options for effective interventions. Transcranial direct current stimulation (tDCS) is a noninvasive neuro-modulatory technique found to be safe and effective in adults for treating symptoms of schizophrenia. There is a paucity of studies of tDCS in adolescents regarding its safety and tolerability. We studied the tolerability of add-on tDCS in adolescents with schizophrenia and other psychotic disorders.
Material and Methods:
Fifteen adolescents with a diagnosis of schizophrenia and other psychotic disorders (ICD 10) with a mean age of 15.25 years, refractory to an adequate trial of antipsychotic and CGI-severity score of 4 (moderately ill) or more were recruited. All patients received 10 sessions of tDCS (2 mA current) with sponge electrodes of 25 sq cm. Adverse effects were measured during each session using a tDCS adverse effect questionnaire, local, general physical, and mental state examination.
Results:
The most common adverse effects reported were itching and burning sensations (33.3% each) followed by tingling sensation in 20% of subjects at the site of electrode application. Among study subjects, 86.6% had at least one adverse effect, and in 80%, the reported adverse effects were mild. 60% of the reported adverse effects were probably due to tDCS.
Conclusion:
Adverse effects reported with tDCS are transient. tDCS was tolerable, safe, easily acceptable, and feasible to administer in adolescents with schizophrenia or other psychotic disorders admitted to the hospital.
Keywords
Introduction
Childhood-onset schizophrenia (COS) has an onset before the 13th birthday and an incidence of less than 0.04%, which rises steeply during adolescence.1,2 The onset of schizophrenia during childhood or adolescence can have a profound impact on the developing brain causing neuro-developmental deficits. The onset of schizophrenia in childhood is characterized by moderate to severe impairment across the lifespan and prognosis is overall worse when compared to its adult counterpart.3–5 Treatment of COS and related psychotic disorders is challenging due to limited options for effective intervention. Antipsychotic medication, especially second-generation antipsychotics (SGAs), is the mainstay of treatment, but they have limited efficacy and are associated with adverse effects, such as age-inappropriate weight gain, obesity, hypertension, lipid-glucose abnormalities, and higher cardiovascular morbidity.6–9 Electroconvulsive therapy (ECT) and transcranial magnetic stimulation (TMS) are not commonly used in children because of safety concerns. Transcranial direct current stimulation (tDCS) is one emerging novel intervention.
tDCS is a non-invasive neuro-modulatory technique, acts polarizing resting membrane potential of neurons. When tDCS is applied to the human brain, anodal stimulation enhances cortical activity and excitability while cathodal stimulation can have opposite effects. 10 Encouraging reports are available for their effectiveness in reducing auditory hallucinations, negative symptoms and improving cognitive functions in adults. 11 In addition, when studied in adults it is found to be safe and tolerable without causing any serious adverse effects. 12 However, there is a scarcity of studies evaluating the tolerability of tDCS in the pediatric population. In the only study of tDCS in COS by Mattai et al., mild side effects, such as tingling and itching over the scalp and fatigue were commonly reported. 13 tDCS evaluated in a few other studies with cerebral palsy, dystonia, language disorder and neurotypical children did not report any serious adverse effects.14–17 We conducted this study to assess the tolerability of tDCS used as add-on to ongoing treatment in adolescents with schizophrenia or other psychotic disorders in the Indian setting.
Materials and Methods
We aimed to study the tolerability of tDCS in adolescents with schizophrenia and other psychotic disorders with partial or no response to antipsychotic treatment by evaluating adverse effects in a systematic way. The study had the approval of the institutional ethics committee and it was conducted at the child and adolescent Psychiatry inpatient facility of the National Institute of Mental Health and Neuro Sciences (NIMHANS), Bengaluru, which is a tertiary mental health care center catering to a diverse population.
All adolescents ( <18 years) with schizophrenia and related psychotic disorders who had received an antipsychotic trial in adequate doses for at least six weeks were consecutively assessed for study criteria before selection. Sixteen adolescents having ICD 10 diagnoses of schizophrenia or other Psychotic disorders with CGI-severity score of 4 (moderately ill) or more were recruited after obtaining informed consent from parents and assent from adolescents wherever possible. All subjects were right-hand dominant assessed by the Edinburgh inventory. 18 Patients with intellectual disability, pervasive developmental disorders, central nervous system disorders, seizure disorders and those deemed uncooperative for tDCS were excluded from the study. The psychiatric diagnosis was made by clinical interview of caregiver and adolescent using MINIKID - The Mini International Neuropsychiatric Interview (English version 6.0). 19 Fifteen adolescents including eight girls within the age range of 13-17 years (mean 15.25) and mean age 15.25 years (SD 1.34) completed all 10 sessions of tDCS. A substantial portion of the sample had onset before 13 years of age with the mean age of onset being 12.06 years (SD 2.43) and treatment initiation at 12.31 years (SD 2.18). A total of 43.8% of the sample was currently attending school and an equal number had dropped out. One participant with a comorbid diagnosis of undifferentiated schizophrenia and obsessive-compulsive disorder dropped out after the first session due to restlessness and low sitting tolerance as part of his psychopathology, unrelated to tDCS treatment.
tDCS adverse effect questionnaire by Brunoni et al. was used for the systematic recording of tDCS-related adverse effects which includes 16 items covering all the symptoms reported earlier in the studies. 20 All the assessments were done before starting and after completion of 10 sessions of tDCS. A protocol was developed to manage scalp pain/irritation during the session to facilitate uniformity in handling such side effects. Special precautions were taken to prevent severe adverse events by daily monitoring of vital signs, local area examination and mental status examination.
tDCS Study Protocol
All the subjects were administered tDCS using Neuroconn DC Stimulator Plus (
All study subjects received two sessions of tDCS, each lasting 20 minutes spaced by three hours in a day. Overall, 10 sessions of tDCS over five consecutive working days were administered. A head bath was given in the morning before the first session. No changes in antipsychotics or the addition of any medication were made during this treatment process. During each session, an available parent or caregiver accompanied the subject and a few subjects requested for both parents to be present with them, which was permitted for the smooth conduct of the session.
Results
The most common diagnosis among study subjects was paranoid schizophrenia (56.3%) followed by undifferentiated schizophrenia (18.8%), severe depressive episode with psychotic symptoms (12.5%), schizoaffective disorder (6.3%) and persistent delusional disorder (6.3%). The most common psychopathology in the sample was negative symptoms (93.8%) followed by the presence of delusions, hallucinations, and cognitive complaints in equal numbers (87.5%) measured by the Brief Psychiatric Rating Scale for Children (BPRS-C). 22 The most common type of delusion was persecutory (75%) and hallucinations were auditory (87.5%) measured by Scales for Assessment of Positive and Negative Symptoms (SAPS and SANS).23,24 The average number of adequate antipsychotic trials received by subjects in the study population was 2.37 (SD 0.99), the most common being Risperidone with a mean dose of 4.18 mg (SD 1.47) by 69.3% subjects and Olanzapine 15.36 mg (SD 5.85) by 44.1% subjects. At the time of study participation, 37.8% of the subjects were receiving two or more antipsychotics, the most common being Clozapine 300 mg (SD 189.57) by 31.5% of subjects.
Tolerability of tDCS
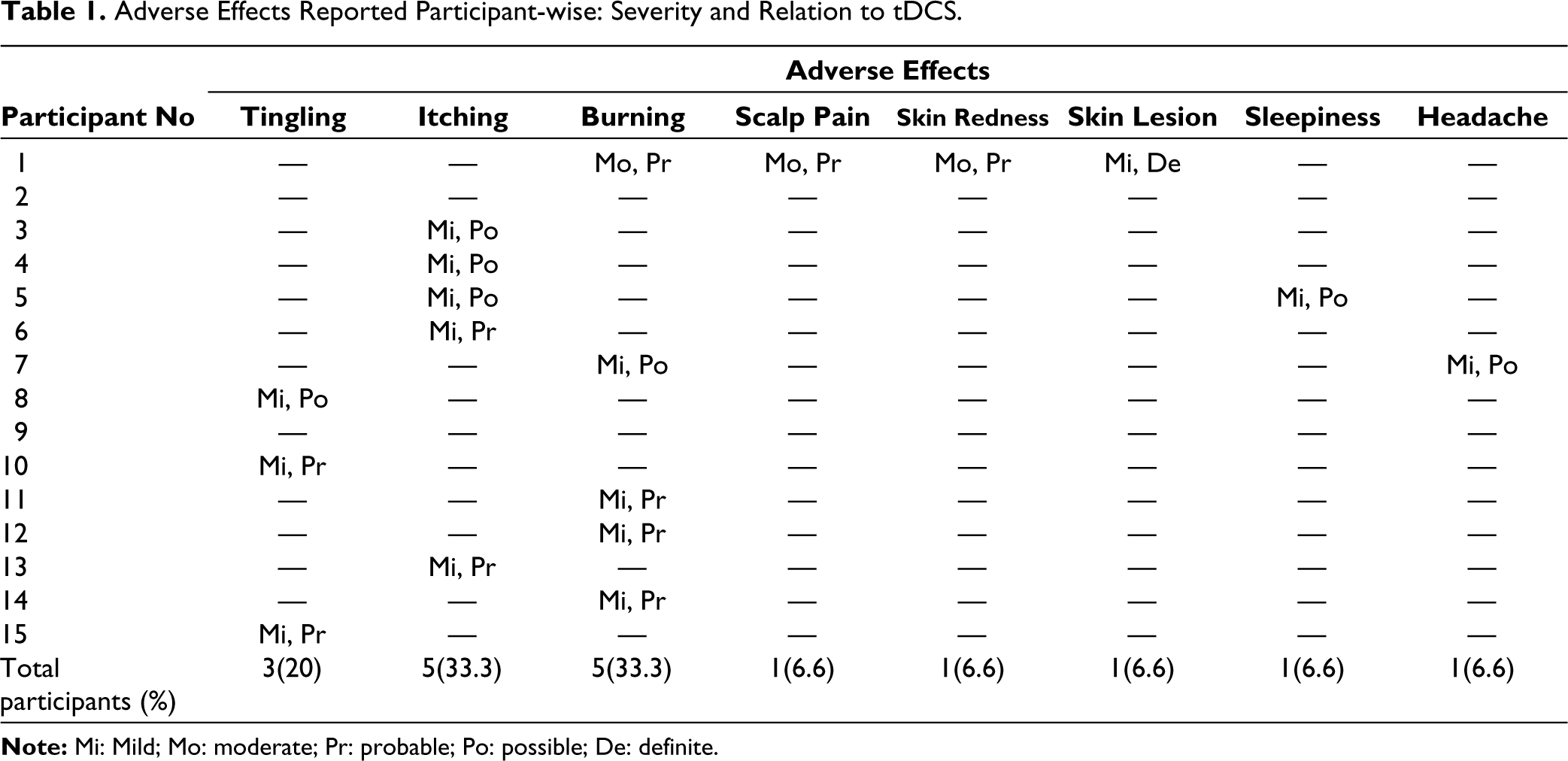
Among the study subjects, 86.6% had at least one adverse effect with tDCS, the most common being an itching and burning sensation (33.3% each) followed by tingling sensation (20%) at the site of electrode application. Scalp pain, skin redness, sleepiness, and headache were reported by one subject each. A superficial, non-painful, non-itchy papule (superficial burn) was seen at the electrode site over the scalp in one subject after the first session, which resolved without any treatment in three days. In subjects reporting a burning sensation, it was transient and disappeared after reassurance, hence the same strength was continued, which was tolerated well in subsequent sessions. None of the subjects required a reduction in current intensity due to adverse effects or reported any severe adverse effects requiring discontinuation of tDCS. Novel adverse effects, such as significant behavioral change, memory impairment, switch to hypomania or mania, involuntary movements, seizures and myoclonic jerks were not reported by any of the subjects during the mental status examination and daily assessments.
When it comes to characterization of severity of adverse effects, 80% had mild and 6.3% had moderate adverse effects. Of the reported adverse effects, 60% were probably due to tDCS and adverse effects such as headache and sleepiness were not regarded as due to tDCS by parents. Adverse effects were more common in the initial sessions of starting tDCS and mild to very mild in nature (initial few seconds or minutes only) as illustrated in Table 1. The adverse effects reported were most common in the first three sessions of tDCS, with 80% of the subjects reporting in, each session most common being itching and burning sensation followed by a tingling sensation. The reported adverse effects started declining after the third session and only four subjects reported tingling, itching, and sleepiness in the last session.
Adverse Effects Reported Participant-wise: Severity and Relation to tDCS.
Discussion
The most common adverse effects reported in our study were mild and these findings are similar to the earlier study of COS by Mattai et al. 13 At least one adverse effect was reported by 13 subjects (86.6%) in our study. This high incidence of adverse effects reports may be attributed to our methodology of recording adverse effects, wherein, we included adverse effects not only spontaneously reported by patients but also after enquiry using the questionnaire. Nearly 50% of those who reported adverse effects had very mild or minimal degree of distress which is not possible to record separately in the tDCS adverse effect questionnaire proposed by Brunoni et al. This pattern of adverse effects is similar to those reported by earlier studies in children.13,25 It is difficult to ascertain the causality of adverse effects like headache and sleepiness with tDCS as they were not regarded as due to tDCS by parents and could be because of other factors of illness or medication treatment itself. The adverse effects were reported more in initial sessions compared to later ones, indicating that the patients may develop tolerance to adverse effects or the reduction of anxiety related to the procedure might have played a role.
All the parents of adolescents meeting the study criteria and approached by us consented to study, and 87.5% of the adolescents also provided assent for the study. Also, none of the participants discontinued or withdrew consent due to the adverse effects. This shows that tDCS is easily acceptable to parents and adolescents with psychotic disorders, possibly due to the ease of administration of tDCS and non-invasiveness of the intervention. During tDCS session few subjects requested for both parents to be present with them which was permitted for smooth conductance of session in presence of a doctor. Continuous monitoring was ascertained for discontinuation of stimulation process due to body movements, especially common in this group of symptomatic adolescents. Subjects also needed to be frequently reassured about adverse effects and remaining time for completion of session. This indicates that administration of tDCS is feasible in adolescents with psychotic disorders with some modifications made to meet developmental needs.
This study has its own limitations in the form of possible bias in noting or reporting adverse effects and lack of a sham control arm. Administration of tDCS in adolescents is not common, hence a priori small sample size calculation was not done, and small size of this study limits generalization of the findings. However, this study showed that in adolescents with schizophrenia and other psychotic disorders admitted to the hospital, tDCS was tolerable, safe, easily acceptable, and also feasible to administer with some modifications meeting the developmental needs.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was accorded Ethical Committee Approval vide NIMHANS Ethics Committee, NIMHANS, Bengaluru No.NIMH/DO/BEHV.SC.DIV/2015 dated 17/2/2015. The study was carried out in accordance with the principles as enunciated in the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research Grant (SR/CSI/158/2012) to Ganesan Venkatasubramanian is supported by the Department of Science and Technology (Government of India) and Venkataram Shivakumar by Indian Council of Medical Research Young Scientist Research Grant. Anushree Bose is supported by the Wellcome Trust/DBT India Alliance and Harleen Chhabra by the Department of Biotechnology, Government of India.
Informed Consent
Written informed consent was taken from all the participants.
