Abstract
Background:
Autism spectrum disorder is a neurodevelopmental disorder of childhood in which there is a deficit in social and communicative skill, and the child shows restricted and repetitive behavior. For children diagnosed with autism spectrum disorder, family-mediated intervention and early intensive behavioral intervention are considered standard of care. The study aimed to compare their efficacy on social skills and other developmental domains in our cultural context.
Methods:
This study was a parallel-arm, open-label, randomized active-controlled clinical trial. Fifty children diagnosed with autism spectrum disorder were randomly assigned to either the family-mediated intervention or early intensive behavioral intervention groups. Communication DEALL Developmental Checklist was applied at 0, 12, and 24 weeks.
Results:
The difference in the mean value of Communication DEALL Developmental Checklist at the end of 24 weeks from the initial value was significantly better in the family-mediated intervention as compared to the early intensive behavioral intervention group in social (mean difference = −16.8, 95% CI = −27.18, −6.49, p = .002) as well as fine motor, cognitive, and overall development. In other developmental domains of Communication DEALL Developmental Checklist, the family-mediated intervention group performed better in comparison to the early intensive behavioral intervention group at different time periods although not statistically significant.
Conclusion:
The results of the study indicated that, throughout the course of 24 weeks of treatment, autism spectrum disorder children in family-mediated intervention showed a distinct improvement in major developmental domains. Family-mediated intervention can be advocated for autism spectrum disorder children in our cultural context in view of improved functioning in major developmental domains in the background of our study findings.
Keywords
Introduction
Neurodevelopmental disorders begin in children during the developmental years and affect the developmental domains. 1 Autism spectrum disorder (ASD) is classified as a neurodevelopmental disorder in current nosology, with core features comprising deficits in speech, language, and social skills, as well as fixated interests. Poor eye contact, lack of social smile, delayed responding, disturbed peer interaction, hyper or hyposensitivity to sensory stimuli, and repetitive, restrictive behavior are some of the features of ASD. Mostly, the diagnosis of ASD is made after three years. 2 One percent of children below 10 years of age are affected by ASD, 3 and worldwide, the prevalence is between 0.60% and 1.00%. 4 The occurrence of ASD is increasing at present. Various forms of psychological and pharmacological treatment are available for ASD. Applied behavior analysis, which uses techniques from learning theories, has been found to have significant positive results in ASD children. 5 Providing intervention to children as early as possible results in significant changes due to the neuroplasticity of the region of interest in the brain. 6 Early Intensive Behavioral Intervention (EIBI) is one approach to treatment that targets preschoolers and toddlers. The intervention is provided intensively in a clinical setting by a team of early interventionists. EIBI has been found to bring positive changes in core developmental domains, and it is beneficial with limited resources.5,7
In low-income countries like India, where there is a lack of proper infrastructure and specialized professionals, the focus of therapy should be on involving the parents or caregivers who will be motivated to provide therapy to their child. Also, the evidence-based studies are mostly from high-income countries. Family (parent)–mediated intervention (FMI) is one such approach to treatment that focuses on training parents or family members in therapeutic techniques so that they can provide therapy to their child even in a home setting. The parents in this situation become the primary therapist. Very few studies have shown that children have benefited where parents were the primary therapist. 8 In summary, both FMI and EIBI have adequate evidence as therapeutics for ASD children, but no direct comparison is attempted. Moreover, these data were not from rural India, which accounts for the majority of our country. Furthermore, in a state like Bihar, located in the eastern part of India, which has the lowest human development index in terms of a healthy life and a decent standard of living, it is of utmost importance to compare the therapeutic effects of both interventions in our cultural context. Thus, the study aimed to assess the efficacy of FMI, which was directly compared with the efficacy of EIBI using the Communication DEALL Developmental Checklist (CDDC) as an outcome measure in these children.
Materials and Methods
Institute Ethics Committee approval was obtained, and our study was registered with the Clinical Trials Registry, India (CTRI/2020/08/027099) before the recruitment of subjects for the study.
Study Population and Eligibility
Children between the age of two and six who were clinically diagnosed as per the DSM-5 with ASD and visited the neurodevelopmental clinic of the referral hospital were included in the study. The INCLEN Diagnostic Tool for Autism Spectrum Disorder (INDT-ASD) was applied to further screen the cases of ASD. Parents who were willing to commit to 24 weeks of therapy were included in our study. Naive children with no exposure to any form of therapy were included in the study. The study excluded children who were already diagnosed with genetic or neurological disorder, or notable motor, medical, or sensory impairments that would prevent them from taking part in the study.
Study Design, Intervention, and Setting and Procedure
The study was parallel design, open-label, active controlled randomized clinical trial. The study spanned over a period of 23 months (September 2021–August 2023). The study began with obtaining written informed consent from the parents. A detailed clinical and medical history was taken using a semi-structured proforma at baseline. By using mixed block randomization, the children were divided into two treatment groups with an equal allocation ratio. The codes were allocated to a series of random numbers, and the allocation process was kept confidential by utilizing an opaque sealed envelope with a serial number.
Fifty percent of ASD children received FMI, which was conducted in the hospital setting by a trained therapist as well as at home setting by a primary caregiver, and another half of the participants received EIBI, which was conducted in the hospital setting by trained therapists. The sessions were conducted at individual and group level with a focus on pre-requisite learning skills combined with the promotion of desired behavior, social skills, and speech and occupational therapy. During breaks, therapists worked on activities of daily living (ADL) like dressing, toilet training, and eating. The intervention in the EIBI group was for 3 h by therapists twice a week. Sessions in the EIBI group were conducted twice a week for 3 h, while in the FMI group, sessions comprised weekly for 3 h by early interventionists along with daily sessions by their parents or caregivers. The number of sessions conducted for EIBI was 48, whereas for the FMI group, the number of sessions conducted was 24 (Figure S1 and Table S1 provided therapy particulars obtained in both the FMI as well as EIBI group). Psycho-education was provided only in the EIBI group regarding the need for early intervention, but no hands-on training was given, whereas in the FMI group, hands-on training was given to the primary caregiver so that therapy could be conducted at home. A family member (mostly one of the parents) acted as the primary therapist and was trained in the techniques by the therapist, which they had to follow at home during the week. In the FMI group, parents were also requested to have audio–video sessions conducted at home to ensure adequate practice of the techniques and compliance with them. Change in developmental domains was checked by using CDDC at 0, after 12, and 24 weeks.
Study Objectives
Our study objectives aimed to compare the effect of FMI versus EIBI on social skills and other developmental domains in children with ASD using the CDDC.
Outcome Measures
The outcome measures comprised differences in social domain and other individual domain (gross and fine motor skills, ADL, receptive and expressive language, cognitive and emotional skills) scores as well as overall developmental domain scores in CDDC from 0 to 12 weeks, 12 to 24 weeks, and 0 to 24 weeks, respectively.
Tools Utilized for Measurement of Study Outcomes
INDT-ASD: It is an Indian-validated tool developed by INCLEN to diagnose autism based upon DSM-IV TR criteria. The age range of the tool is 2–9 years, and it contains two sections. It has 98% sensitivity and 95% specificity. 9
Semistructured proforma: It comprises the sociodemographic and clinical details of ASD children and their informants. Sociodemographic details include information related to age, sex, academic qualification, place of residence, caregiver relationship to the ASD children, and caregiver academic qualification. Clinical details include pregnancy duration, delivery type, history of any late talkers in the family, or presence of any genetic disorder.
CDDC: Karanth et al. 10 have developed a scale for the Indian population. The age range of the scale is 0–6 years. It assesses developmental skills in eight developmental domains, which are gross motor, fine motor, ADL, receptive and expressive language, cognitive skills, social skills, and emotional skills. It uses a 5-point Likert scale. 10
Sample-Size Determination
Karanth et al.,
10
in their study, reported an average CDDC social domain score of 33.7 ± 12.94 in Indian ASD children (2–6 years). We used this score as a guide to determine the sample size of our study. Considering a common standard deviation of 12.4 units with an assumed mean difference (effect size) of 11, with a level of significance of 5%, and a power of 80%, a sample size of 20 would be required for each group (
Data Analysis
JAMOVI version 2.3.24 was used for data analysis. The chi-square/Fisher’s exact test was used to compare the means of categorical variables between the groups. The independent samples t-test was applied to compare the means of continuous variables. Repeated measure ANOVA test was used to compare the change in CDDC scores within the group over time, and independent samples t-test was used to compare change in CDDC scores between groups. Intention-to-treat (ITT) and per-protocol (PP) approaches were used for analysis. A p <.05 was considered statistically significant.
Results
Participant Recruitment and Baseline Sociodemographic and Clinical Profile
Sixty-five children were screened who were diagnosed with ASD. Out of 15 children, eight were excluded because they were over six years of age, and the parents of seven children were not willing to continue therapy. Randomization was done for the remaining 50 ASD children who were allocated to either of the treatment groups. In the FMI group, one child did not continue therapy after three months due to logistic reasons, and one did not continue as parents were not motivated to follow therapy. Thus, 23 children in the FMI group and 25 children in the EIBI group were included in the PP analysis, while 25 children each in both groups were included in ITT analysis (Figure 1). However, no major differences in the results were observed in the ITT versus PP analysis due to only two ASD children dropping out of the FMI group in our study after randomization. Here, we present the ITT and PP analysis results, which are provided in the supplementary file (Tables S2 and S3).
CONSORT Flow Chart Illustrating Timeline of the Trial.
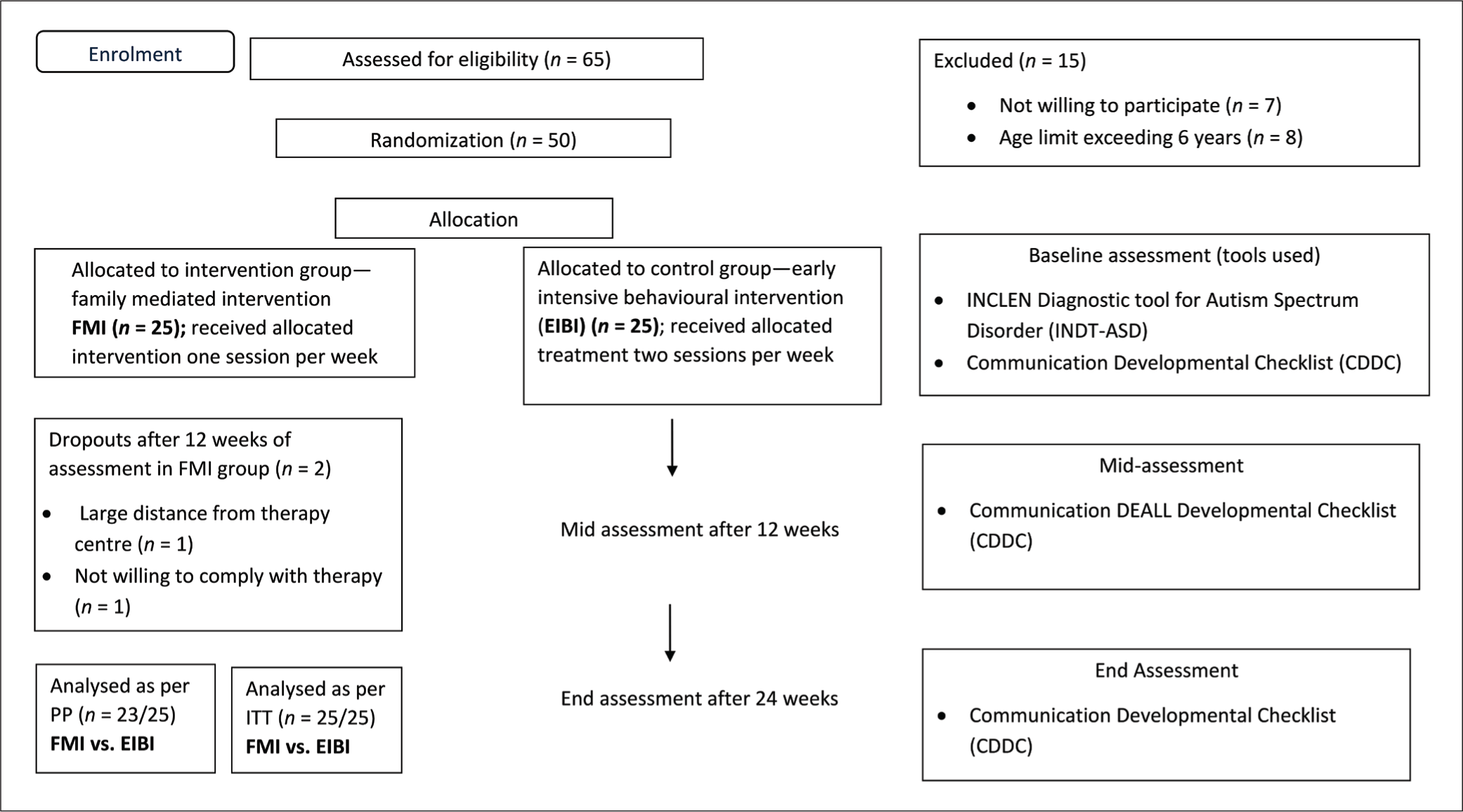
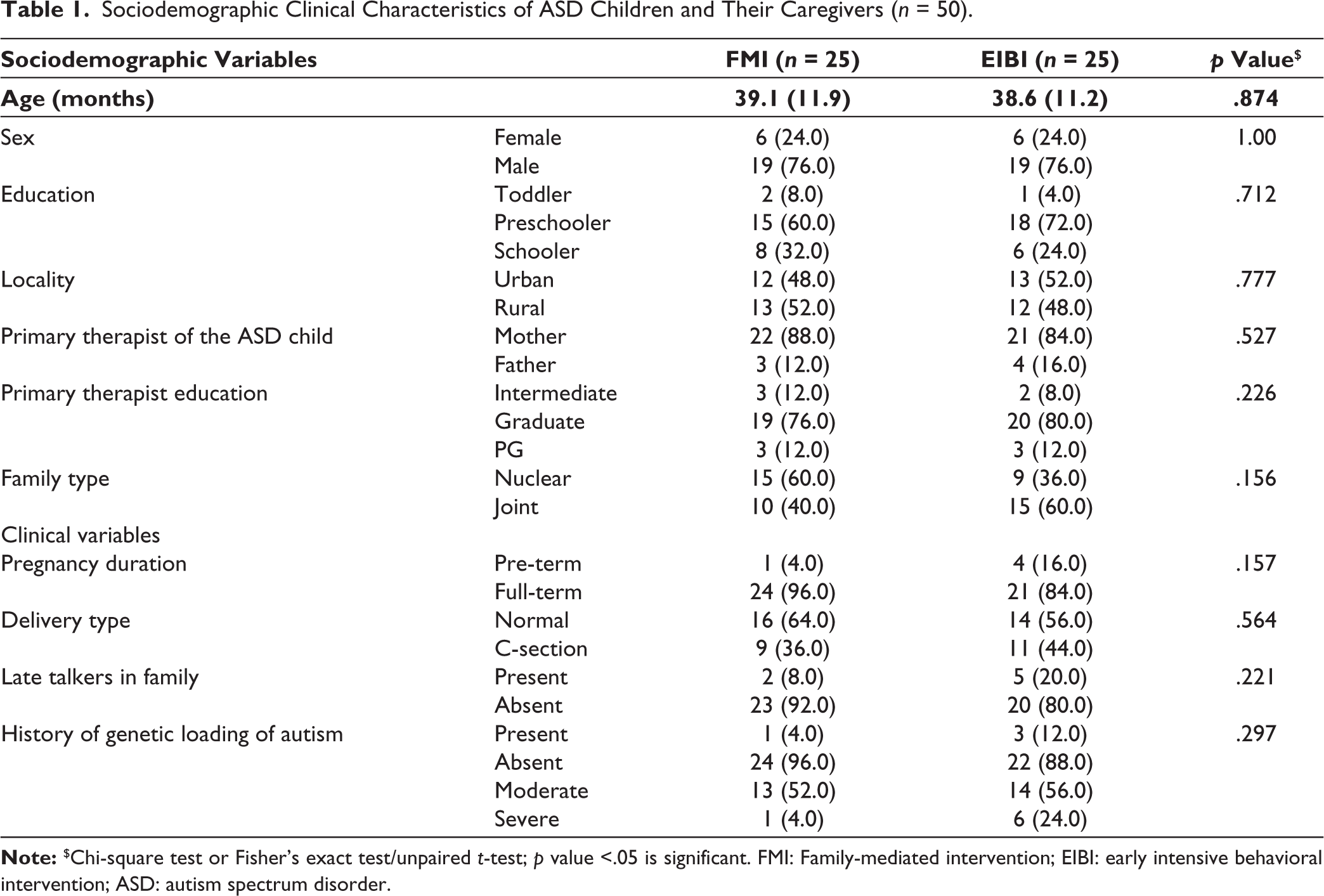
The sociodemographic and clinical profile of both groups was not significantly different during the initial evaluation (Table 1). The mean age of ASD children in FMI group was 39.1 ± 11.9 months; similarly, mean age of ASD children in the EIBI group was 38.6 ± 11.2 months. There were more males (FMI 76% and EIBI 76%) and preschoolers (FMI 60% and EIBI 72%) in both groups. Mother (FMI 88% and EIBI 84%) was the primary therapist in most of the cases in both groups and was educated up to graduation. Most of the children in both groups were full term (FMI 96% and EIBI 84%) and had normal delivery (FMI 64% and EIBI 56%).
Sociodemographic Clinical Characteristics of ASD Children and Their Caregivers (n = 50).
Change in CDDC Score
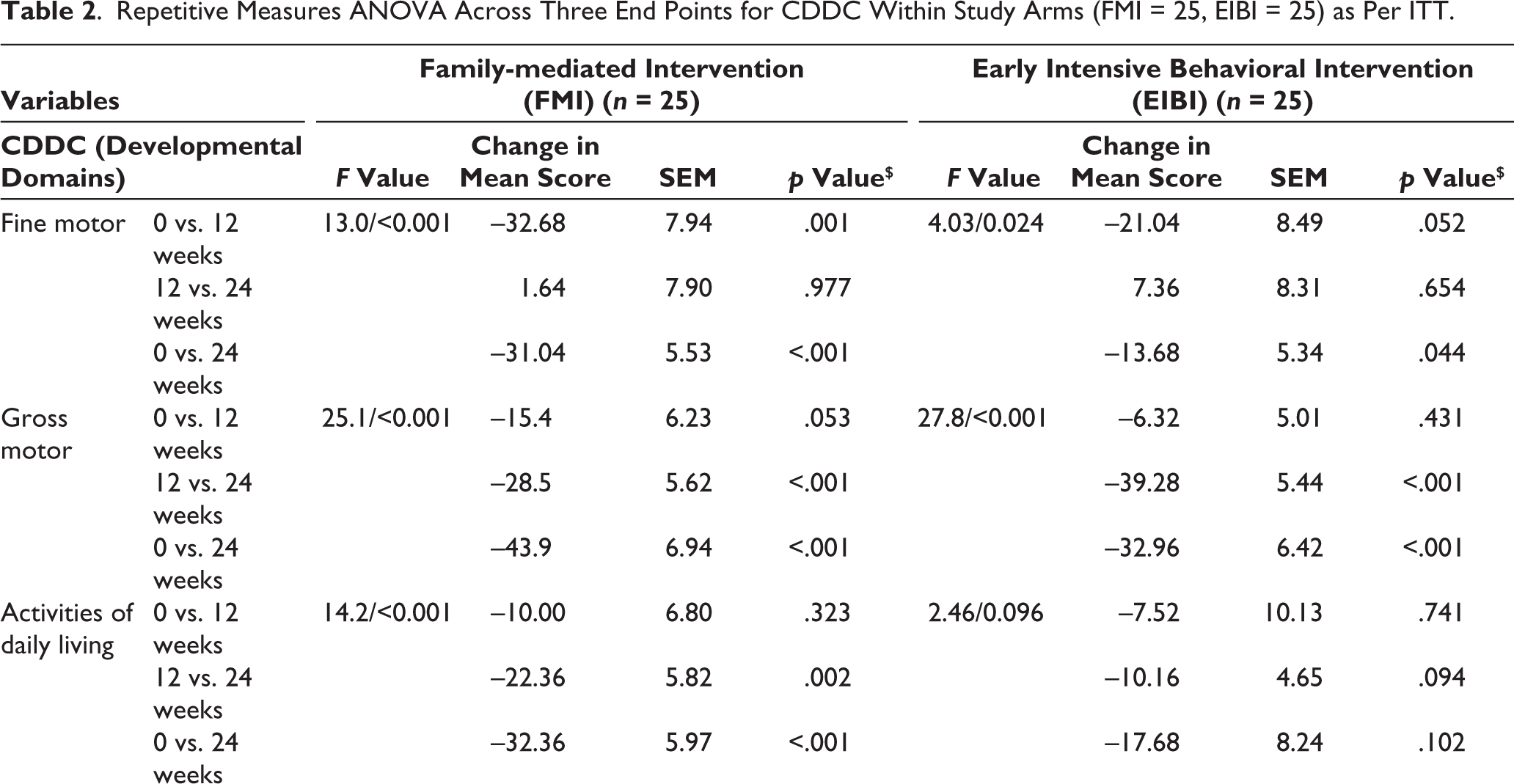
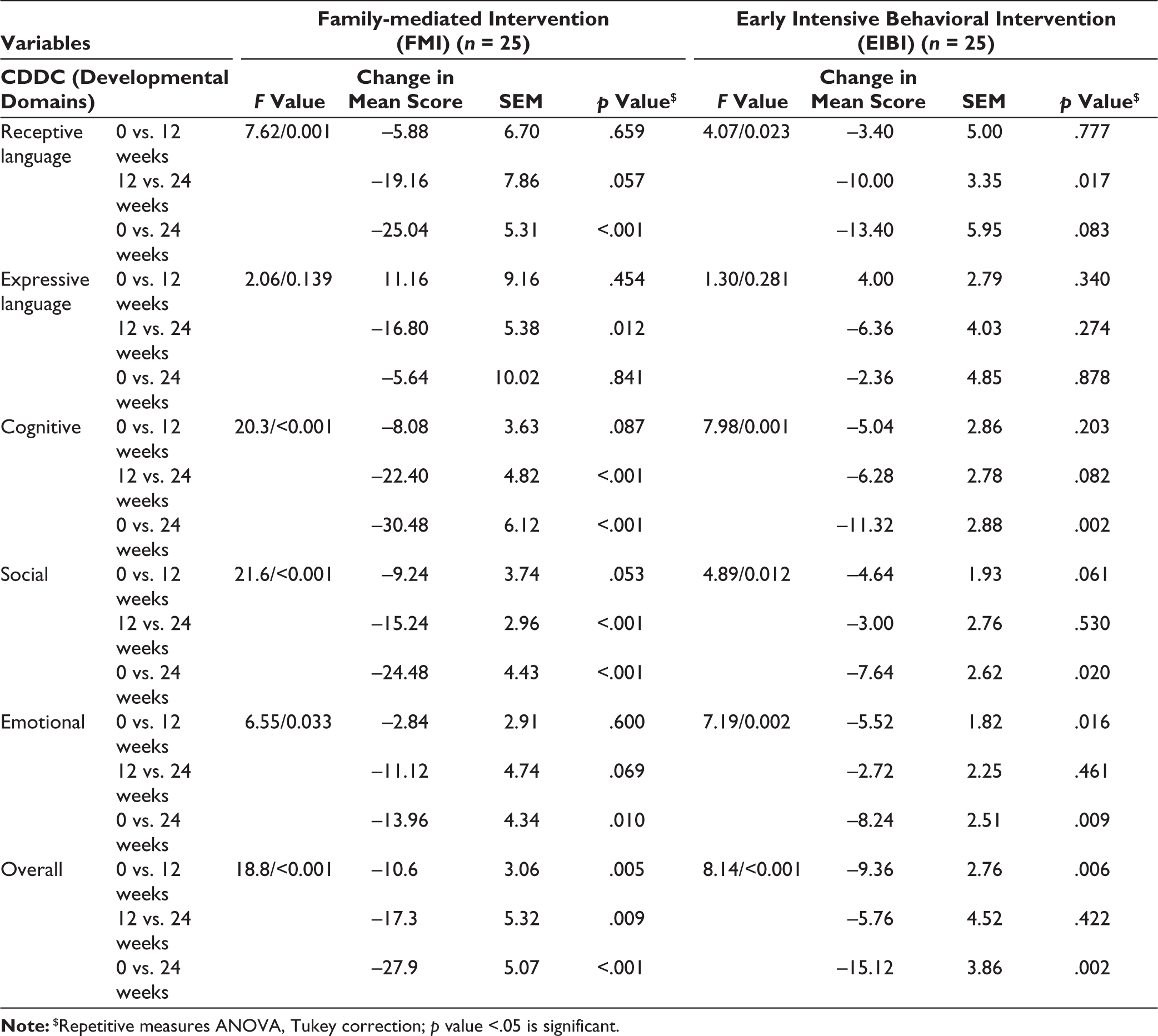
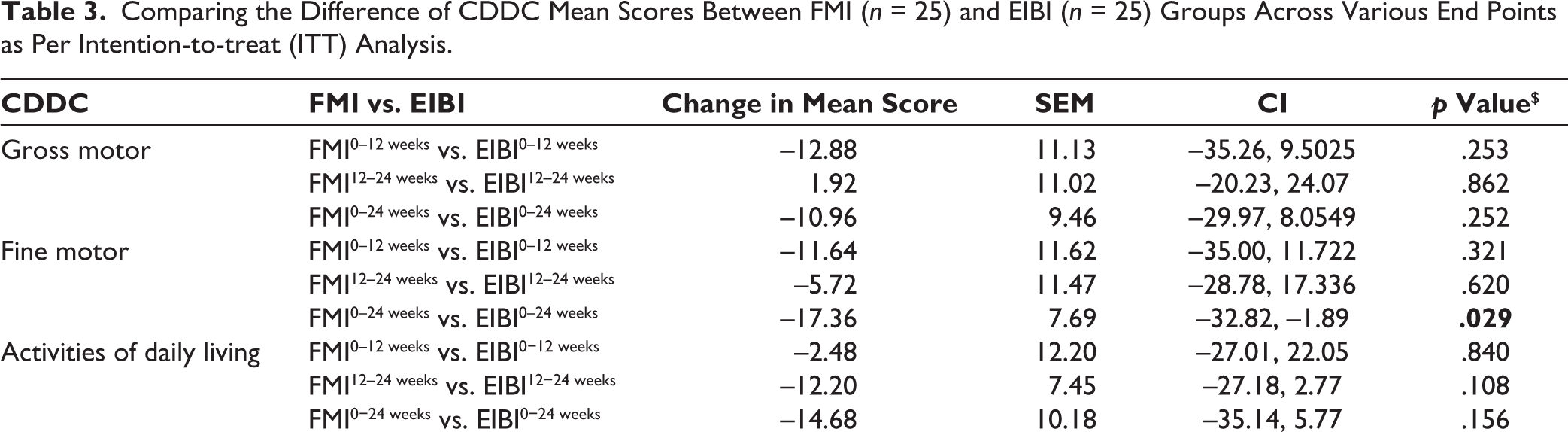
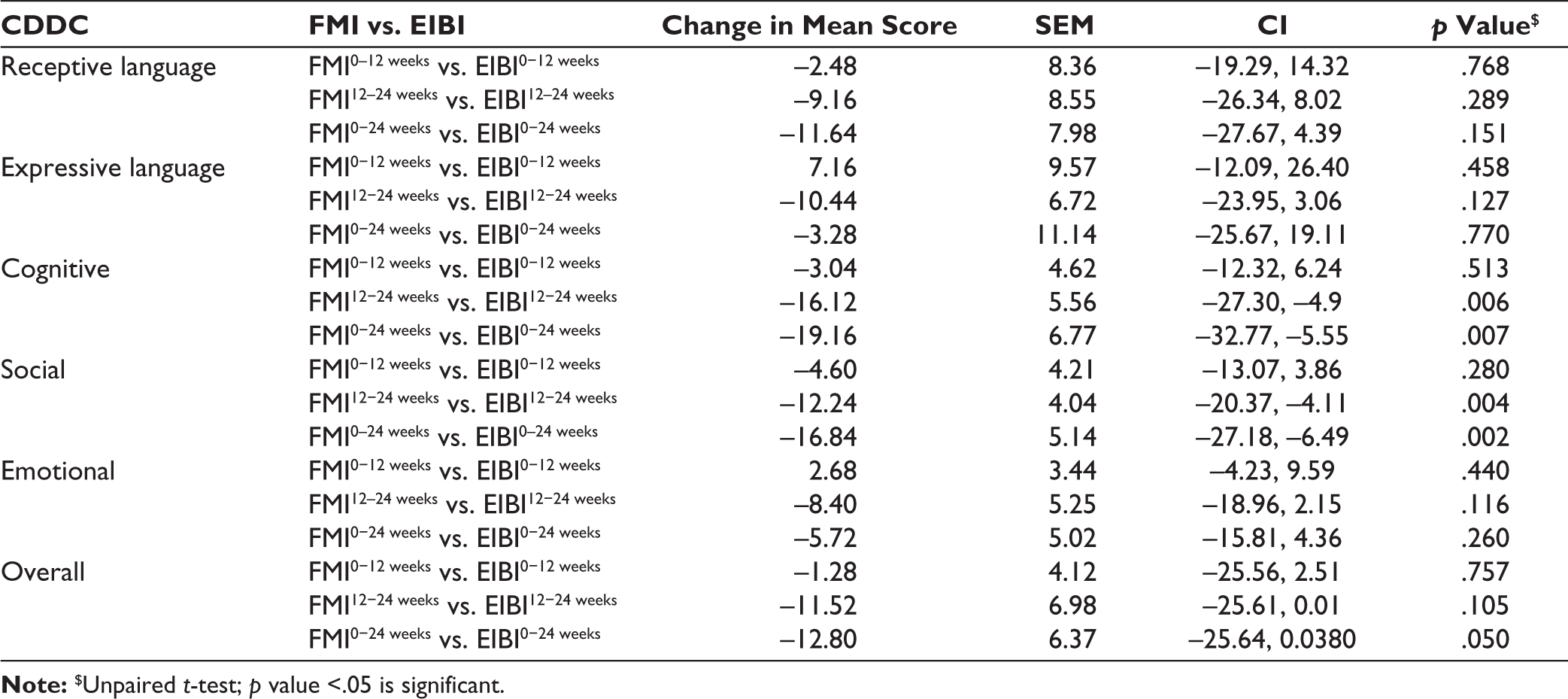
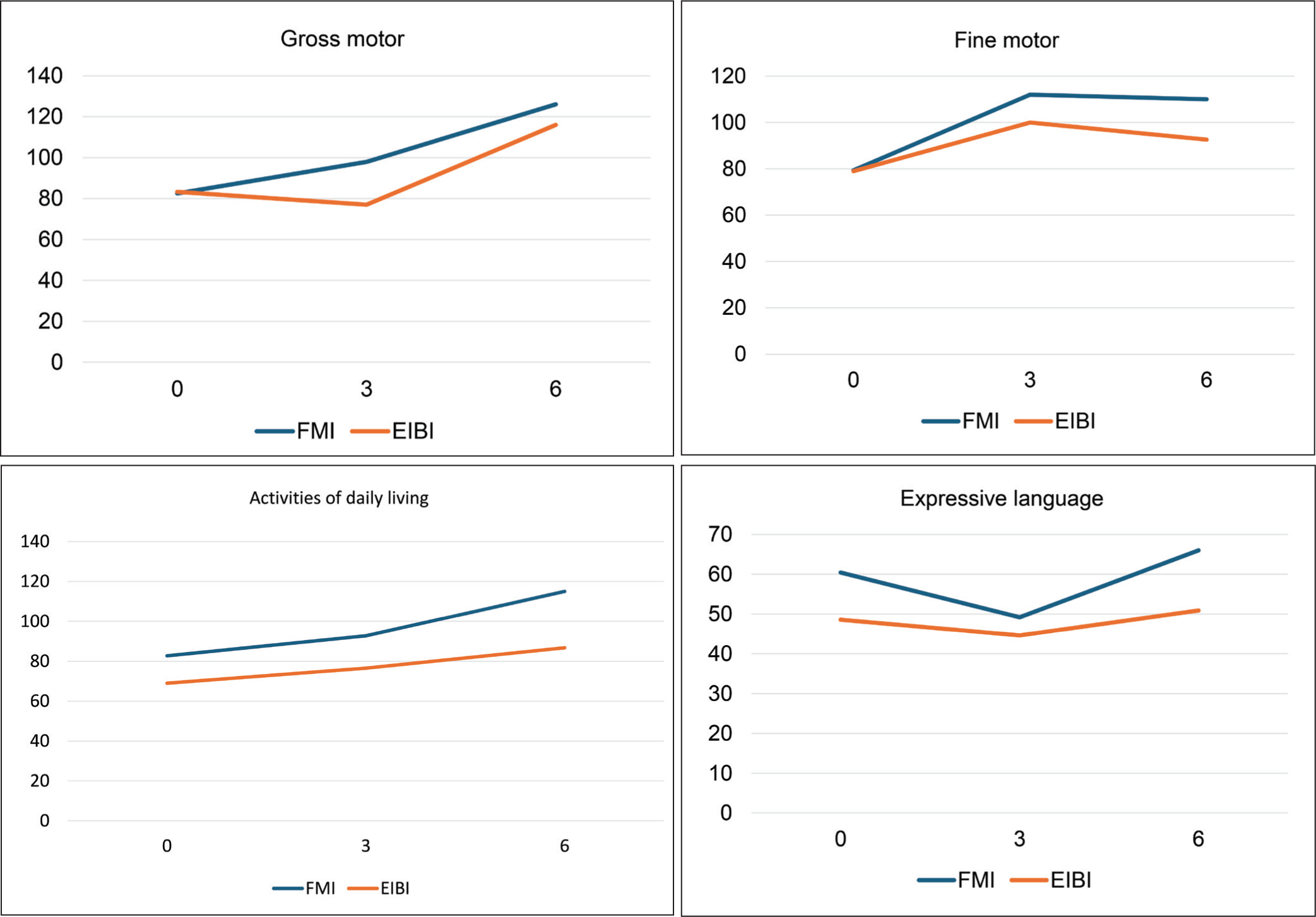
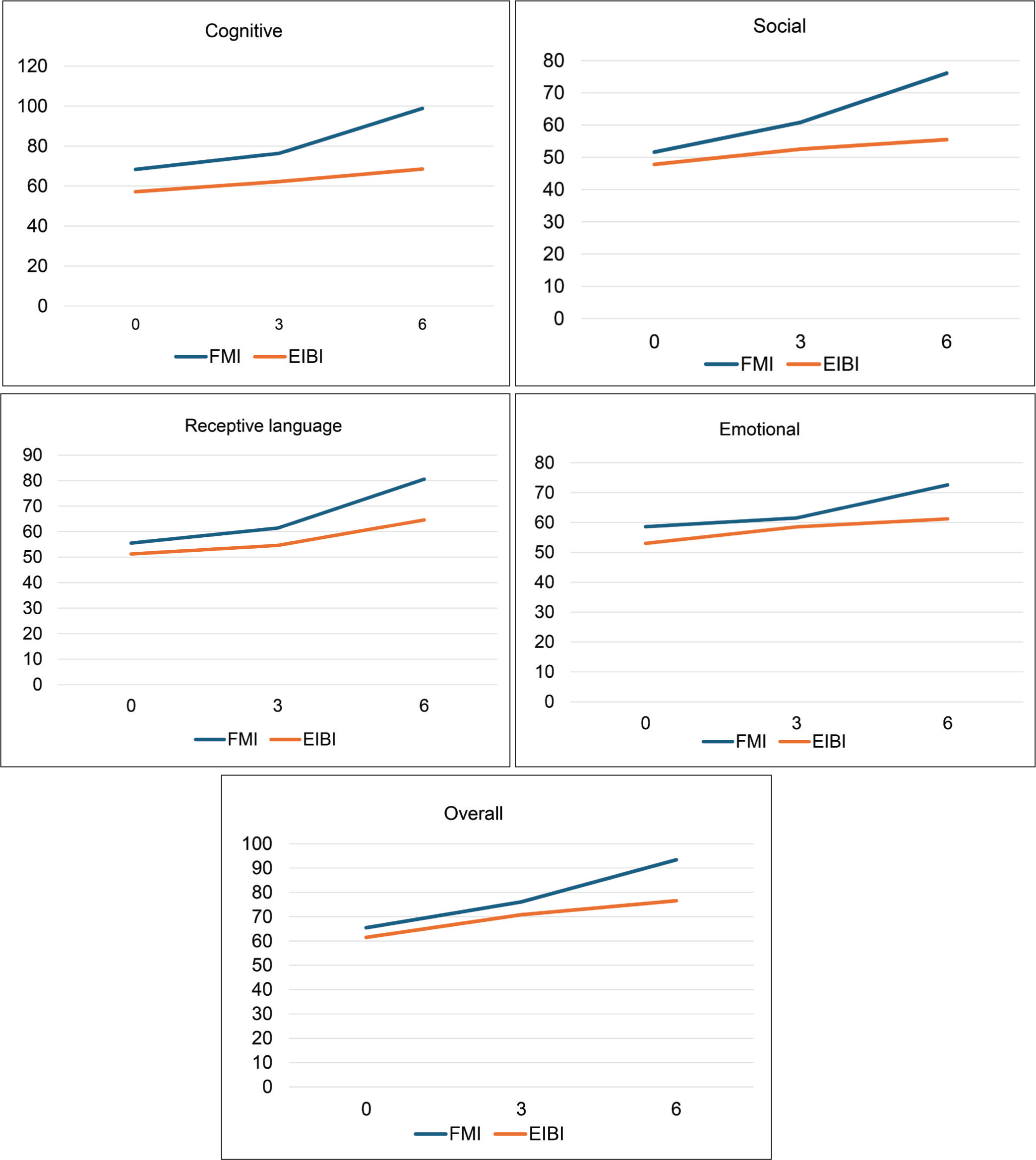
By the end of 12 weeks, overall CDDC scores were higher, and by the end of 24 weeks, the scores had further increased compared to scores at the initial evaluation. The scores from baseline were statistically significant in both the FMI and EIBI groups at the end points (12 and 24 weeks). Individual domain score improvements from baseline to six months were statistically significant for all the domains in both groups except ADL (in EIBI group) and expressive language (in both groups) (Table 2). Change in mean score of CDDC at the end point of 24 weeks from the initial evaluation was significantly better in FMI as compared to EIBI group in social (mean difference = −16.8, 95% CI = −27.18, −6.49, p = .002) as well as fine motor, cognitive, and overall development (mean difference = −12.8, 95% CI = −25.64, 0.038, p = .050). FMI group performed numerically better with regard to other domains too but it was not statistically significant (Table 3). Mean CDDC scores at different end points are displayed in Figure 2.
Repetitive Measures ANOVA Across Three End Points for CDDC Within Study Arms (FMI = 25, EIBI = 25) as Per ITT.
Note:$Repetitive measures ANOVA, Tukey correction; p value <.05 is significant.
Comparing the Difference of CDDC Mean Scores Between FMI (n = 25) and EIBI (n = 25) Groups Across Various End Points as Per Intention-to-treat (ITT) Analysis.
Comparison of Mean Scores of CDDC Developmental Domains at Different End Points Between Groups.
Discussion
Our study aimed to compare FMI and EIBI in context to developmental domains using CDDC as an outcome measure in ASD children. In EIBI, children were given extensive therapy in a clinical setting by specialized therapists. In low-resource settings where there is an unavailability of specialized therapists, conducting EIBI is difficult. In such cases, FMI is effective where parents work as primary therapists. Involving family is beneficial because they are better able to help their child which also reduces their anxiety and frustrations.
In our study, CDDC was used to measure the efficacy in eight developmental areas, namely, fine motor, gross motor, ADL, language-receptive/expressive, cognition, and socio-emotional domain. Children with ASD typically show poor communication and socio-emotional skills. 10 Our study findings demonstrated notable betterment in all the developmental domains in both study arms, although our study arm (FMI) outperformed the control arm (EIBI) in all major developmental domains. The results significantly favored FMI in CDDC in our study. This might be attributable to the difference in direct caregiving and therapeutic intervention by parents in the FMI group. It might emphasize that FMI must be internalized by the parent so that it takes lesser time for the parent to operationalize the intervention program, and it would influence the positive outcome in ASD child. 11 Significant gain was noticed in motor, communication, and socio-emotional domains and prerequisite learning skills as expressed by parents. CDDC was also used to measure the efficacy for social competence in toddlers and preschoolers with ASD in which pretend play or eye-to-eye contact was used as a therapeutic modality. There was a significant improvement in social competence scores in CDDC like our study.12–14 Further, we could not find any study in which FMI or EIBI was used as therapeutic modality where CDDC was used as an outcome measure. We also could not find any study in which all major developmental domains were measured using these two therapeutic modalities. Furthermore, our study findings suggest significant improvement in social as well as fine motor, cognitive, and overall development in FMI group over specialist intervention which is the novel finding of our study. One of the key findings of our study is that there was minimal improvement in expressive language in both groups which indicated longer duration of intervention is required for better speech production and vocabulary in children with ASD. Further literature review suggests that about one-third of individuals with autism do not develop enough speech to meet their daily communication needs, which supports our finding. 15
There were few limitations which is worth highlighting for our study. First, the adequate period of assessment of developmental domains in ASD children may have been constrained by 24 weeks of follow-up. Our study findings suggest that a more extensive course of treatment is required in both treatment group, especially for improvement in expressive language domain. Our study was an open-label design, that is, blinding of researcher and subjects was not done due to the nature of intervention, although full caution was taken to avoid any bias. Lastly, it cannot be ascertained that the parents regularly followed therapy at home, although we asked the parents to bring a video recording of their intervention provided so as to minimize the compliance issue.
To conclude, there is a significant betterment in functioning in major developmental domains in ASD children with FMI over the period of 24 weeks. FMI is much more effective in low resource settings like India, as observed in our study. FMI may be considered as an effective treatment modality in ASD children in our cultural context in view of improved functioning in major developmental domains. A large-scale randomized controlled trial is recommended for the better generalization of our study results.
Supplemental Material
Supplementary material for this article is available online.
Footnotes
Clinical Trial Registration
Clinical Trial Registration Number: CTRI/2020/08/027099
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This single-center study was supported by the Indian Council of Medical Research (ICMR Grant No. 5/7/790-PRG/2012-RCH; project code: 2020-2886).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent were received and obtained respectively before initiating the study from all participants. The study was accorded Ethical Committee Approval vide Ethics Committee (Institutional Ethics Committee, All India Institute of Medical Sciences, Patna) No. AIIMS/ Pat/IEC/ 2020/468; dated May 29, 2020. Written informed consent was taken from all the participants. The study was carried out in accordance with the principles as enunciated in the Declaration of Helsinki.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
