Abstract
Introduction:
Autism spectrum disorder (ASD) is a neurodevelopmental disorder with impairments in language acquisition, social functioning, and restricted and repetitive behaviors. There is no definitive management for the ASD spectrum. The management options available are not beyond criticism. Hence, the search for new options is actively in progress. Noninvasive brain stimulation interventions such as repetitive transcranial magnetic stimulation (rTMS) appears to be a promising tool for the treatment of ASD. However, the available literature on TMS use in ASD is preliminary.
Methods:
This was a hospital-based longitudinal study conducted over 50 patients with ASD of 6 to 16 years of age. The Indian Scale for Assessment of Autism (ISAA) was used for the assessment of the extent of disability in these children. They were given 10 sessions of rTMS over a period of 10 days over the left dorsolateral prefrontal cortex (l-DLPFC) and reassessed for any improvement in ISAA score after 1 day and 3 months of completion of all the sessions of rTMS.
Results:
There was no statistically significant change in the total ISAA score or any of the 6 domains of ISAA after 1 day and 3 months of giving 10 sessions of TMS. There were no significant adverse effects after giving rTMS. In a few patients, mild headache, neck pain, or discomfort at the stimulation site was noted.
Conclusion:
This study has shown that low-frequency rTMS does not have any significant improvement on the various symptoms of ASD. However, there are no major adverse effects after giving rTMS. Also, it points toward the need for further studies.
Keywords
Introduction
Autism spectrum disorder (ASD) is a phenotypically heterogeneous group of neurodevelopmental syndromes, with polygenic heritability, characterized by a wide range of impairments in social communication, and restricted and repetitive behaviors. 1 ASD occurs more often in boys than girls, with a 4:1 male-to-female ratio. 2 The reported prevalence rates of autism and its related disorders have been increasing worldwide over the past decades, from approximately 4 per 10,000 to 1 per 100 children. 3 The clinical, social, and financial burden of ASD is very high. Studies with pharmacologic agents have had disappointing results and often demonstrate an unacceptable side effect burden with long-term use. 4 Given the chronic handicap of the child, valid and reliable biomarkers for diagnosis and also some modes of effective treatments are needed. Over the past quarter-century, neuroscience techniques have been developed and applied to ASD to study brain structure and function. Additionally, clinical trials of therapeutic interventions aimed at modulating brain functioning have also been evaluated. Noninvasive brain stimulation interventions such as repetitive transcranial magnetic stimulation (rTMS) have increasingly been considered in the treatment of ASD. 5 Transcranial magnetic stimulation (TMS) is a method where localized intracranial electrical currents, large enough to depolarize a small population of neurons, are generated by rapidly changing extracranial magnetic fields. 6 TMS can be applied in single pulses, pairs of pulses, or repeated trains of pulses (rTMS). 7 Studies have indicated that low-frequency or “slow” rTMS (≤1 Hz) increases inhibition of stimulated cortex, whereas high- frequency rTMS (>1 Hz) increases excitability of stimulated cortex. Such lasting inhibitory or facilitatory effects of rTMS are thought to occur by various mechanisms, including synaptic changes resembling experimental long-term depression and long-term potentiation as well as larger shifts in network excitability, activation of feedback loops, and activity-dependent metaplasticity. 8
In the majority of the studies, the dorsolateral prefrontal cortex (DLPFC) was targeted, which corresponds to the intersection between Broadmann areas 9 and 46, in the middle frontal gyrus. This area is connected to the orbitofrontal cortex and many other regions, and it is closely related to executive function and working memory. Experimental studies in monkeys and humans have demonstrated the crucial involvement of the DLPFC in the cerebral network of working memory. 9
There have been very few studies on the role of TMS in ASD. Previously, 23 studies described the effects of rTMS on ASD symptoms and ASD-related cognitive deficits, 4 of which were case reports,10–13 7 were noncontrolled prospective intervention trials,14–18 and the remaining 12 were controlled clinical trials.19–30
The overwhelming majority of studies recruited patients with an IQ higher than 80. The 4 trials 27 were all performed on subjects with severe cognitive impairment, while few others16, 17, 23 included patients both with and without cognitive impairment.
Meta-analyses of these studies which mostly comprised high-frequency studies showed a significant but moderate effect on repetitive and stereotyped behaviors, social behavior, and the number of errors in executive function tasks, but not other outcomes. Most studies had a moderate-to-high risk of bias, mostly due to lack of subject and evaluator blinding to treatment allocation.
Among the studies in low-frequency rTMS, 1 study had shown low-frequency, subthreshold rTMS to DLPFC (left or sequential bilateral) have significant improvements in irritability and repetitive behaviors, normalization of electroencephalogram (EEG) components related to target detection, and error monitoring and enhanced autonomic balance. 31 Another study has shown that low-frequency rTMS to DLPFC minimizes early cortical responses to irrelevant stimuli and increases responses to relevant stimuli. Improved selectivity in early cortical responses leads to better stimulus differentiation at later-stage responses. 32 However, these studies consisted of few participants (between 12 and 25) and focused more on electrocortical functioning and endophenotypes rather than the gamut of symptomatology.
There have been very limited studies that focus on the safety of the procedure in children. Studies by Kumar et al, 33 Schrader et al, 34 and Rossi et al, 35 looked into the safety of rTMS in children in general. Seizures are the most serious possible rTMS-related adverse event. Overall, the risk of seizure is considered to be less than 0.01%. 35 Studies have identified the potential risk factors that could contribute to an increase in the probability of inducing a seizure during rTMS treatment include neurological insult or pre-existing condition, including multiple sclerosis, stroke, and traumatic brain injury, interrupted sleep pattern, and a history of seizures. 36 Subject risk factors increased seizure risk substantially. 37 Some patients have also experienced presyncope reactions following stimulation. Other more common side effects that have been associated with TMS are considered relatively minor and include headache, neck pain, discomfort at the site of stimulation, and transient increases in auditory thresholds. TMS can also cause transient or long-lasting changes in cognition or mood. 34
Studies have also shown that individuals with ASD have a greater than average prevalence of epilepsy, approximately 30%. 38 Hence, during rTMS in ASD children, more caution toward seizure side effects may be necessary.
To the best of our knowledge, no such study was done in India. This study based in a tertiary care hospital in Eastern India was planned with the following objectives:
To assess the improvement of social relationships and reciprocity, emotional responsiveness, speech- language and communication, behavior patterns, sensory aspects, and cognitive components of ASD after 1 day of giving rTMS. To reassess the persistence of improvement of the children after 3 months of low-frequency rTMS therapy. To assess the adverse effects in the participants receiving rTMS.
This study will fill the void of such physical intervention studies in ASD in this part of the world, considering both safety and efficacy.
Methods
Study Design and Site
This was a hospital-based single group longitudinal pre-post design, analytical study of the immediate, and intermediate efficacy of rTMS in patients with ASD. The study was conducted in the Outpatient of Psychiatry Department, IPGME&R, Kolkata. The protocol was submitted to and approved by the Ethics Committee of IPGME&R, Kolkata. Informed consent was taken from at least 1 parent of each child participating in the study. Children above 7 years were approached for a verbal assent, but due to developmental disability none of them could be communicated meaningfully for obtaining a valid assent.
The sample size was 43 as calculated by Epi Info. 39 ASD prevalence was taken as 1 in 100 3 and 5% error and 99.9% confidence interval. Some allowance was taken to allow for dropouts. Hence, total recruitment target was 50 children with ASD.
Participants and Procedure of the Study
The inclusion criteria of the study was children with ASD of 6 to 16 years of age attending Outpatient Department of Psychiatry, IPGME&R, Kolkata between May 2018 and June 2019. They were given a study number and chosen for the evaluation of exclusion criteria according to a predetermined random number list. The exclusion criteria were patients (where applicable) or their guardians not willing to participate, patients with a known case of seizure disorder, patients with any form of hearing impairment, patients with cochlear implants, patients with ferromagnetic implants in the body, any comorbid emotional disorders (as neurodevelop- mental disorders have significant comorbidities, only emotional disorders like anxiety, depression, and bipolarity and ADHD that was diagnosed by a child and adolescent psychiatrist according to DSM-5, and treated accordingly were excluded. Otherwise, other neurodevelopmental disorders like intellectual deficiency disorders, motor coordination disorders, etc were kept in this study), extremely violent and agitated patients, and any serious and unstable physical illness. A written consent for participation in the study was obtained from parents and guardians of those who satisfied inclusion and exclusion criteria, and were recruited in the study. This process continued till 50 patients were recruited in the study.
Before initiating treatment with rTMS, the severity of autism in the participants was determined using the ISAA. Initial socio-demographic developmental profiles of the children were also taken. The handedness of the children was asked as a part of this questionnaire. The participants were given 10 sessions of low-frequency rTMS over 10 consecutive days, with 1 session every day, over the left (dominant) DLPFC with an intensity of 100% of motor threshold, frequency of 1 Hz given continuously throughout the session, for 10 min with no intertrain interval (ie, 600 pulses per day). To locate the DLPFC, the first step was to find the site of the motor cortex that represents the abductor pollicis brevis. For doing this, we had to find the vertex (midpoint between nasion and inion and between both auditory canals) and go down 5 cm in the desired direction (left or right) and point to the external auditory meatus. The DLPFC would be 5 cm forward from this point, in a parasagittal plane. After completing all the 10 sessions of TMS, they were reassessed for any improvement in ISAA score after 1 day and 3 months.
During this period, that is, from pretreatment evaluation to 3 months posttreatment evaluation, no other pharmacological or nonpharmacological treatment was started for treatment of naïve patients. For those children who were already on medications, no changes were made in the doses of prescribed medicines, no new medicines were added, and no nonpharmacological therapies were started or put on hold. It was decided that if we had to start any pharmacological or nonpharmacological treatment for any patient, he/she would be eliminated from the study.
Tools for Assessment
Indian Scale for Assessment of Autism
This is an objective assessment tool for persons with autism which uses observation, clinical evaluation of behavior, testing by interaction with the subject, and also information supplemented by parents or caretakers to diagnose autism. ISAA consists of 40 items rated on a 5-point scale ranging from 1 (never) to 5 (always). The 40 items of ISAA are divided into 6 domains:
Social Relationship and Reciprocity Emotional Responsiveness Speech-Language and Communication Behavior Patterns Sensory Aspects Cognitive Component
Results
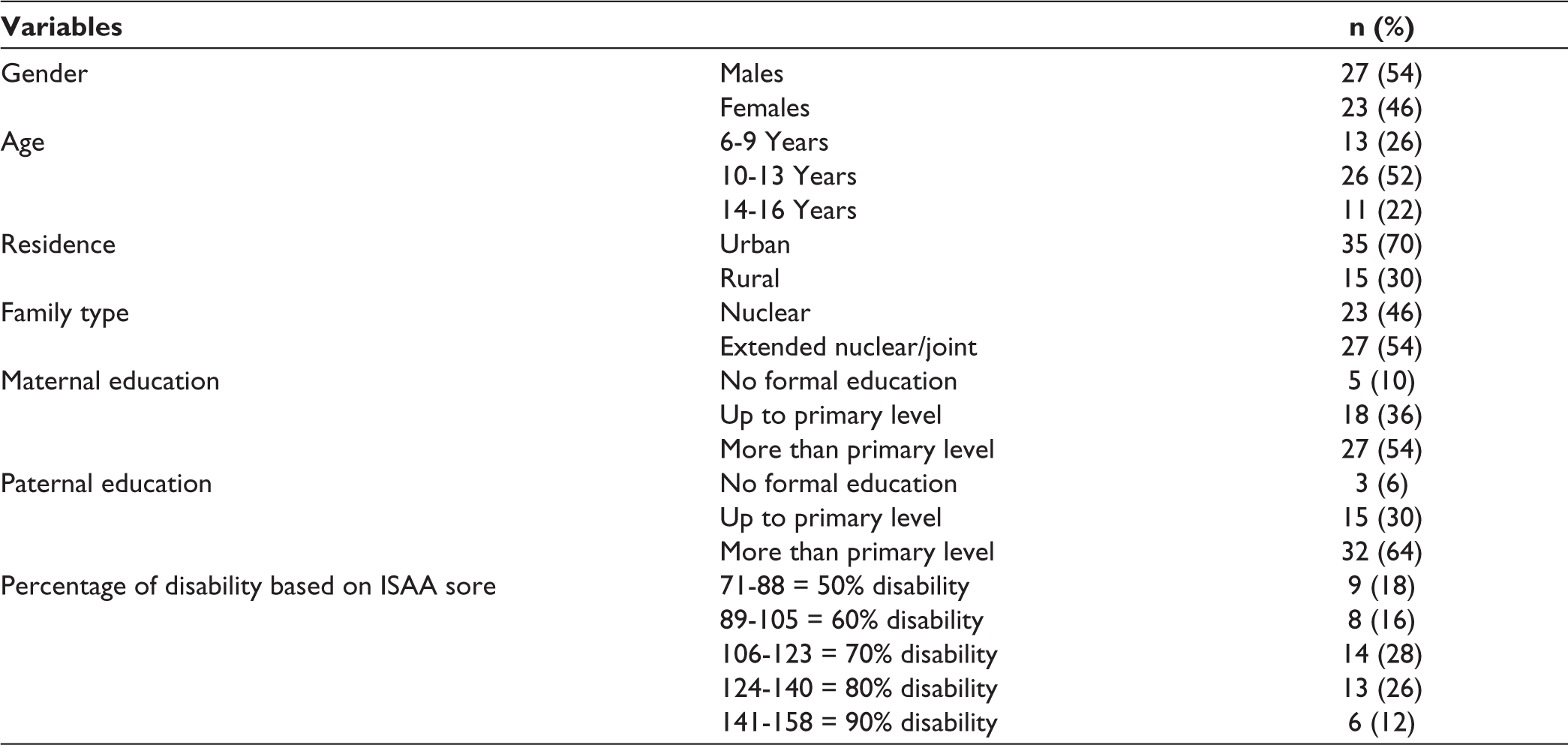
The sociodemographic details of the participant were cal- culated in terms of percentages (Table 1). Comparison among the mean pretreatment ISAA score and mean posttreatment ISAA score was done by paired t test, P value ≤.05 was considered as statistically significant. Data were analyzed in 2 parts. In the first part, the comparison was done among the mean pretreatment ISAA score and mean posttreatment ISAA score after 1 day of giving rTMS. In the second part, the comparison was done among the mean pretreatment ISAA score and mean posttreatment ISAA score after 3 months of giving rTMS.
Sociodemographic and Clinical Profile of the Participants.
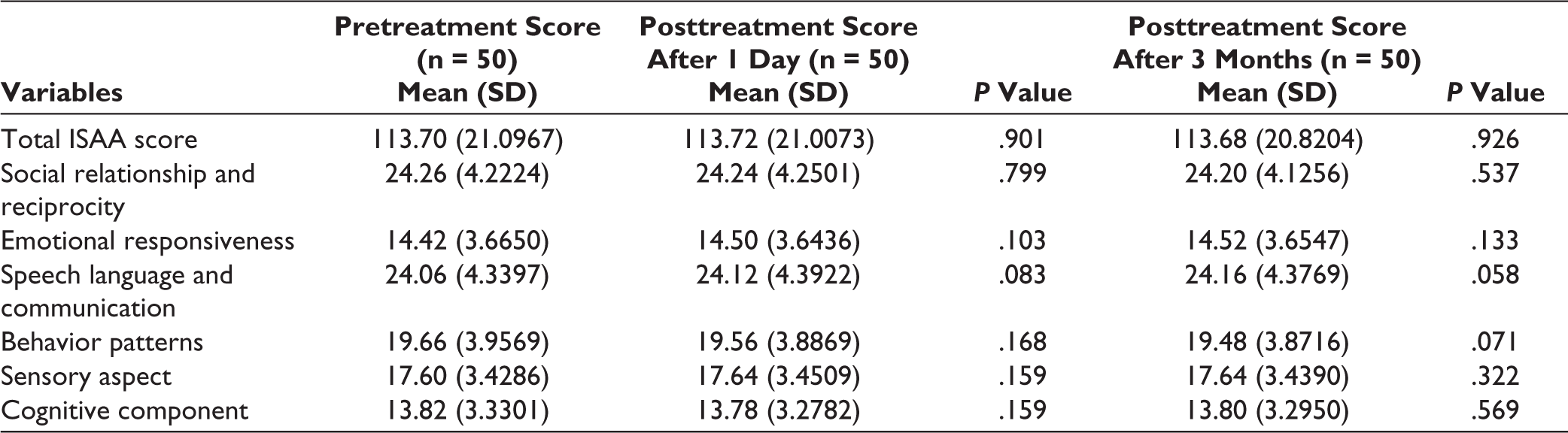
As depicted in Table 2, there was no statistically significant change in the mean pretreatment ISAA score after 1 day and 3 months of giving rTMS. Considering the 6 domains of ISAA (social relationship and reciprocity, emotional responsiveness, speech-language and communication, behavior patterns, sensory aspects, and cognitive component), the difference of pretreatment score and posttreatment score after 1 day and 3 months of giving rTMS was statistically not significant.
Changes in Various Domains of ISAA Score 1 Day and 3 Months After Giving RTMS.
To look for any adverse effects after giving rTMS, any change in behavior of the children during or after the TMS sessions was carefully noted (Table 3). Children with less severe cognitive impairment were assessed and interviewed by us and their caregivers for the emergence of any adverse effects, especially those which were looked into in the previous studies. It was seen that out of 50 participants, 38 had no change in behavior after giving rTMS, 5 had a mild headache, 4 had neck pain, and 3 had mild discomfort at the stimulation site. These changes were transient, occurring shortly after the completion of TMS sessions, and resolved spontaneously without any intervention. There was no history of seizure, syncopal attack, or severe headache among the participants after giving rTMS. No residual adverse effects were reported by the caregivers during the 1 day and 3 months post rTMS period.
Adverse Effects After Giving rTMS.

The study entertained all neurodevelopmental comorbidities to match real-life conditions, but the primary measure was ASD symptoms and the other confounders like pharmacological and other nonpharmacological measures were kept constant throughout the study so that its effects could be minimized.
Discussion
Data were analyzed in 2 parts. In the first part, the comparison was done among the mean pretreatment ISAA score and mean posttreatment ISAA score 1 day after giving rTMS. It is seen that the changes in total ISAA score or each of the 6 domains were not significant after 1 day of giving 10 sessions of TMS. In the second part, the comparison was done among the mean pretreatment ISAA score and mean posttreatment ISAA score after 3 months of giving rTMS. It was seen that the changes in total ISAA score or each of the 6 domains are not significant after 3 months of giving 10 sessions of rTMS.
Some of the above findings are inconsistent with the findings of some earlier studies. Baruth et al 23 assessed the effects of rTMS on evoked gamma frequency oscillations in ASD. Following 12 sessions of bilateral “slow” rTMS to the DLPFC rTMS application, individuals with ASD showed significant improvement in discriminatory gamma activity between relevant and irrelevant visual stimuli and significant improvement in the responses on behavioral questionnaires. Casanova et al 40 assessed the effects of rTMS on event-related potential Indices of Attention in Autism. Twelve sessions of low-frequency rTMS applied bilaterally to the DLPFC found a significant improvement in both early and later stages of visual processing and significant reductions in both repetitive behavior and irritability according to clinical behavioral questionnaires. Sokhadze et al 32 examined the effects of low-frequency rTMS on novelty processing as well as behavior and social functioning in 13 individuals with ASD. They found that low- frequency rTMS to DLPFC minimizes early cortical responses to irrelevant stimuli and increases responses to relevant stimuli which leads to better stimulus differentiation at later-stage responses. In contrast, low-frequency rTMS appears to have no significant effect on reaction time, a correlate of the psychomotor slowness that is characteristic of ASD. 41 These studies have mostly looked into electrophysiological parameters and endophenotypes. The current study is one of the very few studies that looked into clinical symptomatology of ASD with low-frequency rTMS and probably the only one that applied rTMS on DLPFC. As it failed to produce and direct changes in symptomatology in this study, further studies focusing on specific areas of symptoms can be undertaken.
Regarding adverse effects, the above findings are consistent with the findings of some earlier studies. Rossi et al 35 studied the most serious possible TMS-related adverse event, that is, induction of a seizure and found that the overall risk of seizure is considered to be less than 0.01% across all patients and all paradigms. However, the risk varies based on factors including inter-pulse interval, the intensity of stimulation, and risk factors in the participants. To date, no seizures have been reported during TMS in any individual with ASD, and given the paucity of TMS safety data in ASD, it is currently reasonable to default to the safety guidelines established by the “Safety of TMS Consensus Group.” 35 According to Grossheinrich et al, 42 some patients have also experienced presyncope reactions following stimulation, but it is hard to disentangle the direct effects of stimulation from that of a vasovagal response to anxiety or discomfort in these cases. A meta-analysis by Rajapakse and Kirton 43 reviewed the studies to date involving all rTMS protocols in children (approximately 1,000 children have been studied across all rTMS protocols to date) and highlighted its minimal risk, excellent tolerability, and increasingly sophisticated ability to interrogate neurophysiology and plasticity making it an enviable technology for use in pediatric research with future extension into therapeutic trials. Only a few studies focused specifically on the tolerability of rTMS in children with ASD, one of them is a case report.12, 27 The current study supports that low-frequency rTMS is safe in children with ASD with the largest sample size among the available studies.
It may be possible that high-frequency (>1 Hz) rTMS may have some benefits due to increased excitability of the stimulated cortex. 9 But individuals with ASD have a greater than average prevalence of epilepsy. 38 Hence, as the population is vulnerable and low-frequency variant is better tolerated than high-frequency rTMS, 44 only low-frequency rTMS was used. It may also be possible that a greater number of pulses per day may have some additional benefits,39, 45, 46 but again due to the risk of seizure, only 600 pulses of rTMS were given per day.
The findings of this study should be considered in light of several limitations. Randomization was done before em- ploying exclusion criteria for ease of implementation. That leaded to a greater number of random number generation for achieving expected sample size. The sample size of the ASD cases taken in the study was small and thus data on these cases may not be representative of the disease under evaluation. Extremely violent and agitated patients were excluded from the study. Thus, data about behavior patterns in ASD may not be representative. It was not assessed whether different medication status affect the outcomes. It was beyond the scope of this study to investigate predictors or moderators of outcomes such as medication. Handedness is difficult to assess through objective tests in ASD children, considering their developmental challenges. It was assessed only through parental observation. Berksonian selection bias had always been a problem from clinic-based studies like this which compromises generalizability.
While assessing the safety of rTMS, patients with difficulties in speech and communication or intellectual disability were unable to express the problems which they faced during the treatment. During the follow-up visits, the caregivers were interviewed for the emergence of any adverse effect, which was not very reliable. Hence, the mild adverse effects of rTMS could be undermined in the study.
Though there are studies with high-frequency rTMS in DLPFC. Studies with low-frequency rTMS in DLPFC focuses mainly on endophenotypes measurements like EEG and cortical responses. The strength of this study is that with low-frequency rTMS, this study ventured direct assessment of clinical parameters stimulating DLPFC. It was planned to explore the intervention at clinical effectiveness level. This study also confirmed the safety of low-frequency rTMS in children with ASD. It is also the first of its kind in the Indian population as far as it could be searched in prominent databases. This will be a stepping stone for further studies with high- frequency or high pulse rate studies. In the future, isolated studies on ASD excluding intellectual disabilities can also be undertaken. Pharmacological and non-nonpharmacological parameter stratification can also be done in future with larger studies with multiple groups. Not only that but stimulation of different areas of the brain can also produce good results as evidenced by other studies that can also be explored in future studies.47, 48
Conclusion
From this study, we can conclude that low-frequency rTMS does not have any significant improvement on the various symptoms of ASD at a clinical level. However, in a majority of the patients, there are no major adverse effects after giving low-frequency rTMS. As this study was done in a smaller population, larger studies should be conducted in future in a significantly larger population to assess the effectiveness of the intervention in a larger sample. Also, as the procedure was found to be safe in children, a longitudinal study may be undertaken to determine the stimulation location, frequency, as well as number and timing of sessions, which can be the difference between facilitating or suppressing cortical functioning or having no effect at all. Endophenotype improvement could be assessed from similar studies in future. Such negative effect studies can render the medical fraternity with a balanced view about a new modality of treatment in a vulnerable population. But, at the same time, negative side effects encourage researchers to take up new larger projects in this vulnerable population. Future studies with high-frequency rTMS can be ventured as this study found that rTMS is safe on ASD kids. Population-based studies can be more representative.
Footnotes
Author Contributions
MP, NM, AR, and PKS: Have been involved in drafting the manuscript or revising it critically for important intellectual content; MP and AR: Have made substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data; MP, NM, AR, and PKS: Have given final approval of the version to be published.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent were received and obtained respectively before initiating the study from all participants.
