Abstract
Background
Depression, often accompanied by cognitive impairment, remains a major clinical challenge due to the limited efficacy and side effects of conventional anti-depressants. The herbal pair of Ginseng and Polygonatum has demonstrated synergistic effects in tonifying qi, nourishing yin, and improving neurological function. Emerging evidence suggests this combination may offer multi-targeted benefits in alleviating depressive symptoms and cognitive deficits with fewer adverse effects.
Objectives
This study employs network pharmacology, molecular docking, and animal experiments to investigate the mechanisms underlying the anti-depressant efficacy of the Ginseng–Polygonatum herbal pair.
Materials and Methods
The network pharmacology was adopted to analyze Ginseng and Polygonatum to identify core genes and perform gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyzes. A chronic unpredictable mild stress (CUMS) rat model of depression was established, and after administering Ginseng–Polygonatum extract via gavage, behavioral tests were performed to assess depressive-like symptoms in the CUMS rats. Hematoxylin–eosin (H&E) staining was used to detect neuronal damage in the hippocampal tissue, and the expression levels of interleukin-1 beta (IL-1β), cysteine-aspartic protease 1 (Caspase-1), phosphoinositide 3-kinase (PI3K), protein kinase B (AKT), glycogen synthase kinase 3 beta (GSK3β), B-cell lymphoma 2 (Bcl-2), and Bcl-2-associated X protein (Bax) in the rats’ hippocampus were measured.
Results
The key components of the Ginseng and Polygonatum herbal pair against depression include kaempferol, aposiopolamine, frutinone A, panaxadiol, and n-coumaroyltyramine. The critical targets identified are PIK3CA, AKT1, IL-1β, Bcl-2, Caspase-1, and ICAM1. Molecular docking results indicate that these core components exhibit strong binding affinity to the key targets. Additionally, animal experiments demonstrate that the Ginseng and Polygonatum herbal pair can improve cognitive impairment in CUMS rats by regulating the PI3K/AKT/GSK3β signaling pathway and inhibiting cell apoptosis.
Conclusion
Ginseng and Polygonatum herbal pair can regulate multiple targets through various active components, influencing several signaling pathways. It modulates biological processes and related pathways, such as inflammatory responses and metabolic functions, to improve depressive symptoms and cognitive impairment in CUMS rats. This effect is likely mediated by the regulation of the PI3K/AKT/GSK3β signaling pathway and the alleviation of cell apoptosis.
Introduction
Depression is a chronic, recurrent, and potentially life-threatening mental disorder (Wang et al., 2019). The World Health Organization (WHO) ranks severe depression as the third leading cause of global disease burden. Neurocognitively, it is characterized by impaired cognitive flexibility and reduced prefrontal inhibition (Disner et al., 2011). Cognitive dysfunction often persists after symptom remission, accounting for 85%–94% of depressive episode duration (Conradi et al., 2011), with deficits noted in flexibility, inhibitory control, and verbal fluency (Wagner et al., 2015). Addressing these cognitive impairments has become an urgent therapeutic priority.
Adult hippocampal neurogenesis, vital for memory and learning (Chesnokova et al., 2016), is disrupted in depression, contributing to cognitive and emotional dysfunction. Depression is also linked to impaired neuroplasticity, brain volume loss, neuronal apoptosis, and neuroinflammation—all of which play key roles in its progression (Zhang et al., 2022). The phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT)/glycogen synthase kinase 3 beta (GSK3β) pathway is closely associated with these processes. Its inhibition is linked to cognitive deficits (Emamian et al., 2004), as it regulates neuronal apoptosis and central sensitization (Treede et al., 1992), making it a critical target in depression-related cognitive impairment.
Ginseng and Polygonatum form a classical traditional Chinese medicine (TCM) herbal pair used to treat depression. Traditionally valued for nourishing qi and yin, calming the mind, and enhancing cognition, this combination is frequently prescribed by experts like Dr. Hao Wanshan and Professor Wang Zhonglin. Modern studies show that ginsenosides suppress inflammation by reducing IL-6, interleukin-1 beta (IL-1β), tumor necrosis factor-alpha (TNF-α), and nuclear factor kappa B (NF-κB) activity (Jiang et al., 2022). Ginsenoside Rg1 modulates microglia and cytokines, promoting neuroplasticity (Shi et al., 2019). Polygonatum sibiricum polysaccharide attenuates inflammation-induced depressive behaviors (Shen et al., 2021) and protects neurons via the PI3K/AKT pathway (Zhang, Cao, et al., 2015; Zhang, Wang, et al., 2015). However, the synergistic mechanisms of Ginseng–Polygonatum in treating depression remain unclear.
In this study, a chronic unpredictable mild stress (CUMS)-induced rat model of depression was established. Through network pharmacology and data mining, the potential targets and pathways of Ginseng–Polygonatum were analyzed. Behavioral tests and PI3K/AKT/GSK3β signaling markers were examined to explore the pharmacological mechanisms underlying its anti-depressant and cognitive-enhancing effects. This research provides a scientific basis for the clinical use of Ginseng–Polygonatum and offers a reference for integrative therapeutic strategies.
Materials and Methods
Network Pharmacology Analysis
Screening of Active Ingredients and Target Proteins of Ginseng–Polygonatum
Initially, the active compounds and significant targets of Ginseng–Polygonatum were retrieved utilizing the Traditional Chinese Medicine Systems Pharmacology (TCMSP) database (Ru et al., 2014). Integration of drug targets obtained from TCMSP, and the utilization of the Bioinformatics Online Tool (
Construction of a Common Drug Target for Active Ingredients of Ginseng and Polygonatum
Using the human gene databases GeneCards (Stelzer et al., 2016) and Online Mendelian Inheritance in Man (OMIM), we searched with the keyword “depressive” to identify human genes associated with depression. The predicted targets of Ginseng–Polygonatum and depression-related disease targets were then input into the Bioinformatics and Evolutionary Genomics online tool to generate Venn diagrams, thereby identifying the overlapping targets shared between the “drug” and “disease” categories.
Prediction of Depression Disease Targets and Construction of Drug-disease Common Targets
The acquired target data were uploaded to the STRING database (Szklarczyk et al., 2021) to construct a protein–protein interaction (PPI) network. The minimum required interaction score was set to medium confidence (0.900) for subsequent target screening.
Construction of the Ginseng–Polygonatum “Drug Active Ingredient-Disease Target” Network
The refined key target files were uploaded to Cytoscape 3.10.1 software (Doncheva et al., 2019) to generate a network diagram illustrating the compound-target-disease interactions.
Gene Ontology (GO) Function Enrichment Analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Enrichment Analysis
The targets of Ginseng–Polygonatum were analyzed for GO and KEGG enrichment using Metascape. The R language was then used to visualize key gene functions and pathways involved in depression. The top 20 KEGG pathways with the lowest p values and their genes were imported into the Bioinformatics Online Tool to generate GO and KEGG bar charts and a bubble chart.
Animals
Fifty male specific pathogen-free (SPF)-grade Sprague–Dawley (SD) rats (8 weeks old, 200 ± 20 g) were obtained from Liaoning Changsheng Biotechnology Co., Ltd. (License No. SCXK [Liao] 2020-0001). Rats were housed at the Animal Experimental Center of Liaoning University of TCM under controlled conditions (24°C–28°C, 40%–60% humidity). The study was approved by the Animal Ethics Committee of the university (approval no. 21000042022048).
Drugs and Reagents
Ginseng and Polygonatum were purchased from Beijing Tong Ren Tang Co., Ltd. (batch numbers: 20220819 and 20220828) and authenticated by Professor Kaiyun Jiang of the Department of Chinese Medicine, Liaoning University of TCM. Fluoxetine hydrochloride capsules were obtained from Eli Lilly Suzhou Pharmaceutical Co., Ltd. (batch no. 22273A). IL-1β enzyme-linked immunosorbent assay (ELISA) kits were supplied by Shanghai Guangrui Biotechnology Co., Ltd. (ER2736). Quantitative real-time polymerase chain reaction (qRT-PCR) reagents included the PrimeScript reverse transcription (RT) Reagent Kit with genomic DNA (gDNA) Eraser and TaqMan Buffer (TB) Green Premix Ex Taq II (Takara Bio Inc., Japan; RR047A, RR820A).
Primary antibodies used were cysteine-aspartic protease 1 (Caspase-1) (Merck, SAB5700660), PI3K (Abcam, ab302958), p-PI3K (Abcam, ab182651), AKT (Bioswamp, PAB34089), p-AKT (Abcam, ab81283), GSK3β (CST, 5675), p-GSK3β (CST, 9327), B-cell lymphoma 2 (Bcl-2) (Affinity, AF6139), Bcl-2-associated X protein (Bax) (CST, 2772), and β-actin (Affinity, AF7018) as internal controls.
Instruments
The following instruments were used in this study, all provided by the Engineering Technology Center for Innovative Chinese Medicine, Liaoning University of TCM: tissue grinder (CK1000D, Thmorgan Corp.), animal motion tracking system (NOLDUS, Netherlands), high-speed refrigerated centrifuge (Stratos, Thermo Scientific), microplate reader (Multiskan FC, Thermo Fisher), electrophoresis apparatus (EC250-90, Thermo Electron Corp.), and paraffin microtome (Leica).
Methods
Grouping, Modeling, and Drug Administration
Fifty rats were randomly divided into five groups (n = 10 each): blank, model, Western medicine (fluoxetine), low-dose Ginseng–Polygonatum, and high-dose Ginseng–Polygonatum. All rats underwent a 7-day acclimatization period. Rats in the blank group were group-housed with free access to food and water, without any external stress. The other four groups were exposed to CUMS for 21 days based on established protocols (Xu & Li, 2003), with two randomly selected stressors applied daily, including: 5 min cold-water swim (4°C), 24 h wet bedding, 24 h fasting, 24 h cage tilting (45°), 2 min tail pinch, reversed light-dark cycle (24 h), 5 min heat stress (45°C swim) (Liu et al., 2024), and 24 h exposure to unfamiliar objects.
After model establishment, Ginseng and Polygonatum decoction pieces (1:1 ratio) were prepared. Herbs were soaked in cold water for 30 min, decocted twice in 500 mL water (30 min each), and the filtrates were combined and concentrated. The resulting aqueous extracts were administered at concentrations of 5.67 g/kg (low dose) and 17.01 g/kg (high dose). Dosing was based on the adult clinical dosage (9 g/day), the 2020 edition of the Chinese Pharmacopoeia, and human-to-rat dose conversion (Zhang, Cao, et al., 2015; Zhang, Wang, et al., 2015). The low-dose group received the equivalent adult dose, while the high-dose group received three times that amount. Rats in the model and blank groups received normal saline. Fluoxetine dosage was calculated based on standard instructions using a human-to-rat conversion ratio of 1:6.
Behavioral Tests
Body Weight Measurement
After adaptation, the general condition of the rats was observed. Body weight was recorded on day 21 post-modeling and day 35 after drug administration.
Open Field Test
After modeling, an open field test was conducted in a 100 × 100 cm black box. Each rat was gently placed at the center, and its movement was recorded by a top-mounted camera for 5 min to assess total distance and average speed. After each test, the box was cleaned, and the data were analyzed using EthoVision XT 13 software.
Forced Swimming Test
After modeling and treatment, rats underwent a forced swimming test. They were placed in a cylinder (water at 25°C ± 2°C), allowed 30 s to adapt, and then observed for 6 min. Immobility—defined as floating with minimal limb movement and no horizontal motion—was recorded. After the test, rats were dried and returned to their cages, and the water was changed between tests.
Sucrose Preference Test
Before the experiment, rats underwent a 3-day adaptation period, during which two bottles were placed on opposite sides of each cage—one containing 1% sucrose solution and the other regular water. The positions of the bottles were switched daily. After adaptation, rats were deprived of food and water for 24 h. The main sucrose preference test followed the same procedure: rats were given access to both bottles for 1 h, and their intake was recorded. Sucrose preference was calculated as:
Sucrose Preference (%) = Sucrose Intake/(Sucrose Intake + Water Intake) × 100%.
Sample Collection
Following the final administration, rats were anesthetized intraperitoneally with 20% Urlatan solution (100 g/mL). Brain extraction was performed on a sterile workbench. Brains were immediately placed on ice, and the hippocampus was rapidly dissected according to the brain atlas guidelines. The hippocampal tissues were then stored in cryogenic tubes at −80°C for subsequent analysis.
Hematoxylin–Eosin (H&E) Staining to Detect Pathological Changes in Hippocampal Tissues
Hippocampal tissues were fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned at 6 µm. Sections were dewaxed, rehydrated, and stained with H&E. After differentiation with acid alcohol, dehydration in ethanol, and xylene clearing, slides were sealed with neutral gum. Histopathological changes were then observed under a microscope.
Immunohistochemical Staining to Detect the Expression Levels of Caspase-1 in the Hippocampus of Rats in Each Group
Paraffin-embedded hippocampal sections were deparaffinized, rehydrated, and treated with hydrogen peroxide for 10 min, followed by phosphate-buffered saline (PBS) rinsing. Sections were incubated overnight with primary antibodies against Caspase-1 (1:1,000). After washing with PBS, 3,3′-diaminobenzidine (DAB) staining and hematoxylin counterstaining were performed. Sections were then dehydrated in graded ethanol, cleared in xylene, air-dried, and coverslipped. Three random fields (200×) per section were imaged under an optical microscope. ImageJ software was used to calculate average optical density (AOD), defined as the integrated optical density (IOD) divided by the pixel area.
ELISA to Detect the Content of IL-1β in the Hippocampal Tissues of Rats in Each Group
Hippocampal tissues were thawed and centrifuged at 3,000 rpm for 20 min (radius: 16 cm), and the supernatant was collected. Standard samples were prepared, added to the wells, incubated, and washed. Enzyme-labeled reagents were then added, followed by further incubation and washing. A chromogenic reaction was carried out at 37°C in the dark for 10 min. After stopping the reaction, absorbance was measured at 450 nm. A standard curve was generated to calculate sample concentrations.
RT-PCR to Detect PI3K, AKT, GSK3β, Bcl-2, and Bax Messenger Ribonucleic Acid (mRNA) in the Hippocampus of Rats in Each Group
Total RNA was extracted from hippocampal tissue using the TRIzol method. RNA purity and concentration were assessed with a NanoDrop 2000. Complementary DNA (cDNA) synthesis was performed according to the manufacturer’s instructions. Quantitative PCR (qPCR) was conducted following Takara’s protocol: pre-denaturation at 95°C for 30 s, denaturation at 95°C for 5 s, annealing/extension at 60°C for 30 s, for 30 cycles. Amplification curves were monitored, and relative mRNA expression was calculated using the 2–∇∇Ct method. Primers were synthesized by Takara, and sequences are listed in Table 1.
Primer Sequence.

Western Blot Analysis of PI3K, AKT, GSK3β, Bcl-2, and Bax Protein Expression in Rat Hippocampal Tissues
Hippocampal tissue was lysed with radioimmunoprecipitation assay (RIPA) buffer, centrifuged, and the protein concentration was measured by bicinchoninic acid (BCA). Proteins were denatured, transferred to membranes, blocked, and incubated overnight with the primary antibody at 4°C. After washing, membranes were incubated with the Horseradish Peroxidase (HRP)-secondary antibody for 1 h at room temperature, washed, then developed using enhanced chemiluminescence (ECL). Bands were analyzed with ImageJ, and protein levels were normalized to the internal control.
Statistical Analysis
The statistical analysis of experimental data was performed utilizing GraphPad Prism 9.5.1 software. Quantitative data conforming to normal distribution were described as mean ± standard deviation
Results
Integrated Analysis of Network Pharmacology
Ginseng–Polygonatum Pair: Active Ingredients and Their Targets
Using the TCMSP database with criteria oral bioavailability (OB) ≥ 30% and drug-likeness (DL) ≥ 0.18, 22 active ingredients from Ginseng and 8 from Polygonatum were identified (30 total, Table 2). Target genes were obtained via UniProt: 256 for Ginseng and 40 for Polygonatum, with 122 overlapping genes, indicating shared regulatory pathways.
Active Ingredients for Ginseng and Polygonatum.
Construction of a Common Drug Target for Active Ingredients of Ginseng and Polygonatum
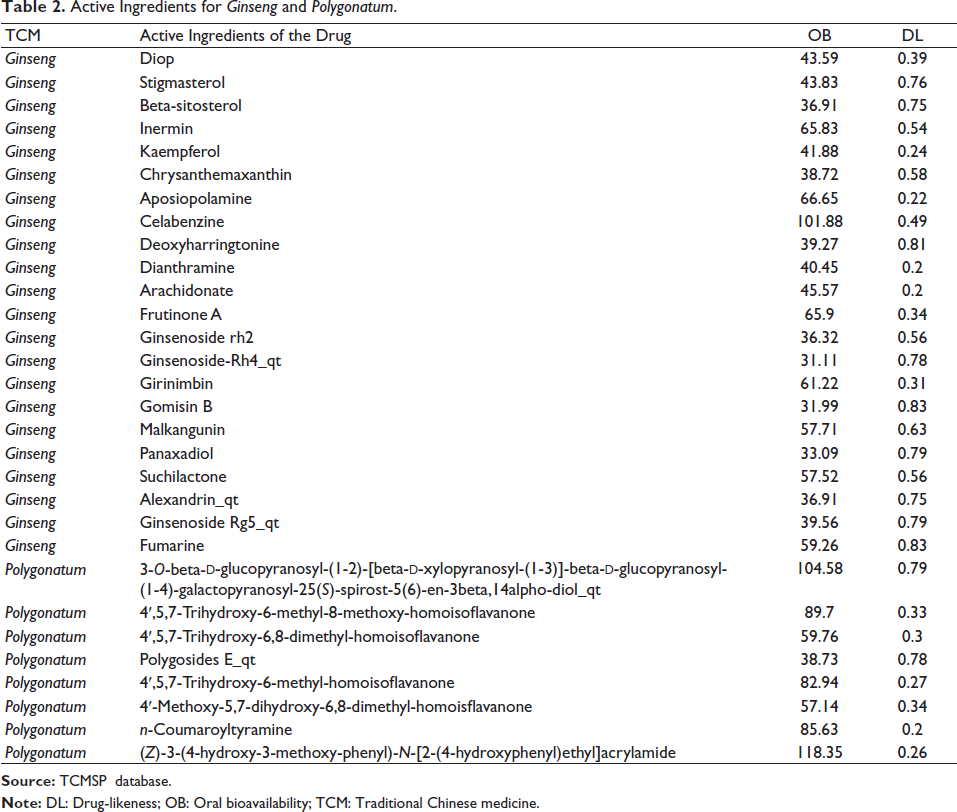
Using the TCMSP database, 121 active ingredient targets for Ginseng and Polygonatum were retrieved. A Venn diagram drawn with R revealed 16 common targets (Figure 1A).
(A) Ginseng–Polygonatum Co-active Ingredient Target Venn Diagram. (B) Drug-disease Common Target Venn Diagram. (C) Protein–protein Interaction (PPI) Network. (D) Ginseng–Polygonatum “Drug Active Ingredient-Disease Target” Network. (E) Gene Ontology (GO) Enrichment Analysis: Cellular Component (CC). (F) GO Enrichment Analysis: Molecular Function (MF). (G) GO Enrichment Analysis: Biological Process (BP). (H) Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analysis.
Prediction of Depression Disease Targets and Construction of Drug-disease Common Targets
Using GeneCards and OMIM, 15,874 depression-related target genes were retrieved. A Venn diagram created with R identified 114 common targets between the drugs and the disease (Figure 1B).
Construction of Protein Interaction Networks and Selection of Core Genes for Ginseng–Polygonatum in Depression Treatment
The 112 selected Ginseng–Polygonatum target genes were imported into the STRING database for PPI analysis (Figure 1C), generating a network of protein interactions. Key depression-related targets identified included TNF, AKT1, PTGS2, PRKACA, IL-1β, BCL2, and CASP1 (Figure 1D).
GO Enrichment Analysis
GO analysis revealed the mechanisms of Ginseng–Polygonatum targets across three categories: cellular component (CC, Figure 1E), molecular function (MF, Figure 1F), and biological process (BP, Figure 1G).
KEGG Enrichment Analysis
KEGG functional enrichment analysis of core Ginseng–Polygonatum target genes was performed using R. The top 20 pathways were visualized as horizontal bar graphs. Results showed enrichment in pathways such as lipid and atherosclerosis, cancer, and Alzheimer’s disease (Figure 1H).
Effects of Ginseng–Polygonatum on Body Weight of Depressive Rats
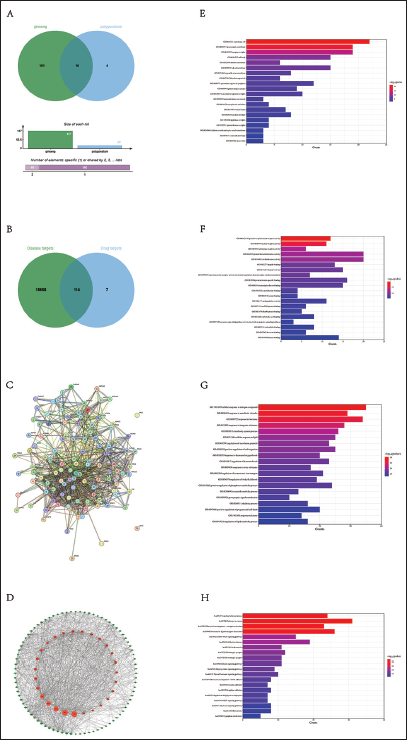
After 21 days of modeling, all groups except the blank group showed significant weight loss (p < .01), confirming successful model induction. Model rats exhibited symptoms such as lethargy, reduced appetite, low activity, and gastrointestinal issues. Following treatment, rats receiving Western medicine or Ginseng–Polygonatum (both doses) showed significant weight gain compared to the model group (p < .01) and improved overall condition. These findings suggest that Ginseng–Polygonatum may help counteract depression-induced weight loss (Figure 2A).
Behavioral Results
Effects on Sucrose Preference Rate in Rats
After depression induction, rats in the model group showed a significant reduction in sucrose preference compared to the blank group (p < .01). Following treatment, sucrose preference significantly increased in both the Western medicine and Ginseng–Polygonatum groups (p < .01). These results suggest that Ginseng–Polygonatum, at both doses, effectively improves anhedonia in depressed rats (Figure 2B).
Effects on Immobility Time on the Swimming Test
Following drug administration, the model group showed a significant increase in immobility time compared to the blank group (p < .01). In contrast, both the Western medicine and Ginseng–Polygonatum groups (low- and high-dose) demonstrated significantly reduced immobility times compared to the model group (p < .01). These findings suggest that Ginseng–Polygonatum effectively alleviates depressive-like behavior in rats, as reflected in improved performance in the forced swim test (Figure 2C).
Effects of Ginseng–Polygonatum on Central Area Residence Time and Movement Distance in Depressive Rats
Compared to the blank group, rats in the model group showed significantly reduced central area residence time, frequency of central crossings, and total movement distance (p < .01). However, rats treated with Western medicine and both doses of Ginseng–Polygonatum exhibited a marked increase in central area residence time compared to the model group (p < .05, p < .01) (Figure 2D–2F).
Observation of Hippocampal Neuron Morphology by H&E Staining
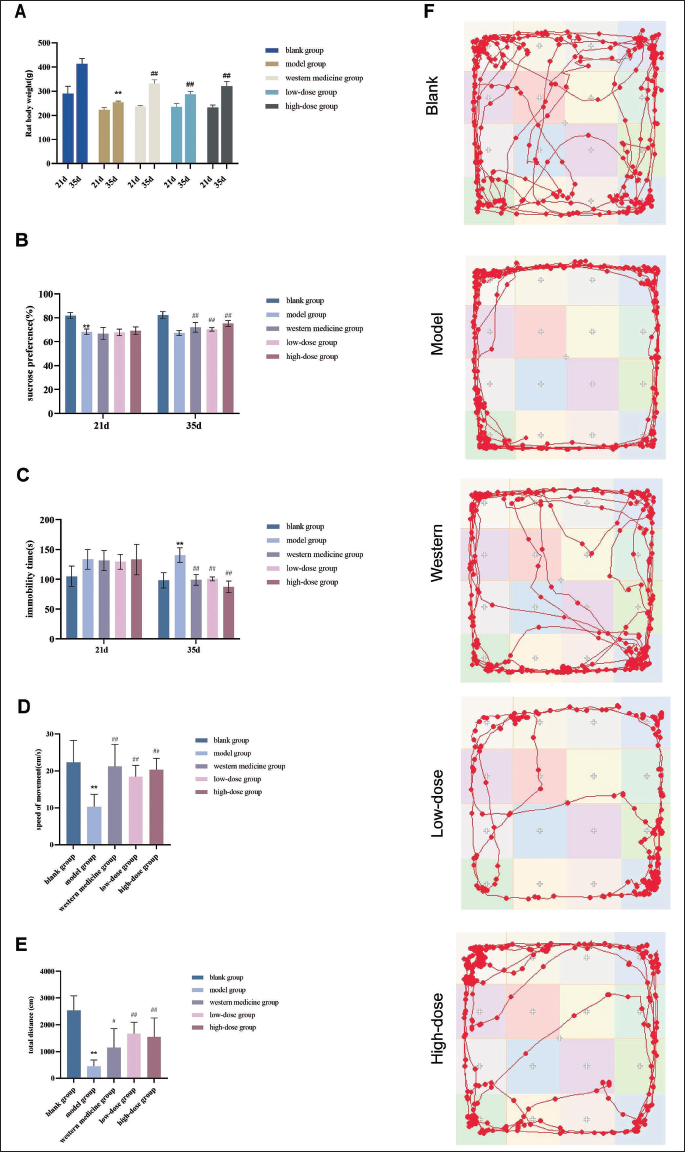
Under 100× and 200× magnification, hippocampal tissue morphology was examined. In the blank group, neurons showed normal structure, with regular morphology, round nuclei, and orderly arrangement. In contrast, the model group exhibited a marked reduction in neuronal number, irregular morphology, nuclear shrinkage, and a disordered arrangement. Compared to the model group, both Ginseng–Polygonatum (low- and high-dose) and Western medicine groups showed increased neuronal numbers, improved morphology, and more organized cell arrangement. All treatment groups demonstrated varying degrees of histological improvement in hippocampal neurons (Figure 3A).
Effects of Ginseng–Polygonatum on Caspase-1 Protein Expression in the Hippocampus of Depressive Rats
In the model group, hippocampal neurons were sparsely and irregularly arranged, with significantly elevated Caspase-1 expression compared to the blank group (p < .01). In contrast, the Western medicine, low-dose, and high-dose Ginseng–Polygonatum groups showed more orderly neuronal arrangement and significantly reduced Caspase-1 expression (p < .01) (Figure 3B).
Effects of Ginseng–Polygonatum on IL-1β Levels in the Hippocampus of Depressive Rats
Compared to the blank group, IL-1β levels in the hippocampus were significantly elevated in the model group (p < .01). Treatment with Western medicine and high-dose Ginseng–Polygonatum significantly reduced IL-1β levels compared to the model group (p < .05, p < .01) (Figure 4A).
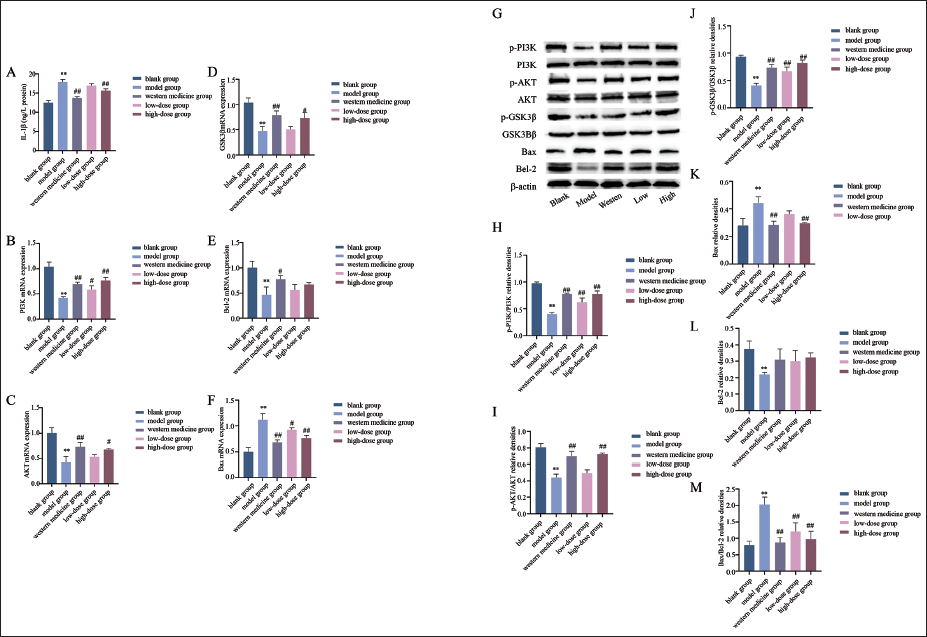
Effects of Ginseng–Polygonatum on PI3K, AKT, GSK3β, Bcl-2, and Bax mRNA Expression in the Hippocampus of Depressive Rats
Compared to the blank control, the model group showed a significant increase in hippocampal Bax mRNA and decreases in PI3K, AKT, GSK3β, and Bcl-2 mRNA (p < .01). Treatment groups exhibited a decrease in Bax and increases in PI3K, AKT, GSK3β, and Bcl2 mRNA, with significant differences seen in the Western medicine and high-dose groups (p < .05, p < .01) (Figure 4B–4F).
Effects of Ginseng–Polygonatum on the Expression of PI3K, AKT, GSK3β, Bcl-2, and Bax Proteins in the Hippocampus of Depressive Rats
Compared to the blank group, the experimental group showed significant decreases in p-PI3K/PI3K, p-AKT/AKT, p-GSK3β/GSK3β, and Bcl-2 levels (p < .01), while Bax and Bax/Bcl-2 levels significantly increased (p < .01). Both the Western medicine and high-dose Ginseng–Polygonatum groups demonstrated significant increases in p-PI3K/PI3K, p-AKT/AKT, and p-GSK3β/GSK3β, along with significant decreases in Bax and Bax/Bcl-2 compared to the experimental group (p < .01) (Figure 4G–4M).
Discussion
Depression in TCM is primarily classified under the category of “Yu syndrome,” characterized by frequent recurrence, high disability, and difficulty in cure. With the rapid increase in clinical neurological disorders, the clinical symptoms of depression patients are generally categorized into three dimensions: emotional, physical, and cognitive (Malhi & Mann, 2018). According to the “Research Domain Criteria (RDoC)” proposed by the National Institute of Mental Health in the US (Knight & Baune, 2019), cognitive dysfunction has been established as one of the core diagnostic criteria for depression.
In TCM, depression is described as “qi stagnation and spirit injury.” The brain, regarded as the residence of the original spirit (Yuan Shen), is believed to be the center of cognitive awareness. Cognitive disturbances in depression are thus attributed to disorders of brain structure. Neurodegenerative changes, including hippocampal volume loss, are frequently observed in depression and are supported by animal models (Czéh & Lucassen, 2007). Studies have confirmed that patients with depression often show damage in the hippocampal CA1 region, contributing to neurological dysfunction, cognitive decline, and worsening depressive symptoms (Chen et al., 2022).
Our experimental studies demonstrated that Ginseng–Polygonatum extract exerts potential anti-depressant effects in CUMS-induced depressive rats. After 14 days of intragastric administration, rats showed significant improvements in locomotor activity and memory, indicating that it may alleviate anhedonia and cognitive impairment associated with depression.
As the smallest unit in TCM compatibility, herbal pairs highlight the targeted and adaptable nature of traditional treatments. Ginseng and Polygonatum, both medicinal and edible, are widely used in both therapy and daily health care. Their pairing, first documented in the Compendium of Materia Medica, reflects the balance of yin and yang—Ginseng tonifies qi and yang, while Polygonatum nourishes yin. They enhance therapeutic effects with good safety and suitability for long-term use, offering a potential alternative to conventional anti-depressants.
The inflammatory hypothesis of depression suggests that stress can trigger inflammation, where IL-1β activates the NF-κB pathway, promoting the release of cytokines like IL-6 and TNF-α. This amplifies inflammation and leads to damage in the hippocampus and prefrontal cortex—regions crucial for learning and memory—contributing to depressive symptoms and cognitive decline (Strenn et al., 2015). Experimental results showed significantly increased hippocampal IL-1β levels in the model group versus the control, supporting the neuroinflammatory hypothesis and confirming successful model establishment.
In the initial network pharmacology analysis, the key focus was placed on the PI3K/Akt/GSK3β signaling pathway, along with apoptosis-related targets such as Bcl-2 and Bax. The PI3K pathway is a promising target for treating neurodegenerative and psychiatric disorders, including Alzheimer’s disease, depression, and anxiety (Duman & Voleti, 2012). Dysregulation of this pathway impairs neuronal function and hippocampal stem cell activity (Emamian et al., 2004). AKT, as a key downstream effector, is often downregulated in depression, highlighting the relevance of this signaling cascade (Lottering & Lin, 2021). GSK3β, a major target of PI3K/AKT signaling, is closely linked to depression; its inhibition reduces tau phosphorylation, neurofibrillary tangles, and cognitive impairment (Xie et al., 2024).
GSK3β also regulates hippocampal neuron apoptosis. Apoptosis, a programmed cell death process, is tightly regulated by Bax and Bcl-2, key proteins involved in depression-related neurodegeneration. Bax promotes apoptosis, while Bcl-2 is anti-apoptotic. The PI3K/AKT pathway inhibits apoptosis via multiple mechanisms: suppressing GSK3β activity (Gai et al., 2016), activating NF-κB to promote cell survival (Dou et al., 2015), and preventing mitochondrial cytochrome C release through Akt activation (Gibson et al., 2002).
Previous and current experiments by our research group have demonstrated elevated NF-κB, Caspase-1, and IL-1β levels in the hippocampus of CUMS-induced depressive rats. Inflammasomes regulate Caspase-1 activation in immune cells, leading to IL-1β maturation and inflammation (Wang et al., 2020). Bcl-2 not only prevents apoptosis but also reduces IL-1β release by inhibiting Caspase-1, thereby alleviating neuroinflammation. The Bax/Bcl-2 ratio serves as an indicator of cell apoptosis. After treatment with Ginseng and Polygonatum, this ratio significantly decreased in CUMS rats, indicating that the PI3K/AKT pathway effectively inhibits neuronal apoptosis.
Conclusion
The experiment suggests that the combination of Ginseng–Polygonatum may exert anti-depressant and cognitive protective effects in rats, possibly by regulating cell apoptosis via the PI3K/AKT signaling pathway. This study presents a novel development of a medicinal and edible homologous herbal pair, supporting the application of the “medicine and food homology” theory in treating modern mental disorders. It also offers a preliminary scientific basis for potential dietary interventions in depression. However, the study is limited by a small sample size and lacks differentiation based on TCM syndromes. Further research with larger samples and clinical validation is needed.
Footnotes
Abbreviations
AKT: Protein kinase B; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; Caspase-1: Cysteine-aspartic protease 1; CUMS: Chronic unpredictable mild stress; GSK3β: Glycogen synthase kinase 3 beta; IL-1β: Interleukin-1 beta; PI3K: Phosphoinositide 3-kinase.
Acknowledgments
The authors would like to express their sincere gratitude to Yang Li and Cairong Ming for their valuable technical assistance and experimental support. Special thanks to the Engineering Technology Center for Innovative Chinese Medicine, Liaoning University of Traditional Chinese Medicine, for their insightful discussions and suggestions throughout the course of this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal study was reviewed and approved by the Experimental Animal Ethics Committee of Liaoning University of TCM, with an ethics review permit number (21000042022048).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the innovative talents in colleges and universities of the China Postdoctoral Science Foundation (No. 2021MD703843); the Liaoning Provincial Higher Education Innovative Talent Support Program (No. LR2019039); the Natural Science Foundation of Liaoning Province (2024-BS-135); and the Open fund of Key Laboratory of Ministry of Education for TCM Viscera-State Theory and Applications, Liaoning University of Traditional Chinese Medicine (No. zyzx2104).
Informed Consent
The informed consent has been obtained for the study.
