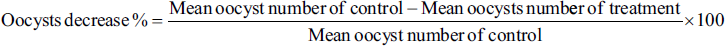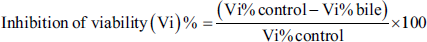Abstract
Background
Plant products are a reliable source of traditional and contemporary medications.
Objectives
This research investigates the impact that the Calotropis procera plant has on the Eimeria magna oocysts and sporozoites.
Materials and Methods
Twelve well-plates of 3 mL containing 1 × 104 unsporulated oocysts were revealed to six treatments: 2.5% potassium dichromate solution as the non-treated control, four concentrations of C. procera extract (12.5, 25, 50, and 100 mg/mL), and toltrazuril 25 mg/mL as traditional medicine, in addition to 250, 500, 750, and 1,000 µg/mL concentrations for anti-sporozoites activities. The mixture was examined (after 24, 48, 72, and 96 h) for oocysticidal activities and after 12 and 24 h for anti-sporozoite activities.
Results
The results showed that the extract contained five chemical components. The extract exhibited notable inhibition of E. magna oocysts at 100 mg/mL doses, with suppression rates of approximately 89% after 96 h, nearly similar to the utilized reference drug, toltrazuril. While C. procera extract exhibited inhibition, the highest suppression of 89% of E. magna sporozoite viability was observed at a concentration of 1,000 µm/mL at 24 h. The inhibitory rate increases with a more extended incubation duration and a higher concentration ratio.
Conclusion
The findings demonstrated that C. procera extract possesses inhibitory properties for oocyst sporulation, confirming that using the extract to treat coccidiosis is effective.
Introduction
Rabbits are regarded as a significant and nutritious source of animal protein. They are vulnerable to significant infections that might diminish their output, resulting in substantial economic losses. Coccidiosis is a significant protozoan illness caused by Eimeria species (Abd El-Ghany, 2020). This condition poses substantial challenges in commercial and small-scale rabbit farming, leading to economic losses and animal welfare concerns (Oseni & Lukefahr, 2014). A variety of circumstances, such as the parasite’s life cycle, the host’s immune responses, and the limitations of available treatments, make managing coccidiosis difficult (Cox, 1998). Current treatments for coccidiosis primarily involve the use of anti-coccidial drugs, such as sulfonamides and ionophores (Martins et al., 2022). However, these treatments have notable limitations, such as drug resistance and side effects, including toxicity and disruption of gut microbiota (Alexander et al., 2017). Given the associated risks of conventional treatments for coccidiosis, there is an increasing trend toward exploring medicinal herbs as alternative solutions.
Calotropis procera, belonging to the Asclepiadaceae family, is an evergreen perennial shrub with unique medicinal properties (Mandila, 2021). Its latex contains triterpenoids, alkaloids, cardenolides, anthocyanins, resins, and proteolytic enzymes (Mali et al., 2019). C. procera thrives in arid environments and open spaces with little competition (Khan et al., 2023). Treatments for leprosy, fever, menorrhagia, malaria, and snake bites have all been linked to the giant milkweed (McGaw et al., 2022). With known benefits in various ailments, including analgesic, anti-cancer, anti-coagulant, anti-inflammatory (Jain et al., 2014), and anti-microbial properties, C. procera has demonstrated its efficacy in preventing coccidian and inhibiting oocyst sporulation in Eimeria papillata (Mpofu, 2022; Murshed et al., 2022). Recent studies have also highlighted its oocysticidal activity against Eimeria stiedae (Murshed et al., 2023).
The Eimeria genus is a predominant cause of coccidiosis, a widespread and impactful parasitic infection affecting animals. Particularly in local rabbit populations, it leads to substantial mortality rates, contributing to annual losses reaching up to 13 billion dollars in poultry farming (Blake et al., 2020; Pakandl et al., 1995). Eimeria magna, identified as a slightly pathogenic species developing in the duodenum, adds to the complexity of this issue (Pakandl, 2005). The life cycle of Eimeria involves both external and endogenous phases, with its classification as an obligatory parasite in the Apicomplexa phylum. Entering the host through the oral route, these parasites infect and proliferate within mucosal epithelia and various digestive tract sections, causing gastrointestinal damage, inflammation, bloody or watery feces, and other symptomatic manifestations, morbidity, and death (Chapman, 2014). According to scientific studies, this pathogen has the potential to disrupt the microbiota of the intestinal tract and make the environment of the intestinal tract more conducive to the growth of pathogenic bacteria like Clostridium perfringens, which can ultimately lead to necrotic enteritis (Madlala et al., 2021). Eimeria infection destroys the host’s mucosal cells, leading to increased cell permeability, leakage of nutrients and plasma proteins, reduced digestion, and decreased protein absorption. These side impact contribute to coccidiosis’s clinical and subclinical manifestations (Yang et al., 2020). Dehydration, anemia, hypoproteinemia, and malabsorption of nutrients are among the potential outcomes of induced villi atrophy, which can also lead to electrolyte imbalance. The consequence of the problem is that commercial rabbits suffer economic costs (Montoro-Huguet et al., 2021). Managing these disorders necessitates costly therapeutic interventions, including chemoprophylaxis and live-attenuated vaccines, with notable downsides such as potential parasite resurgence and medication resistance (Madlala et al., 2021). Given the existing challenges, searching for novel agents with unique modes of action is imperative (Muthamilselvan et al., 2016). Natural products, such as C. procera, have emerged as potential supplementary and alternative means of controlling coccidiosis. C. procera’s anti-coccidial action suggests it may be a useful substitute for chemotherapy drugs for Eimeria species (Murshed et al., 2023).
Considering this background, this research investigates the impact of C. procera in handling rabbit E. magna oocysts and sporozoites through in vitro trials, providing valuable insights into potential alternative therapies for coccidiosis.
Materials and Methods
Collected Plant Leaf
Extracts from the leaves of C. procera were obtained from the herbal located in the desert of Riyadh, Saudi Arabia, and the plant’s identification was validated by a taxonomist in the Botany Department of King Saud University. The leaves were desiccated for three days in an incubator at 43°C, thereafter pulverized into a powder, and immersed by blending with 80% methanol for 30 h at +4°C with periodic agitation. Whatman filter papers were employed to purify the leaf extract. The generated extract was subsequently concentrated and dried using a rotary vacuum evaporator (Yamato RE300, Tokyo, Japan) at 40°C and reduced pressure. The crude extracts were stored at −20°C until utilized in an experiment. Distilled water was employed to dissolve the material for the various trials (Begashaw et al., 2017).
Infrared Spectroscopy
After preparing the sample to be analyzed, a minute portion of the extract was homogenized and combined with an excessive quantity of potassium bromide powder (1:99 wt%, 102648742, Sigma–Aldrich, Berlin, Germany). The material was subjected to a rough mashing process before being placed in a pellet-forming die. An optical spectrometer manufactured by Thermo Scientific and referred to as the NICOLET 6700 Fourier-transform infrared spectrometer (FT-IR) was utilized to analyze the infrared spectrum. The ability to forecast which components had the potential to be the most active was made. The word “several waves” (cm−1) is utilized to represent the greatest significant number of waves that are absorbed. At a temperature of 25°C, spectra were recorded with a resolution of 4 cm, and the spectral range extended from 4,000 to 400 cm−1 (Mansoori et al., 2020).
Parasite
The study was conducted by Veterinary and Sanitary Expertise (Saudi Arabia) at the King Saud University Parasitology Laboratory of the Department of Zoology. Samples of the rabbits’ feces were analyzed when it was discovered that they were naturally infected with Eimeria. The feces were examined for oocysts of Eimeria spp. The type was obtained from E. magna oocysts (Ogedengbe, 2011).
Eimeria magna Oocysts Sporulation Test by C. procera In Vitro
To determine the impacts different concentrations of C. procera have on the sporulation of E. magna oocysts, an experiment was conducted both in vitro. In this study, C. procera was introduced into a mixture of 2.5% potassium dichromate that included 1 × 104 oocysts. The ultimate concentration of the mixture ranged from 5% to 1.25%. It was decided not to treat the oocysts that were in the control group. Every treatment was carried out three times to ensure accuracy. After each petri dish had been incubated at a temperature of 28°C for 4 days, the sporulation percentage was determined for each treatment by counting the number of oocysts that had sporulated and those that had not sporulated using the McMaster technique. Additionally, morphological alterations, malformations, and the percentage of destruction were evaluated for each therapeutic intervention.
Experiment Application
Unsporulated E. magna oocysts collected from the duodenum of infected rabbits were used for oocyst sporulation analysis. E. magna oocyst sporulation was conducted in an aqueous solution of 2.5% potassium dichromate (K2Cr2O7) (104 oocysts/mL) with or without 100 µg/mL of extracts at 25°C for 70 h. An examination under a microscope and a count of the number of sporulation and inhibitory oocysts in a total of 100 oocysts were used to determine the percentage of sporulated oocysts.
Preparation of Sporulated Oocysts
After obtaining E. magna oocysts from the duodenum, these oocysts were cleaned and concentrated using the flotation method. The sporulated oocysts were stored in K2Cr2O7 at a concentration of 2.5% at a temperature of 4°C until they were utilized to conduct experimental infections. The E. magna field isolates were maintained alive by undergoing periodic transmission (passage) through young rabbits in the Animal House of the Zoology Department, which also houses the Parasitology Laboratory, according to the method used by Schito et al. (1996).
Anti-sporozoite Activity of C. procera Under In Vitro Conditions
Phosphate-buffered saline (PBS) at pH 7.5 was used to clean oocyst samples that had been stored in potassium dichromate solution. The centrifuge was used in Falcon tubes with 10 mL suspended at 1,008 g for approximately 15 min, 3–6 times until the K2Cr2O7 solution was completely removed. The oocysts were incubated in a water bath for 1 h at a temperature of 42°C, shaking the tubes. The parasite suspension with 1,000 sporozoites was then split into two parts, each containing 125, 250, 500, and 1,000 µg/mL of the extract. These parts were then put into a 3 mL Petri dish with 24 wells. To facilitate comparison, a reference treatment of toltrazuril at a dose of 30 µg/mL was used. In addition, the potassium dichromate solution served as a control. Each treatment was repeated three times under the same conditions. The results were recorded after 12 and 24 h to determine the viable sporozoites. A McMaster chamber was utilized to carry out sporozoite counting. After determining the number of viable and non-viable sporozoites, the viability of viable sporozoites was calculated from a total of 100 using the following mathematical law:
Statistical Analysis
Using a statistical program SPSS 22 (SPSS, Chicago, IL, USA), one-way analysis of variance (ANOVA) was carried out for every experimental group, along with Duncan’s post hoc test for any further multiple comparisons. A significant level was set at p ≤ .05. To evaluate the correlations between variables, a straightforward linear correlation study was conducted using Pearson’s approach.
Results
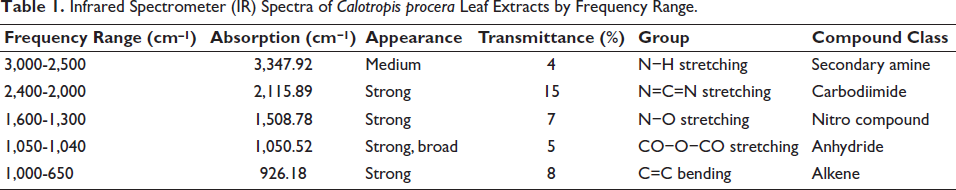
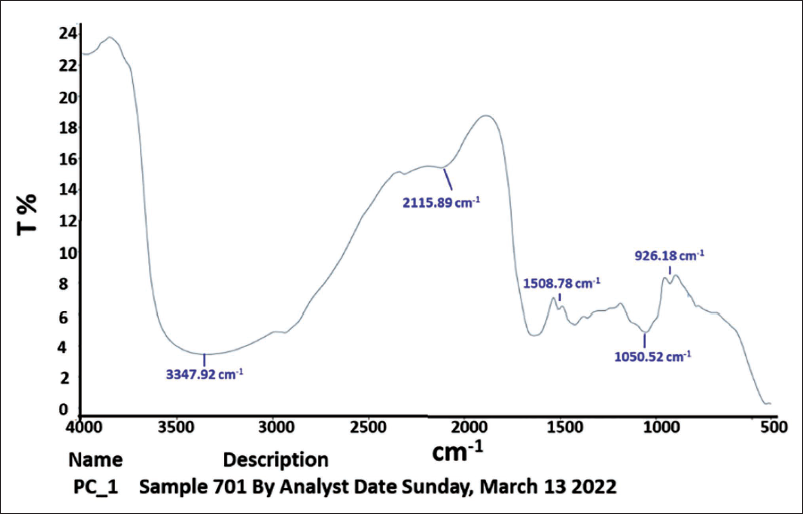
To conduct spectroscopy on the leaf extracts of C. procera, an FT-IR spectrometer was employed. The results of the analysis revealed that the principal bands were located at 3,347.92, 2,115.89, 1,508.78, 1,050.52, 1,050.52, and 926.18 cm−1, respectively, as illustrated in Figure 1. The active phytochemical components were produced in the following manner: N−H stretching, N=C=S stretching, N−O stretching, CO−O−CO stretching, and C=C bending, with an absorbance ranging from 400 to 4,000/cm−1 (Table 1).
Infrared Spectrometer (IR) Spectra of Calotropis procera Leaf Extracts by Frequency Range.
The Examination of the Leaf Extract of Calotropis procera was Conducted Using an FT-IR Spectrometer for Infrared Spectroscopy.
The effects of different doses of C. procera during different periods on preventing E. magna sporulation were tested. The methanolic extract showed that the longer the incubation periods, the greater the rate of inhibition, and the opposite for the sporulation rate. As the incubation time increased, the rate of sporulation inhibition continued to vary depending on the extract concentration (p ≤ .05) in the treatment groups. In general, the best dose for inhibiting the parasite was 100 mg/mL compared with the control group and the reference treatment (Figure 2).
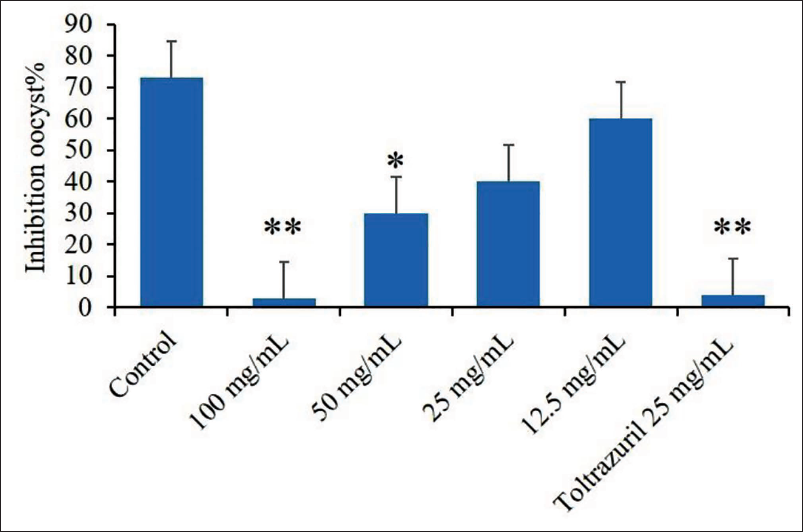
The experimental groups showed highly significant rates of inhibition with increasing dose, and the statistical analysis showed that there were statistically significant differences between the 100 mg/mL dose compared to the control group (p < .01) and between the reference drug of toltrazuril of 30 µg/mL compared to the control group (p < .05). Meanwhile, the rest of the other doses did not show any statistically significant differences (Figure 3).
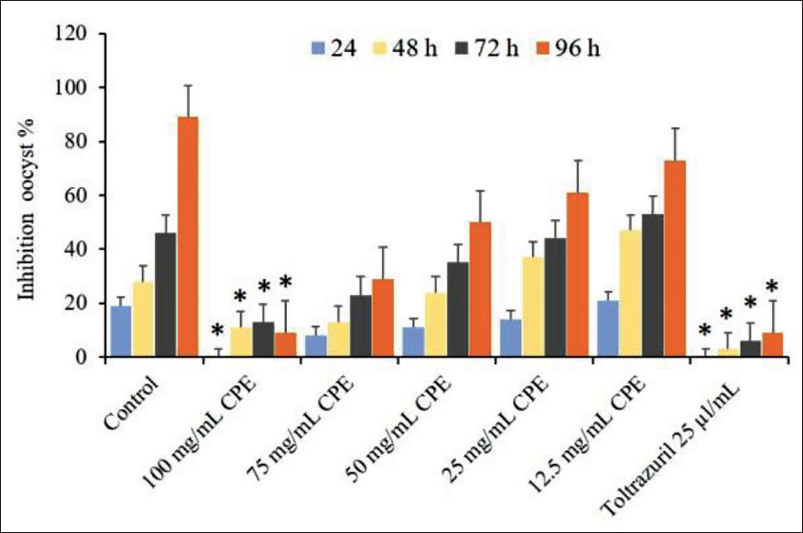
Figure 4 apparition the main effects of sporulation time and experimental groups on sporulation and non-sporulation (%) of E. magna oocysts sporulation in vitro. It demonstrates that the sporulation percentage increased with incubation time and, conversely, the non-sporulation percentage decreased. The rate of sporulation inhibition rose considerably with incubation time up to 96 h (p < .05), showing that the rate of sporulation inhibition differed significantly between 24, 48, and 72 h exposures (Figure 4).
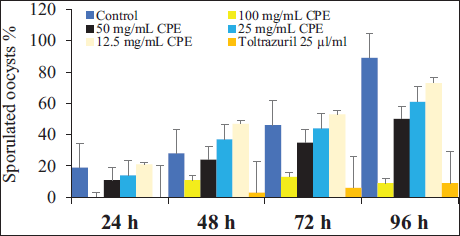
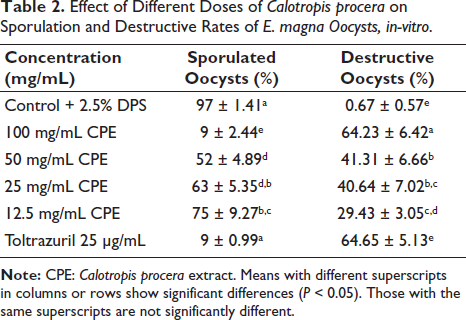
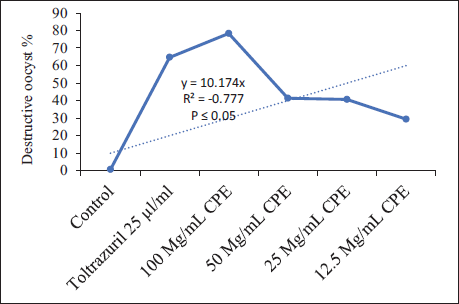
The microscopic examination and counting of oocysts treated with C. procera at various concentrations resulted in deformed oocysts with cracked walls and lysis. The sporulation rate for the control after 96 h was 97% ± 1.41%. At the doses of 100 mg/mL of C. procera and the reference drug 25 µg/mL of toltrazuril (p ≤ .05), the sporulation rate was significantly inhibitory. The inhibition rates of C. procera and the reference drug relative to the control were 97%, 9%, 52%, 63%, 75%, and 9%, respectively. Also, the oocysticidal effect was confirmed by the higher ratio of degenerated oocysts 0.67%, 64.23%, 41.31%, 40.64%, 29.43%, and 64.65% for control, 100, 50, 25, 12.5 mg/mL of extract, and 25 µg/mL of toltrazuril, respectively compared to the control (Table 2).
Effect of Different Calotropis procera on Sporulation and Destructive Rates of E. magna Oocysts, in-vitro.
There was a negative correlation between sporulation and concentration percentage. The linear equation showed the highest coefficient of determination (R2) p value is .000244, The result is significant at p < .05. for sporulation. The p value is .039412. The result is significant at p < .05, for the destruction (Figure 5).
Anti-sporozoite Activity of C. procera: In Vitro
A test was conducted to determine the sporozoite vitality rate in C. procera according to the length of the incubation period and the extract concentration rate in measuring the vitality. It was observed after 12 and 24 h of incubation that 1,000 µg/mL of extract and 25 µg/mL toltrazuril showed statistically significant changes (p < .05) in the viability rate of E. magna sporozoites when compared to the control treatment (K2Cr2O7). In contrast, the sporozoites at lower extract concentrations (500, 250, and 125 µg/mL) displayed varying degrees of vitality (Figure 6).
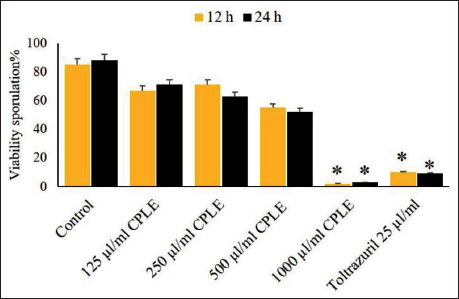
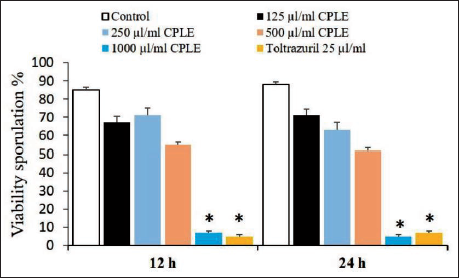
Figure 7 illustrates that the experimental groups have a substantial effect on sporozoite vitality rates, which rise with increasing incubation time, and vice versa for non-vitality percentage. Therefore, the sporozoite inhibition rate increased considerably with incubation time up to 12 h (p < .05); the sporozoite inhibition rate did not change significantly between 12- and 24-h exposure (Figure 7).
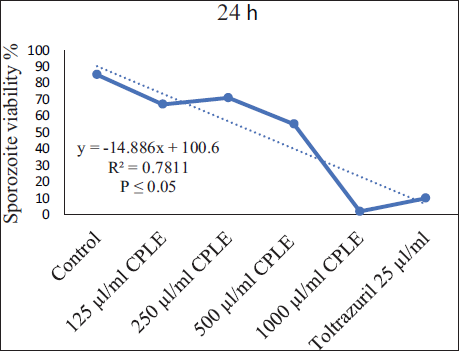
The viability % of sporozoites exhibited a negative correlation with increasing concentration (y = −18.8 × + 15.133, R² = 0.9453, p < .05), while there were no significant differences at the 1,000 µg/mL concentration of extract and the reference drug (25 µg/mL of toltrazuril). This suggests that the efficacy of the extract at this concentration is comparable to the established reference drug. This finding raises interesting questions about the potential of C. procera leaf extract (CPLE) as a promising alternative in the context of E. magna control, especially considering its equivalence to toltrazuril (Figure 8).
Discussion
Coccidiosis, a globally prevalent disease, significantly hampers rabbit productivity, leading to substantial financial losses. The associated morbidity and mortality from these parasitic infections not only impair livestock productivity but also pose a severe health risk (Nowland et al., 2015). Understanding the current species and prevalence of intestinal parasites becomes crucial for minimizing financial losses in the rabbit sector, assessing infection risks, and implementing effective control measures (Nwosu et al., 2007). Numerous authors have explored the impact of diverse plant extracts on oocyst viability and the duration of sporulation (Fatoba & Adeleke, 2018). These extracts have proven effective in inhibiting sporulation and reducing the vitality of Eimeria oocysts (Khan et al., 2020). The consistent application of natural extracts in animal farms, including rabbits and poultry, could emerge as a therapeutic and preventive strategy to diminish the survival rate of oocysts belonging to the genus Eimeria and prevent them from maturing (Kuralkar & Kuralkar, 2021). The impact of natural product extracts on the oocysts of these parasitic protozoa has been the subject of extensive research. In our previously published work, experiments detailed the effects of Nerium oleander leaf extract against E. magna and Eunotia exigua oocysts, bile against Eimeria columbae and E. papillata oocysts, and C. procera leaf extracts against E. stiedae oocysts (Murshed et al., 2023). In a methanolic extract exposed to oocysts for 96 h, the results demonstrated a significant inhibition, ranging from 90% to 98%.
The effectiveness of C. procera extract against E. magna can be attributed to several mechanisms: Alkaloids found in C. procera, such as calotropin and uscharin, have been demonstrated to have cytotoxic effects on different pathogens (Singh et al., 2024). Flavonoids are known for their antioxidant properties; flavonoids can disrupt cellular processes in parasites (Chagas et al., 2022). The extract may inhibit the germination of Eimeria oocysts by interfering with environmental cues necessary for activation (Risco-Castillo, 2022). Flavonoids and other active compounds inhibit metabolic pathways in E. magna (Murota et al., 2018). For instance, targeting enzymes involved in glycolysis or mitochondrial function reduces ATP production, impairing the parasite’s ability to thrive and reproduce (Lyu et al., 2023). Phytochemicals such as flavonoids could play a role in modulating signaling pathways that trigger oocyst activation (Memariani et al., 2024). They may interfere with the energy metabolism of E. magna by inhibiting key enzymes involved in glycolysis and other metabolic pathways (Morad et al., 2023). Tannins have astringent characteristics and can bind to proteins, potentially compromising the structural integrity of the parasite’s surface proteins and enzymes that are essential to life (Bah & Etonihu, 2024; Chithra et al., 2023). Saponin compounds can alter membrane permeability, leading to cell lysis (Berlowska et al., 2015). Saponins from C. procera may damage the oocyst membranes of Eimeria, making it easier for the contents inside to leak out and eventually kill the parasite (Murshed et al., 2022). The anti-parasitic activity of C. procera extract against E. magna oocysts and sporozoites appears to be multifaceted, involving a combination of phytochemical interactions that disrupt cellular integrity, inhibit metabolic processes, and potentially enhance host immunity.
This study investigated the impact of C. procera extract over varying durations (24, 48, 72, and 96 h) and concentrations (12.5%, 25%, 50%, and 100%) on E. magna oocyst sporulation in vitro. The highest effectiveness of the tested concentrations resulted in approximately 91% inhibition of sporulation after 96 h of exposure to a 100% C. procera concentration. In contrast, the control group (K2Cr2O7) exhibited a high level of oocyst sporulation. The efficacy of different extract concentrations varied depending on the concentration percentage and incubation period of the oocysts. Comparing extract concentrations of 50%, 25%, and 12.5% to the control group, the highest rates of inhibition and lowest rates of sporulation were observed, while lower concentrations showed no statistically significant differences. These findings align with Murshed et al. (2023), who reported that C. procera may reduce E. stiedae oocysts at high concentrations. Extended incubation times and higher doses often improved the inhibition rate. Moreover, at a dose of 1,000 µg/mL, C. procera exhibited the maximum inhibition rate of Eimeria steal sporozoite viability (92%) and the lowest rate of inhibition (8%) at 125 µg/mL. These results are consistent with (Murshed et al., 2022), who found that oocyst inhibition in potassium dichromate could reach approximately 96% at higher concentrations of C. procera. The outcomes indicated a gradual and dose-dependent decrease in oocyst sporulation. The extract inhibited oocyst sporulation at doses of 50%, 25%, and 10%, with corresponding inhibition rates of 71.7%, 33.11%, and 19.88%. Sporulation and inhibition were directly related over an extended period (Gupta et al., 2002; Murshed et al., 2023). The effectiveness of other extracts varied based on concentration percentage and incubation time. Generally, the extract concentration of 100% exhibited the highest rates of inhibition and the lowest rates of sporulation compared to the control group. Lower concentrations (50%, 25%, and 12.5%) of C. procera did not show statistically significant differences. These findings concur with Murshed et al. (2022), who observed that sheep bile could suppress E. stiedae oocysts at high concentrations. Increased incubation time and larger doses generally enhanced the inhibition rate. Additionally, C. procera showed the highest rate of inhibition of E. stiedae sporozoite viability (92%) at 1,000 µg/mL and the lowest rate of inhibition (8%) at 125 µg/mL. The current findings align with Murshed et al. (2023), who found that higher concentrations of extract could inhibit oocysts by approximately 96% in potassium dichromate. Moreover, the results demonstrated the ability to suppress oocyst sporulation in a dose-dependent manner over time. In concentrations of 50%, 25%, and 10% of C. procera, the inhibition rates were 71.7%, 33.11%, and 19.88%, respectively. Therefore, a direct association between inhibition and sporulation was observed over an extended period (Bezerra et al., 2017; Murshed et al., 2023).
The results align with Remmal’s study, where the effects of eight plant extracts, essential oils, and their combination were investigated as potential treatments for coccidial infection, particularly against E. tenella (Remmal et al., 2013). Remmal independently assessed the primary components of essential oils, including carvacrol, isoeugenol, thymol, eugenol, and carvone, and found their effectiveness against coccidian. In another study, the anti-coccidial effects of essential oils from Artemisia argyi and Camellia sinensis, along with extracts from Punica granatum L., Plantago asiatica L., Bidens pilosa L., Acalypha australis L., Pteris multifida Poir, and Portulaca oleracea L., were explored for their potential as anti-coccidial agents. These substances demonstrated the ability to inhibit E. tenella invasion both in vitro and in vivo (Han et al., 2022). A study by Cedric et al. (2018) suggested that P. macrophylla extracts might have influenced the decreased sporozoite viability by interfering with calcium-mediated signaling in the sporozoites. Vitis vinifera leaf extracts also affected E. papillae oocysts, and these are consistent with our results. The findings affirm that C. procera effectively prevents the formation of oocysts.
Conclusion
The elimination of parasites is critical to the health of organisms, which in turn can contribute to the development of sustainable production methods that maintain the health of consumers. It is concluded that the extract of C. procera leaves played an important role in inhibiting oocysts and preventing their sporulation, and it also has a destructive effect against oocysts. Additional empirical research is required to elucidate the pharmacological and therapeutic characteristics of the plant to identify active ingredients. The development of substantial medicinal drugs derived from the active phytochemical constituents of C. procera will be enabled.
Footnotes
Abbreviations
ANOVA: Analysis of variance; CPE: Calotropis procera extract; CPLE: Calotropis procera leaf extract; E. magna: Eimeria magna; FT-IR: Fourier-transform infrared spectrometer; K2Cr2O7: Potassium dichromate; PBS: Phosphate-buffered saline.
Author Contributions
Mutee Murshed: Conceptualization, methodology, software, writing—original draft preparation, writing—review and editing. Jameel Al-Tamimi: Validation, data curation, writing—review and editing. Hind Alzaylaee: Visualization, validation, funding acquisition, project administration. Saleh Al-Quraishy: Conceptualization, methodology, investigation, supervision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The research adhered to ethical guidelines for animal use established by the Kingdom of Saudi Arabia, as outlined by the Ethics Committee at King Saud University, with Ethics Endorsement ID: KSU-SE-21-86.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Thank you to the Project No. PNURSP2025R401, Princess Nourah Bint Abdul Rahman University, Riyadh, Saudi Arabia.