Abstract
Background
Alzheimer’s disease (AD) is a neurological disorder characterized by behavioral and memory deficits, along with cognitive impairments. The majority of AD cases are often present in individuals aged 60 and above.
Objectives
The present work was conducted to investigate the neuroprotective properties of sinensetin against aluminum chloride (AlCl3)-induced Alzheimer’s disease (AD) in rats.
Materials and Methods
The rats were given AlCl3 (175 mg/kg) orally for 25 days to induce AD. Sinensetin (20 and 40 mg/kg) was orally given to the AD rats from the 25th to the 36th day. After treatments, behavioral changes were evaluated using the open-field and Morris water maze assays. The inflammatory cytokines, oxidative stress markers, acetylcholinesterase (AChE), and Aβ protein were evaluated using assay kits. The histopathology study was conducted on brain tissues.
Results
The findings of the behavioral assessments revealed that sinensetin improved the memory and learning capacity of the AD rats. The levels of oxidative and inflammatory markers were decreased, while antioxidant enzyme activities were elevated in the brain tissues of the sinensetin-treated AD rats. The increase in BDNF and PPAR-γ levels and the reduction in p38MAPK and NF-κB levels were noted in the sinensetin-treated AD rats. The results of histopathological analysis revealed the neuroprotective properties of sinensetin. The outcomes of this work exhibited that the sinensetin treatment of the AD rats resulted in improved memory and learning, reduced oxidative stress and inflammation, an increase in BDNF/PPAR-γ levels, and a reduction in p38MAPK/NF-κB levels in the brain tissues.
Conclusion
Therefore, it can be concluded that sinensetin shows promise as a therapeutic candidate to treat AD in the future.
Introduction
Alzheimer’s disease (AD) is a neurological disorder characterized by behavioral and memory deficits, along with cognitive impairments. Over 50 million people worldwide suffer from dementia, and AD accounts for 60%–70% of these incidences, according to WHO data (World Health Organization, 2017). The frequency of AD is projected to increase in the next few years due to the rising global aging population (Gaugler et al., 2021). It is projected that by 2050, there will be a total of 106.8 million individuals diagnosed with AD worldwide. AD poses a significant public health challenge with significant socio-economic consequences. The majority of AD cases are often present in individuals aged 60 and above (Mahaman et al., 2023).
AD has been associated with environmental pollution, specifically with the involvement of various contaminants, including carbon monoxide, ozone, and particulate matter. There is substantial evidence linking aluminum (Al) toxicity to AD (Mesole et al., 2020). Al is a pivotal cause of several neurodegenerative disorders by influencing multiple biomolecules associated with neurotoxicity (Mehrbeheshti et al., 2022). Therefore, the induction of AD in rats using aluminum chloride (AlCl3) serves as a reliable model for investigating the neuroprotective properties of various sample drugs against AD. Due to the prevalence of Al in the environment, everyday activities, and food, it is nearly inevitable to be exposed to it (Ramakrishna et al., 2025). Utilizing this model replicates the defensive and remedial properties of certain compounds in countering AD in humans.
The progression of neurodegenerative diseases has been demonstrated to be impacted by inflammation and oxidative stress (Behl et al., 2021). The accumulation of AlCl3 is tightly connected with the progression of neurodegenerative disorders due to its capacity to stimulate reactive oxygen species (ROS) accumulation and produce oxidative neurotoxicity (Madi et al., 2020). The characteristic features of AD are the abnormal accumulations of hyperphosphorylated tau protein, known as intracellular neurofibrillary tangles (Venugopalan et al., 2021). Multiple studies have demonstrated that several pathways are involved in the progression of AD, such as oxidative stress (Srivastava et al., 2021).
The brain is very susceptible to oxidative stress due to its elevated oxygen usage and inadequate antioxidant system. Therefore, the neurotoxic effects of Al may be attributed to the excessive generation of ROS, leading to significant damage to neurons due to the dysfunction in the antioxidant defense mechanism. This process stimulates the generation of free radicals, leading to high protein and DNA oxidation and lipid peroxidation. This leads to substantial oxidative damage and can lead to brain trauma (Srivastava et al., 2021).
While current medications do not provide a cure or reversal of the neurodegeneration associated with AD, they enhance memory loss and cognitive skills. Cholinesterase inhibitors have been associated with significant side effects (Saeed et al., 2024). Additional systemic consequences associated with cholinergic activity include an urgent need to urinate, a slow heart rate, and fainting. Therefore, it is advisable to closely monitor the patient and adjust the dosage accordingly (Prajapati et al., 2025). Hence, it is important to explore novel approaches for the advancement of pharmacotherapy for AD. Researchers and the pharmaceutical industry have been more interested in a range of natural compounds that have been found to have neuroprotective benefits. The growing preference of scientists for natural products is driven not just by their potential but also by their favorable safety profile. There is a particular focus on natural substances for the development of “multiple-target lead” (MTD) to treat AD (Ayaz et al., 2019).
Sinensetin is a polyethoxylated flavonoid that is present in Orthosiphon aristatus, Orthosiphon spicatus, and other citrus fruits. The concentration of this compound is higher in the peel of citrus fruits compared to other parts of the fruit (Wan et al., 2022). Recently, several previous studies have described that sinensetin has successfully protected pulmonary fibrosis (Wan et al., 2022), suppressed angiogenesis in liver cancer (Li et al., 2022), attenuated cartilage damage, alleviated osteoarthritic symptoms (Liu et al., 2022), and showed immunomodulatory activity (Wang et al., 2022). Patel and Patel (2022) have highlighted the therapeutic effects of sinensetin against various cancers and human complications, including breast cancer, colon cancer, gastric cancer, colitis, diabetes, inflammation, and obesity. However, the therapeutic effects of sinensetin against AD were not studied. Hence, the current work was performed to examine the neuroprotective effects of sinensetin against AlCl3-induced AD in rats.
Materials and Methods
Chemicals
The chemicals and reagents, such as sinensetin, AlCl3, NaCl, and others, were obtained from Sigma-Aldrich, USA. The marker-specific assay kits used for biochemical assays were obtained from Abcam, USA, RnD Systems, USA, and Elabscience, USA, respectively.
Experimental Rats
The 6–8-week aged male Wistar rats weighing over 180–210 g were used in this work and were caged in a sterile enclosure located underneath the well-structured cabin, maintained at a 22–24°C temperature, and subjected to a 12-h light/dark series. During the study, all rats were given unrestricted access to water and food (Bio-Serv, USA).
Experimental Design
The rats were distributed into four groups, labeled as Groups I–IV. Group I rats were the control. Group II rats were given AlCl3 (175 mg/kg) orally for 25 consecutive days to induce AD. From the 25th to the 36th day, they were given 0.9% NaCl at a dosage of 5 mL/kg. An aqueous suspension of AlCl3 was given orally at a dosage of 0.5 mL/100 g. Group III and Group IV rats were exposed to AlCl3, as indicated in Group II, for a duration of 25 days. Additionally, they were orally treated with 20 and 40 mg/kg of sinensetin, respectively, from the 25th to the 36th day.
Open Field Test
A plexiglass device with a wooden floor, measuring 100 cm in width, 100 cm in diameter, and 40 cm in height, was divided into 25 squares. The behavioral changes in rats were observed and recorded for a duration of 5 min using a video camera. The following interpretations were recorded: (a) The count of square boxes traveled only includes where the rat enters the box with all four paws on the ground. The number of squares the rat entered with all four paws was recorded. (b) Grooming, which is the act of licking paws and fur and body scratching. (c) Rearing is the act of smelling, standing straight on the hind limbs, and leaning against a wall with the forelimbs. (d) The identification of emotional expression involved the detection of feces (fecal pellet counts) (Seibenhener & Wooten, 2015).
Morris Water Maze (MWM) Test
The MWM test was employed to examine learning and memory (Vorhees & Williams, 2006). Briefly, a cylindrical water tank with a height of 60 cm and a diameter of 150 cm was filled with tap water until it reached half of its capacity. To achieve opacity, a water-soluble white paint with non-toxic properties was introduced into the water. The pool was effectively partitioned into four quadrants, each of which was almost equal in size and aligned with the cardinal directions (north, south, east, and west). A 10 cm-diameter escape platform was concealed 2 cm under the water’s surface, positioned at a fixed place in the quadrant’s center. The rats were given 60 s to locate the concealed platform and a 20-s rest period before the next trial. If they had delayed their arrival at the platform by 60 s, they were carefully positioned on it and given 20 s of relaxation. The duration required to locate the platform (escape latency) was documented. On the 4th day, the turbid water was substituted with transparent water, and a probing test was conducted by removing the platform and enabling the rats to swim unrestrictedly for 60 s. The duration of time spent within the designated quadrant was monitored.
Analysis of Acetylcholinesterase (AChE)
To estimate the AChE activity, the hippocampus tissues were obtained from the rats and immersed in a sucrose buffer solution with a concentration of 0.25 M for 30 min. The tissues were homogenized and then subjected to centrifugation at 10,000 rpm, and the resulting supernatant was employed to analyze the activity of AChE utilizing a spectrophotometric method.
Quantification of Aβ Protein
The levels of Aβ protein were examined in the brain tissues of the experimental rats using commercially available assay kits, following the procedures recommended by the manufacturer (Elabscience, USA).
Analysis of Oxidative Stress Markers
The malondialdehyde (MDA) levels and the activity of the antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) were examined using assay kits, following the suggested procedures by the manufacturer (Abcam, USA).
Analysis of Inflammatory Cytokines
The interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α levels in the brain tissues were examined using assay kits following the instructions provided by the manufacturer (RnD Systems, USA).
Analysis of PPAR-γ, NF-κB, p38MAPK, and BDNF Levels
The PPAR-γ, NF-κB, p38MAPK, and BDNF levels in the brain tissues of the experimental rats were studied using assay kits provided by Abcam, USA, following the manufacturer’s protocols.
Histopathological Analysis
The hippocampal sections of the experimental rats were excised and cut into small pieces. These pieces were subsequently immersed in Bouin’s fixative solution for a duration of 24 h. Subsequently, the sections underwent paraffinization and were then cut into slices with a thickness of 4–6 µm, stained using hematoxylin and eosin, and subsequently evaluated in a blinded manner using an optical microscope.
Statistical Analysis
The assay outcomes were shown as the Mean ± SD of triplicate assays, which were analyzed using GraphPad software (version 9). The study analyzed statistical differences between the experimental groups using a one-way analysis of variance (ANOVA), subsequently using Tukey’s post hoc test with p < .05 as significant.
Results
Effect of Sinensetin on the Memory and Learning Ability of the AD Rats Analyzed by Open Field Test
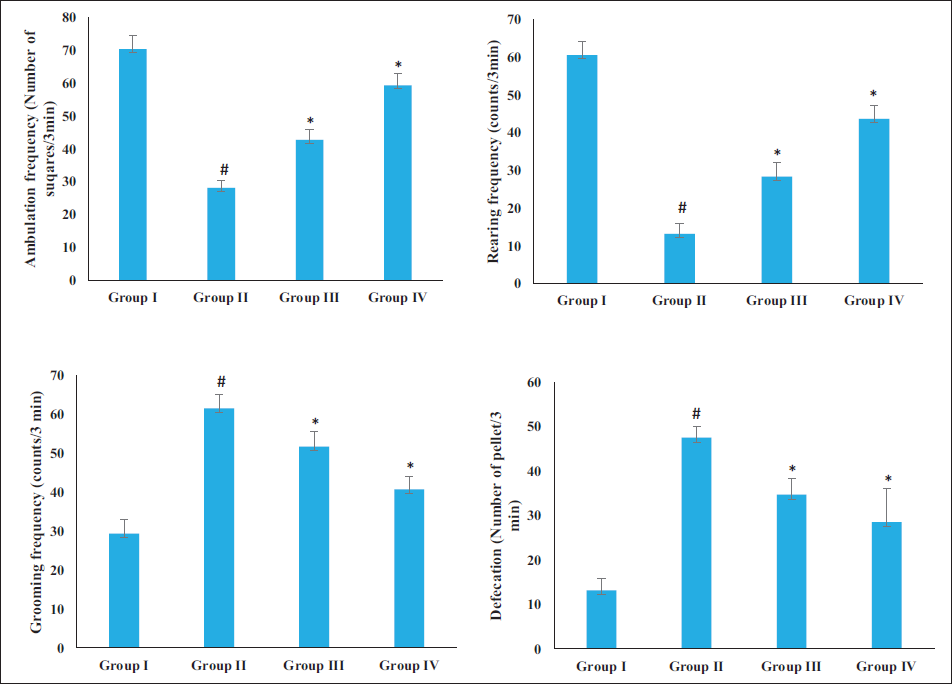
The changes in the behavior of both experimental rats were examined by the open field test, and the results are illustrated in Figure 1. AD rats induced by AlCl3 exhibited reduced rearing and ambulation frequencies and increased defecation (with an increase in fecal pellets) and grooming frequency than the control. In addition, the 20 and 40 mg/kg of sinensetin in the AD rats led to an increase in the frequency of ambulation and rearing while reducing the frequency of grooming and defecation. These results suggest that sinensetin regulates the behavioral changes in AD-induced rats.
Effect of Sinensetin on the Aluminum Chloride (AlCl3)-induced Behavioral Changes in the Alzheimer’s Disease (AD) Rats Analyzed by Open Field Test. The Values of the Treated Groups and the Control Group Were Statistically Analyzed by One-way Analysis of Variance (ANOVA) Followed by Tukey’s post hoc Test Using SPSS Software, and Values are Shown as Mean ± SD; #p < .01 Indicates That Values are Significant from Control, *p < .05 Indicates That Values are Significant from the AD-induced Group.
Effect of Sinensetin on the Memory and Learning Capacity in the AD Rats Analyzed by MWM Assay
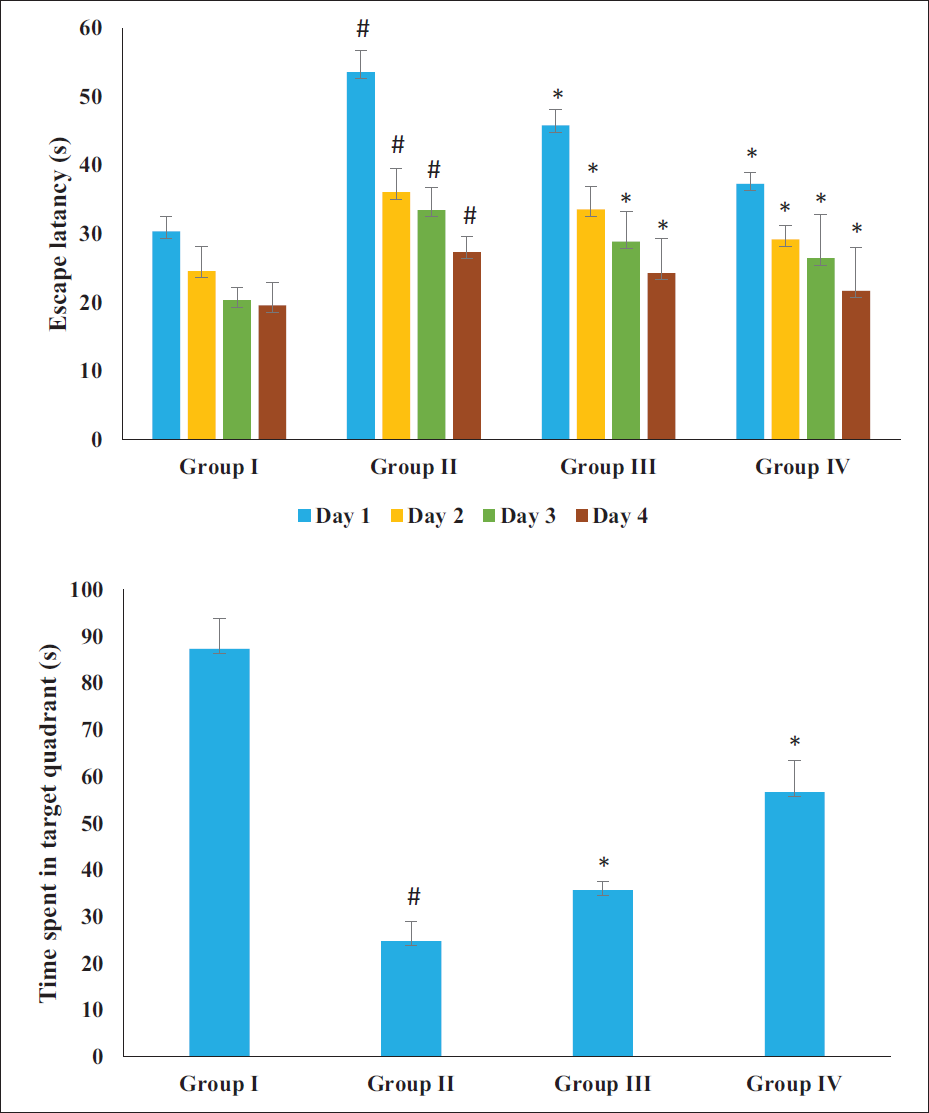
The results of the MWM test demonstrated the impairments in learning and memory in the AD rats induced by AlCl3 (Figure 2). The obvious increase in escape latency revealed that the cognitive abilities of the AD rats were severely impaired. The administration of AlCl3 significantly enhanced the escape latency and inhibited their memory, as seen by the decrease in the duration spent in the target quadrant. The sinensetin treatment had a significant effect on reducing these behavioral deficits. Treatment of the AD rats with 20 and 40 mg/kg of sinensetin resulted in reduced escape latency and significantly augmented time spent in the target quadrant. These findings highlight the beneficial activity of sinensetin in modulating behavioral deficits in AD rats.
Effect of Sinensetin on the Aluminum Chloride (AlCl3)-induced Behavioral Changes in the Alzheimer’s Disease (AD) Rats Analyzed by Morris Water Maze (MWM) Assay. The Values of the Treated Groups and the Control Group Were Statistically Analyzed by One-way Analysis of Variance (ANOVA) Followed by Tukey’s post hoc Test Using SPSS Software, and Values are Shown as Mean ± SD; #p < .01 Indicates That Values are Significant from Control, *p < .05 Indicates That Values are Significant from the AD-induced Group.
Effect of Sinensetin on the Aβ Protein Level and AChE Activity
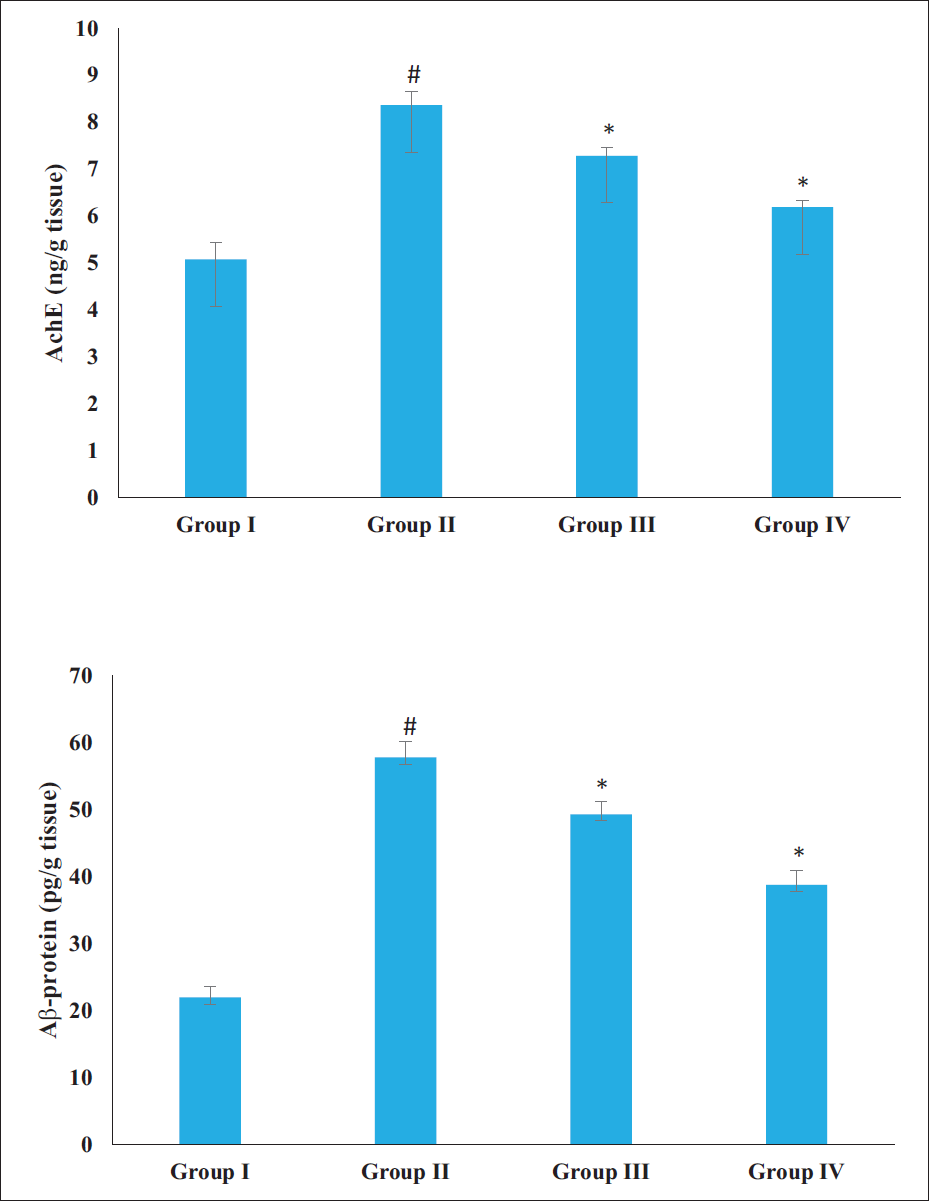
The AChE activity and Aβ protein level in the brain tissues were drastically augmented in the AD rats induced by AlCl3, compared to the control. Figure 3 showed that the treatment of rats with AD with 20 and 40 mg/kg of sinensetin demonstrated a diminution in the AChE activity and Aβ protein levels. Therefore, it was clear that the sinensetin treatment may prevent amyloid plaque formation and promote neurotransmission in AD rats by inhibiting the Aβ protein and AChE activity, respectively.
Effect of Sinensetin on the Aβ Protein Level and Acetylcholinesterase (AChE) Activity in the Brain Tissues of Aluminum Chloride (AlCl3)-induced Alzheimer’s Disease (AD) Rats. The Values of the Treated Groups and the Control Group Were Statistically Analyzed by One-way Analysis of Variance (ANOVA) Followed by Tukey’s post hoc Test Using SPSS Software, and Values are Shown as Mean ± SD; #p < .01 Indicates That Values are Significant from Control, *p < .05 Indicates That Values are Significant from the AD-induced Group.
Effect of Sinensetin on the Oxidative Stress Markers
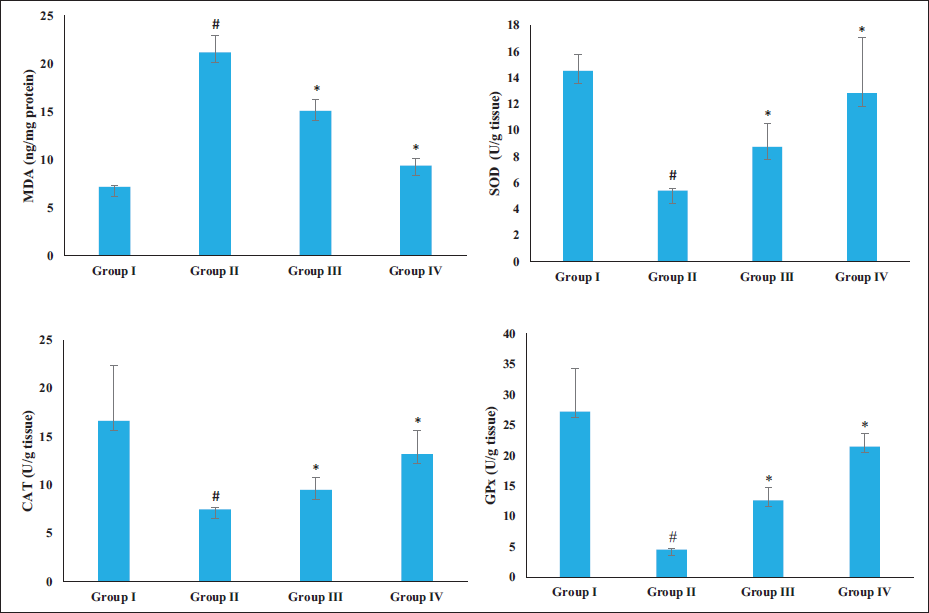
The levels of MDA and the SOD, GPx, and CAT activities were analyzed, and the results of this analysis are presented in Figure 4. The rats with AD induced by AlCl3 showed an upsurge in the MDA level and a reduction in the SOD, GPx, and CAT activities than the control. Notably, when the AD rats were given a dosage of 20 and 40 mg/kg of sinensetin, there was a considerable reduction in the MDA levels and an improvement in the SOD, GPx, and CAT activities in the brain tissues. These results highlight the antioxidant potential of sinensetin.
Effect of Sinensetin on the Oxidative Stress and Antioxidant Enzyme Activities in the Brain Tissues of Aluminum Chloride (AlCl3)-induced Alzheimer’s Disease (AD) Rats. The Values of the Treated Groups and the Control Group Were Statistically Analyzed by One-way Analysis of Variance (ANOVA) Followed by Tukey’s post hoc Test Using SPSS software, and Values are Shown as Mean ± SD; #p < .01 Indicates That Values are Significant from Control, *p < .05 Indicates That Values are Significant from the AD-induced Group.
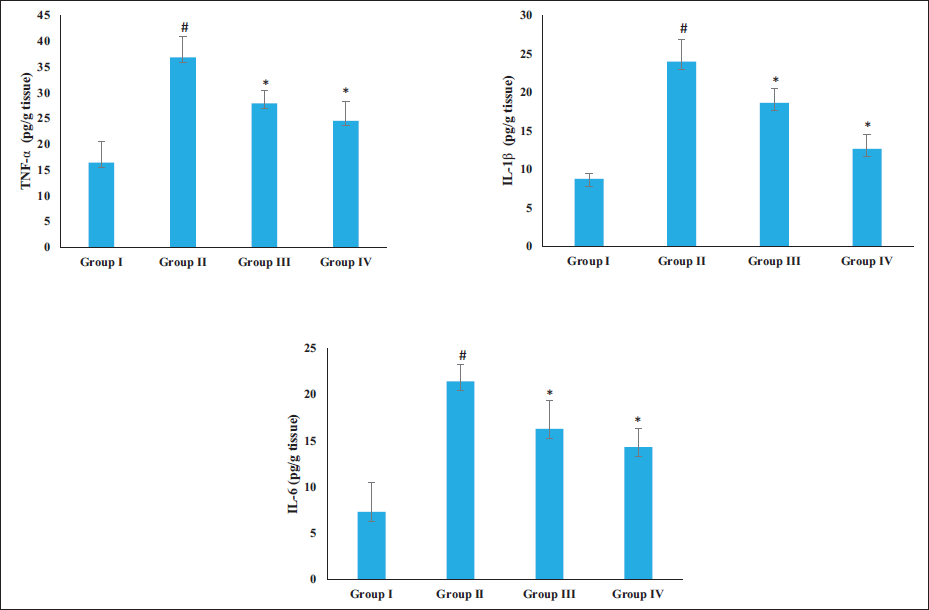
Effect of Sinensetin on the Inflammatory Cytokine Levels
The IL-1β, IL-6, and TNF-α levels in the brain tissues of both control and treated rats are shown in Figure 5. These cytokines were more abundant in the brain tissues of AD rats. The treatment of the AD rats with 20 and 40 mg/kg of sinensetin exhibited a reduction in these cytokine levels. The sinensetin treatment successfully decreased these cytokines, which proves its anti-inflammatory activity.
Effect of Sinensetin on the Inflammatory Cytokine Levels in the Brain Tissues of Aluminum Chloride (AlCl3)-induced Alzheimer’s Disease (AD) Rats. The Values of the Treated Groups and the Control Group Were Statistically Analyzed by One-way Analysis of Variance (ANOVA) Followed by Tukey’s post hoc Test Using SPSS Software, and Values are Shown as Mean ± SD; #p < .01 Indicates That Values are Significant from Control, *p < .05 Indicates That Values are Significant from the AD-induced Group.
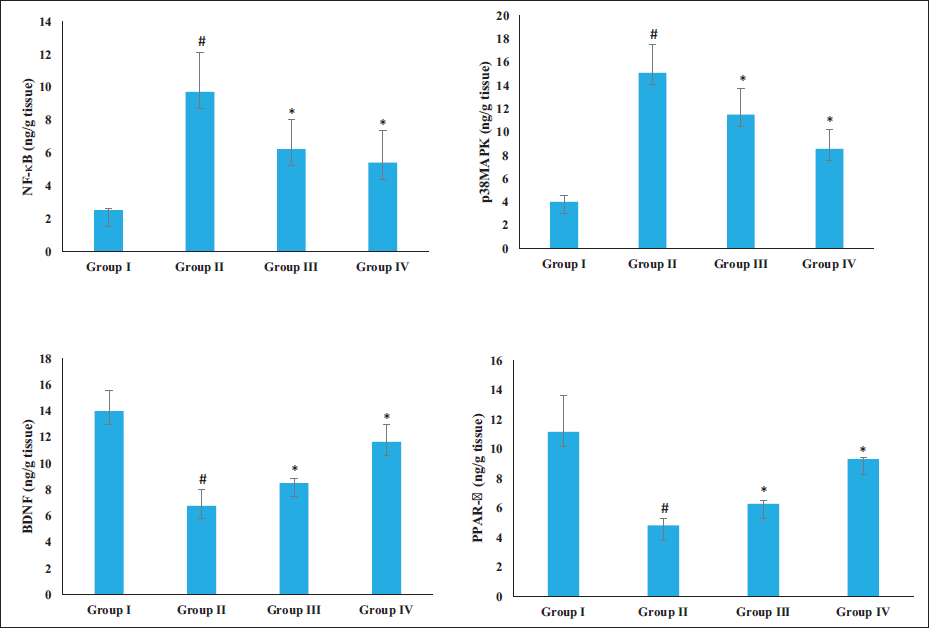
Effect of Sinensetin on the PPAR-γ, NF-κB, p38MAPK, and BDNF Levels
Figure 6 demonstrates the PPAR-γ, NF-κB, p38MAPK, and BDNF levels in the brain tissues of the experimental rats. The rats exposed to AlCl3 exhibited a considerable upsurge in NF-κB and p38MAPK and a diminution in PPAR-γ and BDNF levels in their brain tissues. The sinensetin treatment remarkably reduced the NF-κB and p38MAPK levels while increasing PPAR-γ and BDNF levels.
Effect of Sinensetin on the PPAR-γ, NF-κB, p38MAPK, and BDNF Levels in the Brain Tissues of Aluminum Chloride (AlCl3)-induced Alzheimer’s Disease (AD) Rats. The Values of the Treated Groups and the Control Group Were Statistically Analyzed by One-way Analysis of Variance (ANOVA) Followed by Tukey’s Post Hoc Test Using SPSS Software, and Values are Shown as Mean ± SD; #p < .01 Indicates That Values are Significant from Control, *p < .05 Indicates That Values are Significant from the AD-induced Group.
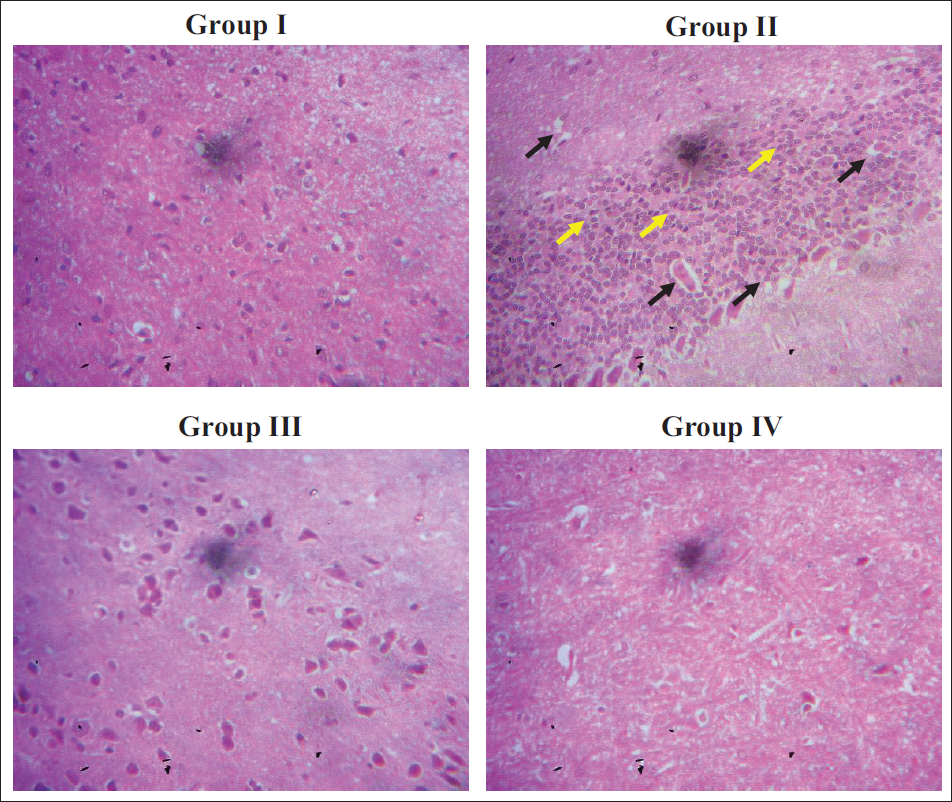
Effect of Sinensetin on the Brain Histopathology
The hippocampus of the control rats demonstrated normal tissue structures. In contrast, the rats challenged with AlCl3 exhibited degenerating cells in the hippocampus tissues, along with the presence of microglia cells and portions of a decreased cell population (Figure 7). The hippocampus of the rats that received sinensetin had hippocampal structures with reduced changes. Both doses (20 and 40 mg/kg) of sinensetin likewise showed protective effects, as indicated by the decreased histological changes and preservation of normal cellular structures.
Effect of Sinensetin on the Brain Histopathology of the Aluminum Chloride (AlCl3)-induced Alzheimer’s Disease (AD) Rats. Group I: Normal Control Rats; Group II: AlCl3-induced AD Rats; Group III: AlCl3-induced AD + 20 mg/kg Sinensetin-treated Rats; Group IV: AlCl3-induced AD + 40 mg/kg Sinensetin-treated Rats. Black Arrows: Cell Degenerations; Yellow Arrows: Influx of Microglia Cells.
Discussion
AD is an age-associated neurodegenerative disorder that occurs with age and is characterized by permanent and progressive deterioration of the brain. It is characterized by memory loss, cognitive impairment, and behavioral problems that hinder normal physical activities. The signs are caused by the permanent death of nerve cells and the development of aberrant extracellular protein clumps, which is a significant characteristic of the disease (Heilman & Nadeau, 2022). The association between Al and AD was well known. Deposits of Al in various parts of the brain hinder memory and cognitive functioning while promoting the accumulation of Aβ protein (Saba et al., 2017).
The initiation of the pathophysiology of AD begins with Aβ accumulation. The Aβ aggregation stimulates an overproduction of proinflammatory cytokines (Hampel et al., 2021). Aβ can easily spread throughout the brain tissue and initiate a series of harmful effects, including neuronal cell death. It is worth noting that the reduced Aβ clearance led to its buildup in the brain and has a substantial impact on the development of AD (Ju et al., 2022). Exposure to Al has been found to enhance the expression of Aβ secretases in the brain. This suggests that Al poisoning is responsible for the development of Aβ (Osman et al., 2019). The present findings proved that the sinensetin treatment diminished the Aβ protein level in the AD rats. As a result, it was clear that sinensetin can prevent the formation of amyloid plaques.
Al also alters the cholinergic transmission, hence impacting the amount of ACh in the brain. Prolonged exposure to Al leads to a decline in memory and cognition by causing changes in the neurons (Klotz et al., 2017). AChE is a vital enzyme that maintains the integrity of cholinergic neuronal membranes and breaks down ACh. Exposure to Al leads to the breakdown of cholinergic function by modifying cholinergic signaling and transmission (Al-Otaibi et al., 2018). It has been shown that prolonged administration of Al results in the reduction of AChE and the deterioration of cholinergic fibers. The inhibitors of AChE are employed to treat AD. AChE inhibitors are utilized to reinstate normal cholinergic activity in individuals with AD (Xin et al., 2013). Supportively, the findings of this study proved that the treatment of AD rats with sinensetin resulted in drastically reduced AChE activity in the brain tissues. This result proves that the sinensetin treatment can restore neurotransmission in AD brains by inhibiting AChE activity.
The MWM is the preferred method for studying the anxiety and cognition of experimental animals. The animals are tasked with locating the concealed platform while the elapsed time is measured. It was already reported that Al intoxication led to memory and behavioral impairments in the experimental animals (Kumar & Gill, 2014). In this work, the increased escape latencies and reduced time spent in the target quadrant during the MWM test in rats treated with AlCl3 suggest a decline in spatial memory, which is symptomatic of impaired learning and memory (Malik et al., 2022). Interestingly, the sinensetin treatment successfully modulated the behavioral changes in the AD rats, which were assessed using both MWM and open field tests.
The activation of microglia and the subsequent production of a significant amount of proinflammatory cytokines are key factors in the pathogenesis of AD, which results in neuroinflammation. This process leads to neuronal death and degeneration (Didonna, 2020; Mohamed et al., 2021). Inflammatory cytokines are components of the body’s immune system that detect and respond to inflamed tissues. The earlier study findings indicated that rats exposed to AlCl3 exhibited elevated levels of cytokines (Jangra et al., 2015). Similarly, the current findings also showed an upsurge in the brain levels of IL-6, IL-1β, and TNF-α cytokines. Nonetheless, the sinensetin substantially diminished these cytokines in the AD rats.
Neurodegenerative disorders arise from a disproportion of free radicals and antioxidants, which trigger oxidative stress (Amanzadeh et al., 2021). In addition, the brain does not possess a strong antioxidant mechanism and includes fatty acids that promote peroxidation. Additionally, it consumes a significant quantity of oxygen, rendering it more vulnerable to free radicals (Small & Duff, 2008). Hence, the implementation of an antioxidant therapy that effectively decreases ROS holds great potential for mitigating the progression of AD. MDA serves as a marker for the presence of oxidative stress (Snezhkina et al., 2019). The enzyme CAT utilizes its defensive mechanism against oxidative stress to convert hydrogen peroxide into water and oxygen. The SOD enzyme produces superoxide anions as a protective response to oxidative stress (Chtourou et al., 2015). Moreover, GSH-Px plays a pivotal role in averting oxidative stress. It was already stated that SOD, CAT, and GPx activities were linked to the body’s capacity to remove free radicals from its surroundings (Stankovic et al. 2020). In this study, the rats treated with AlCl3 exhibited a significant elevation in brain MDA levels, along with a notable reduction in SOD, GPx, and CAT activities, indicating the presence of oxidative stress. However, the sinensetin treatment remarkably increased the antioxidant enzyme activities while reducing the MDA level in AD rats, which confirms its antioxidant activity.
MAPKs are crucial for the regulation of cell growth and differentiation. Furthermore, it has been demonstrated that MAPKs can initiate apoptosis (Ijomone et al., 2021). Out of all these kinases, p38 MAPKs have received significant interest. The p38MAPK pathway is activated due to several stress stimuli, such as ROS and inflammatory cytokines. The involvement of p38MAPK in AD pathologies has been well documented, indicating that targeting the p38MAPK pathway could be crucial for AD treatment (Oliveira et al., 2013). The MAPK pathway is the major factor in activating NF-κB. It regulates both innate and adaptive immunological activities as well as inflammation (Capece et al., 2022). Additionally, the fact that inhibiting NF-κB reduces neurotoxicity serves as evidence of a connection between NF-κB and neurotoxicity (Shih et al., 2015). In this work, the findings demonstrated that sinensetin substantially reduced the p38MAPK and NF-κB levels in the AD rats. These results highlight the beneficial roles of sinensetin in mitigating neuroinflammation and AD progression in rats via inhibiting p38MAPK and NF-κB levels.
BDNF plays a crucial role in promoting development and facilitating the differentiation of neuronal cells and cognitive functioning. BDNF protects neurons against damage and can impede neuroinflammation by regulating NF-κB. Research has indicated that Al might cause a reduction in BDNF expression (Johnson & Sharma, 2003). Patients with AD have lower BDNF levels in their brain tissues, which also causes a decline in their learning and memory capacities (Xie et al., 2017). To improve memory and cognitive capacities, it is necessary to restore the amount of BDNF. BDNF is also involved in the prevention of pro-inflammatory mediators, which contributes to its neuroprotective effect (Fang et al., 2019). The present study proved that the sinensetin treatment successfully increased BDNF levels.
PPARs are transcription factors that are selectively expressed in the brain, especially in neurons and microglial cells (Warden et al., 2016). PPAR-γ, a subtype of PPARs, is present in microglia and astrocytes and plays a critical role in regulating inflammation and several pathways in the brain. Furthermore, PPAR-γ has a crucial role as a target, as it interacts with various signaling pathways involved in AD. It was highlighted that increased PPAR-γ activity can reduce neuroinflammation, boost BDNF production, decrease AChE activity, and enhance memory, cognition, and learning abilities (Yin et al., 2018). In this work, the observed results proved that the sinensetin treatment effectively increased the PPAR-γ level in the AD rats, thereby facilitating the improvement of memory, cognition, and learning capacity.
Conclusion
The present work showed that sinensetin treatment in AlCl3-induced AD rats led to behavioral enhancements, decreased oxidative stress and inflammation, increased levels of BDNF and PPAR-γ, and lowered levels of p38MAPK and NF-κB in brain tissues. These results demonstrate the antioxidant, anti-inflammatory, and neuroprotective properties of sinensetin. Consequently, it may be inferred that sinensetin demonstrates promise as a therapeutic option for the treatment of AD in the future. Nevertheless, more research is advisable in the future to elucidate the fundamental neuroprotective mechanisms of sinensetin.
Footnotes
Acknowledgments
This project was supported by the Researchers Supporting Project number (RSP2025R230), King Saud University, Riyadh, Saudi Arabia.
Availability of Data and Materials
All the data are available from the corresponding author upon request.
Consent for Publication
All authors have agreed to the manuscript data and the author’s positions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The institutional animal ethics committee of the Xi’an GaoXin Hospital, Xi’an, China has approved all the protocols carried out in this research work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
