Abstract
Background
Osteoarthritis (OA) is a common degenerative joint disorder characterized by pain, stiffness, and reduced mobility, impacting the quality of life. Extracts from turmeric (Curcuma longa) and Boswellia (Boswellia serrata) have shown significant anti-inflammatory effects. Rhuleave-K has been formulated as a natural combination of turmeric extract, Boswellia extract in sesame oil using Speed Technology.
Objectives
The present clinical investigation assessed the efficacy and safety profile of Rhuleave-K in comparison to a placebo, among subjects with mild-to-moderate osteoarthritis (OA).
Materials and Methods
This randomized, double-blind, placebo-controlled study enrolled 100 subjects with mild to moderate OA symptoms (Kellgren–Lawrence (KL) grade 2–3) and pain intensity >5 on the Numerical Rating Scale (NRS). Participants were administered either 500 mg of Rhuleave-K soft gel capsules (turmeric extract, Boswellia extract, and sesame oil as key components) as a test product or a matching placebo once daily for 12 weeks. The primary outcome was the change in the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores at the end of the study. The secondary outcomes included Osteoarthritis Research Society International (OARSI) battery tests and NRS scores. Data were recorded at baseline and on day 5, week 6, and week 12. Safety assessments, including blood biochemistry and vital signs, were performed at baseline and week 6 and week 12.
Results
The test group showed a significant improvement in the total WOMAC score of 55% (p < .001), whereas there was a negligible change (0.72%) in the placebo group. The WOMAC pain reduction was 75% (p < .001), WOMAC stiffness was 67% (p < .001), and WOMAC functional limitation was 48% (p < .001) from baseline. There were significant improvements in the OARSI battery of tests, such as the 40 m fast-paced walk by 10%, nine-step stair negotiation (9SSN) by 1%, 30 s chair stand test by 34%, timed up and go test by 18%, and 6 min walk test by 8% from baseline. The range of motion, as measured by goniometry, showed significant improvement (mean difference 2.06° ± 1.24°, p < .001) in flexion, whereas there was no change in the placebo group. C-reactive protein (CRP) showed a clinically significant 77% decrease, and complete blood count (CBC), liver function test (LFT), and renal function test (RFT) showed statistical significance, but no clinically significant changes, and no adverse events were evident in either the test or the placebo group during the study period. The treatments were well tolerated in the present study.
Conclusion
Rhuleave-K, at a daily dose of 500 mg for 12 weeks, significantly decreased pain, joint stiffness, difficulty in physical function, and mobility associated with mild-to-moderate OA compared with placebo. The improvement was observed as early as 6 weeks and can be a safe and suitable addition to the current management of OA.
Introduction
Osteoarthritis (OA) is a common and widespread age-related joint condition that progressively affects the musculoskeletal system, and it is a significant cause of disability in the elderly population (Loeser et al., 2016; Mobasheri, 2011; Verma et al., 2022). OA symptoms include pain, morning stiffness, joint swelling, limited range of motion, reduced physical function, and restrictions in social activities and work capacity (Clynes et al., 2019). Although current OA treatments (analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), and cortisone injections) offer some relief from pain and inflammation, they do not address the underlying imbalance between catabolic and anabolic processes that progresses OA (Haroyan et al., 2018; Jones et al., 2019), and are not recommended for long-term use.
Native to the Indian subcontinent and Southeast Asia, Turmeric (Curcuma longa) is an herb belonging to the ginger family, Zingiberaceae. The rhizome of this herbaceous plant has been used for centuries in Ayurveda, Siddha, and traditional Chinese medicine (Verma et al., 2022). Curcumin has attracted significant interest in anti-inflammatory studies (Kunnumakkara et al., 2023) because of its potential to inhibit the production of inflammatory molecules (prostaglandins) and neutrophil activity, both of which are considered to play a key role in the inflammatory process (Makuch et al., 2021). Studies have shown significant and efficient anti-inflammatory responses in patients treated with turmeric-derived volatile oils and curcumin (Toden et al., 2017).
Boswellic acids are biologically active constituents derived from the gum resin of Boswellia serrata, a medium-to-large-sized, branching tree commonly used in herbal and Ayurvedic medicine for the treatment of various disorders, such as asthma, rheumatism, dysentery, and skin ailments (Ammon et al., 1993; Iram et al., 2017; Singh & Atal, 1986; Vieira et al., 2025). Boswellic acid works by specifically inhibiting an enzyme (5-LOX), which generates inflammatory molecules called leukotrienes. Administration of formulations containing boswellic acid reportedly shrinks inflamed tissues and improves blood supply to the affected area without any reported toxicity or adverse effects. Leukotrienes contribute to inflammation by promoting free radical-induced damage, disrupting calcium balance, enhancing cell adhesion, and attracting inflammatory cells to the affected areas of the body. In vitro studies have shown that an extract of B. serrata resin has significant anti-inflammatory capability in an animal model (Ammon et al., 1993; Singh & Atal, 1986).
The effectiveness of multitarget therapy and synergistic interactions between different biologically active components has also been reported to be beneficial in neurological health (Xu et al., 2022). An open-label study conducted on 88 participants showed that a high-dissolution oil formulation (Rhuleave-K), constituting turmeric and Boswellia, significantly relieved joint pain during exercise and was found to be similar to acetaminophen administration (Rudrappa et al., 2020). Studies also reported the effective and safe management of musculoskeletal pain by the turmeric-Boswellia formulation (Rhuleave-K) in a short period (Antony et al., 2022; Rudrappa et al., 2022). The combination of turmeric and Boswellia extracts, owing to the natural compounds such as curcuminoids and boswellic acids, inhibits the production of prostaglandin E2 (PGE2) and leukotrienes (LTB4, LTC4, LTD4, and LTE4). Additionally, sesamin can inhibit cyclooxygenase enzymes, which are responsible for the production of prostanoids (Antony et al., 2022; Dey et al., 2022; Solanki et al., 2024; Yashaswini et al., 2017). Inhibition of both the cyclooxygenase and lipoxygenase pathways is a potentially unique property exhibited by this synergistic combination in inhibiting both prostaglandins and leukotrienes (Yashaswini et al., 2017). However, the optimal combination and concentration of these herbal extracts for effective pain relief in OA is yet a topic of ongoing research.
The present randomized, double-blind clinical study was conducted to compare the efficacy of a turmeric-Boswellia formulation in sesame oil, using proprietary SpeedTech technology (Rhuleave-K®), and a placebo among subjects in the treatment of mild to moderate OA. The main goal of this study was to evaluate the effectiveness of the formulation using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) total score questionnaire in adult subjects with knee OA from baseline to day 5, week 6, and week 12.
Materials and Methods
Study Design
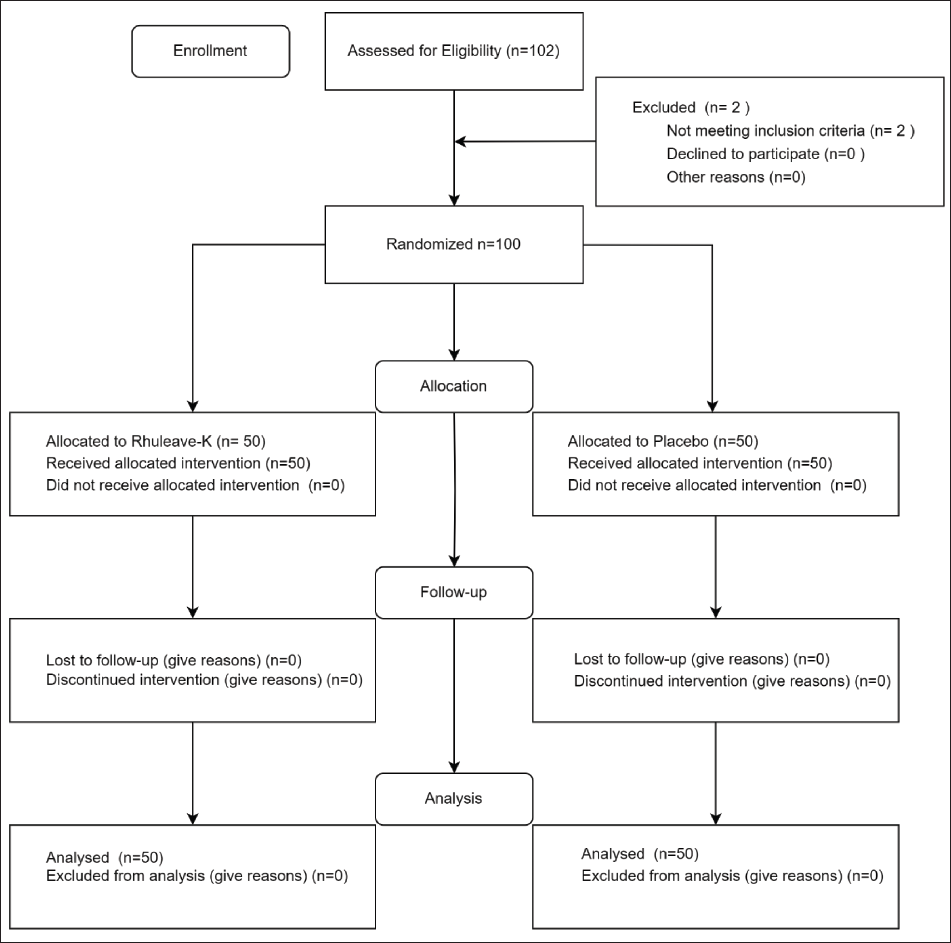
This was a phase 2, multicentric, double-blind, placebo-controlled, parallel-arm study involving 100 participants with mild to moderate OA. Male and female participants aged 18–70 years were included in the study in a 1:1 ratio. Participants diagnosed with a primary complaint of knee pain intensity of five or greater on the Numerical Rating Scale (NRS), with an X-ray-based Kellgren–Lawrence (KL) classification of OA grade 2–3, a WOMAC pain score of two or more, a stiffness score of one or more, a physical function score of 10 or more, and a body mass index (BMI) score of 18–29 kg/m2 were included in the study. The main exclusion criteria were a history of inflammatory arthropathy, rheumatoid arthritis, severe OA (defined as KL grade four-based X-ray imaging), underlying back pain, fibromyalgia, gout, history of knee surgery within the past 6 months, and expectations of surgery in the next 4 months. Participants meeting the inclusion and exclusion criteria were enrolled in the study. A CONSORT flow chart of the study design is shown in Figure 1.
Participants Flow Diagram.
Treatments Administered, Dosage, and Duration
The turmeric-Boswellia extract in sesame oil (Rhuleave-K, test) and placebo were manufactured by Arjuna Natural Pvt. Ltd. (Aluva, Kerala, India). The test and placebo products were administered as 500 mg soft gel capsules orally, once daily, along with or after breakfast, for 12 weeks (84 days). The active ingredients of Rhuleave-K were turmeric extract (95%), 28%, B. serrata extract (10%) in the excipient base of sesame seed oil, standardized to contain total curcuminoid NLT (26.6%), and acetyl-11-keto-β-boswellic acid (AKBA) NLT (1%). The placebo consisted solely of inactive substances, specifically a mixture of polysorbate-80 and polyethylene glycol (PEG) 400 in a 1:1 ratio, contained in each soft gel capsule.
Randomization, Blinding, and Unblinding
An independent statistician generated the randomization master list using WinPepi software version 11.65. Participants were stratified by gender (male and female) in a 1:1 ratio, with the intervention being randomized within each stratum in a 1:1 ratio. The allocation was concealed using bottles of identical shapes and sizes, each with an alphanumeric allocation code (R01–R100). The randomization list was kept confidential and provided to the pharmacist for the serial distribution of the study treatments. Both the subjects and investigators were blinded to the treatment allocation. Blinding was achieved through the use of opaque soft gel capsules, which are identical in size, shape, and color.
Outcome Measures
The primary efficacy parameter was the total WOMAC score. The WOMAC assesses five pain-related items (with scores ranging from 0 to 20), two items for stiffness (scored 0–8), and 17 items evaluating functional limitation (scored 0–68). Higher scores reflect greater severity of pain, stiffness, and functional impairment. A decrease in the total WOMAC score is desirable, and it represents symptom improvement. The scores were summed to represent the total WOMAC score (Roos et al., 1999).
The secondary efficacy parameters considered in this study included physical performance measures for OA, based on function-based physical tests recommended by the Osteoarthritis Research Society International (OARSI) (Dobson et al., 2013).
30 s Chair Stand Test (30 s CST)
This measures lower-body strength by counting the number of stands completed within 30 s. An increase in the number of stands is desirable, which reflects improved strength and physical function.
40 m (4 × 10 m) Fast-paced Walk Test (40 m FPWT)
This assesses the walking speed of the participants over a distance of 40 m. An increase in walking speed is desirable, indicating improved mobility and endurance.
Timed Up and Go Test (TUG)
This assesses the time taken to stand from a chair, walk 3 m, turn, and return to their chair. A decrease in time indicates improved balance and mobility, which is desired.
Nine-step Stair Negotiation (9SSN)
This evaluates the time taken to ascend and descend nine steps. A decrease in time is desirable, reflecting better functional performance.
6-min Walk Test (6WT)
This measures the distance covered in 6 min, which is desirable and reflects overall endurance and mobility.
Pain intensity was measured using an NRS. Participants were asked to rate their pain intensity from 0 to 10, with 0 indicating no pain and 10 indicating the worst pain imaginable. A decrease in the NRS score is desirable, indicating reduced pain intensity.
A standard protractor goniometer measured the knee’s range of motion. The subject was placed in a supine position, and the plane of movement was sagittal. Knee flexion and extension were also assessed. The knee was straightened to the maximum extent possible for extension. The goniometer should read 0° if it is fully extended or a slightly negative value if hyperextended (the knee bends backward slightly). During flexion, the knee was comfortably bent. The normal knee flexion range is approximately 0°–135° (Shelbourne et al., 2007). An improvement in the flexion angle and knee extension is desirable, reflecting improved joint mobility.
Changes from baseline in primary and secondary efficiency parameters were compared on day 5, week 6, and week 12 of treatment, estimating the significant variability between the test group and placebo. Safety parameters include blood biochemistry of liver function tests (LFTs), renal function tests (RFTs), complete blood count (CBC), and C-reactive protein (CRP). Vital signs such as blood pressure, heart rate, pulse rate, respiratory rate, and temperature were assessed at baseline and after 12 weeks to monitor adverse effects and to ensure safety. A decrease in CRP levels is desirable, reflecting reduced systemic inflammation. Variabilities within and between interventions were statistically assessed.
Sample Size Calculation
The sample size estimation for the hypothesis (H0: µ1 = µ2 vs. H1: µ1 ≠ µ2) was calculated using a parallel two-group design. The comparison was made using a two-sided Mann–Whitney U (Wilcoxon rank-sum) test, with a type I error rate (α) of 0.05, an effect size of 0.7, and a power of 80%. The number thus calculated was 33 in each group. Factoring in a 30% dropout, the sample size per group was 47. For this study, we have rounded the sample size to 50 per group, totaling a sample size of 100. The sample size was computed using Noether’s formula (Noether, 1987), using the rankFD package in R version 4.3.2.
Statistical Analysis
Statistical analysis was performed by an independent statistician using the R 4.2.3 package. The Shapiro–Wilk test confirmed non-normality across the dataset, and hence, non-parametric tests were used.
Within-group variability was assessed using the Wilcoxon signed-rank test. The magnitude of change in study outcomes was quantified as the absolute change over time from the baseline observation. The absolute change value for each participant was calculated on day 5, week 6, and week 12, by subtracting the baseline observation from the corresponding values at each time point. These absolute change values for the study outcomes were then used in the Mann–Whitney U test to assess the between-group variability in outcomes between the test and placebo groups. All statistical analyses were performed at a 95% significance level.
To have a comprehensive analysis of the evaluation of the treatment effects on the primary outcome variable (WOMAC score), a mixed-model repeated measures (MMRM) analysis was performed. This model was selected considering its ability to handle repeated observations and missing data without the need for listwise deletion. Such characteristics of MMRM make it well-suited for analyzing longitudinal data with treatment (test vs. placebo) and time (baseline, day 5, week 6, and week 12) as fixed effects to assess within and between variability.
WOMAC sub-scores (stiffness, functional limitation, and pain) and other secondary outcome variables, such as OARSI scores, goniometry, NRS pain scores, and within-group variability, were studied using the Wilcoxon signed-rank test. In contrast, between-group variability was analyzed using the Mann–Whitney U test. For safety parameters, evaluations were done exclusively for within-group variability using the Wilcoxon signed-rank test.
Results
In this study, 102 subjects were initially screened, and 100 participants were selected based on the inclusion and exclusion criteria. None of the participants were lost to follow-up, and there were no dropouts during the study, ensuring complete data collection from all participants. A detailed overview of the study plan and participant allocation is shown in the flowchart (Figure 1).
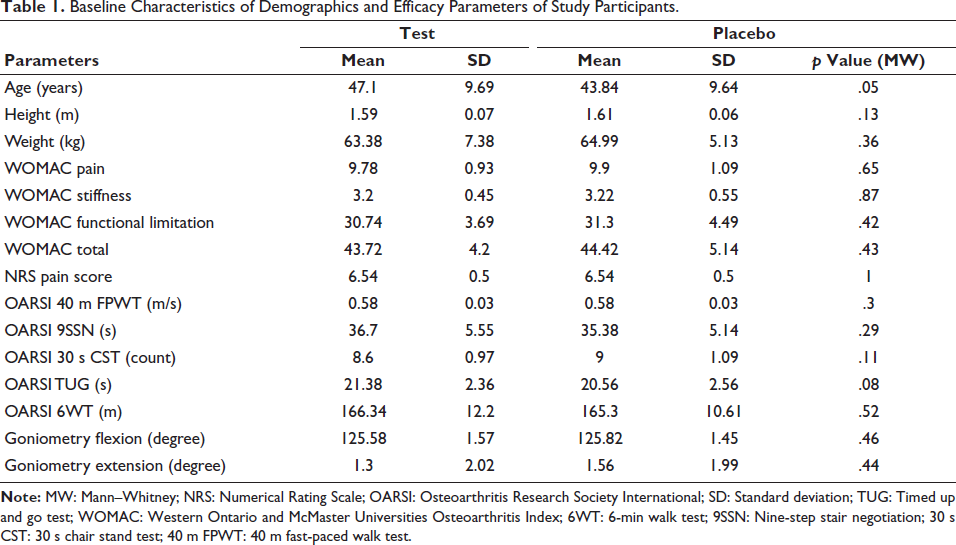
There was no significant variability in the overall demographic characteristics, as well as the primary and secondary efficacy parameters, between the treatment and placebo groups at baseline (Table 1). The mean age of participants was 47.1 ± 9.69 years in the test group and 43.84 ± 9.64 years in the placebo group, with no significant difference (p = .05). The mean height of participants was 1.59 ± 0.07 m in the test group and 1.61 ± 0.06 m in the placebo group, with no significant difference (p = .13). The mean weight of participants was 63.38 ± 7.38 kg in the test group and 64.99 ± 5.13 kg in the placebo group, with no significant difference (p = .36). Additionally, the WOMAC, Numerical Pain Rating Scale (NRS), OARSI, and goniometry parameters showed no significant differences between the test and placebo groups at baseline.
Baseline Characteristics of Demographics and Efficacy Parameters of Study Participants.
WOMAC Total Score
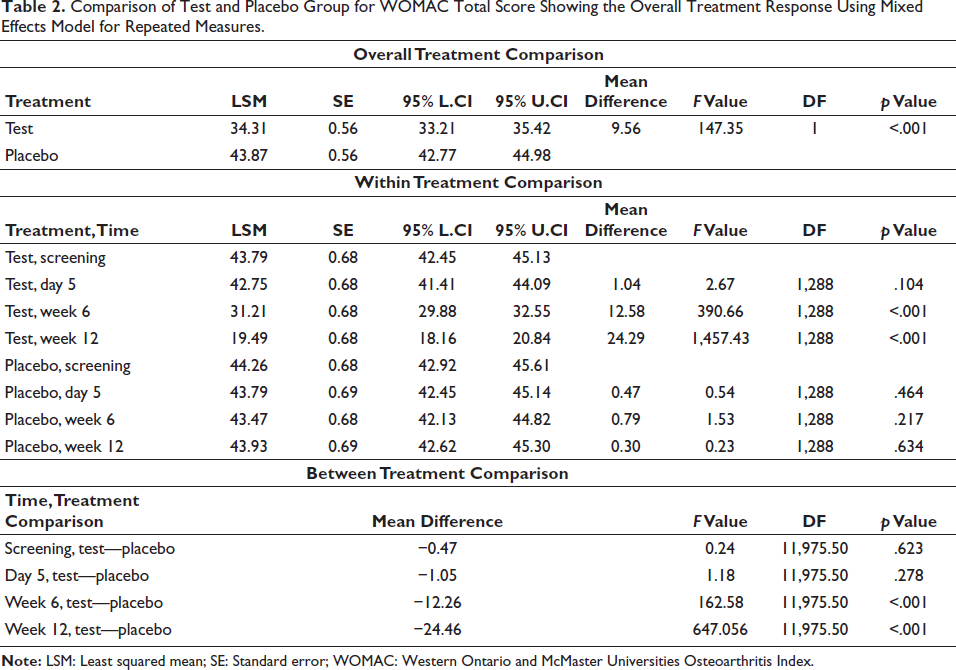
An overall analysis of the WOMAC total score using the MMRM analysis provided strong evidence that the test group was able to significantly reduce the total WOMAC score (mean = 34.31 ± 0.56) compared to the placebo group (mean = 43.87 ± 0.56). The mean difference was 9.56 (p < .001), indicating a potential benefit for the test group (Table 2). The total WOMAC score of the test group was found to be significantly decreased at day 5, week 6, and week 12 compared to baseline (p < .001). However, variability in the total WOMAC score was not significant in the placebo group (p > .05).
Comparison of Test and Placebo Group for WOMAC Total Score Showing the Overall Treatment Response Using Mixed Effects Model for Repeated Measures.
WOMAC Pain
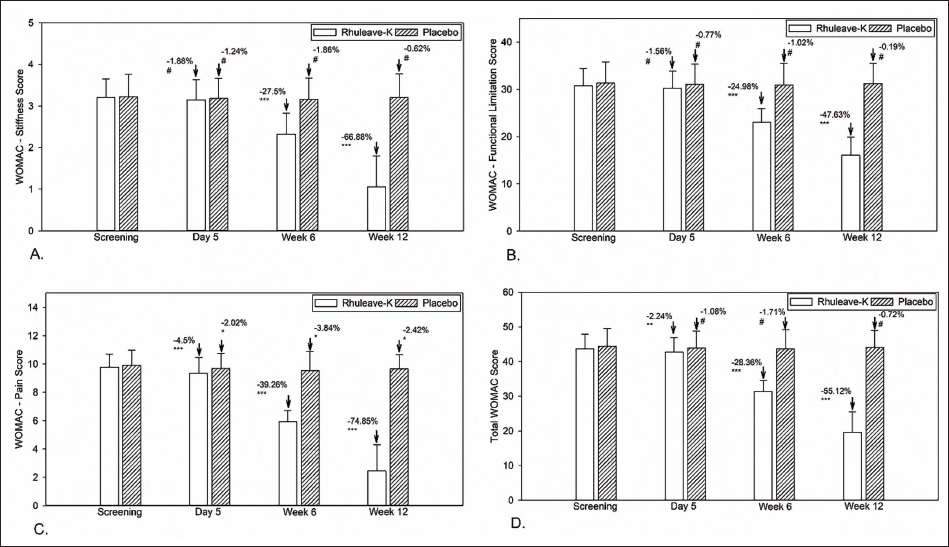
The WOMAC pain score showed significant variability at day 5, week 6, and week 12 compared to baseline in both the test and placebo groups. Still, they were remarkably different according to the percentage of pain reduction. At the end of the study, the test group showed a 75% reduction in pain intensity from baseline, whereas the placebo group showed only a 2.4% change. The test group was significantly more effective than the placebo (p < .001) at week 6 and week 12 (Figure 2 and Table 3).
Descriptive Statistics and Within and Between Group Analysis of Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Osteoarthritis Research Society International (OARSI), Goniometry, and Numerical Rating Scale (NRS) Scores.
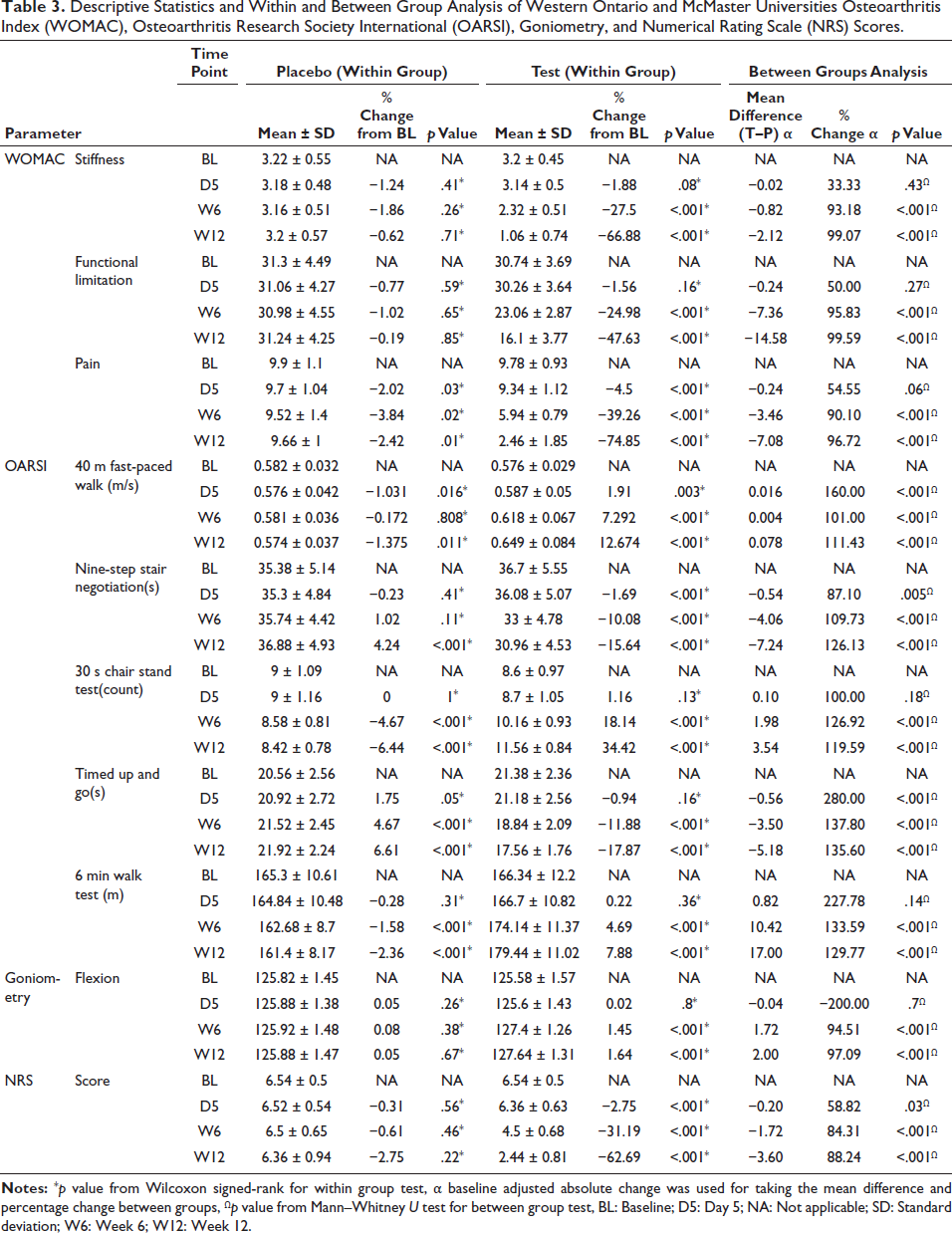
WOMAC Stiffness
No significant reduction in stiffness was observed in either the test group (−1.88%, p = .08) or the placebo group (−1.24%, p = .41) on day 5 compared to the baseline. An improvement was recorded in the test group (mean change = −27.5%, p < .001) at week 6 compared to baseline (mean change = −1.86%, p = .26). There was a more significant reduction in the test group at week 12 than at baseline (−66.88%, p < .001) and no significant variability was observed in the placebo group. The test group was significantly more effective than the placebo (p < .001) at week 6 and week 12 (Figure 2 and Table 3).
WOMAC Functional Limitation
The treatment and placebo groups did not show any significant variability in functional limitations on day 5 compared to the baseline observation (p > .05). Functional limitation scores at week 6 and week 12 from baseline showed significant variability in the test group (−24.98% and −47.63%, respectively, p < .001). In contrast, the placebo group showed no statistically significant variability at week 6 and week 12 (−1.02% and −0.19%, respectively, p > .05). The test group was significantly more effective than the placebo (p < .001) at week 6 and week 12 (Figure 2 and Table 3).
OARSI Battery of Tests Score
40 m (4 × 10 m) FPWT (40 m FPWT)
In the 40 m distance in the paced walking test, subjects in the test group were found to have significantly increased walking speed on day 5 (1.87%, p < .001), week 6 (7.19%, p < .001), and week 12 (12.54%, p < .001) compared to the baseline observation. In contrast, the placebo group reported a significant decrease in 40 m distance walking speed at day 5 (−1.1%, p = .016) and week 12 (−1.49%, p = .011), but the reduction in speed reported by the placebo group during week 6 was not statistically significant (−0.257%, p = .81). The test group was significantly more effective than the placebo (p < .001) at day 5, week 6, and week 12 (Figure 3 and Table 3).
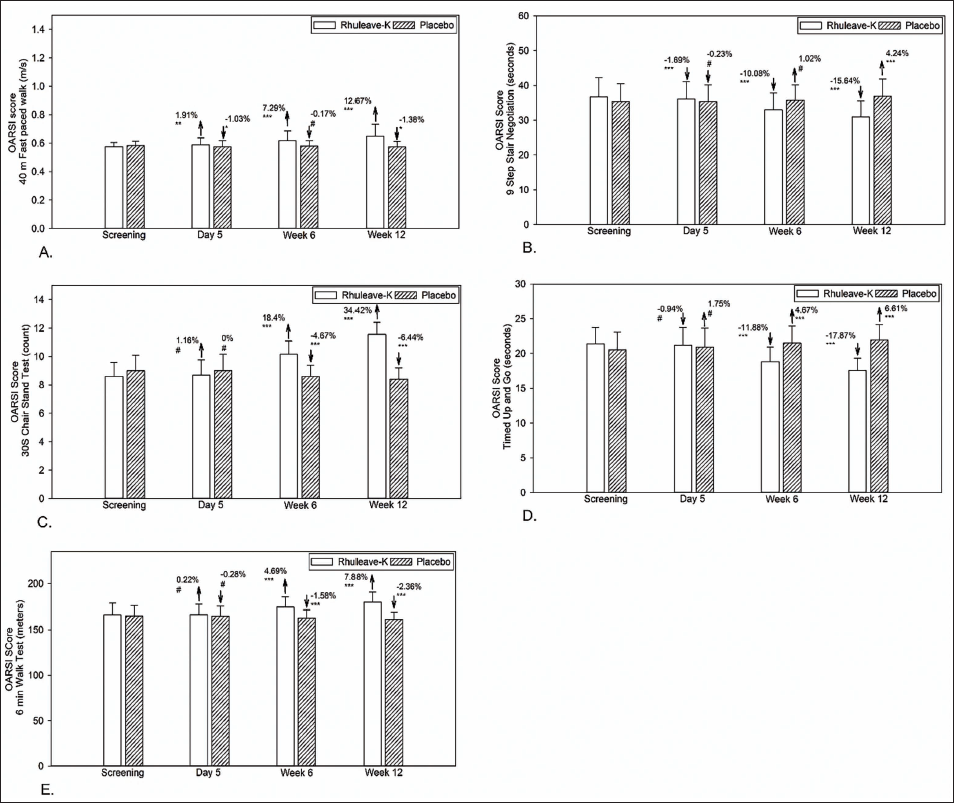
9SSN
The time taken for 9SSN reduced significantly on day 5 (−1.69%, p < .001), week 6 (−10.08%, p < .001), and week 12 (−15.64%, p < .001) in the test group. In contrast, the placebo group reported an insignificant reduction on day 5 (−0.23%, p = .41), but showed a significant increase at week 6 (1.02%, p = .11) and week 12 (4.24%, p < .001). The test group was significantly more effective than the placebo (p < .001) at day 5, week 6, and week 12 (Figure 3 and Table 3).
30 s CST
The variability in the number of standings from a sitting position in the 30 s CST on day 5 from baseline was not significant in either the test or placebo group (p > .05). While the placebo group reported a significant reduction in the count at week 6 (−4.67%, p < .001) and week 12 (−6.44%, p < .001), the participants in the test group showed a significant increase in the count of the 30 s CST count at week 6 (18.14%, p < .001) and week 12 (34.42%, p < .001). The test group was significantly more effective than the placebo (p < .001) at week 6 and week 12 (Figure 3 and Table 3).
TUG
The variability in response time for the TUG on day 5 from baseline was not significant in either the test or placebo group (p > .05). There was a significant reduction in the response time in the test group at week 6 (−11.88%, p < .001) and week 12 (−17.87%, p < .001) compared to baseline. The placebo group reported a significant increase in response time at week 6 (4.67%, p < .001) and week 12 (6.61%, p < .001) compared to baseline. The test group was significantly more effective than the placebo (p < .001) at day 5, week 6, and week 12 (Figure 3 and Table 3).
6WT
The distance covered by participants in the 6WT in the test group showed significant improvement at week 6 (4.69%, p < .001) and week 12 (7.88%, p < .001) from the baseline data. In contrast, the placebo group showed a significant reduction in the distance covered at week 6 (−1.58%, p < .001) and week 12 (−2.36%, p < .001). The variability in the distance covered on day 5 from baseline was not significant in either the test or placebo group (p > .05) (Table 2). The test group was significantly more effective than the placebo (p < .001) at week 6 and week 12 (Figure 3 and Table 3).
Goniometry
The observations on day 5 from baseline showed no significant improvement in the knee flexion angle in both the test and placebo groups (p > .05). There was a significant improvement in the knee flexion angle in the test group at week 6 (1.45%, p < .001) and week 12 (1.64%, p < .001) from baseline. No significant improvement in knee flexion angle was reported in the placebo group at week 6 (0.08%, p = .38) and week 12 (0.05%, p = .67). There was no change in the knee joint extension angle from baseline to week 12; hence, the analysis was not possible. A group comparison of the absolute changes in flexion scores showed significant variability in the effect of the test on improving the knee flexion angle and movement among the participants, compared to the placebo group (p < .001) (Table 3).
NRS of Pain
Variability in the NRS pain scale scores observed in the test and placebo groups on day 5, week 6, and week 12, compared to the baseline, are shown in Figure 4. It is evident from the analysis that there was a significant reduction in pain in the test group on day 5 (−2.75%, p < .001), week 6 (−31.19%, p < .001), and week 12 (−62.69, p < .001) from baseline. The placebo group showed no significant variability in the NRS pain scale scores among the participants during day 5 (−0.31%, p = .56), week 6 (−0.61%, p = .46), and week 12 (−2.75%, p = .22) from baseline (Table 3).
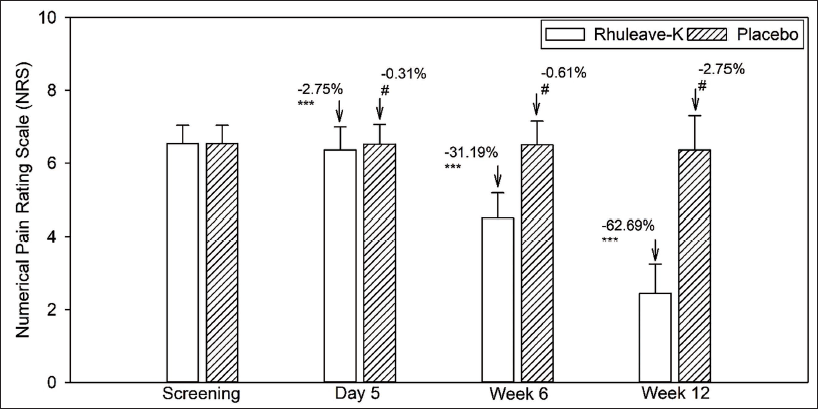
A group comparison of the absolute changes in the NRS pain scale showed significant variability in the effect of the test in improving pain relief among the participants compared with the placebo group (p < .001), respectively (Table 2).
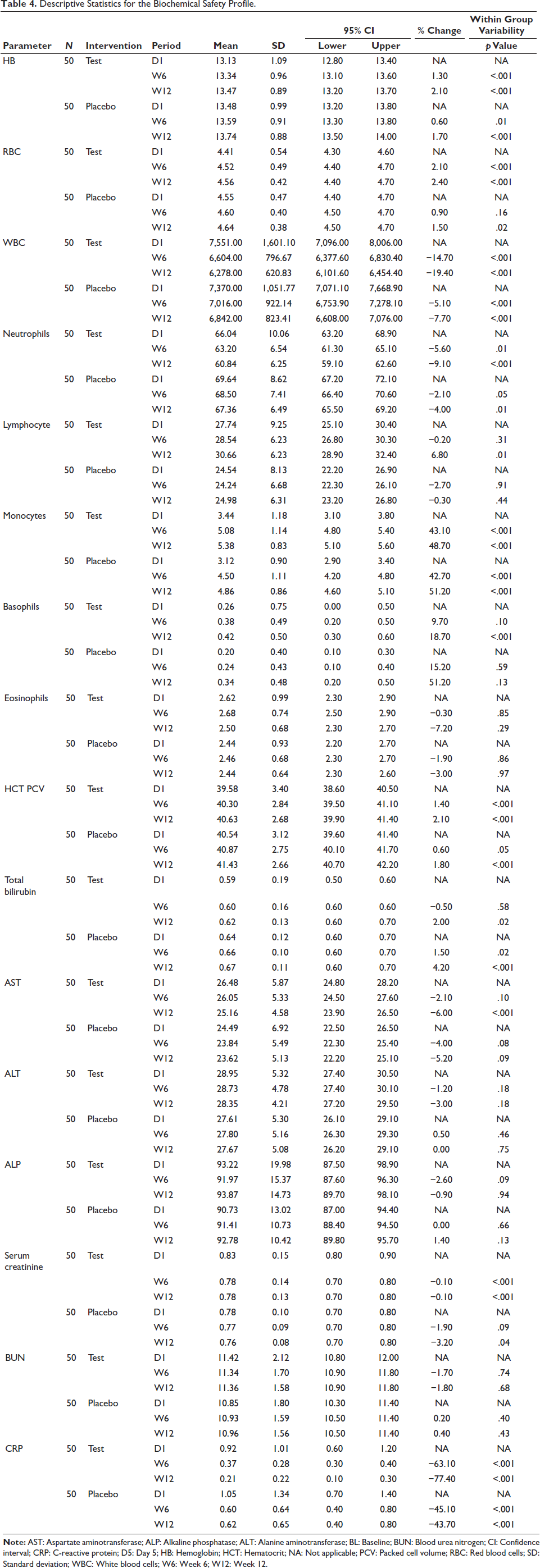
Safety Parameters
It is evident from this study that the treatments administered to participants in the test and placebo groups were well tolerated. CRP levels were significantly reduced, indicating that the extract had considerable anti-inflammatory properties. It was evident from the safety analysis that parameters such as blood urea nitrogen (BUN), alkaline phosphatase (ALP), alanine aminotransferase (ALT), and eosinophil count showed no significant variability during treatment. Based on the evidence of no adverse effects related to the treatments administered, all measured blood parameters remained within normal limits, indicating the safety profile of the treatment (Table 4).
Descriptive Statistics for the Biochemical Safety Profile.
Discussion
A comprehensive assessment of the effectiveness of a turmeric-Boswellia formulation (Rhuleave-K®) and its safety in the management of mild-to-moderate OA was conducted and compared with a placebo. The results from this study demonstrate that the test product significantly reduces WOMAC and other functional index scores compared to placebo, indicating its effectiveness in OA. The participants reported a significant reduction in OA-related symptoms such as knee stiffness and pain. Furthermore, there was a significant improvement in the knee flexion angle, indicating improved joint activity among participants receiving the test, compared to those receiving a placebo. The numerical pain relief scores at week 6 and week 12 also demonstrated the potential pain-relieving effects of the test compared to the placebo. In addition, the test group showed improved functional activities, thereby improving the quality of their daily general activities. This test formulation has also, in earlier studies, shown very good pain management and had improved effects in the treatment of inflammation and musculoskeletal pain (Murthy et al., 2022; Rudrappa et al., 2020).
Extensive documentation from various studies has highlighted the protective effects of curcuminoids against arthritis and cartilage dysfunction (Guan et al., 2022; Nicoliche et al., 2020; Panahi et al., 2016). The primary mechanism of action of curcuminoids involves the inhibition of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), thereby suppressing several key regulators of inflammation such as cyclooxygenase-II, activator protein-1, c-Jun N-terminal kinase (JNK), mitogen-activated protein kinase (MAPK), and PI3K/Akt (Shakibaei et al., 2007). This inhibition effectively reduces the release of pro-inflammatory cytokines such as tumor necrosis factor-α, interleukin-1β (IL-1β), interleukin-6 (IL-6), macrophage chemotactic protein-1, and PGE2. These anti-inflammatory effects have been observed in studies using cultured chondrocytes (Chowdhury et al., 2008). Moreover, curcuminoids potentially inhibit matrix metalloproteinase enzymes, which are crucial for cartilage degradation. This action promotes extracellular matrix accumulation, prevents cartilage breakdown, and reduces inflammation-induced apoptosis (Zhou et al., 2020).
An important aspect of this study is the utilization of proprietary-speed technology in the formulation of turmeric-Boswellia extract in black sesame seed oil. This technology solubilizes the activities of turmeric and Boswellia extracts in sesame seed oil, which is termed a carrier of activities in the classical texts of Ayurveda (Abbas et al., 2022). This combination optimized the overall performance of the test better than the regular combination of turmeric and Boswellia extracts, as can be seen from the results of this study. In addition, B. serrata extract has been identified as a specific non-redox inhibitor of 5-LOX and can inhibit leukotriene biosynthesis, which in turn helps alleviate joint stiffness, pain, and improve physical comfort (SenGupta et al., 2011). The observed improvement in joint stiffness and pain among OA participants treated with the test can be attributed to the combined action of turmeric and B. serrata extract.
The findings from this study highlight the significant effect of the administration of the test product on symptomatic improvements associated with OA and the improved daily functioning of patients. With a robust sample size of 100 participants, this study provides substantial evidence supporting the safety and potential effectiveness of this test in addressing mild-to-moderate OA symptoms. However, it is essential to acknowledge certain limitations, as the study focused only on patients with mild-to-moderate OA symptoms; hence, the results cannot be considered conclusive for extreme OA symptomatic cases. The relatively short treatment duration of 12 weeks and the reliance on subjective participant-reported data without biomarker analysis or inclusion of a positive control group are noteworthy constraints. Future research should comprehensively address these limitations.
Conclusion
Rhuleave-K at a 500 mg dosage for 12 weeks significantly decreased pain, joint stiffness, difficulty in physical function and mobility, and significantly decreased CRP levels associated with mild-to-moderate OA compared to placebo. The improvement was observed as early as 6 weeks, and this supplement can be considered a safe option as a regular supplement for OA management.
Footnotes
Abbreviations
ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; BUN: Blood urea nitrogen; CBC: Complete blood count; CRP: C-reactive protein; LFTs: Liver function tests; LT: Leukotrienes; NRS: Numerical Rating Scale; OA: Osteoarthritis; OARSI: Osteoarthritis Research Society International; PG: Prostaglandin; RFT: Renal function tests; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
Acknowledgments
The authors acknowledge Arjuna Natural Pvt. Ltd., Kerala, India, for providing the test product (Rhuleave-K®) and placebo capsules.
Authors Contribution
AG was involved in the study design, investigation, and analysis, and wrote the original draft of the manuscript. AA and SA contributed to the study design and planning and reviewed the manuscript. All the authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study was approved by the Institutional Ethics Committee of the Nirmal Hospital on October 14th, 2023 (Ref: IEC/Nirmal Hospital, Jhansi/2023/05, IEC/Nirmal Hospital, Jhansi/2023/06, and IEC/Nirmal Hospital, Jhansi/2023/07). The clinical trial was prospectively registered in the Clinical Trial Registry of India with registration number CTRI/2023/10/059301 (registered on October 30th, 2023). All participants signed a written informed consent form before participating in this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
