Abstract
Objectives
To evaluate the efficiency of the turmeric-Boswellia formulation (TBF, Rhuleave-K) in reducing acute low back pain (LBP) among healthy subjects.
Materials and Methods
This research aimed to assess the effectiveness of TBF compared to a placebo through a single dose of 1,000 mg, administered in a randomized, double-blinded, placebo-controlled trial with 76 participants experiencing acute low back pain. Efficacy was evaluated using objective outcome measures, namely the numerical rating scale (NRS), categorical pain relief scale (PRS), the onset of analgesia, and the short-form McGill questionnaire. NRS and PRS measurements were taken 30 minutes post-dose at rest, during movement, and under pressure application at the afflicted area. The onset of analgesia was assessed 6 hours post-dose. Quantitative analysis of the Sum of Pain intensity Difference (SPID) and Total Pain Relief (TOTPAR) values was conducted based on the NRS and PRS scores, respectively.
Results
Pain relief began quickly and all participants in the TBF group reported a significantly noticeable reduction in pain at about 76.4 minutes, compared to the 190 minutes in the placebo group. Similarly, 92.7% of participants in the TBF group experienced mean meaningful pain relief (MPR) as early as 224.1 minutes, compared to the zero MPR responders in the placebo group. There was a statistically significant difference in the time to pain relief between the TBF and placebo groups, with significant improvement in the TBF group (log-rank hazard ratio, p < 0.001). None of the individuals in the placebo group demonstrated a minimum of 50% maximum TOTPAR in rest, movement, and pressure. Whereas, 75.71% of the TBF group experienced at least 50% maximum TOTPAR in rest, 88.09% in movement, and 90.48% in pressure. The effectiveness of the treatment, as measured by the number needed to treat (NNT) for achieving at least 50% of MPR in the TBF group, was 1.2 at rest and 1.1 during both movement and pressure.
Conclusion
Oral administration of TBF (Rhuleave-K) to individuals with acute low back pain significantly reduces pain, providing meaningful relief within 4 hours of administration. This highlights the substantial analgesic effectiveness of TBF in the context of acute LBP.
Introduction
Low back pain (LBP) is one of the major global health concerns, irrespective of the region and socio-economic status, leading to discomfort and disabilities of different scales. LBP reportedly affects nearly 80% of individuals at some point in their lives. This widespread ailment transcends mere physical discomfort, translating into a hefty economic burden through lost workdays and decreased productivity (Pai & Sundaram, 2004). LBP is responsible for a significant loss of workdays in the industrialized part of the world (Alonso-García & Sarría-Santamera, 2020; Gaskin & Richard, 2012). Research indicates that the lifetime prevalence of LBP varies between 51.0% and 84.0% (Henschke et al., 2015). Unlike many other pain conditions, LBP has been extensively studied in terms of its epidemiology. Regarding age, it seems that the prevalence of certain pain conditions, such as LBP, rises from childhood through adolescence, reaching its peak among adults (Calvo-Muñoz et al., 2013; Hestbaek et al., 2006; Taimela et al., 1997). Though there is a general concern that consistent physical activities can increase the prevalence of back pain, there is no clear scientific evidence showing that consistent physical activities alone are risk factors for back pain. However, there is evidence of the effect of conventional painkillers, like paracetamol, in providing temporary relief to afflicted patients. Their potential efficacy remains questionable, and there are serious concerns regarding the long-term use of these painkillers and their potential side effects (Chou et al., 2015). Hence, there are no universal recommendations for the long-term use of paracetamol for treating LBP (Saragiotto et al., 2019). This necessitates the exploration of a safe and effective alternative therapy, particularly those rooted in natural medicine (Patidar et al., 2014).
Among the various natural extracts commonly used in conventional medical practices, turmeric (Curcuma longa) and Boswellia serrata have a rich legacy in Ayurvedic and traditional Chinese medicine for their potent anti-inflammatory properties (Governa et al., 2018; Pinzon et al., 2019). Curcumin, a bioactive compound in turmeric, possesses documented efficacy in managing LBP and inflammation (Xiao et al., 2017). Similarly, B. serrata extracts, rich in Boswellic acids, exhibit potent anti-inflammatory and analgesic effects (Sharma et al., 2010). Sesame oil, with its sesamin and sesamol content, further potentiates the anti-inflammatory activity of these key ingredients (Monteiro et al., 2014). However, traditional formulations often face a significant obstacle—bioavailability. This study delves into the potential of a proprietary speedtech formulation of turmeric-Boswellia-sesame oil (TBF/Rhuleave-K) to revolutionize LBP relief. This novel formulation employs cutting-edge strategies to unlock the full potential of these natural ingredients. Unlike paracetamol, whose efficacy for LBP remains debatable (Saragiotto et al., 2019), this study focuses on a natural extract-based product contributing to acute LBP relief.
This study aimed to evaluate the effectiveness of a proprietary speedtech formulation of turmeric-Boswellia-sesame oil (TBF/Rhuleave-K) in relieving symptoms of acute LBP, compared to a placebo. This could be achieved through a randomized, double-blind, placebo-controlled trial involving healthy subjects experiencing acute LBP. The study aimed to evaluate the effectiveness of the TBF formulation in providing rapid and meaningful pain relief (MPR).
Materials and Methods
Study Design
The study design involved a randomized, placebo-controlled, double-blinded multicenter approach to ensure rigorous investigation. Individuals experiencing acute LBP triggered by vigorous exercise were recruited. The participants were carefully selected to represent a diverse group, and their health status was thoroughly assessed before enrollment.
Study Procedure
Individuals willing to provide informed consent and who met all specific criteria for inclusion and exclusion were included in the study. All selected individuals were then queried about detailed aspects of their pain history, including the date of occurrence, onset time, pain duration, the specific body part affected, and any prior instances of similar pain during exercise within different timeframes (<24 hours, 24–48 hours, and >48 hours), the pain severity, its duration, and treatment history.
Study Inclusion/Exclusion Criteria
This study included both men and women participants aged 18–65, who had exercise-induced acute LBP, registering a resting numerical rating scale (NRS) of 5 or higher on a 0–10 scale within the 24 hours preceding their visit to the site. The participants comprised mainly regular gym attendees, while the remainder engaged in home exercises. Individuals excluded from this study were those who required parenteral therapy or surgery for acute muscle spasms, were hospitalized for the management of painful acute soft tissue injuries (including grade 2 and 3 sprains or strains) of the upper or lower extremities, and those with a history of other issues like rheumatoid arthritis, osteoarthritis, or recent use of products for pain and inflammation in the week before the study.
Intervention and Dosing
Participants meeting the specified criteria (inclusion/exclusion) were randomly assigned in the trial with the experimental intervention, TBF (Rhuleave-K, 1,000 mg) (equivalent to 500 mg × 2 soft gels), which includes 266 mg of curcuminoids and 10 mg of 3-acetyl-11-keto-boswellic acid (AKBA), or intervention with a matching placebo 1,000 mg (500 mg × 2 soft gels) produced by Arjuna Natural Pvt. Ltd. in Aluva, India.
The TBF formulation comprises extracts from turmeric (C. longa L.), B. serrata, and black sesame (Sesamum indicum) seed oil. Dried rhizome of turmeric (C. longa L. Zingeberaceae), which was identified by a qualified botanist, was extracted using ethyl acetate to derive the turmeric oleoresin. Curcuminoids were then crystallized from the oleoresin to derive the turmeric extract containing 95% total curcuminoids, confirmed by the high-performance liquid chromatography (HPLC) analysis method based on the US Pharmacopoeia. B. serrata gum resin was extracted with ethyl acetate, which contains 10% of AKBA, analyzed by the HPLC method. Using proprietary technology, the active ingredients of C. longa and B. serrata were consistently dissolved in sesame seed oil. The final TBF formulation contains a minimum of 26.6% curcuminoids and 1% AKBA. Both the TBF and placebo were enclosed in vegetarian soft gel (reddish-brown) capsules of size “0”. The placebo was made from maltodextrin blended with an excipient base consisting of polysorbate-80, propylene glycol, and polyethylene glycol-400, all within acceptable daily intake levels.
Randomization, Blinding, and Unblinding
With the help of an independent statistician, the randomization chart was prepared using WinPepi version 11.65 (2016), and the individuals were randomly assigned to either the TBF or placebo groups. To ensure confidentiality, the allocation was concealed through the use of opaque bottles and alphanumeric codes, making the trial products identifiable only through the specific allocation codes. The pharmacist responsible for disbursing the study products received the hidden randomization code list. Both the randomization schedule and study materials were kept under the pharmacist’s restricted access to reduce the risk of bias in selection, and they were administered one after the other. No other personnel involved in the study were included in the dispensing procedure. The investigators and participants were blinded by using a placebo designed to match the size, color, packaging, and labeling of the actual treatment. All staff involved in the study were kept unaware of the treatment identities. Sealed, opaque envelopes containing package inserts revealing the identity of the study products were provided to the pharmacist. At times of emergencies requiring unblinding, the investigator would notify the pharmacist, who would then provide the envelope corresponding to that participant. The investigator would also communicate the need for unblinding to the sponsor and document the events accordingly.
Outcome Assessment
The primary measure assessed was the change in the sum of pain intensity difference (SPID) at 6 hours at rest (SPID6rest), which was calculated using the NRS. Secondary measures included the time taken for perceptible pain relief (PPR) and MPR using the double-stopwatch method to estimate the onset of pain relief (analgesia). Additional secondary outcomes comprised the SPID at movement (SPID6move) and pressure (SPID6pres), total pain relief at 6 hours at rest (TOTPAR6rest), movement (TOTPAR6move), and pressure (TOTPAR6pres) using the PRS. The number needed to treat (NNT) was also estimated as an added outcome of the PRS score for rest movement and pressure. Safety assessment measures were not studied in detail considering the 6-hour duration of the study. Physical examination, vital signs, and treatment-emergent adverse events, such as hypersensitivity or events reported by the patients, were recorded to assess the safety of the study.
NRS: The NRS comprises an 11-point scale ranging from 0 (“no pain”) to 10 (most severe pain imaginable). Participants were requested to assess their pain intensity using a numerical value between 0 and 10. The NRS evaluations were conducted during periods of rest, movement of the affected area, and application of pressure to the affected region, as per the screening report. Individual participants included in the study were those with an NRS (rest) score of 5 or more. Following the administration of medication, participants’ pain intensity ratings were recorded at rest, during movement, and under pressure, every 30 minutes for up to 6 hours post-dose to calculate SPID over 6 hours.
PRS: The PRS, a categorical scale coded 0–4, represents “no relief” to “complete relief”. TOTPAR indicates the area under the curve representing the duration of the analgesic effect. Following medication administration, participants assessed pain relief using PRS at rest (PRSrest), during movement of the affected area (PRSmove), and under pressure on the area affected (PRSpres), every 30 minutes for 6 hours to evaluate TOTPAR over 6 hours.
Onset of analgesia: The “double stopwatch” method was used to measure the onset of analgesia. Following medication administration, two stopwatches were started simultaneously and provided to the participant. When the individual participant reported the first pain relief point (PPR), the first stopwatch was stopped, while the second stopwatch stopped when the participant achieved complete pain relief, which is referred to as MPR. Times for PPR and MPR were recorded in hours and minutes, rounding up to the next minute if above 30 seconds. If the pain relief (PPR/MPR) was not achieved within 6 hours, it was noted as censored at that time point.
Short form of McGill pain questionnaire (SFMPQ): The MPQ allows for the expression of both aspects, affective domain (quality-wise) and sensory domain scale (intensity-wise), with responses provided at baseline and end of the study. Moreover, subjects reported their present pain intensity (PPI) on a 0–5 score. The pain intensity was measured using a 0–100 mm horizontal visual analog scale (VAS), with the no-pain endpoint (score of 0) and worst possible pain (score of 100).
Statistical Analysis
Pain intensity was evaluated for 6 hours following the oral intake of a single dose of the study medication, using NRS. The NRS-derived endpoints (PID and SPID) were statistically analyzed using linear mixed models for repeated measures, t-tests, or Mann–Whitney tests. A longitudinal mixed model for repeated measures was used to examine the PID, which indicates the change from baseline (PIbaseline − PItime). This model incorporated fixed effects for treatment, time, treatment-by-time interaction, and a random effect for participants. Variability between the treatment and the placebo was assessed using the least square mean (LSM), along with 95% confidence intervals (CIs) and two-sided p values.
Kaplan–Meier (KM) curves were employed to analyze the onset of analgesia (PPR and MPR), and a comparison was made using the log-rank test. The test statistic was then compared to a χ2 distribution with 1 degree of freedom. Median time to onset, Restricted Mean Survival Time (RMST), and RMST ratio (placebo/TBF) were derived from the KM analysis. The graphical verification of the proportional hazard assumption was performed using the Schoenfeld residual test, and survival curves were observed. The log-rank test, Gehan generalized Wilcoxon procedure, and Tarone–Ware were employed to determine the variability in survival probabilities between the groups. Mann–Whitney U test was used to assess the difference between groups in the TOTPAR. The NNT was calculated only when at least 50% of maximum TOTPAR and statistical significance was obtained from the Wald chi-square test.
Results
This study was designed to understand the effects of a single prescribed dose of the study medication administered to participants on a single day. There was complete adherence to treatment, with 100% compliance among participants. A total of 76 participants who reported acute LBP were included in the assessment to check the capability of treatment (TBF) in improving the LBP, compared to the placebo, with 35 corresponding to the placebo group and 41 to the group receiving the TBF. In the placebo group, there were 18 male participants and 17 female participants, while in the TBF group, there were 17 male participants and 24 female participants. None of the individual participants were reported with any adverse effects during the study period. There were no dropouts during the study. LBP among the participants treated with the test product and the placebo during the baseline observation was not statistically significant (p > 0.05) in all three categories (rest, movement, and pressure). Baseline observation also showed no significant gender-wise difference in the distribution of LBP in all three categories (p > 0.05).
NRS on Rest, Movement, and Pressure
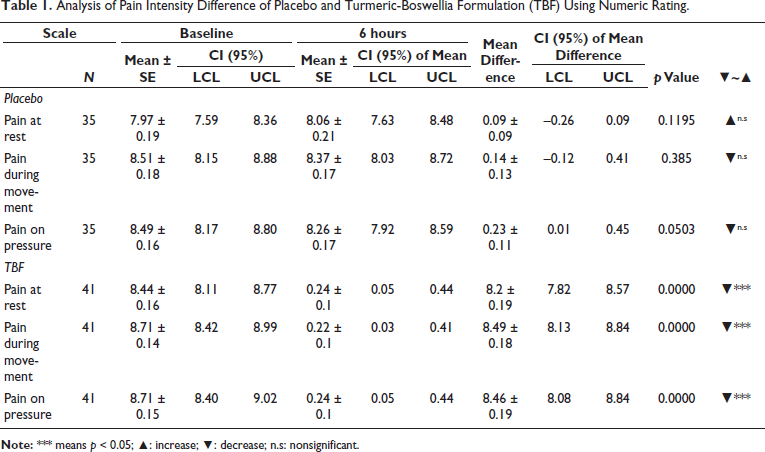
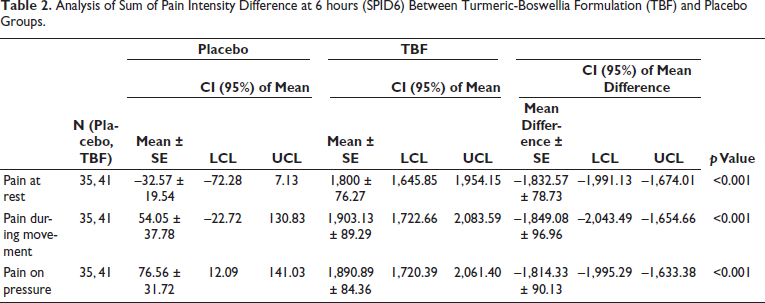
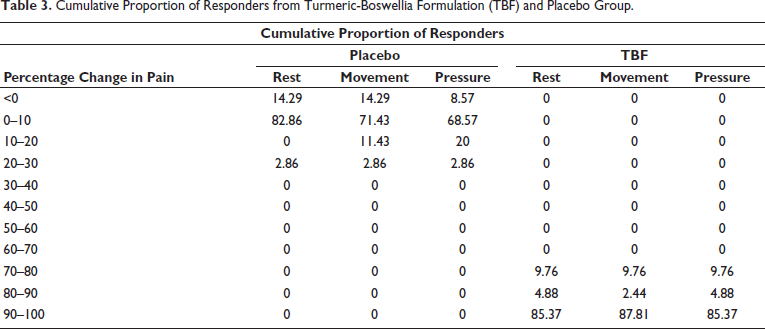
Analysis of the PID showed that TBF administration significantly reduced pain for all categories (rest, movement, and pressure), whereas no significant decrease in pain was recorded in the placebo group (Table 1). For pain at rest, the mean NRS score decreased by 8.2 ± 0.19 points in the TBF group compared to a 0.09 ± 0.09 point increase in the placebo group (p < 0.0001). Similarly, for pain during movement, the mean NRS score decreased by 8.49 ± 0.18 points in the TBF group compared to a 0.14 ± 0.13 point decrease in the placebo group (p < 0.0001). For pressure pain, the mean NRS score decreased by 8.46 ± 0.19 points in the TBF group compared to a 0.01 ± 0.45 point decrease in the placebo group (p < 0.0001). The response of pain among the participants from the treatment using the administration of TBF and placebo is summarized here as SPID. SPID explains the treatment response over the study period (360 minutes). From the analysis, it is clear that the response of participants under TBF treatment showed significantly better SPID response than the placebo at rest, movement, and pressure (Table 2). The results showed that TBF was significantly more effective than placebo in relieving pain intensity during rest (p < 0.001), pain during movement (p < 0.001), and pressure pain (p < 0.001). From the CPRA estimate for treatment and placebo, it is clear that subjects in the TBF group showed a higher percentage of improvement in LBP when compared to the placebo group (Table 3). The NRS analysis showed that the superior effect of TBF in relieving LBP compared to placebo is statistically and clinically significant.
Analysis of Pain Intensity Difference of Placebo and Turmeric-Boswellia Formulation (TBF) Using Numeric Rating.
Analysis of Sum of Pain Intensity Difference at 6 hours (SPID6) Between Turmeric-Boswellia Formulation (TBF) and Placebo Groups.
Cumulative Proportion of Responders from Turmeric-Boswellia Formulation (TBF) and Placebo Group.
Onset of Analgesia
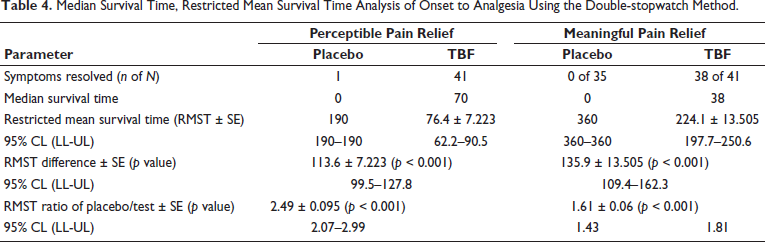
Participants in the TBF group showed a significantly higher onset of analgesia with 100% of participants experiencing PPR compared to the 2.86% of participants experiencing PPR in the placebo group. While participants in the TBF group experienced 92.7% MPR, none of the participants in the placebo group experienced MPR. This shows that the acute LBP relief was significantly higher in the TBF group.
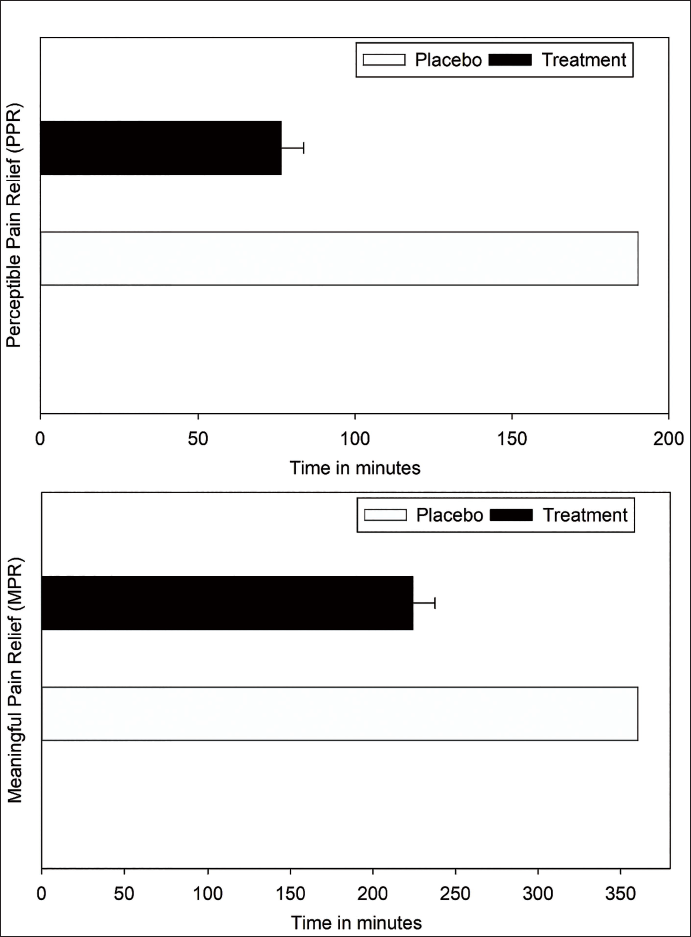
The median time for pain relief was 70 minutes in the TBF group but was not estimated for the placebo group due to relatively lower pain relief (Table 4). The RMST analysis showed a PPR of 76.4 ± 7.223 minutes and MPR of 224.1 ± 13.505 minutes for the TBF group, compared to 190 minutes (PPR) and 360 minutes (MPR) for the placebo group (Figure 1). From the RMST ratio, it is evident that the TBF group experienced a PPR of 2.49 times and achieved MPR 1.61 times faster than the placebo group, illustrating the significant improvement in acute LBP relief from TBF administration. Also, a low RMST value in the MPR and PPR for the TBF group (MPR = 224.1 ± 13.51, PPR = 76.4 ± 7.2) compared to the placebo group (MPR = 360, PPR = 190) indicates a lower pain duration in the TBF with an overall pain improvement than the placebo group.
Median Survival Time, Restricted Mean Survival Time Analysis of Onset to Analgesia Using the Double-stopwatch Method.
Comparison of Mean Time to Achieve Perceptible Pain Relief (PPR) and Meaningful Pain Relief (MPR) in the Treatment (Turmeric-Boswellia Formulation (TBF)) and Placebo Groups.
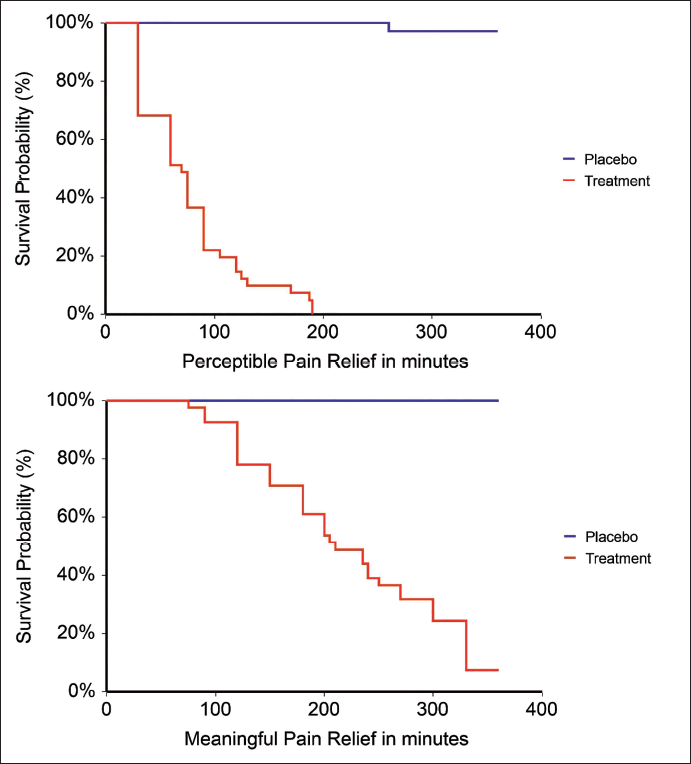
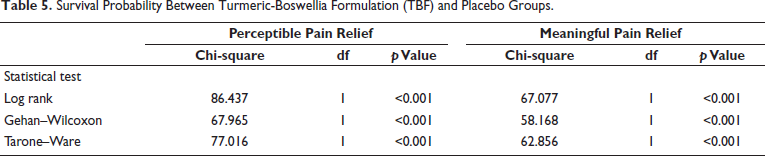
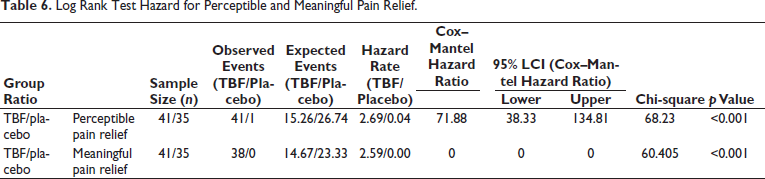
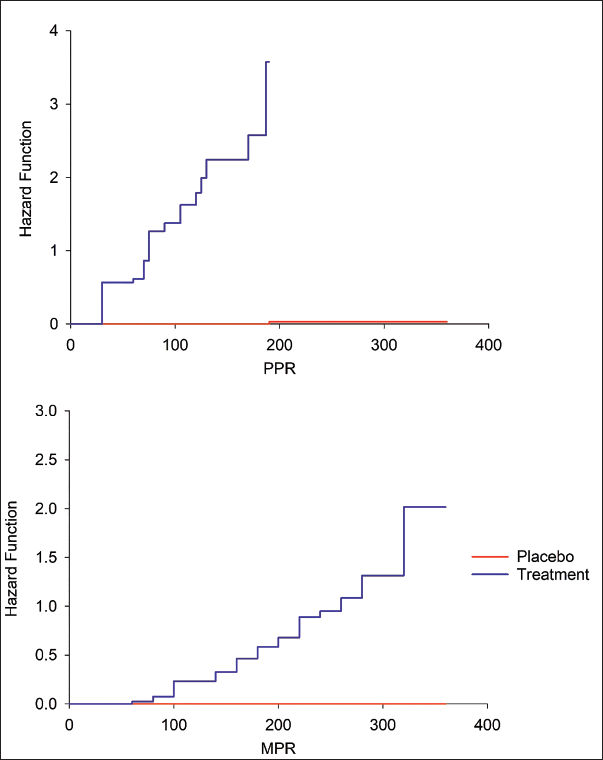
The KM survival plots for PPR and MPR also showed superior performance of TBF in achieving faster acute LBP pain relief among studied participants compared to the placebo group (Figure 2). For MPR, the earliest pain relief was reached within 75 minutes (1 participant) and a total of 38 participants achieved MPR in 360 minutes, whereas, in the placebo group, none of the participants reached MPR in 360 minutes. While, 13 participants in the TBF group experienced PPR as early as 30 minutes and 41 participants within 190 minutes, only one participant in the placebo group achieved PPR during the study period. The TBF group exhibited significantly improved survival outcomes compared to the placebo group, as evidenced by the results of the log-rank test, Gehan generalized Wilcoxon procedure, and Tarone–Ware test (p < 0.001), as detailed in Table 5. The log-ranked hazard ratio for PPR and MPR also showed statistically significant variability in the observed relief of acute LBP between TBF and placebo groups (p < 0.001), with high PPR and MPR achievement in the TBF group (Table 6). The cumulative hazard ratio plots for PPR and MPR also evidence the superior performance of TBF in faster relief of acute LBP for both PPR and MPR (Figure 3). It is hence clear that the effect of TBF in relieving acute LBP is statistically and clinically significant.
Kaplan–Meier Survival Plot of Onset of Analgesia Observed Among Participants in Treatment (Turmeric-Boswellia Formulation (TBF)) and Placebo Groups.
Survival Probability Between Turmeric-Boswellia Formulation (TBF) and Placebo Groups.
Log Rank Test Hazard for Perceptible and Meaningful Pain Relief.
Cumulative Hazard Ratio of Onset of Analgesia Observed Among Participants in Treatment (Turmeric-Boswellia Formulation (TBF)) and Placebo Groups.
Total Pain Relief
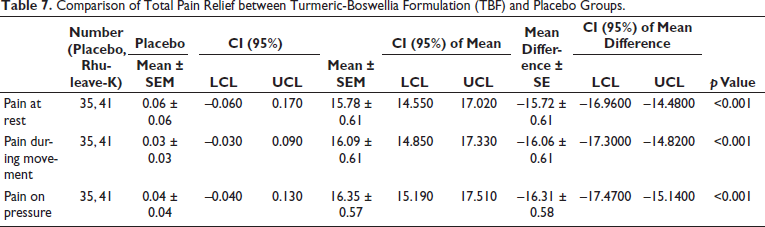
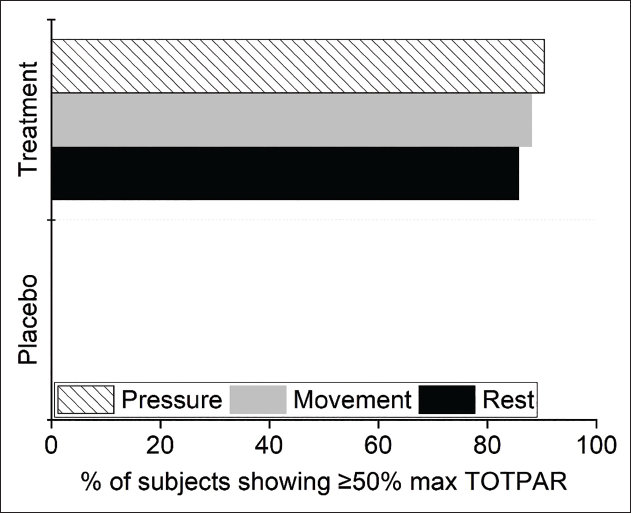
The total PRS calculated as TOTPAR6 for rest, movement, and pressure evidenced statistically significant variability between the TBF and placebo groups (Table 7). TBF group showed significantly better efficiency in relieving LBP compared to placebo during rest (mean difference placebo-TBF = –15.72 ± 0.6; p < 0.001), movement (mean difference placebo-TBF = –16.06 ± 0.61; p < 0.001) and pressure (mean difference placebo-TBF = –16.31 ± 0.58; p < 0.001). The Wald chi-square test gave statistically significant variability in the proportion of subjects showing at least 50% maximum TOTPAR between the placebo and TBF group at rest, pressure, and movement (p < 0.001). No participants from the placebo group demonstrated a minimum of 50% maximum TOTPAR in rest, movement, and pressure, whereas, 75.71% of the TBF group experienced at least 50% maximum TOTPAR in rest, 88.09% in movement, and 90.48% in pressure (Figure 4). Treatment efficacy (NNT) in the TBF group for at least 50% of MPR was 1.2 in rest and 1.1 in both movement and pressure.
Comparison of Total Pain Relief between Turmeric-Boswellia Formulation (TBF) and Placebo Groups.
Percentage of Responders with At Least 50% Maximum Total Pain Relief (TOTPAR).
Short Form of McGill Pain Questionnaire
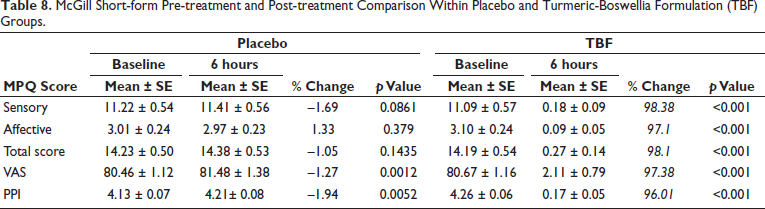
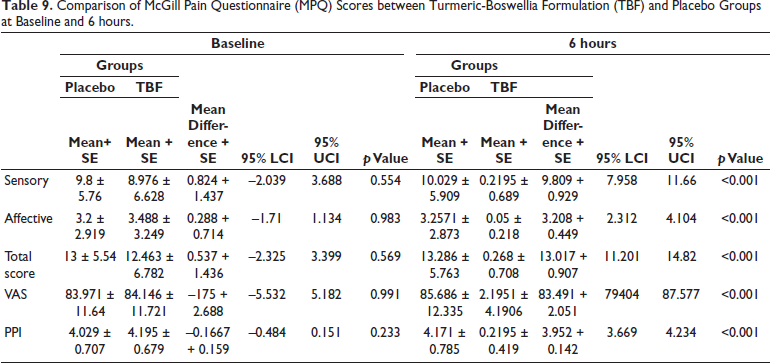
There is a statistically significant reduction in sensory, affective, and total scores in the TBF group during the observations at 6 hours after the treatment compared to the baseline (p < 0.001). Similarly, VAS and PPI records from the TBF group during 6-hour post-treatment showed a statistically significant reduction compared to the baseline score (p < 0.001), whereas the placebo group showed no statistically significant variability in the MPQ scores (p > 0.05) during observations at 6 hours compared to the baseline observation (Table 8). The VAS and PPI scores in the placebo group showed a statistically significant increase during post-treatment compared to the baseline (p < 0.01). Between-group analysis of the MPQ showed no significant variability at baseline (p > 0.05) in the sensory, affective, and total scores of MPQ. The VAS and PPI also showed no significant variability between TBF and placebo during the baseline (p > 0.05). The TBF group revealed a significantly higher reduction in pain-related symptoms (sensory, affective, and total) than the placebo group at the 6th-hour observation (p < 0.001). Similarly, VAS and PPI records from the TBF group during 6-hour post-treatment showed a statistically significant reduction (p < 0.001), compared to the placebo group (Table 9).
McGill Short-form Pre-treatment and Post-treatment Comparison Within Placebo and Turmeric-Boswellia Formulation (TBF) Groups.
Comparison of McGill Pain Questionnaire (MPQ) Scores between Turmeric-Boswellia Formulation (TBF) and Placebo Groups at Baseline and 6 hours.
Discussion
Physical exertion tasks are often integral components of various occupations, especially those involving heavy manual labor involving lifting, moving, and carrying physical loads. Manual handling and postures while carrying heavy materials potentially result in whole-body vibrations resulting in acute LBP. The relationship between manual handling and acute LBP among labor workers has been the subject of extensive research. Equally, stress on the body from sports activities also causes acute LBP, and there are some explicit studies and research focusing on acute LBP among athletes. These studies brought light to the specific physical activities that potentially contribute to back-related issues. Biomechanical studies identified lifting, bending, and twisting, biomechanically stressful activities that pose a higher risk of stressing the spine and causing acute LBP. Such biomechanical investigations were extensively conducted across various workplaces to identify the prevalence and risk of such activities and to understand more about the potential preventive measures. In the context of addressing the challenges posed by manual materials handling and its impact on acute LBP, this study explored the effects of TBF on exercise-induced acute LBP.
Rhuleave-K is developed using natural bioactives from turmeric-Boswellia which is designed using patented speed tech, which creates a powerful combination to deliver faster action. Curcuminoids are bioactive components in turmeric, which include curcumin (C21H20O6, 368.4 g/mol), desmethoxycurcumin (DMC) (C20H18O5, 338.4 g/mol), and bis-desmethoxycurcumin (DMC) (C19H16O4, 308.3 g/mol). The active components of the B. serrata gum resin are boswellic acids. The most potent anti-inflammatory agents are 11-keto-β-boswellic acid (KBA) (C30H46O4, 470.7 g/mol) and AKBA (C32H48O5, 512.7 g/mol).
Rhuleave-K is reportedly a successful herbal therapeutic intervention in relieving pain, which has comparative pain-relieving capabilities as that of acetaminophen. Such capabilities of TBF (Rhuleave-K) are being assessed in this study to potentially mitigate the impact of exercises and manual jobs on acute LBP. This approach aligns with the broader goal of identifying practical solutions and interventions to reduce the incidence of work-related LBP issues among individuals engaged in manual materials handling. As the body of research expands, this baseline assessment on the impact of TBF in treating LBP will help in providing immediate effects of manual handling on lower back health, but also the long-term implications. Beneficially, a recent study showed the safety, efficacy, and potential benefit of a herbal pain relieving formula that can be well integrated into occupational health strategies.
The relationship between certain strenuous work and sports like weightlifting and gymnastics in causing risk of disk degeneration, vertebral damage, and back pain has been investigated, with evidence suggesting a potential connection. Apart from physical stress, some studies showed the significant role of psychological factors in synergy with physical factors in causing occupational LBP. Furthermore, the neurohormonal changes associated with stress may influence metabolic activity in various tissues of the back, potentially contributing to back pain. The synergy of physical and psychological factors highlights the importance of understanding and managing lower back-related issues. While physical risk factors may be particularly relevant in the initial onset of back pain, the combined influence of psychosocial and physical factors should not be underestimated, especially in the context of occupational settings. The present study contributes to this understanding by investigating the onset of pain relief in subjects treated with TBF utilizing two distinct methods, PPR and MPR. The study demonstrates that subjects under TBF treatment experience a significantly shorter duration of pain, even with comparable pain intensity as that of placebo, suggesting the potential efficacy of TBF in managing and alleviating pain associated with strenuous physical activities, offering valuable insights for interventions in individuals prone to lower back-related issues. While all participants in the TBF group achieved measurable pain relief during the study period, none from the placebo group achieved MPR. This is evident in the efficacy of TBF administration in acute LBP relief.
Pain perception is intricately linked to structures that contain nerve endings, as these specialized receptors, known as nociceptors, are responsible for detecting noxious stimuli. In addition to the more generalized nociceptors, there are specialized nerve endings located in tendons, ligaments, and joint capsules. These nerve endings are typically sensitive to mechanical stimuli and play a crucial role in proprioception. However, under certain conditions, such as injury or excessive mechanical stress, these specialized nerve endings can also contribute to the sensation of pain. Furthermore, mechanoreceptors present in tendons and muscle insertions can give rise to pain. These receptors are responsive to mechanical forces and movements, and when subjected to abnormal or excessive stimuli, they can transmit signals perceived as pain. The presence of nociceptors in tendons, ligaments, joint capsules, blood vessels, and fascia, as well as the sensitivity of muscle spindles, highlights the complexity of pain perception. Studies showed that C. longa extracts have anti-nociceptive action by the activation of both opioid and nonopioid mediating systems. Also, there is extensive research on the antioxidant, anti-inflammatory, and antinociceptive activities of C. longa. Similarly, B. serrata extracts also reportedly have high efficacy in pain relief treatments. The present study highlights the efficacy of Rhuleave-K (a combination of C. longa and B. serrata extracts) in effectively controlling both the affective (emotional) and sensory domains of acute pain in the lower back part of the body.
The assessment of pain intensity using the NRS and PRS supports the efficacy of TBF in relieving acute LBP. The statistically significant reduction in acute LBP among participants in the TBF group during rest, movement, and pressure shows the significant efficacy of TBF in relieving acute LBP. Also, significant improvements in pain intensity and relief, as measured by the SPID and TOTPAR compared to placebo, highlight the positive outcomes associated with TBF administration. Moving beyond the subjective reports of pain, the study recognizes the importance of assessing functionality in individuals with LBP. Clinical evaluation of dysfunction, disability, and capacity often relies on observing and understanding patient performance. The close connection between physical function and behavior underscores the challenge of wholly separating the two in both conceptualization and practical assessment. Notably, the study recognizes that nonspecific LBP is often linked to disturbed function or painful musculoskeletal dysfunction. To comprehensively understand the functionality of TBF, the study extends its assessment to pain intensity and relief scores under movement and pressure application. The findings consistently demonstrate the significant effectiveness of TBF in alleviating pain across various measures, reinforcing its positive impact on both the sensory and functional aspects of musculoskeletal pain. These results contribute valuable insights into the potential therapeutic benefits of TBF in managing LBP and improving overall functionality in individuals with musculoskeletal conditions.
The curcumin content in Rhuleave-K potentially affects the transient receptor potential channel, vanilloid subfamily member 1 (TRPV1), and helps in reducing pain by selectively targeting pain-associated neurons. TRPV1 is found in sensory neurons and is known to play a crucial role in detecting and transmitting pain signals. Curcumin, a natural compound with anti-inflammatory and antioxidant properties, is believed to inhibit TRPV1-mediated pain. The exact mechanism by which curcumin inhibits TRPV1-mediated pain is associated with the disruption of calcium influx. Calcium is an essential ion for neuronal function, and alterations in its homeostasis can affect the ability of neurons to transmit signals. When TRPV1 is activated for a prolonged period or with sufficient intensity, the resulting calcium influx may lead to degeneration and apoptosis (programmed cell death) of the neurons. This process essentially exhausts the neurons involved in pain transmission, contributing to pain relief. The significant reduction in acute LBP as obtained from various metrics like NRS, SPID, pain survival, and TOTPAR from this study among the participants with the administration of Rhuleave-K (TBF) highlights the efficacy of the single dose of Rhuleave-K in reducing the body exertion induced acute LBP with rapid effect.
Conclusion
In summary, the results and inference from this randomized double-blinded placebo-controlled trial give significant support for the effect of the turmeric-Boswellia formulation (TBF, Rhuleave-K) in reducing acute LBP among healthy participants. The findings showed a markedly fast onset of analgesia, with a significant number of participants obtaining clinically meaningful relief in a relatively shorter period of time after administration of TBF. Importantly, the TBF group displayed statistically significant improvement in pain relief in comparison to the placebo group as seen by subjective measures like NRS and PRS. It is noted that the TBF group reported MPR across diverse activities such as resting, moving, and pressure application with a favorable NNT, which affirms the clinical efficacy of this treatment. These findings strengthen the possibility of the TBF as a potential therapeutic strategy for the treatment of acute LBP and demand further investigation in clinical practice.
Abbreviations
LBP: Low back pain; NNT: Number needed to treat; NRS: Numerical rating scale; MPR: Meaningful pain relief; PID: Pain intensity difference; PPI: Present pain intensity; PPR: Perceptible pain relief; RMST: Restricted mean survival time; SF-MPQ: Short form of McGill pain questionnaire; SPID: Sum of pain intensity difference; TOTPAR: Total pain relief; VAS: Visual analog scale.
Footnotes
Acknowledgments
The authors acknowledge Arjuna Natural Pvt. Ltd., Kerala, India for providing the test product (Rhuleave-K) and placebo capsules.
Author Contributions
Girish HR was involved in the study design, investigation, and analysis of the study, and wrote the original draft of the manuscript. Sanjeev Kumar Kare and Ajay Gupta contributed to the study design, planning, and investigation and reviewed the manuscript. All authors read and approved the final manuscript.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This study protocol was reviewed and approved by Rajalakshmi hospital Institutional Ethics Committee (RHIEC), Vidyaranyapura, Bangalore (approval number RH/IEC/AP-056/2020 dated March 21, 2020), Vagus Institutional Ethics Committee, Malleshwaram, Bangalore (approval dated May 14, 2020), Santhosh Hospital Institutional Ethics Committee, Promenade Road, Bangalore (approval number SHIEC/CC/MAR_2020/03 dated March 19, 2020), Institutional Ethics Committee Government Medical College and Government General Hospital, Srikakulam, Andhra Pradesh (approval dated September 12, 2020), Institutional Ethics Committee Nirmal Hospital, Jhansi, Uttar Pradesh (Approval dated September 3, 2020), and Sudbhawana Hospital Ethics Committee, Lanka, Varanasi (approval dated August 29, 2020).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Before participating in the study, a written voluntary informed consent form was signed by all the participating subjects.
Statement of Ethics
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The clinical trial was prospectively registered in the Clinical Trial Registry of India with registration number CTRI/2020/06/025601 dated 04/06/2020.
