Abstract
Background
Inflammatory diseases, including autoimmune disorders, and chronic inflammatory conditions, are driven by dysregulated immune responses, particularly involving macrophage activation. Macrophages play a critical role in modulating inflammation through the production of pro-inflammatory mediators such as nitric oxide (NO), reactive oxygen species (ROS), tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6). Vicenin-2 (VCN-2), a flavonoid with potential immunomodulatory properties, has garnered attention for its therapeutic potential. This study investigates the bidirectional immunomodulatory effects of VCN-2 on lipopolysaccharide (LPS)-induced RAW264.7 mouse macrophage cells to establish a foundation for its development as a novel anti-inflammatory agent.
Objectives
This study investigates the bidirectional immunomodulatory effects of vicenin-2 (VCN-2) on lipopolysaccharide (LPS)-induced mouse macrophage RAW264.7 cells, aiming to establish a basis for developing VCN-2 as a novel anti-inflammatory agent in diseases such as skin inflammation, autoimmune disorders, and chronic inflammatory conditions.
Materials and Methods
LPS-induced mouse macrophage RAW264.7 cells were used as an inflammation model. The CCK-8 method was used to detect the activity and toxicity of VCN-2. The neutral red assay was used to measure the phagocytic ability of RAW264.7 cells. The Griess assay was used to detect nitric oxide (NO) levels. Flow cytometry was used to measure reactive oxygen species (ROS) levels. Enzyme-linked immunosorbent assay (ELISA) was used to measure tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) levels. Quantitative reverse transcription polymerase chain reaction (qRT-PCR) was used to detect the expression of inducible nitric oxide synthase (iNOS), TNF-α, and IL-6 messenger ribonucleic acid (mRNA). Western blot (WB) was used to detect the expression of phosphorylated p38MARK (p-p38).
Results
VCN-2 activates RAW264.7 cells and promotes cell proliferation. As the concentration of VCN-2 increases, its proliferative effect increases. In the resting state, VCN-2 improves cell proliferation by increasing cell number and cell volume. In the inflammation model, VCN-2 repairs cell morphology and restores cell proliferation. VCN-2 promotes phagocytosis of resting cells while attenuating phagocytic activity in inflammatory models. VCN-2 increases ROS levels in resting cells and reduces ROS levels in LPS-induced cells. VCN-2 increases NO, IL-6, and TNF-α content by promoting iNOS, IL-6, and TNF-α gene transcription levels, regulating macrophage immune function. In the inflammation model, VCN-2 significantly downregulates the transcription levels of iNOS, IL-6, and TNF-α genes, reduces NO, IL-6, and TNF-α content, and alleviates the inflammatory response. VCN-2 upregulates macrophage p38 in the resting state. In terms of phosphorylation expression, VCN-2 downregulates the phosphorylation expression of p38 in inflammatory model cells.
Conclusion
VCN-2 enhances RAW264.7 cell proliferation, improves cell morphology, and amplifies phagocytic capacity. VCN-2 modulates the transcription of iNOS, IL-6, and TNF-α mRNA through the dual regulation of p38 phosphorylation, influencing the secretion of NO, IL-6, TNF-α, and ROS in RAW264.7 cells under varying conditions. This study demonstrates that VCN-2 exerts a bidirectional regulatory effect on macrophage immune function, supporting its potential as a therapeutic agent for inflammatory and immune-related diseases.
Introduction
Inflammation and immunity, though distinct, share intricate connections through common cellular and molecular pathways. Immune cells, such as T lymphocytes, natural killer cells, and monocytes/macrophages, play a pivotal role in both inflammatory and immune responses. The cytokines secreted by these cells, including tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and IL-1β, function as crucial mediators in these processes (He & Dai, 2011). Anti-inflammatory immune drugs, capable of modulating these responses, are essential in treating various inflammatory immune diseases. These drugs, particularly those derived from natural plant sources, are gaining attention due to their efficacy, reduced toxicity, and minimal side effects (Ma et al., 2013). Recent studies highlight the critical role of macrophages in orchestrating inflammatory responses, particularly in chronic diseases like psoriasis and rheumatoid arthritis, where dysregulated cytokine production drives the pathology (Boutet et al., 2021; Kamata & Tada, 2022).
Vicenin-2 (VCN-2), a flavonoid component widely found in Chinese medicine and medicinal plants, has demonstrated a range of biological activities, including anti-viral, anti-spasmodic, and anti-tumor properties (Kang et al., 2015; Zhang et al., 2020). Recent research underscores VCN-2’s potential as an anti-inflammatory agent, with studies showing its ability to modulate immune responses in various cellular models (Sulaiman & Lakshmanan, 2022). Our in vitro experiments were conducted on human dermal fibroblast (HDF) cells exposed to ultraviolet B rays (UVB) and found that VCN-2 has significant anti-UVB toxicity and can reduce photodamage by inhibiting the mitogen-activated protein kinases (MAPKs) signaling pathway (Duan et al., 2019). As the understanding of skin photoaging mechanisms evolves, immune modulators are gaining prominence in both prevention and treatment. Our in vivo study revealed that VCN-2 modulates immune function and exerts anti-inflammatory effects. This is achieved through mitigating damage to the thymus and spleen in photoaging mice, regulating macrophage polarization in the skin, and curbing the production of pro-inflammatory factors. Consequently, VCN-2 appears to counteract skin photoaging by protecting immune organs and altering macrophage immune status (awaiting publication). Furthermore, VCN-2’s flavonoid structure suggests potential similarities with other immunomodulatory flavonoids like quercetin, which have shown promise in attenuating inflammatory signaling (Singh et al., 2021). In conclusion, VCN-2, a natural plant-derived component, is hypothesized to possess bidirectional immunomodulatory effects. Although VCN-2’s pharmacological benefits are documented, its bidirectional effects on macrophage function remain underexplored, particularly in inflammatory diseases like skin photoaging, psoriasis, and autoimmune conditions. This gap presents a unique opportunity to investigate the role of VCN-2 in inflammatory and immune responses, particularly in the context of skin diseases such as skin photoaging, where inflammation and immunosuppression are key pathogenic factors.
Macrophages, derived from bone marrow and differentiated from monocytes, are central to immune regulation and inflammatory responses (He et al., 2012). They are involved in various functions, such as phagocytosis, antigen presentation, and cytokine secretion (TNF-α, IL-6, IFN-γ) (Epelman et al., 2014). The mouse macrophage RAW264.7 serves as an important model for studying cellular inflammation and immunity (Ji et al., 2020). Lipopolysaccharide (LPS) is the main component of the cell wall of Gram-negative bacteria and can activate M1 macrophages to release a variety of enzymes, mediators, and cytokines such as inducible nitric oxide synthase (iNOS), nitric oxide (NO), TNF-α, and IL-6 (Xie et al., 2019; Zhu & Ma, 2021), ultimately causing inflammation and even shock. Therefore, stimulating RAW264.7 cells with LPS as a common cellular inflammation model is the best choice to explore the anti-inflammatory effects of drugs.
This study focuses on elucidating the bidirectional immunomodulatory effects and underlying mechanisms of VCN-2 in LPS-induced RAW264.7 cells. Through this investigation, we aim to establish a theoretical foundation for the use of VCN-2 in treating various inflammatory immune diseases, mainly skin diseases, autoimmune disorders, and chronic inflammatory conditions, potentially leading to the development of novel anti-inflammatory immune therapies.
Materials and Methods
Cell Culture and Viability Assessment
The mouse monocyte macrophage leukemia cell line, RAW264.7, was purchased from the Cell Bank of the Chinese Academy of Science (Shanghai, China). RAW264.7 cells were cultured in Dulbecco’s modified Eagle medium (DMEM) (Cat. No. 11995065, Gibco BRL., Gaithersburg, MD, USA) containing 10% fetal bovine serum (FBS, Cat. No. B040100, ZSGB Bio. Co. Ltd., Beijing, China) and 1% penicillin-streptomycin (Cat. No. C0222, Beyotime Biotech.) at 37°C, in the humidified condition with 5% CO2. The DMEM medium was replaced with internal of 2–3 days, and cells were passaged when they achieved 80% confluency. The cell viability was evaluated using the CCK-8 assay. The LPS pre-treated RAW264.7 cells were cultured in 96-well plates for 24 h and then treated with or without VCN-2 (CAS No. 23666-13-9), and cultured for a further 24 h. This 24-h treatment duration was selected based on preliminary time-course experiments showing optimal inflammatory and immunomodulatory responses, consistent with prior studies (Lee et al., 2021). Then, the cytotoxicity of 20, 40, 80, 100, 120, 240, and 480 µg/mL VCN-2 was post-incubating with CCK-8 (Cat. No. C0037, Beyotime Biotech.) at a dosage of 10 µL/well at 37°C for 4 h. The 96-well plates were gently shaken to dissolve the formazan product, and the absorbance was measured at 450 nm with a microplate reader (Model: Varioskan LUX, Thermo Fisher Scientific).
Establishment of Inflammation Model and Morphological Analysis
The RAW264.7 cells were randomly divided into four groups, including the normal control group (RAW, without any treatment except for DMEM), the RAW+LPS group (also as RAW264.7 cell drug model, cells treated with 1 µg/mL LPS), the RAW+LPS+VCN-2 group (cells treated with 1 µg/mL LPS and 80 µg/mL VCN-2), and the RAW+VCN-2 group (cells treated with 80 µg/mL VCN-2). Then, the cells in the above groups were cultured for the following experiments. After 24 h of culture, the cells in different groups were observed under an inverted fluorescence microscope (Nikon, Japan), pictures were taken, and samples were collected for subsequent experiments.
Assessment of Immunological and Inflammatory Markers
The biochemical characteristics of RAW264.7 cells, including NO content, reactive oxygen species (ROS) activity, and pinocytosis ability, were measured. The NO content was determined using the Nitric Oxide Detection Kit (Cat. No. S0021S, Beyotime Biotech.) as instructed by the manufacturer. The pinocytosis ability was evaluated by staining the RAW264.7 cells with 0.1% neutral red for 30 min, washing with PBS three times, and measuring at 540 nm using a microplate reader (Model: Varioskan LUX, Thermo Fisher Scientific). The ROS activity was determined with the Reactive Oxygen Species Assay Kit (Cat. No. S0033S, Beyotime Biotech.) as described protocol of the manufacturer, with a CytoFLEX flow cytometry (Beckman Coulter Inc., Brea, CA, USA).
Real-time Quantitative Polymerase Chain Reaction (RT-qPCR) Assay
Total ribonucleic acids (RNAs) in RAW264.7 cells were extracted using the RNAiso Plus (Cat. No. 9108Q, Takara, Tokyo, Japan) following the protocol of the manufacturer. The total RNA (2 µg) was used to synthesize the complementary DNA (cDNA) with a Hifair II 1st Strand cDNA Synthesis Kit (Cat. No. 11121ES60, Yeasen Biotech. Co. Ltd., Shanghai, China) as instructed by the manufacturer. The gene transcription of target genes was detected using Hieff UNICON Universal Blue qPCR SYBR Green Master Mix (Cat. No. 11184ES03, Yeasen Biotech. Co. Ltd.) and quantified with an AB StepOnePlus Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The reaction condition was carried out as follows: 95°C for 2 min, 40 cycles of 95°C for 10 s and 60°C for 30 s. The previously reported 2–∆∆Ct method (Livak & Schmittgen, 2001) was used to calculate the relative gene transcription of target genes, and the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the control gene. The PCR primer sequences for iNOS, TNF-α, IL-6, and GAPDH were designed and listed in Table 1.
The Primers for the Polymerase Chain Reaction (PCR) Assay.
Western Blot (WB) for Examining Phosphorylated-p38 Expression
Collect cells, add 800 µL radioimmunoprecipitation assay (RIPA) lysis buffer, and collect the supernatant. Quantify protein by bicinchoninic acid (BCA) method, prepare protein sample, prepare polyacrylamide gel, perform SDS-PAGE (sodium dodecyl sulfate–polyacrylamide gel electrophoresis) gel electrophoresis, transfer protein to polyvinylidene difluoride (PVDF) membrane by wet method, dilute the corresponding primary antibody (rabbit polyclonal, p-p38, cell signaling, USA) with blocking solution to a concentration of 1:1,000. The final dilution concentration of the internal reference primary antibody (rabbit polyclonal, GAPDH, Abcam, UK) was 1:1,000, and then it was incubated for 1.5 h. Wash with membrane washing buffer four times, 5 min each time; dilute the secondary antibody (horseradish peroxidase (HRP) conjugated goat anti-rabbit IgG, BlueSky, China) to a concentration of 1:1,000 with blocking solution, and then incubate for 1 h. The samples were washed four times with washing buffer for 5 min each. Enhanced chemiluminescence (ECL) substrate luminescence treatment was then performed. The gray values of the protein bands were analyzed using the ImageJ software.
Statistical Analysis
Statistical analyses were conducted using the GraphPad PRISM software (version: 7.0, GraphPad Software Inc., San Diego, CA, USA). Data were assigned as Mean ± Standard Deviation (SD), and analyzed using one-way analysis of variance (ANOVA) followed by the LSD test, when comparing the differences among multiple groups. p value less than .05 was considered to be a significant difference.
Results
VCN-2 Modulates NO and ROS Levels in Resting and LPS-induced Macrophages
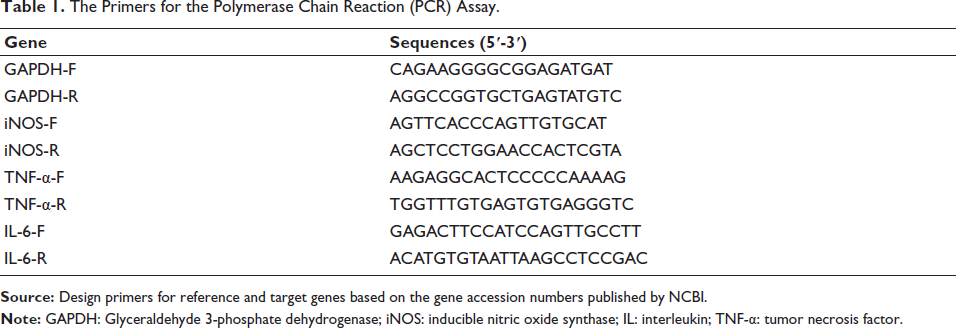
The CCK-8 results showed that VCN-2 demonstrated a good inflammatory protective effect at low concentration, compared with that of the RAW group (Figure 1A, all p < .001). With the increase in VCN-2 concentration, its protective effect increased (Figure 1A). However, due to the poor solubility of VCN-2, the solvent dimethyl sulfoxide (DMSO) in drugs with a concentration of more than 80 µg/mL had a certain toxic effect on cells. Therefore, 80 µg/mL VCN-2 was selected for the following experiments. Our findings indicated that the NO content in LPS-induced RAW264.7 cells (RAW+LPS group) was significantly higher compared to that in the RAW group (Figure 1B, p < .001). However, VCN-2 treatment (RAW+LPS+VCN-2 group) remarkably decreased the NO content in LPS-induced RAW264.7 cells compared to that in the RAW+LPS group (Figure 1B, p < .001). The flow cytometry for ROS results identified that the ROS value was significantly higher in LPS-induced RAW264.7 cells compared to that in the RAW group (Figure 1C, p < .0001). While VCN-2 treatment markedly reduced the ROS value compared to that in LPS-induced RAW264.7 cells (RAW+LPS group) (Figure 1C, p < .001). Meanwhile, VCN-2 treatment (RAW+LPS+VCN-2 group) could also significantly block the LPS-induced iNOS gene transcription in RAW264.7 cells compared to that in the RAW-LPS group (Figure 1D, p < .0001).
Moreover, VCN-2 treatment (RAW+VCN-2 group) could significantly increase NO content (Figure 1B, p < .05), ROS value (Figure 1C, p < .001), and iNOS gene transcription (Figure 1D, p < .0001) in RAW264.7 cells compared to that in the RAW group. These findings indicate that VCN-2 enhances immune function in resting cells by moderately increasing NO and ROS, while attenuating excessive production in inflammatory states, reflecting its bidirectional regulatory role.
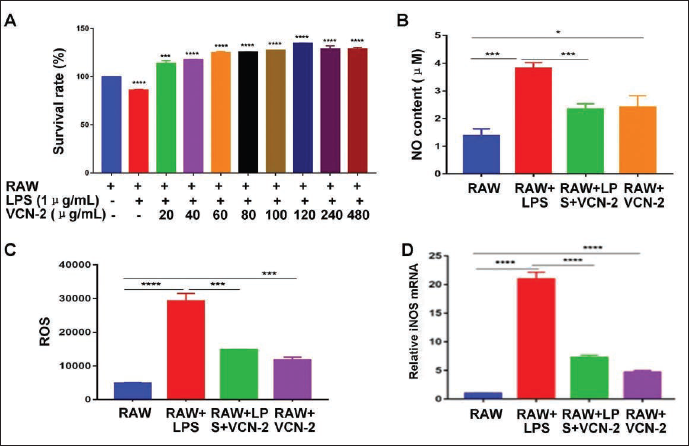
VCN-2 Restores Morphology in LPS-induced Macrophages
In the resting state, RAW264.7 cells are round or oval-shaped, densely packed, and without significant protrusions. After adding an 80 µg/mL concentration of VCN-2, the number of cells increased, with regular morphology and larger volume compared to normal cells (Figure 2, RAW+VCN-2). Following LPS induction, most cells became irregularly shaped and deformed, with a decreased proliferation ability (Figure 2, RAW+LPS). Upon adding VCN-2, there was an increase in cell number and volume, and the cell morphology became regular, with most cells appearing round or oval-shaped, and pseudopodia were observed (Figure 2, RAW+LPS+VCN-2).
Vicenin-2 (VCN-2) Ameliorated RAW264.7 Morphology with or without Lipopolysaccharide (LPS).
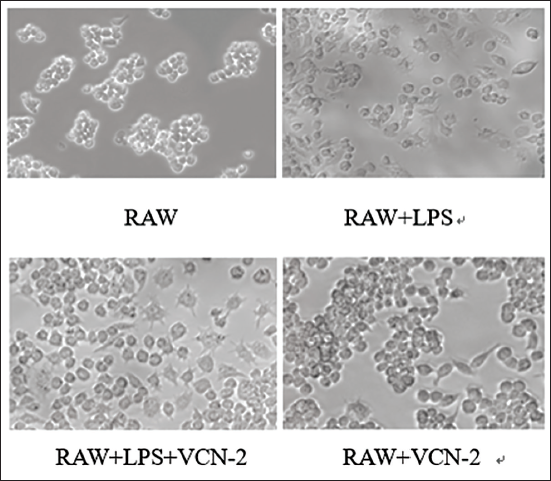
VCN-2 Enhances Phagocytic Capacity in Resting Macrophages and Attenuates It in Inflammatory Models
The results showed that the LPS induction (RAW+LPS group) decreased the pinocytosis ability of RAW264.7 cells compared to that in the RAW group (Figure 3, p < .01). However, VCN-2 treatment (RAW+LPS+VCN-2 group) showed significantly higher pinocytosis ability compared to that in LPS-induced RAW264.7 cells (RAW+LPS group) (Figure 3, p < .05). Therefore, VCN-2 could remove harmful substances in LPS-induced RAW264.7 cells by promoting the pinocytosis ability.
VCN-2 Exhibits Bidirectional Regulation of Cytokine Expression
Both the RT-PCR assay and the enzyme-linked immunosorbent assay (ELISA) assay indicated that VCN-2 treatment (RAW+VCN-2 group) significantly upregulated IL-6 gene transcription and IL-6 levels in RAW264.7 cells (Figure 4B, p < .01). While, TNF-α gene transcription (Figure 4C, p < .0001) and TNF-α levels in RAW264.7 cells (Figure 4D, p < .01) were remarkably upregulated in RAW264.7 cells of the RAW+VCN-2 group compared to those in the RAW group. Therefore, VCN-2 could play immune-enhancing role in RAW264.7 cells through upregulating both IL-6 and TNF-α expression.
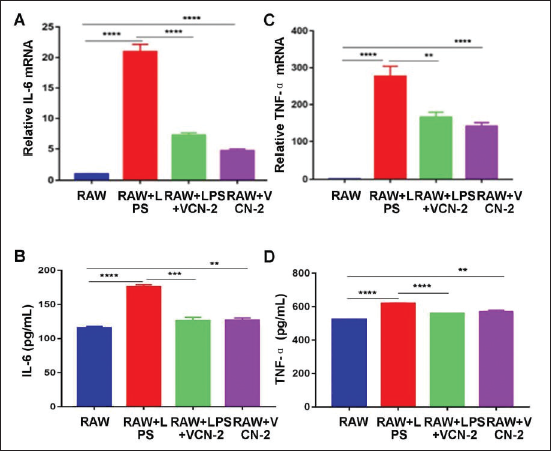
Moreover, VCN-2 treatment obviously downregulated IL-6 gene transcription (Figure 4A, p < .0001) and IL-6 levels (Figure 4B, p < .001) in RAW264.7 cells than those in the RAW+LPS+VCN-2 group. Meanwhile, the TNF-α gene transcription (Figure 4C, p < .01) and TNF-α levels (Figure 4D, p < .0001) in RAW264.7 cells were predominantly downregulated in the RAW+LPS+VCN-2 group than those in the RAW+LPS group. These results suggest that VCN-2 could play an anti-inflammatory role in LPS-induced RAW264.7 cells through down-regulating both IL-6 and TNF-α expression.
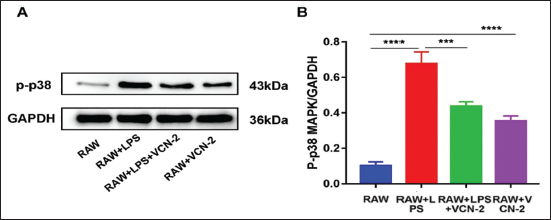
VCN-2 Regulates the Activation of p38MAPK in RAW264.7 Cells
p38 is a member of the MAPK family and a key protein in the MAPK signaling pathway. The expression of phosphorylated p38 was analyzed by WB detection. As shown in Figure 1, compared with the RAW group, the p-p38 protein expression in the RAW+LPS group was significantly higher than that in the RAW group (Figure 5A and 5B, p < .0001), indicating that LPS promotes the activation, that is, phosphorylation, of the p38 protein in RAW264.7 cells. Compared with the RAW group, the expression of p-p38 in the RAW+VCN-2 group was significantly increased (Figure 5A and 5B, p < .0001). The results showed that VCN-2 upregulated the expression of p-p38 in RAW264.7 cells in the resting state. The average values of p-p38/GAPDH in the RAW+LPS group and the RAW+VCN-2 group were 0.676 and 0.353, respectively. Compared with the RAW+LPS group, the RAW+LPS+VCN-2 group significantly reduced the expression of p-p38 in RAW264.7 cells (Figure 5A and 5B, p < .001), that is, VCN-2 down-regulated the phosphorylation of p38 in RAW264.7 cells induced by LPS.
Discussion
Many researchers underscore the pivotal role of macrophages in immune regulation. The phagocytic capability of macrophages is a direct indicator of the body’s immune strength. Macrophages release toxic molecules like ROS, which can induce intracellular oxidation and enzyme degradation, thereby exerting cytotoxic effects. NO, another crucial macrophage secretion, not only maintains the macrophages’ stable immune status but also participates in intricate immune activities alongside other immune cells. Macrophages secrete a variety of cytokines, including TNF-α, IL-6, and IL-1β, which are integral to immune signaling, regulation, and the inflammatory response (Gordon, 2007). This study demonstrates that VCN-2, a flavonoid, exerts bidirectional immunomodulatory effects on RAW264.7 macrophages by modulating p38MAPK phosphorylation, offering potential therapeutic applications for inflammatory diseases such as psoriasis, rheumatoid arthritis, and inflammatory bowel disease. At a concentration of 20 µg/mL, VCN-2 enhanced the proliferation rate of RAW264.7 cells to 113.59%, signifying its ability to activate these cells at low concentrations and to induce proliferation. A dose-dependent increase in cell proliferation was observed within the 20–120 µg/mL concentration range, indicating that VCN-2 stimulates macrophage immune function by boosting cell proliferation and activity. Using VCN-2 at a concentration of 80 µg/mL to construct a RAW264.7 cell drug model revealed that VCN-2 administration increases cell count and volume and enhances cell proliferation, thereby augmenting the immune function of RAW264.7 cells. Furthermore, VCN-2’s addition in the presence of LPS directly influences RAW264.7 cells, aiding in cell morphology repair, cell membrane restoration, and proliferation recovery. These actions collectively contribute to reduced immune stress and manifest anti-inflammatory effects.
The phagocytic capacity of macrophages is a direct reflection of the body’s immune status. Our experiments focused on the impact of VCN-2 on the phagocytosis of neutral red by RAW264.7 cells. The results demonstrate that VCN-2 significantly enhances the phagocytic ability of these cells. This finding suggests that VCN-2 may bolster immune function by augmenting macrophage phagocytosis. Upon stimulation by LPS, RAW264.7 cells can differentiate into M1 macrophages, characterized by a potent pro-inflammatory response. However, excessive phagocytosis by M1 macrophages can lead to self-damage (Kim et al., 2007). Our findings indicate that VCN-2 attenuates this overactive phagocytic activity in the inflammation model. This suggests that VCN-2 can mitigate LPS-induced inflammatory stimulation and immune stress, thereby reducing cellular damage. Macrophages are known to activate multiple signaling pathways, such as nuclear factor-kappa B (NF-κB), MAPKs, transforming growth factor-β (TGF-β), and others for phagocytosis by binding to surface receptors like Toll-like receptors (TLRs) (Chen et al., 2021). Xin et al. (2019) found that ginsenoside Rb1 enhances macrophage phagocytosis via the p38/Akt pathway, and that the inhibition of p38 can reverse this effect. Correlating these findings with our WB results, we hypothesize that the modulation of macrophage phagocytosis by VCN-2 might be associated with the level of p38 phosphorylation. VCN-2’s opposing effects on ROS levels reflect its context-dependent role. In resting cells, VCN-2 moderately increases ROS, likely enhancing immune surveillance and phagocytosis, as low-level ROS acts as a signaling molecule (Lehnert & Iyer, 2002). In LPS-induced cells, VCN-2 reduces excessive ROS, mitigating oxidative stress and inflammatory damage, possibly via p38MAPK inhibition. This dual regulation suggests VCN-2 maintains immune homeostasis, making it a candidate for diseases with oxidative stress components, such as skin photoaging or chronic inflammation.
NO, an endogenous, soluble gas, plays multifaceted roles in biological processes. In the immune system, NO production is triggered in macrophages upon exposure to pathogens and pro-inflammatory factors. This NO production can further stimulate macrophages to secrete cytokines and generate superoxide anions, effectively inhibiting and neutralizing pathogenic microorganisms during the early innate immune responses (Macmicking et al., 1997). NO’s role extends to controlling the metabolic reprogramming of macrophages, proving it is critical for adaptive immune responses (Palmieri et al., 2020). Macrophages can also kill target cells through the NO and other molecules they produce, thereby producing a variety of biologically active substances to exert their immunoregulatory effects. The immune regulatory function of macrophages is enhanced after activation, but excessive activation can cause inflammatory diseases and damage the body. NO is synthesized by NOS, with iNOS being a prominent form in immune responses (Xue et al., 2018). Various cells can produce iNOS, which is calcium-independent and inducible by cytokines or other stimuli. Macrophage activation leads to increased iNOS production, which, in turn, enhances NO secretion and expression. iNOS expression in macrophages is pivotal in regulating the differentiation and function of immune cells through key transcription and activation signaling molecules. A variety of cells can produce calcium-independent iNOS following induction by cytokines or other stimuli. Activation of macrophages can also induce the production of iNOS, thereby promoting the secretion and expression of NO. Studies have shown that macrophages express iNOS and regulate the differentiation and function of immune cells by participating in key molecules in transcription or activation signaling pathways (Mao et al., 2013). A study in an experimental model of endotoxic shock showed that iNOS deficiency inhibited macrophage activation, leading to more severe inflammation, suggesting that macrophage-derived iNOS selectively regulates macrophage differentiation. Studies have also shown that iNOS contributes to macrophage-mediated regulation of pro-inflammatory cytokines (Giordano et al., 2011). In conclusion, the expression of iNOS in macrophages selectively regulates macrophage differentiation, thereby controlling the innate immune response and playing an important role in the dynamic balance of innate immune regulation. Our study reveals that VCN-2 elevates NO levels in resting RAW264.7 cells, with a concurrent increase in iNOS mRNA expression. This indicates that VCN-2 boosts iNOS gene expression, thereby augmenting NO secretion. Such stimulation enhances macrophage immune functions, including inhibitory and cytotoxic effects against various pathogens. Notably, the NO levels induced by VCN-2 are significantly lower than those in inflammatory models, reducing the likelihood of excessive inflammatory responses. In LPS-induced inflammatory cells, VCN-2 notably downregulates iNOS gene levels and reduces NO content, thereby mitigating the damage caused by inflammatory reactions. These findings suggest that VCN-2 exerts a bidirectional immunomodulatory effect.
ROS have been extensively studied for their biological roles in both normal physiological processes and various diseases. Current understanding recognizes ROS as a “double-edged sword” due to its dose-dependent effects. At moderate levels, ROS are essential for cell proliferation and function. However, excessive ROS can lead to an overwhelming oxidative stress response, damaging intracellular proteins, lipids, and nucleic acids, and potentially inducing tissue and organ damage, apoptosis, or necrosis. Particularly in inflammatory responses, macrophages produce substantial amounts of ROS, which can trigger the release of pro-inflammatory cytokines (Scortegagna et al., 2003). Our study reveals that VCN-2 enhances ROS production in resting RAW264.7 cells, albeit at levels significantly lower than those observed in inflammatory models. This indicates a potential up-regulatory effect of VCN-2 on immune responses. Conversely, VCN-2 significantly inhibits excessive ROS production in LPS-stimulated RAW264.7 cells, suggesting a protective role against oxidative stress and subsequent inflammatory responses.
TNF-α is recognized for its bidirectional regulatory effects on inflammation, varying according to different microenvironments within the body (Zelová & Hošek, 2013). In a healthy state, an appropriate secretion of TNF-α mediates immune responses and enhances macrophage immune activity, contributing to the overall immune function. However, excessive release of TNF-α can provoke the production of inflammatory mediators and oxygen radicals, exacerbating inflammatory responses (Salomon et al., 2018). Similarly, IL-6, a pleiotropic cytokine, plays a critical role in regulating acquired immunity. It influences the production of certain complement components, drives the differentiation of activated B cells into various immunoglobulins, and guides T-cell differentiation towards function-specific lineages (Hirano, 2014; Mihara et al., 2012). In summary, TNF-α and IL-6 regulate immunity through diverse mechanisms, facilitating macrophage activation and antigen presentation. Nevertheless, overactive immune responses can precipitate inflammatory diseases and bodily harm. Severe infections can stimulate macrophages to produce high levels of IL-6, IL-1, and TNF-α in response to LPS exposure. These cytokines, in concert with excess NO, activate leukocytes and endothelial cells, leading to the release of oxygen radicals, proteases, and other inflammatory mediators. This cascade can culminate in sepsis, illustrating the critical role of LPS in severe infections, shock, and mortality (Rahim et al., 2019). Our study demonstrates that VCN-2 upregulates the gene transcription levels of TNF-α and IL-6 in resting RAW264.7 cells, enhancing the production of these cytokines, with a more pronounced effect on IL-6. This suggests that VCN-2 elevates cellular immunity in RAW264.7 cells, modulating immune function. In LPS-induced cells, VCN-2 significantly alters cytokine secretion. Excessive cytokine expression under LPS stimulation leads to an inflammatory stress response. VCN-2 effectively downregulates the transcription levels of TNF-α and IL-6, mitigating TNF-α and IL-6 production. Although cytokine levels do not return to baseline, the reduction is sufficient to alleviate inflammatory damage, indicating that VCN-2 regulates immunity through its anti-inflammatory effects.
This study comprehensively demonstrates that VCN-2 exerts a bidirectional regulatory effect on macrophage immune function under varying conditions. Specifically, VCN-2 enhances RAW264.7 cell proliferation, improves cell morphology, and amplifies phagocytic capacity. Additionally, it increases the production and gene expression levels of NO, ROS, iNOS mRNA, and cytokines such as TNF-α and IL-6. In inflammatory models, VCN-2 exhibits a protective role by diminishing the levels and gene expression of ROS, NO, iNOS mRNA, TNF-α, and IL-6. This reduction correlates with a decreased apoptotic impact of inflammation on macrophages, thus helping to maintain cellular homeostasis and exerting immunoregulatory functions. The dual regulatory capabilities of VCN-2 on macrophage immunity highlight its potential as a therapeutic agent. The findings provide a solid theoretical foundation for the further development, utilization, and clinical application of VCN-2 in immunomodulatory therapies. The dual regulatory capabilities of VCN-2 highlight its potential as a therapeutic agent for diseases like psoriasis, rheumatoid arthritis, and inflammatory bowel disease, where macrophage dysregulation is central.
p38, a key member of the MAPK family, is integral to normal immune and inflammatory responses. The p38MAPK pathway is known to be activated by various stimuli, ranging from strong inducers like oxidative stress (Coulthard et al., 2009), UV (Jinlian et al., 2007), and pressure (Shiratsuchi & Basson, 2005), to milder triggers such as heat stress (Park et al., 2005) and plant-derived immunomodulatory compounds (Li et al., 2018; Xu et al., 2016). VCN-2 modulates p38 phosphorylation context-dependently: upregulating p-p38 in resting cells to enhance immune function and downregulating it in LPS-induced cells to suppress inflammation. This likely involves VCN-2’s interaction with TLR4 or upstream kinases, as seen with other flavonoids (Feng et al., 2020). Activation of p38 leads to its phosphorylation, facilitating the activation of a cascade of transcription factors, enzymes, and signaling proteins that are crucial for regulating macrophage immunity. We have demonstrated that VCN-2 can inhibit the expression of p38 in vitro, countering the inflammatory response induced by UVB radiation (Duan et al., 2019). Lee et al. (2015) reported that VCN-2 inhibits the activation of TLR4, subsequently reducing p38 phosphorylation, diminishing macrophage inflammatory responses, and exerting anti-inflammatory effects. The involvement of the p38MAPK pathway in the immunostimulatory impact of VCN-2 on resting macrophages, however, remains to be fully elucidated. Studies have shown that p38 plays a role in regulating stress-responsive cell functions. For example, mild heat stress can activate p38, promoting cytokine secretion by macrophages and enhancing their defensive and regulatory capabilities (Huang et al., 2009; Park et al., 2005). Li et al. (2018) found that the phosphorylation of p38 and c-Jun N-terminal kinase (JNK) in the MAPK pathway is involved in the bidirectional immunomodulatory effect of Astragalus total flavonoids on RAW264.7 cells. In exploring the mechanism of VCN-2’s bidirectional immunoregulatory effect, this study focused on the phosphorylation levels of p38. Results indicate that VCN-2 can appropriately upregulate the expression of p-p38 in resting RAW264.7 cells, yet significantly downregulate the p-p38 expression in LPS-induced RAW264.7 cells. This suggests that VCN-2 may regulate the transcription levels of iNOS, IL-6, and TNF-α mRNA by modulating the phosphorylation level of p38, thus influencing the secretion of NO, IL-6, TNF-α, and ROS in RAW264.7 cells under different conditions. Consequently, this study reveals a potential link between VCN-2 and the p38MAPK signaling pathway, contributing to its immunomodulatory role.
Future studies will validate these findings in vivo using mouse models of skin inflammation (e.g., imiquimod-induced psoriasis) and systemic inflammation (e.g., LPS-induced sepsis) to assess VCN-2’s effects on macrophage polarization and cytokine levels. Chronic administration studies (4–12 weeks) will evaluate safety and long-term immunomodulatory effects. Additionally, p38MAPK inhibitors (e.g., SB203580) will be used to confirm the pathway’s role, enhancing the translational potential of VCN-2 for clinical applications.
Footnotes
Abbreviations
IL-6: Interleukin-6; iNOS: Inducible nitric oxide synthase; LPS: Lipopolysaccharide; MAPKs: Mitogen-activated protein kinases; NF-κB: Nuclear factor-kappa B; NO: Nitric oxide; NOS: Nitric oxide synthase; ROS: Reactive oxygen species; TGF-β: Transforming growth factor-β; TLR: Toll-like receptor; TNF-α: Tumor necrosis factor-α; UV: Ultraviolet ray; UVB: Ultraviolet B rays; VCN-2: Vicenin-2; WB: Western blot.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB). The participant has provided informed consent for the submission of the article to the journal.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Sichuan Medical Youth Innovation Research Project of Sichuan Medical Association (Q23001), PhD Start-up Fund Project of North Sichuan Medical College (CBY23-QDA17), and National Natural Science Foundation Youth Project (82404172).
