Abstract
Background
Asthma is a chronic respiratory disease featured as destruction of epithelial tight junctions. In order to protect the functional integrity of airway epithelial cells, more active and effective prevention and treatment should be sought.
Objectives
This study explored the mechanism by which ligustrazine affects vascular endothelial growth factor (VEGF) on the impairment of airway epithelial function in asthmatic mice through the Janus kinase/signal transducer and activator of transcription 5 (JAK/STAT5) signaling pathway.
Materials and Methods
Asthma model mice were randomly assigned to the model group, tetramethylpyrazine (TMP) low-dose group, medium-dose group, high-dose group, TMP high-dose+α7 nAchR group, TMP high-dose+ruxolitinib group, and TMP high-dose group (through blind selection)+mersalyl group, TMP high-dose+SU56141 group, α7 nAchR+SU56141 group, ruxolitinib+SU56141 group, TMP high-dose+α7 nAchR+SU56141 group. Five animals in each group were given different doses of TMP. Airway ventilation resistance, transepithelial resistance and conductance, reactive oxygen species content, and gene expression of mice in each group were detected; JAK, STAT5, VEGF protein expression, ZO-1 and E-cadherin expression, and hematoxylin and eosin staining were observed.
Results
Ligustrazine can improve the damaged airway epithelial function of asthmatic mice with the involvement of JAK/STAT5 signaling. Further studies found that inhibiting JAK/STAT5 signaling can improve the damaged airway epithelial function of asthmatic mice and is related to VEGF. After using mersalyl and SU56141 for stimulation and inhibition, it was found that inhibiting VEGF can reduce the epithelial inflammatory response induced by asthma and help improve the damaged airway epithelial function. Finally, it was verified that ligustrazine can inhibit the JAK/STAT5 signaling pathway, reduce VEGF expression, and improve the damaged airway epithelial function in asthmatic mice.
Conclusion
High doses of TMP can reduce inflammatory factors and infiltration in mouse airway epithelial tissue and improve tissue structure. In this process, the activity of the JAK/STAT5 signaling pathway is inhibited and VEGF expression is decreased, thus alleviating the impairment of airway epithelial function in mice. This discovery provides new ideas and directions for the treatment of asthma.
Keywords
Introduction
Asthma is a chronic respiratory disease. Its main manifestation is the destruction of epithelial tight junctions caused by damage to airway epithelial function, resulting in the occurrence of inflammatory reactions, thus triggering asthma. Its pathogenesis involves a variety of cytokines and signaling pathways. Therefore, in order to protect the functional integrity of airway epithelial cells, more active and effective prevention and treatment of asthma should be sought (Chowdhury et al., 2021).
Ligustrazine is an extract of the traditional Chinese medicine Ligusticum chuanxiong. It can not only inhibit platelet aggregation and reduce thrombus but can also be used to expand blood vessels and improve microcirculation. In addition, it can also reduce the viscosity of whole blood and inhibit myocardial contraction (Luo et al., 2023). Research by Feng et al. (2023) suggests that ligustrazine may regulate immune and inflammatory responses by inhibiting the production of endothelin (ET) and tumor necrosis factor (TNF), and it can improve chronic obstructive pulmonary disease to a certain extent. It can also improve the patient’s vascular fibrinolytic function, reduce blood viscosity, and prevent the formation of thrombus, thereby alleviating the patient’s symptoms such as dyspnea (Shao et al., 2021). In addition, some studies have shown that tetramethylpyrazine (TMP) can not only regulate Ca2+, CaM, and CaMKⅡβ and inhibit central nervous system excitation, thereby exerting a sedative effect, but it can also exert anti-inflammatory effects, thereby reducing inflammation in pulmonary fibrosis (Li, Li et al., 2022; Li, Zhao et al., 2022). However, can ligustrazine also have a certain control effect on the occurrence of asthma? And how does it affect airway epithelial function needs to be explored further.
Studies by Wu et al. (2022) suggest that the Janus kinase/signal transducer and activator of transcription 5 (JAK/STAT5) signaling pathway can activate inflammatory factors, thereby inducing the activation of inflammatory cells and further aggravating airway epithelial damage. At the same time, relevant studies have found that ligustrazine reduces the expression of Th2 cytokines by inhibiting JAK/STAT5 signaling, thereby alleviating airway inflammation and improving the lung function of patients. In addition, it inhibits the JAK/STAT5 signaling pathway. The activation of the pathway downregulates apoptosis-related genes such as Bax and Bak, thereby inhibiting the occurrence of cell apoptosis and playing an important role in improving respiratory diseases (Luo, Liu et al., 2022; Wu et al., 2022). However, current research is unclear as to whether ligustrazine can play a certain role in epithelial function in the process of asthma by regulating the JAK/STAT5 signaling pathway.
According to reports by Zhou et al. (2022), vascular endothelial growth factor (VEGF) promotes angiogenesis by activating MAPK, PI3K, and others, and is involved in respiratory diseases. At the same time, high expression of VEGF can not only induce the proliferation of airway epithelial cells but can also cause certain changes in the structure of airway epithelial cells, aggravating the course of asthma to a certain extent. Dai et al. (2022) found that activation of JAK/STAT5 signaling promoted VEGF expression to a certain extent, causing the proliferation of airway epithelial cells and further participating in the pathogenesis of asthma. However, whether ligustrazine can affect VEGF through the JAK/STAT5 signaling pathway and participate in airway epithelial function in asthma requires further investigation. Therefore, this study constructed an asthma mouse model to deeply explore the mechanism of ligustrazine on airway epithelial function in asthma, aiming to provide a basis for treatment.
Materials and Methods
Reagents, Instruments, and Animals
Ligustrazine (purity 98%; batch number: 1124-11-4; purchased from Beijing Kezhan Biotechnology Ltd.) is extracted from the plant L. chuanxiong and has various pharmacological effects such as lowering blood pressure, improving cerebral ischemia, and anti-platelet aggregation. cDNA synthesis kit (Shanghai Renjie Biotechnology Ltd.); RT-polymerase chain reaction (PCR) primers, reverse transcription (RT) kit (Shanghai Yiji), purchased from Beijing Ingen Trading Ltd.; reverse transcription reagent kit (Guangzhou Xiangbo Biotechnology Ltd.); primary and secondary antibodies (Heilongjiang Jiufeng Bioengineering Ltd.); phosphate-buffered saline (PBS) buffer (Vander Biotechnology Co., Ltd.); enzyme-linked immunosorbent assay (ELISA) kit (Shanghai Ruifan Biotechnology Ltd.), α7 nAchR (Shanghai Kanglang Biotechnology Ltd.); ruxolitinib (Shanghai Fei Biotechnology Ltd.).
Animals: 60 Sprague-Dawley (SD) mice, male, purchased from Shanghai Lianmai Bioengineering Ltd.
Method
Construction of Asthma Mouse Model
Sixty SD mice were selected, and five were selected as the control group. After raising for 7 days, 200 µL of ovalbumin sensitizer was intraperitoneally injected for sensitization on days 0, 7, 14, and 21 (Kokubo et al., 2022; Luo, Hu et al., 2022). On days 28–32, 50 µL of stimulant per animal per day was instilled through the nose (stimulant: 0.204 mg of ovalbumin was weighed and dissolved in 4.08 mL of normal saline to prepare a 5% ovalbumin solution). 24 hours after the last challenge (day 33), part of the lung tissue of one mouse was taken and sliced for hematoxylin and eosin (HE) staining to observe lung pathological changes. At the same time, the respiratory frequency and depth of the mouse were observed. If the respiratory rate is >100 times/min, it can be determined as asthma.
Group Intervention
Asthma model mice were randomly assigned into model groups: TMP low-, TMP medium-, TMP high-dose group, TMP high-dose+α7 nAchR group, TMP high-dose+ruxolitinib group, TMP high-dose+mersalyl group, TMP high-dose+SU56141 group, α7 nAchR+SU56141 group, ruxolitinib+SU56141 group, TMP high-dose+α7 nAchR+SU56141 group; five animals in each group. Among them, the TMP low-, medium-, and high-dose groups were given 5, 10, and 15 mg/kg TMP by intragastric administration, respectively, and the remaining drugs were administered by intraperitoneal injection (3 mg/kg).
Measurement of Airway Ventilation Resistance
When measuring the airway ventilation resistance of mice, the mice were anesthetized by intraperitoneal injection with sodium pentobarbital solution (4 µL/g), then intubated into the trachea and connected to a ventilator. The airway airflow and pressure were measured under the instrument, and the lung ventilation resistance value of the mouse was calculated (LR = ∆P/∆F). When the mouse’s breathing was stable, the basic value was recorded for 3 min and then atomized with Ach solution with concentrations of 0, 3.125, 6.250, 12.500, 25.000, and 50.000 mg/m3. The relevant data on the airway hyperresponsiveness (AHR) of the mouse were collected. AHR was analyzed.
Epithelial Voltmeter to Measure Transepithelial Resistance (TEER) and Conductance (TRITC)
Mouse bronchoalveolar lavage fluid (BALF) was collected, and cells in BALF were seeded and cultured, followed by the removal of the medium for polarization at air–liquid interface (ALI). TEER was measured using an epithelial voltmeter, and TRITC was calculated as the reciprocal of TEER (1/Ω × cm2).
ELISA
After diluting the obtained sample of mouse serum, equal amounts were added to the enzyme-labeled plate. It was then incubated under constant temperature conditions to fully combine and then washed. Further, enzyme-labeled antibodies were added to incubate; color development was performed after washing; and the reaction was terminated. The levels of pro-inflammatory factors and caspase-3 were calculated through the standard curve.
Flow Cytometry to Detect Changes in Reactive Oxygen Species (ROS)
Mouse sample tissue cells were collected and combined with DCFH-DA for staining (Lv et al., 2022); and a fixative was used to fix the cells. Then, they were rinsed with PBS and centrifuged to collect the cells. Finally, the cell samples were loaded and tested using a flow cytometer (Xiamen Fuliu Biotechnology Ltd.) to detect the ROS level in each cell.
Western Blot Detection of JAK, STAT5, and VEGF Protein Expression
Protein expression was detected by the Western blot method (Belk et al., 2022; Gorvin et al., 2023; Parra-Izquierdo et al., 2022; Raza & Clarke, 2021; Rosa et al., 2023). After preparing the gel, it was incubated through loading, electrophoresis, transfer, and blocking. Then, primary antibodies JAK (ab120951, 1:1,000) and STAT5 (ab32364, 1:1,000), VEGF (ab32152, 1:1,000) and secondary antibody (goat anti-rabbit 1:2,000) for incubation. Using GAPDH as a reference, the blot was analyzed under an enhanced chemiluminescent reagent.
HE Staining to Observe Pathological Tissue of Mouse Lungs
The sample tissues were taken out of the mice after sacrifice. They were washed with physiological saline and blotted dry with filter paper. The airway tissue was embedded in paraffin, sliced into 5 µM slices, put into a dewaxing cylinder, and xylene was used to remove paraffin. Then, it was placed in a hematoxylin dyeing solution for dyeing treatment to decompose further and reverse the blue. Then, it was stained with eosin staining solution for 2 min and finally decomposed with 95% alcohol. The stained sections were observed under a microscope.
Immunohistochemical Analysis
The sample tissue was prepared into slices after being fixed, dehydrated, and embedded. The antigen was combined with the antigen retrieval solution and then blocked by peroxidase, and then added with a secondary antibody for incubation and observation. The reaction between the sample tissue and the sample is observed by displaying the reaction. The binding of antigen and antibody is then analyzed and recorded through a microscope after counterstaining and dehydration.
PCR Detection of VEGF, JAK, and STAT5 mRNA Levels
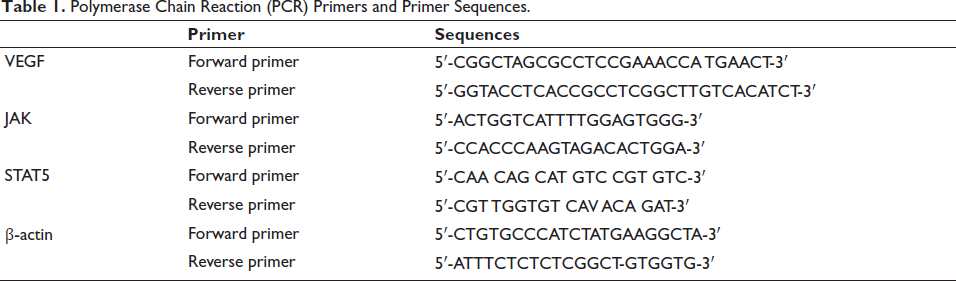
Lung cancer tissue was ground and homogenized with liquid nitrogen to extract total mRNA, and cDNA was reverse transcribed for analysis, with β-actin as a reference. 2−∇∇Ct method analyzed gene level (Livak & Schmittgen, 2001), and the primer sequences are listed in Table 1.
Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Statistical Analysis
The data obtained in each of the above experiments were analyzed using SPSS 21.0 and GraphPad Prism software. If there are no special requirements, p < .05 is used as the test standard.
Results
Successfully Constructed an Asthma Mouse Model
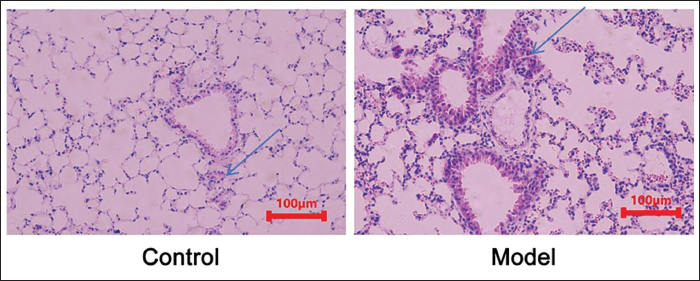
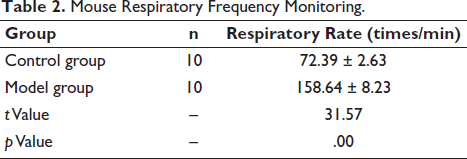
In the control group, there was no proliferation of mouse bronchial epithelial cells and no thickening of the tube wall. At the same time, there was no inflammatory cell infiltration around the bronchus. In the model group, inflammatory cell infiltration appeared, and the bronchial lumen was significantly narrowed and accompanied by wall thickening (Figure 1). The respiratory frequency of mice in the model group increased significantly, manifesting as shortness of breath (Table 2). This shows a successfully constructed asthma model.
Hematoxylin and Eosin (HE) Staining of Mouse Lung Tissue (×200).
Mouse Respiratory Frequency Monitoring.
Ligustrazine Can Improve the Damaged Airway Epithelial Function of Asthmatic Mice and JAK/STAT5 Signaling is Involved
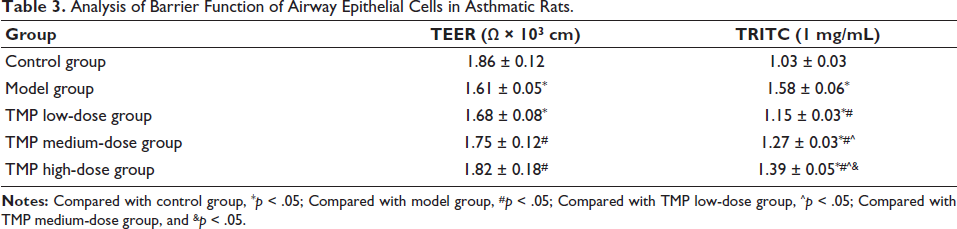
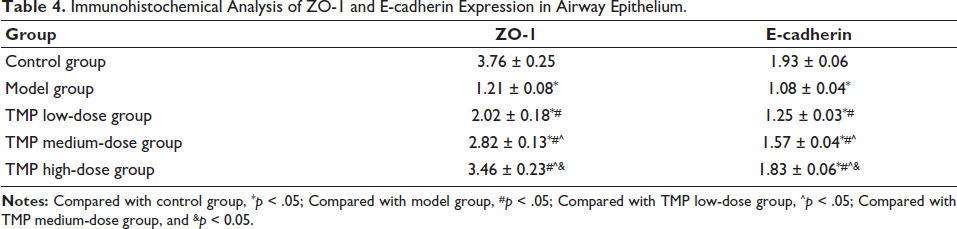
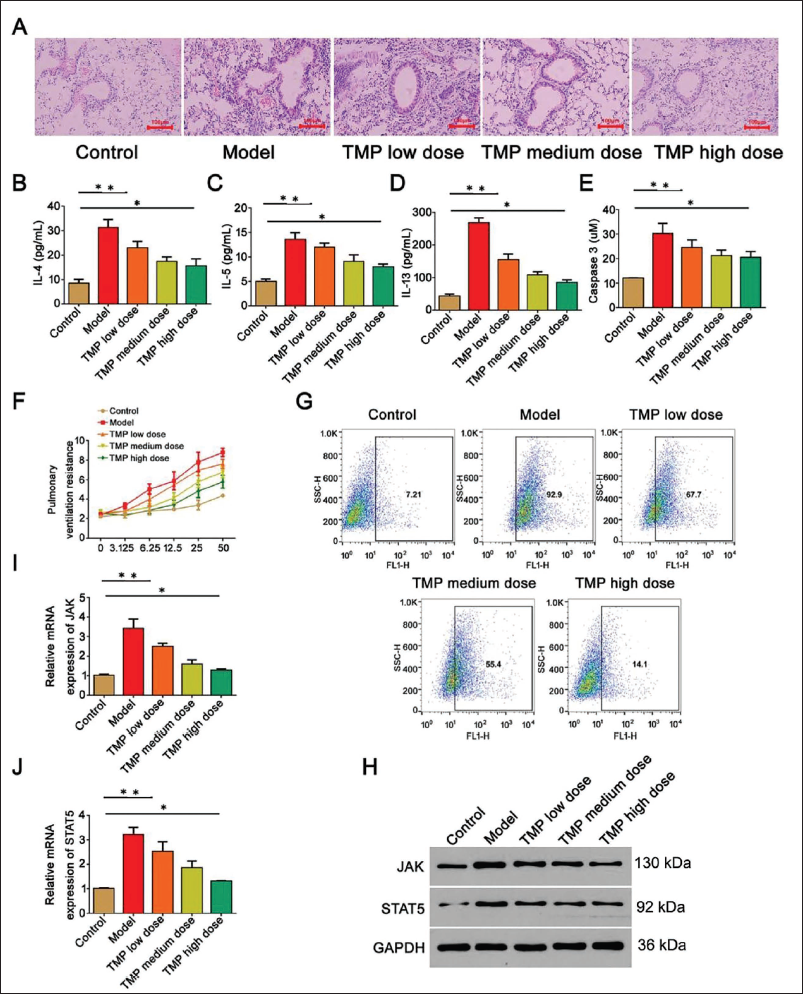
In terms of the barrier function of airway epithelial cells in asthmatic rats, TEER (1.82 ± 0.18) Ω × 103 cm and TRITC (1.39 ± 0.05) 1 mg/mL in the TMP high-dose group were better than model group; meanwhile, expression of ZO-1 (3.46 ± 0.23) and E-cadherin (1.83 ± 0.06) in TMP high-dose group was higher than other groups (Tables 3 and 4), which also illustrates the differences between cells. The connections are tight and have good integrity. Under HE staining of lung tissue, inflammatory infiltration of airway epithelial tissue cells in the TMP high-dose group was reduced, and the tissue structure was improved (Figure 2A). Meanwhile, in the measurement of lung ventilation resistance, the TMP high-dose group was lower than the model group (Figure 3F). Levels of IL-4/5/13, caspase-3, and ROS of the TMP high-dose group were lower than model group (Figure 2B–2E, 2G); from the perspective of gene protein expression, the TMP high-dose group showed lower expression of JAK and STAT5 than the model group (Figure 2H–2J). Therefore, ligustrazine has a certain effect on improving the damaged airway epithelial function of asthmatic mice.
Analysis of Barrier Function of Airway Epithelial Cells in Asthmatic Rats.
Immunohistochemical Analysis of ZO-1 and E-cadherin Expression in Airway Epithelium.
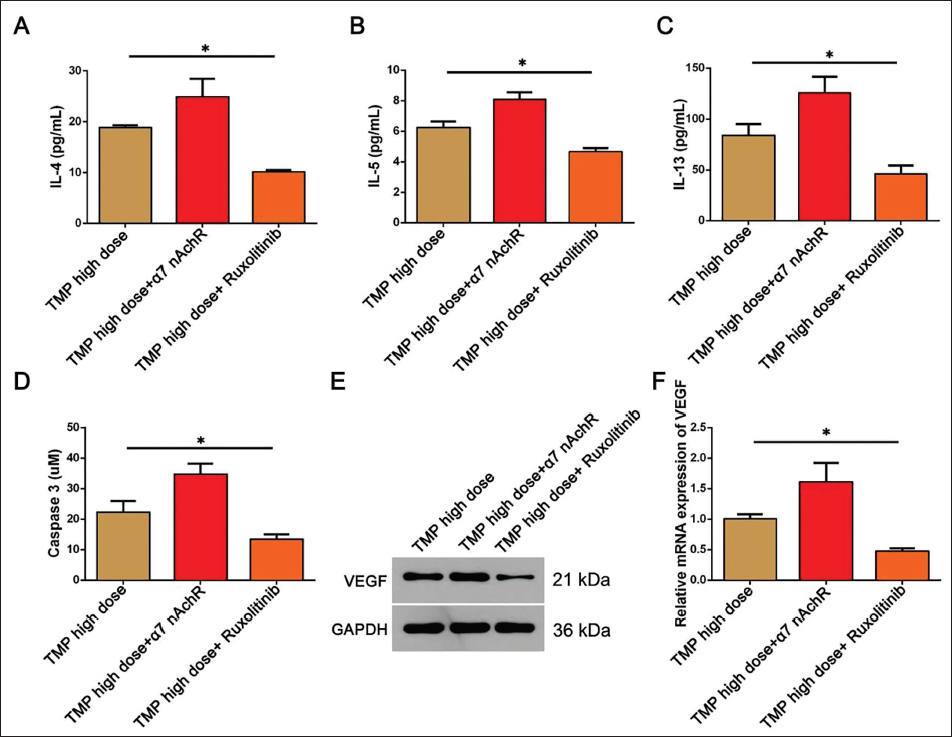
TMP Can Inhibit the JAK/STAT5 Signaling Pathway and Improve the Damaged Airway Epithelial Function in Asthmatic Mice, Which is Related to VEGF
To explore the specific role of JAK/STAT5 signaling in this process, α7 nAchR and ruxolitinib were used for agonism and inhibition on the basis of high-dose TMP. In immunohistochemical analysis, ZO-1 (3.72 ± 0.20) and E-cadherin (1.91 ± 0.08) expression was significantly increased (Table 5); meanwhile, the expression of IL-4/IL-5/IL-13, caspase-3, and VEGF gene proteins was significantly lower in other groups (Figure 3A–3F). Therefore, TMP can improve the damaged airway epithelial function of asthmatic mice by inhibiting JAK/STAT5 signaling, and the effect is further amplified after applying ruxolitinib inhibitor on the basis of TMP high-dose.
Immunohistochemical Analysis of ZO-1 and E-cadherin Expression.
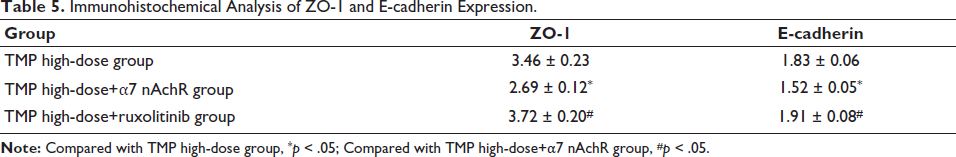
TMP Inhibits VEGF Expression Through JAK/STAT5 Signaling to Improve the Impaired Airway Epithelial Function in Asthmatic Mice
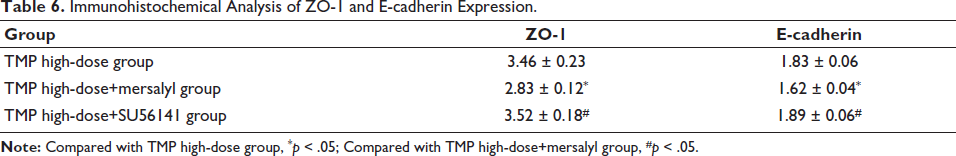
In order to assess the role of VEGF in the impairment of airway epithelial function, we used mersalyl and SU56141 for stimulation and inhibition and compared them with the TMP high-dose group. We found that the TMP high-dose+SU56141 group had an effect on ZO-1 (3.52 ± 0.18) and E-cadherin (1.89 ± 0.06) expression-promoting effect deepened, while the opposite was true in the TMP high-dose+mersalyl group (Table 6); at the same time, the level of IL-4/IL-5/IL-13 in TMP high-dose+SU56141 group was also lower than other groups (Figure 4A–4C). This also shows that inhibiting VEGF expression can reduce the epithelial inflammatory response induced by asthma and help improve the damaged airway epithelial function to a certain extent.
Immunohistochemical Analysis of ZO-1 and E-cadherin Expression.
Ligustrazine Can Improve the Damaged Airway Epithelial Function of Asthmatic Mice by Inhibiting the JAK/STAT5 Signaling Pathway and Reducing VEGF Expression
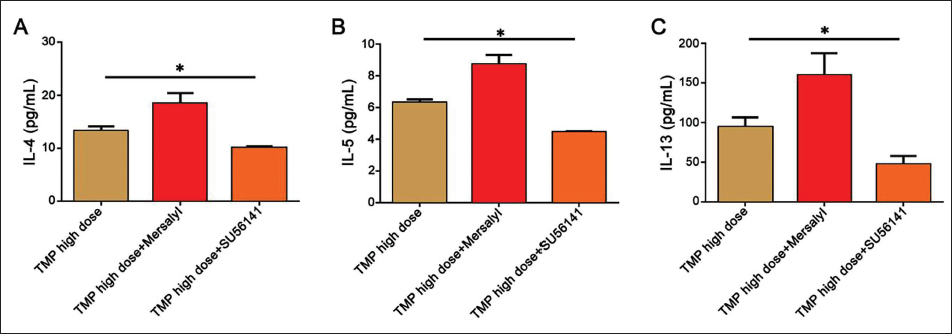
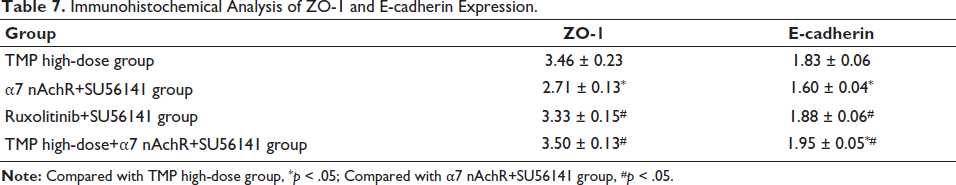
In order to verify the above results, asthma mice were further intervened through the cross-use method of α7 nAchR, ruxolitinib, and SU56141. It was found that the expression of ZO-1 (3.50 ± 0.13) and E-cadherin expression in the TMP high-dose+α7 nAchR+SU56141 group (1.95±0.05) was higher than α7 nAchR+SU56141 group and ruxolitinib+SU56141 group (Table 7); after HE staining, the inflammation in lung in the TMP high-dose+α7 nAchR+SU56141 group was significantly reduced (Figure 5A); and levels of IL-4/IL-5/IL-13, caspase-3, and ROS were reduced and the effect of TMP high-dose+α7 nAchR+SU56141 group was more significant (Figure 5B–5F). At the same time, the lung ventilation resistance of the TMP high-dose+α7 nAchR+SU56141 group was the lowest (Figure 5G). Therefore, this also shows that ligustrazine can improve the damaged airway epithelial function of asthmatic mice by inhibiting JAK/STAT5 signaling and reducing VEGF expression.
Immunohistochemical Analysis of ZO-1 and E-cadherin Expression.
Discussion
Previous studies have shown that TMP has endothelial protective functions and has potential therapeutic value for respiratory diseases (Lin et al., 2022). In acute lung injury (ALI), it can reduce inflammatory cell infiltration and alleviate lipopolysaccharide (LPS)-induced lung tissue damage (Min et al., 2022). TMP promotes nitric oxide production to a certain extent, thereby reducing ovalbumin-induced inflammation in rats. In addition, Wang et al. (2022) found through experiments on rats that TMP can improve the lung function of asthma rat models by inhibiting phosphodiesterase (PDE) activity and reducing serum IgE levels (Wang et al., 2022).
We constructed an asthma mouse model and found that the respiratory frequency of mice in the model group was increased, indicating that the asthma mouse model was successfully constructed. We used different doses of TMP to intervene and found that as the dose of TMP increased, the inflammatory infiltration of mouse airway epithelial tissue cells decreased, the tissue structure was significantly improved, IL-4/IL-5/IL-13 levels were reduced, and pulmonary ventilation resistance was significantly reduced. Caspase-3 and ROS levels were reduced, and TEER and TRITC were increased, showing the effect of improving the barrier function of airway epithelial cells. Caspase-3 is responsible for nuclear DNA fragmentation and is activated upon furin cleavage to allergen-induced release of DN from the airway epithelium, enhancing type 2 immunity in the respiratory tract. Michaeloudes et al (2022). found that in asthma, ROS levels are increased, leading to the occurrence of oxidative stress. Its overproduction activates AMPK, induces mitophagy, and stimulates M2 macrophage polarization in monocytes, thereby leading to asthma airway inflammation and remodeling. Research has found that ZO-1 is involved in the formation of tight junctions between cells. In asthma, reduced ZO-1 expression can indirectly lead to the destruction of tight junctions between cells and the impairment of barrier function (Zhou et al., 2021). Reducing the expression level of E-cadherin will not only weaken the adhesion between cells but also aggravate the damage to airway epithelium (Feng et al., 2022). Therefore, we tested the two and found significantly increased ZO-1 and E-cadherin under the intervention of TMP. In addition, we also found that JAK and STAT5 proteins were significantly inhibited under the intervention of TMP, which suggested that JAK/STAT5 signaling is involved in the improvement effect of TMP on asthma, in accordance with Th17 cells in neutrophilic asthma (Zhang et al., 2020). In terms of airway inflammation, Li pointed out that the JAK/STAT5 signaling pathway is inhibited by Ruxo, which can inhibit the survival of Th17 cells to a certain extent. At the same time, inhibiting JAK/STAT5 signaling can also reduce IFN-γ, alleviate airway inflammation, and reduce IL-4/IL-5 levels, thereby reducing activation and aggregation of inflammatory cells, which is beneficial to reducing airway inflammation response (Shi et al., 2017).
To assess the specific role of JAK/STAT5 signaling in the process of TMP improving the impairment of airway epithelial function caused by asthma, we used α7 nAchR and ruxolitinib to stimulate and inhibit it based on high-dose TMP. We found that under the co-intervention of high-dose TMP and ruxolitinib, ZO-1 and E-cadherin expression was significantly increased and levels of IL-4/IL-5/IL-13 and caspase-3 were reduced, while α7 nAchR reversed the effects of TMP to a certain extent. This shows that TMP can inhibit the JAK/STAT5 signaling pathway and improve the damaged airway epithelial function in asthmatic mice. In addition, we found that while inhibiting the JAK/STAT5 signaling pathway, VEGF protein was significantly reduced, suggesting that VEGF is involved in the process by which TMP inhibits the JAK/STAT5 signaling pathway and improves the damaged airway epithelial function in asthmatic mice.
VEGF plays an important role in inflammation (Díaz-Coránguez et al., 2020). VEGF has been reported to mediate allergic airway inflammation and promote airway epithelial cell proliferation. After binding to VEGFR2, it inhibits the phagocytosis of airway epithelial cells and promotes endothelial cell proliferation, thereby mediating airway inflammation in asthmatic mice (Mu et al., 2020). In one study, it was found that in vivo, VEGF-A was inhibited by WZB117 treatment, which reduced pulmonary edema, injury, neutrophils, and TNF-α levels in LPS-induced ALI. Inhibition of glucose transporter 1 (GLUT1) can downregulate the production of VEGF-A in the alveolar epithelium, thereby alleviating ALI (Liang et al., 2024). At the same time, other studies have shown that VEGF addition downregulated VE-cadherin, β-catenin, occludin, claudin, and ZO-1 proteins in endothelial cells, thereby increasing vascular permeability and leading to the infiltration of inflammatory cells and the occurrence of immune responses (Türkeli et al., 2021).
We also included VEGF agonist mersalyl and inhibitor SU56141 in the experiment and used them simultaneously with high-dose TMP. We found that the addition of SU56141 made TMP inhibit inflammatory factors. The effect and promotion of ZO-1 and E-cadherin expression are deepened, while mersalyl is completely opposite to this. This also shows that inhibiting VEGF can reduce the epithelial inflammatory response induced by asthma and help improve the damaged airway epithelial function to a certain extent. This is because in asthma, neutralizing VEGF can prevent ovalbumin-induced asthma inflammation and reduce levels of IL-4/IL-5/IL-13, thereby reducing AHR. Our study also has some limitations, and we need to further study the performance of clinical samples.
In order to verify the above results, we cross-used α7 nAchR, ruxolitinib, and SU56141 to intervene in asthmatic mice and found that the levels of inflammatory factors were significantly reduced, lung tissue inflammation was significantly alleviated, pulmonary ventilation resistance was significantly reduced, and caspase-3 levels were also reduced considerably. And after adding high-dose TMP on this basis, this effect was further amplified. However, the effect of α7 nAchR and SU56141 was worse than that of high-dose TMP. This also verifies that TMP inhibits VEGF by inhibiting JAK/STAT5 signaling, thereby improving damage to airway epithelial function in asthmatic mice.
Conclusion
In summary, TMP can inhibit JAK/STAT5 signaling, thereby inhibiting VEGF expression and improving the functional damage of airway epithelium in asthmatic mice. This research can help improve the quality and health of asthma patients. However, due to the small sample size, further in-depth exploration needs to be conducted by increasing the sample size.
Footnotes
Abbreviations
ALI: Air–liquid interface; BALF: Bronchoalveolar lavage fluid; ELISA: Enzyme-linked immunosorbent assay; JAK/STAT5: Janus kinase/signal transducer and activator of transcription 5; ROS: Reactive oxygen species; VEGF: Vascular endothelial growth factor.
Acknowledgments
The authors gratefully acknowledge the General Hospital of Northern Theater Command for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethical Committee of the General Hospital of Northern Theater Command.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
No patients were included in this study and this is not applicable.
