Abstract
Background/Objectives
Osteoporosis is a prevalent metabolic bone disorder characterized by a progressive loss of bone density and structural deterioration, significantly affecting the geriatric population, particularly postmenopausal women. Predisposing conditions for osteoporosis include increased lifespan, early menarche, sedentary lifestyles, and dietary habits. Current treatment options for osteoporosis primarily involve antiresorptive and anabolic agents; however, their usage is often limited due to high costs and potential adverse effects. This study investigates the efficacy of Tribulosin, a phytochemical, as a cost-effective alternative to existing antiresorptive drugs in the treatment of osteoporosis.
Materials and Methods
An ovariectomy-induced osteoporosis rat model was established using Sprague Dawley rats, which were treated with Tribulosin for 16 consecutive weeks. The impact of Tribulosin on bone structural integrity was evaluated through measurements of femoral bone weight, length, bone mineral density, and biomechanical properties. Bone remodeling markers were assessed by quantifying serum estradiol levels, bone gamma-carboxyglutamic acid protein, and acid phosphatase using enzyme-linked immunosorbent assay kits. Additionally, serum concentrations of calcium, phosphorus, creatinine, and tartrate-resistant acid phosphatase were measured to analyze the protective effects of Tribulosin against fracture risk.
Results
The osteogenic activity of Tribulosin in ovariectomized rats was assessed by quantifying receptor activator of nuclear factor kappa-B ligand (RANKL), osteoprotegerin, and the transcription factors Osterix and Runt-related transcription factor 2. Moreover, the anti-inflammatory response induced by Tribulosin was evaluated by measuring tumor necrosis factor-alpha and interleukin-1, 1-beta, and 6 levels. The results demonstrated that Tribulosin significantly enhanced femoral bone structural integrity and increased femoral strength. Estradiol levels and serum biomarker levels associated with bone health were elevated following Tribulosin treatment, indicating the ability of Tribulosin to reduce fracture incidence. Tribulosin treatment significantly improved osteogenic marker proteins and attenuated the inflammatory response in ovariectomized rats.
Conclusion
In conclusion, it can be inferred from the results that Tribulosin is a potent curative agent for treating osteoporosis, exhibiting efficacy without side effects. Additional research is necessary to investigate its prolonged impact and the underlying processes influencing bone health.
Introduction
Osteoporosis is commonly recognized as a “silent disease” due to its insidious progression, which often lacks noticeable symptoms until a fracture occurs (Mohammed et al., 2019). This condition is characterized by a significant decrease in bone mineral density (BMD) and deterioration of bone microarchitecture, resulting in increased bone fragility and a heightened risk of fractures (Shrihari & Harpreet, 2024; Sözen et al., 2017). Throughout an individual’s lifespan, bones undergo a continuous remodeling process, where osteoclasts resorb old bone tissue and osteoblasts create new bones. This dynamic process is essential for maintaining mechanical strength and facilitating repair. However, when there is an imbalance in this remodeling activity, specifically when bone resorption outpaces bone formation, it can lead to the pathological changes associated with osteoporosis (Raisz, 2005; Tu et al., 2018). Histological examinations in those with osteoporosis reveal marked reductions in the thickness of trabecular bone, smaller osteons, and enlarged Haversian canals and marrow spaces. Often, this condition remains undiagnosed until it results in low-trauma fractures in vulnerable sites such as the hip, spine, and wrist, which can lead to significant medical complications and increased rates of hospitalization (Porter & Varacallo, 2021).
Osteoporosis is influenced by modifiable factors such as inadequate calcium intake, physical inactivity, vitamin D deficiency, and prolonged use of glucocorticoids (Askari et al., 2019). Non-modifiable etiologies include aging, gender, ethnicity, genetic predisposition, family history, premature menopause, amenorrhea, and delayed onset of menarche (Hannan et al., 2000; Kanis, 1994; Miller, 2016). Additionally, the condition may be exacerbated by prolonged immobilization and the use of certain medications (Alibasic et al., 2020). Osteoporosis can be classified into primary and secondary forms, depending on its underlying cause (Marcucci & Brandi, 2015; Salari et al., 2021). Primary osteoporosis includes type I—postmenopausal, affecting women postmenopause due to estrogen deficiency, leading to trabecular bone loss and increased risk of vertebral and distal radius fractures (Cosman et al., 2014), and type II—senile, occurring in individuals over 70, resulting from the aging of trabecular and cortical bones, often leading to pelvic and proximal femur fractures (Gallagher & Rapuri, 2003; Hannan et al., 2000). Secondary osteoporosis arises from external factors such as medical conditions, medications, and lifestyle choices that negatively impact bone density, including chronic diseases, hormonal imbalances, prolonged glucocorticoid use, and habits like smoking and alcohol consumption (Amarnath et al., 2023).
Osteoporosis is a globally predominant disorder, affecting approximately 200 million individuals, with postmenopausal women constituting the most vulnerable population (Hiligsmann et al., 2019; Keen & Reddivari, 2025; Yu et al., 2022). Osteoporosis continues to escalate alongside the aging individuals, contributing to substantial illness and death rates, particularly among postmenopausal women. Epidemiological data estimate that 10.2 million individuals aged 50 and above in the USA are affected, with projections suggesting a potential increase of more than 30% by 2030 (Özmen et al., 2024). Globally, countries bearing the most significant disease load associated with low BMD include India (25.59%), China (18.75%), the USA (8.35%), Japan (3.29%), and Germany (3.04%), as reflected in disability-adjusted life years and mortality rates (Shen et al., 2022).
Current osteoporosis therapies are broadly categorized into antiresorptive and anabolic agents (Tu et al., 2018). Antiresorptives, such as bisphosphonates, estrogens, and denosumab, inhibit bone resorption, while anabolic agents, such as teriparatide, stimulate bone formation (Camacho et al., 2016). Bisphosphonates are contraindicated in patients with renal impairment due to nephrotoxicity (Patel et al., 2023), and denosumab is not advised in individuals with end-stage renal disease or those on dialysis (Food and Drug Administration, 2015). Raloxifene is contraindicated in women with a history of venous thromboembolism (Evista, 2016). Duavee (conjugated estrogens/bazedoxifene) is used for prevention but carries risks like other estrogen-based therapies. The Women’s Health Initiative demonstrated that estrogen–progestin therapy lowers fracture risk but increases cardiovascular and breast cancer risks, making it unsuitable as a first-line treatment (Keen & Reddivari, 2025; Rossouw et al., 2002). Calcitonin is also not preferred first-line due to limited efficacy in preventing non-vertebral fractures (Anish & Nair, 2024; Trovas et al., 2002). Given the significant side effects and high costs associated with current osteoporosis treatments, there is a pressing need to develop a drug that is both cost-effective and free of adverse effects, particularly for managing osteoporosis in postmenopausal women. This study hypothesizes that Tribulosin, a phytochemical compound, can mitigate ovariectomy-induced osteoporosis in rats.
Materials and Methods
Animals
A total of 24 healthy adult female Sprague Dawley rats (200–250 g) were housed in an animal housing facility maintained with standard laboratory conditions with a temperature of 22 ± 3°C and humidity of 65%. 12 h light–dark cycle and a pathogenic environment were strictly maintained. Ethical committee approval was obtained for all the procedures performed in the present study.
Ovariectomy
A bilateral ovariectomy was conducted in accordance with the established protocol prescribed by Xiong et al. (2023) to establish an ovariectomy-induced osteoporotic rat model. The rats were anesthetized with 35 mg/kg of 3% pentobarbital sodium and positioned on a sterile surgical plate. The dorsal area was shaved and sterilized with 75% ethanol, followed by a 1-cm midline incision made under aseptic conditions. Both ovaries were ligated and removed, after which the incision was closed in layers. In the SHAM group, equal volumes of adipose tissue adjacent to the ovaries were removed instead of performing ovariectomy.
Treatment Regimen
Following a 1-week acclimation phase, the rats were randomly assigned to four groups: the SHAM operated group, the ovariectomy/ovariectomized (OVX) group, which underwent ovariectomy and received no treatment throughout the study, the OVX + low-dose Tribulosin group, which was treated orally with 10 mg/kg body weight of Tribulosin per day following ovariectomy, and the OVX + high-dose Tribulosin group, which received 20 mg/kg body weight of Tribulosin per day orally after ovariectomy. Treatments began 3 weeks after the ovariectomy and continued for a total of 16 weeks. After the experiment, the rats were reweighed and anesthetized with an intraperitoneal injection of a ketamine and xylazine solution. They were then sacrificed using thiopental at a dose of 100 mg/kg. The left femur was removed, preserved in physiological saline, and stored at –20°C for BMD assessment and biomechanical testing.
Assessment of Bone Structural Integrity
The femoral bone was extracted from experimental animals and analyzed for various structural and biomechanical parameters. The femur length and organ coefficient were assessed, with the latter calculated as the ratio of the femur’s wet weight to the total body weight. The bone’s wet weight was recorded, followed by a drying process in an oven to eliminate water content, after which the dry weight was measured.
BMD was measured using dual-energy X-ray absorptiometry (DEXA) in accordance with the manufacturer’s guidelines. Additionally, the biomechanical integrity of the femur was evaluated through a three-point bending flexural test method, as described by Gao et al. (2013). The maximum load-bearing capacity and maximum deflection of the femur were quantified in both control and experimental groups to assess differences in bone strength and flexibility.
Quantification of Bone Remodeling Biomarkers
Biomarkers of bone remodeling, estradiol, bone Gla protein (BGA), and acid phosphatase (ACP) were quantified in the experimental rats. Estradiol and BGA levels in the experimental animals were quantified with enzyme-linked immunosorbent assay (ELISA) kits procured from Cusabio and Elabscience. The acid phosphatase colorimetric assay kit was used to quantify the ACP levels in the experimental animals.
Assessment of Fracture Risk
The serum calcium levels in experimental animals were determined using a colorimetric assay kit obtained from Abnova, while the serum phosphorus, creatinine, and tartrate-resistant acid phosphatase (TRAP) levels were measured with a colorimetric assay kit purchased from Elabscience. All procedures were carried out following the guidelines outlined in the respective kits. The quantification of calcium was based on the formation of a blue-colored complex when free calcium in the sample interacts with phenolsulphonephthalein dye, with absorbance recorded at 612 nm, reflecting calcium concentration. Phosphorus levels were determined by reacting inorganic phosphorus with molybdic acid to form phosphomolybdic acid, which was then reduced to molybdenum blue using a reducing agent. Absorbance was measured at 660 nm. Creatinine levels were determined through a series of chemical reactions that produced a final, pink-colored compound, with its absorbance recorded at 515 nm. TRAP activity was assessed by ACP-mediated catalysis of a chromogenic agent in the kit, generating p-nitrophenol, which was measured at 405 nm.
Analysis of Osteogenic Activity
Osteogenic activity in osteoporosis induced untreated animals and those treated with Tribulosin was analyzed by quantifying significant proteins involved in bone reformation activity. The quantification of receptor activator of nuclear factor kappa-B ligand (RANKL), a promoter of osteoclast activity, and osteoprotegerin (OPG), a decoy receptor for RANKL, was performed in experimental animals using an ELISA kit procured from Abcam. Additionally, the key transcription factors involved in osteoblast differentiation, Runt-related transcription factor 2 (Runx2) and Osterix (Osx), were measured using an ELISA kit obtained from Elabscience. The experiment was conducted in triplicate following the manufacturer’s instructions.
Assessment of Anti-inflammatory Response
Anti-inflammatory response triggered by Tribulosin in ovariectomized rats was analyzed by quantifying the levels of tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6). The levels of the mentioned inflammatory cytokines were quantified using the ELISA kits procured from Abcam. The experiments were done according to the instructions provided in the kits.
Statistical Analysis
The data analysis and comparison were conducted using Statistical Package for the Social Sciences (SPSS) version 22.0 software. Continuous variables were presented as mean ± standard deviation (SD) and evaluated through one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test. A p value of less than .05 was considered indicative of statistical significance.
Results
Tribulosin Improves Bone Structural Integrity in Ovariectomized Rats
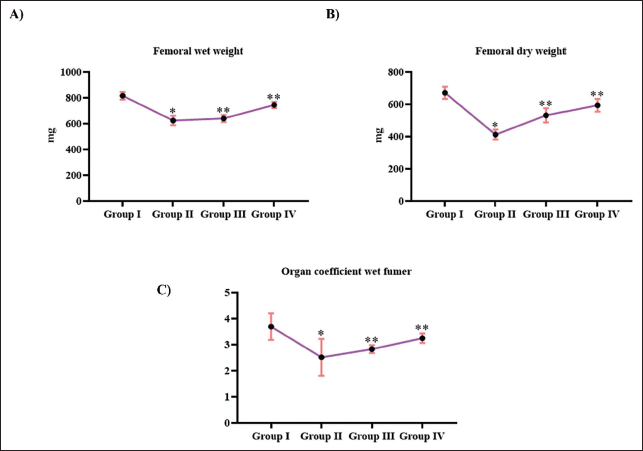
Figure 1 illustrates the bone weight of the ovariectomized, untreated, and Tribulosin-treated animals. The control and treated animals showed significant changes in both dry and wet weight of femur bones. Wet and dry weights of control animals’ femur bone were exhibited to be 810 ± 7 mg and 675 ± 7 mg, respectively. In contrast, ovariectomization in rats significantly decreased the femur bone weight to 584 ± 10 mg in the wet condition and to 410 ± 5 mg in the dried state. Treatment with Tribulosin prevented femur bone weight loss; the animals treated with 10 and 20 mg/kg Tribulosin exhibited 606 ± 11 mg and 725 ± 6 mg of wet weight and 520 ± 9 mg and 575 ± 11 mg, respectively. Tribulosin treatment increased the femur organ coefficient in ovariectomized rats. The treatment with 10 mg/kg of Tribulosin showed 2.7 ± 0.0002, and 20 mg/kg of Tribulosin exhibited 3 ± 0.0002 of organ coefficient. Whereas, the untreated ovariectomized rats showed 2.4 ± 0.0003 of organ coefficient. Control rats showed 3.3 ± 0.0003 of femur organ efficiency.
Tribulosin Attenuated Femur Resorption in Ovariectomized Rats. (A) Femur Wet Weight; (B) Femur Dry Weight; (C) Femur Organ Coefficient of the SHAM Control, Ovariectomized Untreated, Ovariectomized Treated With 10 mg/kg Tribulosin, and Ovariectomized Treated With 20 mg/kg Tribulosin Group Animals. Data Were Presented as Mean ± Standard Deviation (SD) and Evaluated Through One-Way Analysis of Variance (ANOVA), Followed by Tukey’s Post Hoc Test. Statistical Significance Was Considered at a p Value of Less Than .05. “*” Control Versus Ovariectomized Untreated, “**” Ovariectomized Untreated Versus Ovariectomized Treated With 10 mg/kg Tribulosin, Ovariectomized Treated With 20 mg/kg Tribulosin.
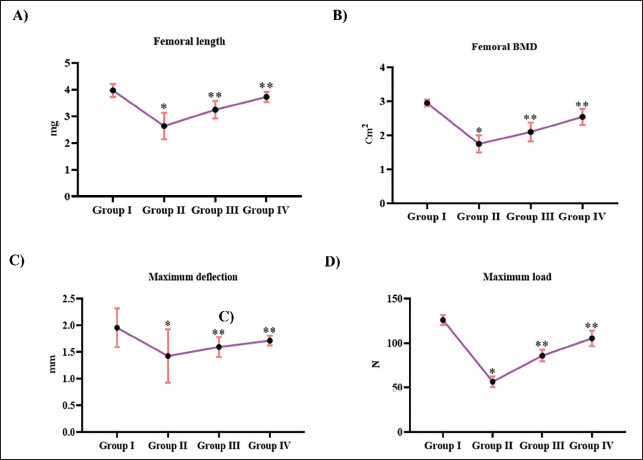
Femur bone length and the BMD were significantly reduced in the ovariectomized rats to 2.7 ± 0.0001 and 1.5 ± 0.0002 cm2, respectively, compared to the control rats, which showed 3.8 ± 0.0002 femur length and 2.4 ± 0.0004 cm2 of BMD. Both the femur length and the BMD were enhanced with Tribulosin treatment to 2.9 ± 0.0003 and 1.8 ± 0.0002 cm2 with 10 mg/kg treatment and 3.3 ± 0.0004 and 1.9 ± 0.0004 cm2 with 20 mg/kg treatment. Tribulosin enhanced the maximum deflection and maximum load capacity to 1.4 ± 0.00002 mm and 85 ± 0.9 N, respectively, with 10 mg/kg treatment and 1.6 ± 0.00005 mm and 90 ± 1.1 N, respectively, with 20 mg/kg treatment. Control rats exhibited significantly higher levels of maximum deflection (1.75 ± 0.00003 mm) and maximum load capacity (120 ± 1.5 N), whereas the ovariectomized untreated rats exhibited reduced levels of maximum deflection (1.25 ± 0.00002 mm) and maximum load capacity (52 ± 0.8 N) (Figure 2).
Tribulosin Improves Bone Structural Integrity in Ovariectomized Rats. (A) Femur Length; (B) Femur Bone Mineral Density; (C) Maximum Deflection; (D) Maximum Load Capacity of the SHAM Control, Ovariectomized Untreated, Ovariectomized Treated With 10 mg/kg Tribulosin, and Ovariectomized Treated With 20 mg/kg Tribulosin Group Animals. Data Were Presented as Mean ± Standard Deviation (SD) and Evaluated Through One-Way Analysis of Variance (ANOVA), Followed by Tukey’s Post Hoc Test. Statistical Significance Was Considered at a p Value of Less Than .05. “*” Control Versus Ovariectomized Untreated, “**” Ovariectomized Untreated Versus Ovariectomized Treated With 10 mg/kg Tribulosin, Ovariectomized Treated With 20 mg/kg Tribulosin.
Tribulosin Induced Osteoblastic Activity in Ovariectomized Rats
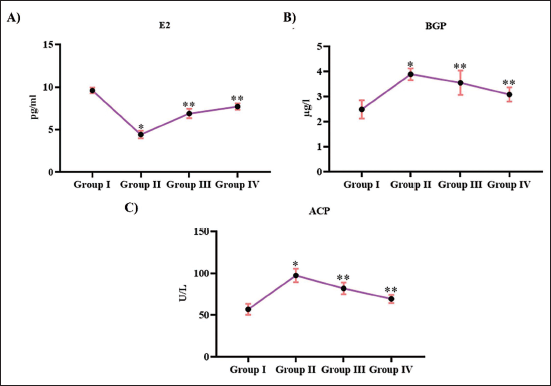
Figure 3 illustrates the effect of Tribulosin on osteoblastic promoters in ovariectomized animals. Treatment with Tribulosin significantly enhanced the levels of estradiol in ovariectomized, which were considerably decreased in the untreated ovariectomized rats. 5.8 ± 0.0003 pg/mL and 7.6 ± 0.0005 pg/mL, respectively, were observed in ovariectomized rats treated with 10 and 20 mg/kg Tribulosin, whereas it was significantly reduced to 3.8 ± 0.0003 pg/mL. Control rats exhibited significantly enhanced levels of estradiol (9 ± 0.0007 pg/mL). Both BGA and ACP levels were enhanced in the ovariectomized untreated rats. 3.8 ± 0.00005 µg/L of BGA and 85 ± 0.006 U/L µU/L of ACP were observed in the ovariectomized untreated animals, whereas the control rats exhibited significantly decreased levels of both BGA (2.3 ± 0.00004 µg/L) and ACP (48 ± 0.004 U/L µU/L). Treatment with 10 and 20 mg/kg Tribulosin in ovariectomized rats decreased the levels of BGA to 3.4 ± 0.00007 µg/L and 2.8 ± 0.00009 µg/L and ACP to 78 ± 0.002 U/L and 68 ± 0.008 U/L, respectively.
Tribulosin Induced Osteoblastic Activity in Ovariectomized Rats. (A) Estradiol; (B) Bone Gla Protein; (C) Acid Phosphatase Levels of the SHAM Control, Ovariectomized Untreated, Ovariectomized Treated With 10 mg/kg Tribulosin, and Ovariectomized Treated With 20 mg/kg Tribulosin Group Animals. Data Were Presented as Mean ± Standard Deviation (SD) and Evaluated Through One-Way Analysis of Variance (ANOVA), Followed by Tukey’s Post Hoc Test. Statistical Significance Was Considered at a p Value of Less Than .05. “*” Control Versus Ovariectomized Untreated, “**” Ovariectomized Untreated Versus Ovariectomized Treated With 10 mg/kg Tribulosin, Ovariectomized Treated With 20 mg/kg Tribulosin.
Tribulosin Attenuated Fracture Risk in Ovariectomized Rats
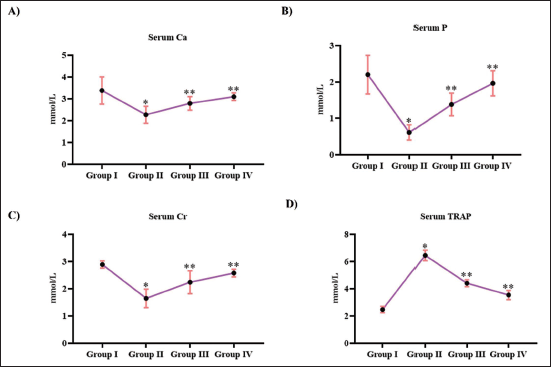
Figure 4 depicts the levels of bone minerals in ovariectomized, untreated, and Tribulosin-treated rats. Ovariectomization significantly reduced the calcium, phosphorus, and creatinine levels in the animals. Ovariectomized, untreated animals exhibited considerably reduced levels of calcium (1.8 ± 0.00002 mmol/L), phosphorus (0.7 ± 0.00004 mmol/L), and creatinine (1.7 ± 0.00006 mmol/L), whereas the control rats exhibited 2.9 ± 0.00003 mmol/L of calcium, 2.1 ± 0.00001 mmol/L of phosphorus, and 2.5 ± 0.00004 mmol/L of creatinine. Both the calcium and phosphorus levels were enhanced to 2.3 ± 0.00005 mmol/L and 2.7 ± 0.00006 mmol/L, respectively, and 1.3 ± 0.00003 mmol/L and 1.6 ± 0.00003 mmol/L, respectively, with 10 and 20 mg/kg Tribulosin treatment. Tribulosin also increased the creatinine levels to 1.9 ± 0.00003 mmol/L and 2.3 ± 0.00002 mmol/L, respectively, with 10 and 20 mg/kg treatment. TRAP levels were increased to 6.2 ± 0.00002 mµ/L with ovariectomization, whereas treatment with 10 and 20 mg/kg Tribulosin reduced the TRAP levels to 4.3 ± 0.00004 mµ/L and 3.8 ± 0.00002 mµ/L, respectively. Control rats exhibited 2.2 ± 0.00002 mµ/L of TRAP levels.
Tribulosin Attenuated Fracture Risk in Ovariectomized Rats. (A) Serum Calcium; (B) Serum Phosphorus; (C) Serum Creatinine; (D) Serum Tartrate Resistant Acid Phosphatase Levels of the SHAM Control, Ovariectomized Untreated, Ovariectomized Treated With 10 mg/kg Tribulosin, and Ovariectomized Treated With 20 mg/kg Tribulosin Group Animals. Data Were Presented as Mean ± Standard Deviation (SD) and Evaluated Through One-Way Analysis of Variance (ANOVA), Followed by Tukey’s Post Hoc Test. Statistical Significance Was Considered at a p Value of Less Than .05. “*” Control Versus Ovariectomized Untreated, “**” Ovariectomized Untreated Versus Ovariectomized Treated With 10 mg/kg Tribulosin, Ovariectomized Treated With 20 mg/kg Tribulosin.
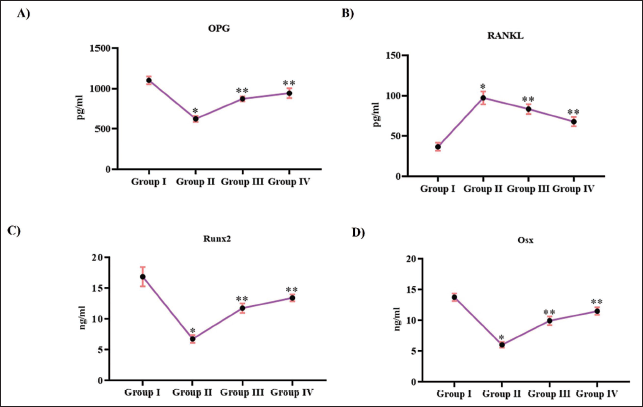
Tribulosin Triggered Osteogenic Activity in Ovariectomized Rats
Figure 5 illustrates the effect of Tribulosin on osteogenic promoters in ovariectomized rats. Ovariectomization in rats considerably decreased the levels of OPG to 600 ±12 pg/mL and increased the RANKL levels to 87 ± 0.004 pg/mL, whereas the control rats exhibited increased levels of OPG 1100 ± 10 pg/mL and decreased levels of RANKL 28 ± 0.002 pg/mL. Treatment with both 10 and 20 mg/kg Tribulosin increased the OPG levels to 800 ± 15 pg/mL and 890 ± 15 pg/mL, respectively, and decreased the RANKL levels to 75 ± 0.003 pg/mL and 58 ± 0.004 pg/mL, respectively. Treatment with Tribulosin increased the levels of transcription factors Runx2 and Osx in the ovariectomized rats. 10 mg/kg Tribulosin-treated exhibited 11 ± 0.008 ng/mL of Runx2 and 8.5 ± 0.009 ng/mL of Osx, whereas treatment with 20 mg/kg Tribulosin exhibited 12.5 ± 0.009 ng/mL of Runx2 and 10 ± 0.008 ng/mL of Osx. Ovariectomized untreated rats exhibited significantly decreased levels of Runx2 (7.5 ± 0.007ng/mL) and Osx (6 ± 0.009 ng/mL), whereas the control rats showed significantly increased levels of Runx2 (16 ± 0.006 ng/mL) and Osx (12 ± 0.008 ng/mL).
Tribulosin Triggered Osteogenic Activity in Ovariectomized Rats. (A) Osteoprotegerin; (B) Receptor Activator of Nuclear Factor Kappa-Β Ligand; (C) Runt-related Transcription Factor 2; (D) Osterix Levels of the SHAM Control, Ovariectomized Untreated, Ovariectomized Treated With 10 mg/kg Tribulosin, and Ovariectomized Treated With 20 mg/kg Tribulosin Group Animals. Data Were Presented as Mean ± Standard Deviation (SD) and Evaluated Through One-Way Analysis of Variance (ANOVA), Followed by Tukey’s Post Hoc Test. Statistical Significance Was Considered at a p Value of Less Than .05. “*” Control Versus Ovariectomized Untreated, “**” Ovariectomized Untreated Versus Ovariectomized Treated With 10 mg/kg Tribulosin, Ovariectomized Treated With 20 mg/kg Tribulosin.
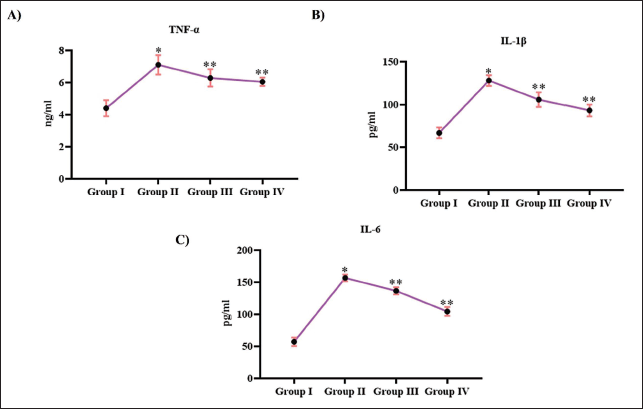
Tribulosin Inhibited the Inflammatory Response in Ovariectomized Rats
Figure 6 depicts the anti-inflammatory response of Tribulosin in ovariectomized rats. Treatment with Tribulosin attenuated the inflammatory cytokines in the ovariectomized rats. TNF-α levels were decreased to 5.8 ± 0.0002 ng/mL and 5.7 ± 0.0004 ng/mL, respectively, in 10 and 20 mg/kg Tribulosin-treated ovariectomized rats, whereas the untreated rats showed 6.9 ± 0.0001 ng/mL of TNF-α level. Both IL-1β and IL-6 were enhanced in ovariectomized rats to 125 ± 0.005 pg/mL and 150 ± 0.002 pg/mL, respectively, whereas treatment with Tribulosin decreased the levels of IL-1β and IL-6 to 98 ± 0.005 pg/mL and 128 ± 0.005 pg/mL with 10 mg/kg treatment and 85 ± 0.006 ng/mL and 80 ± 0.005 ng/mL with 20 mg/kg treatment. 3.9 ± 0.0002 ng/mL of TNF-α, 55 ± 0.003 pg/mL of IL-1β, and 52 ± 0.005 pg/mL of IL-6 levels were observed in the control rats.
Tribulosin Inhibited Inflammatory Response in Ovariectomized Rats. (A) Tumor Necrosis Factor Alpha; (B) Interleukin-1 Beta; (C) Interleukin-6 Levels of the SHAM Control, Ovariectomized Untreated, Ovariectomized Treated With 10 mg/kg Tribulosin, and Ovariectomized Treated With 20 mg/kg Tribulosin Group Animals. Data Were Presented as Mean ± Standard Deviation (SD) and Evaluated Through One-Way Analysis of Variance (ANOVA), Followed by Tukey’s Post Hoc Test. Statistical Significance Was Considered at a p Value of Less Than .05. “*” Control Versus Ovariectomized Untreated, “**” Ovariectomized Untreated Versus Ovariectomized Treated With 10 mg/kg Tribulosin, Ovariectomized Treated With 20 mg/kg Tribulosin.
Discussion
Osteoporosis is a systemic bone condition marked by reduced bone density and deterioration of bone structure, leading to greater fragility and an increased risk of fractures (Agrawal & Garg, 2023). In the context of rising life expectancy, a significant number of women spend more than one-third of their lives postmenopause. The rising incidence of menopause, coupled with the rapidly aging global population, underscores the urgent need to address the vitality and well-being of women in menopause transition (Gregson et al., 2022; Ji & Yu, 2014; Shen et al., 2022). Notably, World Health Organization (WHO) data indicate approximately 30% of postmenopause women were affected by osteoporosis (Xiao et al., 2022), highlighting the essential need for developing effective prevention and treatment approaches for this population.
Tribulosin is a bioactive compound belonging to spirostanol-type saponins found in Tribulus terrestris, a plant commonly used in traditional medicine (Saeed et al., 2024; Ștefănescu et al., 2020). It is known for its potential health benefits, including improving sexual function, physical performance, and providing anti-inflammatory and antioxidant effects (Zhu et al., 2017). Research suggests that Tribulosin may also have applications in treating conditions like diabetes and infertility (Zhang et al., 2019). Tribulosin has demonstrated cytoprotective effects in the cardiac tissue (Zhang et al., 2010) and exhibits antiparasitic activity (Deepak et al., 2002). In this study, we examined the efficacy of Tribulosin in attenuating osteoporosis induced by estrogen deficiency.
The ovariectomized rat is widely recognized as the commonly utilized animal model for researching anti-osteoporotic medications. Ovariectomy induces rapid bone loss, primarily attributed to an imbalance in bone turnover. In estrogen-deficient rats, this imbalance is characterized by bone resorption surpassing bone formation. Over relatively short durations, this model effectively simulates postmenopausal cancellous bone loss, providing a valuable framework for studying therapeutic interventions (Sharma et al., 2012; Turner et al., 2013). In our study, ovariectomized rats exhibited significantly decreased levels of estradiol, accompanied by a considerable reduction in the biomechanical strength of the femur. Untreated ovariectomized rats showed decreased femur length and weight, as well as a significantly reduced femur organ coefficient. Additionally, both BMD and biomechanical strength were markedly diminished in the ovariectomized group, consistent with findings from previous studies, indicating that ovariectomy induces osteoporosis (Shen et al., 2022). Conversely, rats treated with Tribulosin demonstrated an increase in femur biomechanical strength and BMD. These improvements may be attributed to elevated estradiol levels induced by Tribulosin treatment.
Osteocalcin, commonly referred to as BGA, accounts for approximately 15% of the noncollagenous components within the bone matrix (Brown et al., 1984). Osteoblasts, odontoblasts, and hypertrophic chondrocytes predominantly produce it. Initially identified in the 1980s, osteocalcin emerged as a bone-specific marker of formation, underscoring its pivotal role in skeletal biology (Price et al., 1980). Tartrate-resistant ACP is an enzyme predominantly found in osteoclasts, the cells responsible for bone resorption (Igarashi et al., 2002). TRAP is highly expressed in bone tissue, particularly in multinucleated osteoclasts and their precursors (Halleen et al., 2006). Recent research has emphasized TRAP’s critical role in bone resorption; functional studies indicate that inhibiting TRAP activity impairs osteoclast bone resorbing functions. Mouse models deficient in TRAP exhibit disrupted endochondral ossification and mild osteopetrosis, whereas overexpression of TRAP leads to increased bone turnover and mild osteoporosis (Brown et al., 2022; Yılmaz et al., 2024). In our study, Tribulosin treatment decreased the BGA, ACP, and TRAP levels, which were increased in untreated ovariectomized rats. This proves Tribulosin inhibited the activity of osteoclasts in ovariectomized rats.
Estrogen is integral to bone homeostasis, exerting its effects primarily through estrogen receptors expressed on osteoblasts and osteoclasts (Eriksen et al., 1988). It enhances bone formation by stimulating osteoblastic activity and concurrently inhibits bone resorption by promoting apoptosis in osteoclasts (Oursler et al., 1991). Bone metabolism is governed by the dynamic balance between osteoblast-mediated bone formation and osteoclast-mediated resorption (Mano et al., 1996). This balance is tightly regulated by plasma levels of calcium and phosphorus, which are influenced by bone turnover as well as the intestinal absorption and renal reabsorption of these minerals (Xu et al., 2018). Tribulosin treatment considerably increased the levels of serum calcium, phosphorus, and creatinine in the ovariectomized rats. This may be due to the increase in the estradiol levels, which inhibited bone resorption.
Estrogen deficiency enhances bone resorption by upregulating pro-inflammatory cytokines and RANKL, both of which promote osteoclastogenesis. Through the classic genomic pathway, estrogen binds to its receptors in the cytoplasm, forming a complex that translocates to the nucleus. This complex interacts with estrogen response elements in DNA, modulating gene transcription (Bado et al., 2017; Hsu et al., 2024). RANKL typically engages with its receptor, RANK, on osteoclasts and their precursors, thereby facilitating bone resorption. In contrast, OPG acts as a decoy receptor that sequesters RANKL, mitigating its activity and preserving bone integrity (Eastell et al., 2016). Reduced estrogen levels diminish OPG production and the inhibitory effects on RANKL, consequently amplifying osteoclast-mediated bone resorption (Sjögren et al., 2009). Estradiol also contributes to OPG synthesis, further counteracting RANKL-induced osteoclast activation (Hsu et al., 2024). Tribulosin treatment potentially increased the levels of OPG, thereby attenuating the levels of RANKL and preventing osteoclast activity in the ovariectomized rats.
The differentiation of osteoblasts from pluripotent mesenchymal progenitors depends on the precise expression of two essential transcription factors, RUNX2 and OSX1 (Šromová et al., 2023). These osteoblasts play a vital role in both bone formation during skeletal development and in the remodeling of bone. Research by Lin et al. (2019) has shown that the activation of G protein-coupled receptor 30 (GPR30) increases the expression of Runx2, an essential transcription factor for bone formation. Tribulosin treatment in ovariectomized rats not only attenuated the activity of osteoclasts, it promoted the activity of osteoblasts, which is evidenced by increased levels of Runx2 and Osx transcription factors in the Tribulosin-treated ovariectomized rats. Estrogen deficiency causes an increase in interleukin-7 (IL-7), which promotes T cell activation. This activation results in the production of pro-inflammatory molecules such as IL-1, IL-6, and TNF-α, ultimately contributing to the formation of osteoclasts (McLean, 2009). Furthermore, the lack of estrogen enhances T cell activation and osteoclastogenesis by elevating levels of reactive oxygen species (ROS), which in turn stimulate the production of TNF (Weitzmann & Pacifici, 2006). Treatment with Tribulosin exhibited significantly decreased levels of TNF-α, IL-1β, and IL-6. This may be due to an increase in estradiol triggered by Tribulosin, which eventually attenuated the inflammatory response and promoted the activity of osteoblasts.
Conclusion
To conclude, the findings of this study highlight the potential of Tribulosin as an effective and affordable therapeutic agent for osteoporosis treatment. Tribulosin significantly improved bone structural integrity, increased BMD, and enhanced femoral strength in an ovariectomy-induced osteoporosis rat model. Additionally, it promoted osteogenic activity, reduced inflammation, and elevated key serum biomarkers associated with bone health. Given its ability to mitigate fracture risk without adverse effects, Tribulosin presents a promising alternative to existing antiresorptive drugs. However, further research is necessary to determine its long-term efficacy and elucidate its precise mechanisms of action in bone health.
Footnotes
Abbreviations
ACP: Acid phosphatase; ANOVA: Analysis of variance; BGA: Bone Gla protein (also referred to as bone gamma-carboxyglutamic acid protein); BMD: Bone mineral density; DEXA: Dual-energy X-ray absorptiometry; ELISA: Enzyme-linked immunosorbent assay; GPR30: G protein-coupled receptor 30; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; IL-7: Interleukin-7; OPG: Osteoprotegerin; Osx: Osterix; OVX: Ovariectomy/ovariectomized; RANKL: Receptor activator of nuclear factor kappa-B ligand; ROS: Reactive oxygen species; Runx2: Runt-related transcription factor 2; SD: Standard deviation; SPSS: Statistical Package for the Social Sciences; TNF-α: Tumor necrosis factor-alpha; TRAP: Tartrate-resistant acid phosphatase; WHO: World Health Organization.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This work was approved by the Institutional Ethical Committee, Spinal Surgery Department, The Second Affiliated Hospital of Guangdong Medical University, Guangdong Province, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
NA.
