Abstract
Background
Globally, osteoporosis has arisen as a significant health hazard, impacting over 200 million individuals. It is often reported in women, especially those who are in the postmenopausal stage. Even though numerous reasons are linked with osteoporosis etiology, estrogen deprivation is the prime inducer of osteoporosis.
Objectives
The present research was intended to detect the bioflavonoid tangeretin potency against ameliorating osteoporosis in ovariectomized rats.
Materials and Methods
Ovariectomy was performed in healthy female Sprague rats and treated with two different doses of the drug tangeretin. The body weight gain obtained by the experimental animals was noted throughout the experimental period. The uterus and vagina, the organ responsible for estrogen production and activity, were measured. Bone mineral density (BMD) and biomechanical properties of the femur were evaluated to study the impact of tangeretin. Bone biomarkers estradiol, bone GLA protein, and acid phosphatase were quantified to assess the impact of tangeretin on bone turnover. Serum phosphorous, calcium, creatinine, and tartrate-resistant acid phosphatase (TRAP), which are the osteoporosis risk factors, were analyzed in tangeretin-treated ovariectomized rats. Signaling proteins involved in the receptor activator of nuclear factor-κB (RANK)/receptor activator of nuclear factor-κB ligand (RANKL)/osteoprotegerin (OPG) pathway, which is responsible for bone turnover, were quantified.
Results
Tangeretin treatment significantly inhibited excess weight gain and increased the uterus and vaginal weight. BMD and biomechanical analysis of the femur prove tangeretin treatment increased the BMD and upgraded the biomechanical properties in ovariectomized animals. Tangeretin increased the estradiol and decreased the bone GLA protein and acid phosphatase. It also increased serum phosphorous, calcium, and creatinine and decreased the TRAP levels, confirming its ameliorative potency against bone turnover. It inhibited RANKL protein and increased the levels of OPG and its signaling molecules in ovariectomized rats. The anti-inflammatory bioflavonoid tangeretin attenuated the inflammatory stimulating cytokines in ovariectomized rats.
Conclusion
Overall, our analysis confirms tangeretin possesses antiosteoporotic potency which effectively elevates estradiol and attenuates the bone turnover signaling pathway RANK/RANKL/OPG proteins in ovariectomized rats. The results of our research prove the establishment of a treatment strategy using tangeretin can improve bone health and prevent osteoporosis in menopausal women.
Introduction
Osteoporosis, a global silent killer, is characterized by reduced bone mass and density, which leads to weakened bone architecture and a heightened risk of fractures (Shrihari & Harpreet, 2024). This disorder arises due to an imbalance between bone resorption and bone formation, favoring bone loss over bone production and making bones porous and fragile (Salari et al., 2021). Risk factors for osteoporosis are classified into alterable and nonalterable. Alterable risk factors comprise obesity, lack of physical activity, lifestyle changes, calcium deficiency, consumption of alcohol, smoking, and prolonged use of glucocorticoids. Nonalterable risk factors encompass gender, age, race, and genetic traits (Askari et al., 2019). Additionally, certain risk factors are more prevalent in specific genders. For instance, in women, early menopause and the loss of ovarian function before menopause also increase the risk of developing osteoporosis (Pinar et al., 2017).
Osteoporosis is an inevitable morbidity of the geriatric population, which affects more than 200 million individuals globally (Barnsley et al., 2021). International Osteoporosis Foundation reports that osteoporosis-related injury or fracture happens every 3 s, resulting in nearly 8.9 million fractures annually (Sozen et al., 2017). According to the World Health Organization (WHO), osteoporosis is a prevalent condition affecting midlife and geriatric individuals (Fang et al., 2021). Worldwide, its prevalence stands at higher among females than males (Salari et al., 2021). Previous research indicates that over one-third of middle-aged and elderly women globally, compared to one in five men, experience fractures as a result of osteoporosis (Sadeghi et al., 2019). Early menopause and the loss of ovarian function before menopause are prime risk factors for osteoporosis (Rashki et al., 2020). It is anticipated that there will be more than 20 million cases of hip fractures by 2050 (Pisani et al., 2016).
Estrogen deficiency has been reported as the cause of bone loss and osteoporosis in the majority of females at postmenopausal stage. Bone cells are present with abundant number of estrogen receptors, which protects bone from osteoclast activity. Estrogen effectively inhibits receptor activator of nuclear factor-κB ligand (RANKL) activity via promoting osteoprotegerin expression (Cheng et al., 2022) and thereby attenuates the osteoclast formation, bone resorption (Abu-Amer, 2013). Increased secretion of interleukin (IL)-1, IL-6, and tumor necrosis factor (TNF) were detected with estrogen deficiency (Eastell et al., 2016). It also enhances bone turnover and resorption, culminating in osteoporosis (Emmanuelle et al., 2021).
Osteoporosis is treated with drugs such as bisphosphonates, denosumab which attenuates bone resorption or triparatide, and strontium which stimulates osteoblast (Cremers et al., 2019). These drugs target the RANKL ligand of NF-κB receptor, thereby reducing bone resorption and effectively increasing bone density (Deeks, 2018; Liang et al., 2022). In long-term usage, these drugs are reported to cause mandible osteonecrosis and atypical femoral fractures, which hinder the usage of these drugs (Everts-Graber et al., 2022; Ilyas & Camacho, 2019). Phytoestrogenic drugs such as quercetin, biochanin A, daidzein, rutin, and genistein have been proven to possess osteoprotective effects, and all these drugs are safe and cost-effective (Karimi et al., 2023). We analyzed one such phytochemical tangeretin potency to ameliorate osteoporosis in ovariectomized rats. Tangeretin, a significant flavonoid amply found in peels of citrus fruits, exhibits a numerous beneficial pharmacological activity. It is recognized for its antioxidant, anti-inflammatory, antitumor, liver-protective, and neuroprotective properties (Ashrafizadeh et al., 2020).
Materials and Methods
Animals
In the context of this study, female Sprague Dawley rats, aged 10–12 weeks and weighing 200–250 g, were utilized. The rats were housed under laboratory conditions prescribed by the international standards of laboratory animal care. Laboratory rodent pellets, which consist of protein (19.6%), total fat (4.2%), crude fiber (4.6%), acid detergent fiber (7.60%), neutral detergent fiber (15.40%), and total carbohydrate (59.30%), were fed to rats without any restrictions. The animals were treated with humanitarian basis, and only the procedure described and approved by the ethical committee was performed.
Osteoporosis Induction
Osteoporosis is triggered in rats by the surgical removal of ovaries in the experimental rats following the protocol of He et al. (2010). The experimental animals were subjected to anesthesia with 50 mg/kg bwt of pentobarbitone injected intraperitoneally before the initiation of surgery. The rat is positioned on its back, and the incision area was shaved and disinfected. A minor cut was created in the wall of the abdomen typically on the dorsal side, to expose the ovaries. The ovaries, connected to the uterine horns, are carefully excised using precise surgical instruments. Bleeding was controlled, and the incision was then closed with sutures or surgical adhesive. The same procedure was followed for the sham control rats; only difference the ovaries were not exposed. After the surgery, the rat was closely monitored for any signs of pain or infection, and care was taken to promote proper healing. The entire process was conducted in accordance with ethical guidelines to minimize the animal’s discomfort and ensure its well-being.
Treatment Regiment
The rats were habituated to the standard laboratory conditions before the experiment began, ensuring they were accustomed to the environment, and clustered into four each consisting of six rats. First cluster consisted of sham-operated control rats and the remaining three clusters were subjected to ovariectomy. Second cluster of rats was sustained for 3 weeks followed by the ovariectomy procedure. Cluster 3 and 4 rats were treated with 10 and 20 mg/kg body weight of tangeretin/day, respectively, orally for 16 successive weeks. The animals were fasted overnight on the last day of experimental model. The rats were then euthanized, blood samples and the uterus, vagina, and femur bones were collected for the further analysis.
Organ Weight Measurement
The body weight of the experimental rats was monitored regularly for every 7 days throughout the experimental periods and the readings were recorded. The uterus, vagina, and femur bones excised from the rats were weighed to assess the impact of ovariectomy procedure. The femoral bone was subjected to desiccation at 65°C to measure the invariable weight difference of femoral bone in tangeretin-treated and untreated ovariectomized rats. The relative organ coefficient of uterus and femur was also calculated.
Bone Mineral Density (BMD) and Biomechanical Analysis
The excised femur bone was subjected to complete removal of muscle and connective tissue before the initiation of the experiment. The BMD of femur excised was determined using the BMD detector following the manufacturer’s instruction. Biomechanical analysis was performed to detect the parameters such as maximum deflection, maximum load, energy, stiffness, young modules, and maximum stress of the femur bone using the biomechanical testing instrument. The stride distance and the loading velocity were set at 20 mm and 5 mm/s, respectively.
Quantification of Bone Biomarker
Bone biomarkers estradiol, bone GLA protein, and acid phosphatase were quantified in the serum of control and experimental animals using the MyBioSource ELISA kit. The assay was conducted following the manufacturer’s guidelines.
Analysis of Osteoporosis Risk Markers
The biochemical markers predicting the induction of osteoporosis serum phosphorous, calcium, creatinine, and the TRAP were detected in the control and experimental rats. Serum biochemical markers phosphorous, calcium, and creatinine were quantified with the biochemical autoanalyzer, and the levels of serum TRAP were measured with ELISA kit procured from MyBioSource.
Analysis of RANK/RANKL/OPG Signaling
Receptor activator of nuclear factor-κB (RANK)/RANKL/osteoprotegerin (OPG) signaling contributes a critical impact on the bone metabolism. Evidences suggest in postmenopausal women the circulating levels of RANKL are inversely associated with bone turnover and BMD leading to osteoporosis. Therefore, the levels of OPG, RANKL, RunX2, and Osx were quantified in the control and experimental animals using the ELISA kits purchased from CusaBIO.
Assessment of Inflammation
During menopause, due to decreased ovarian function, a rapid rise in inflammatory-inducing cytokines is reported which spontaneously triggers osteoclast activity causing osteoporosis. Hence to assess the impact of tangeretin on ovariectomized rats the levels of TNF-α, IL-1β, and IL-6 were quantified with the ELISA kits, MyBiosource. The test was done according to the guidelines provided in the kit.
Statistical Analysis
The experimental data provided represents the results of independent experiments performed thrice. The data were assessed with GraphPad Prism 5 software and illustrated as mean ± standard deviation (SD), n represents the number of animals. Differences between groups were evaluated using one-way analysis of variance (ANOVA) followed by Bonferroni post-test, with significance set at p < 0.05.
Results
Antiobesity Effect of Bioflavonoid Tangeretin in Ovariectomized Rats
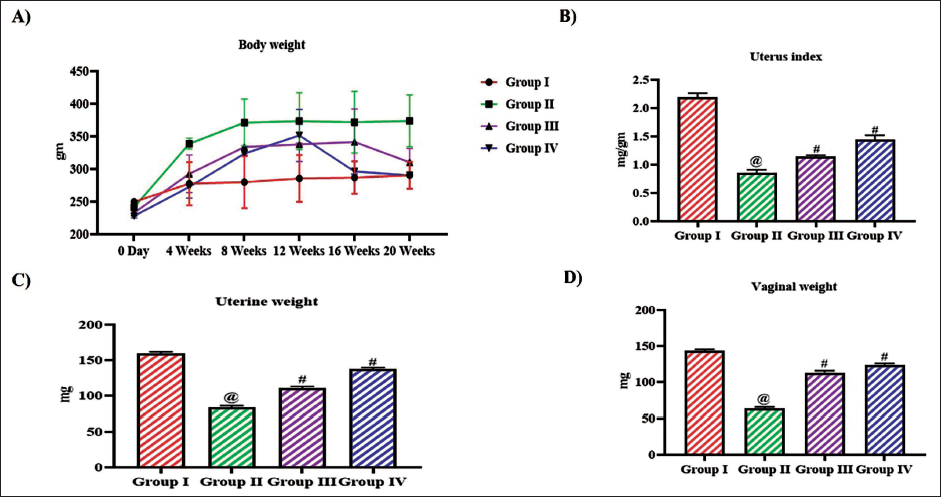
Figure 1 depicts the effect of tangeretin on weight gain in ovariectomized monitored throughout the experimental period. On the 20th week of the treatment period the ovariectomized untreated rats weighed about 368 ± 6 g whereas the control rats weighed about 265 ± 5 g. The weight gain in tangeretin-treated ovariectomized rats was significantly lesser than the untreated rats. 10 mg/kg tangeretin-treated rats weighed about 280 ± 4 g and 20 mg/kg tangeretin-treated rats weighed about 267 ± 5 g. To examine the impact of hormone imbalance in weight gain of ovariectomized rats, the weights of uterus and vagina were measured and the weights were depicted in Figure 1. The uterus index was decreased to 0.8 ± 0.006 mg/g in the untreated ovariectomized rats compared to the control rats which exhibited 2.2 ± 0.008 mg/g. Tangeretin treatment increased the uterus index to 1.1 ± 0.002 mg/g in 10 mg/kg treatment and to 1.4 ± 0.007 mg/g in 20 mg/kg treatment. Tangeretin treatment also increased the vaginal weight to 118 ± 0.5 mg and 126 ± 0.4 mg with 10 and 20 mg/kg body weight, whereas the untreated rats exhibited decreased vaginal weight of 61 ± 0.3 mg.
Antiobesity Effect of Bioflavonoid Tangeretin in Ovariectomized Rats. (A) Total Body Weight Measured Weekly throughout the Treatment Period, (B) Uterus Weight, (C) Uterus Index, (D) Vaginal Weight of Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
Ameliorative Effect of Bioflavonoid Tangeretin on BMD in Ovariectomized Rats
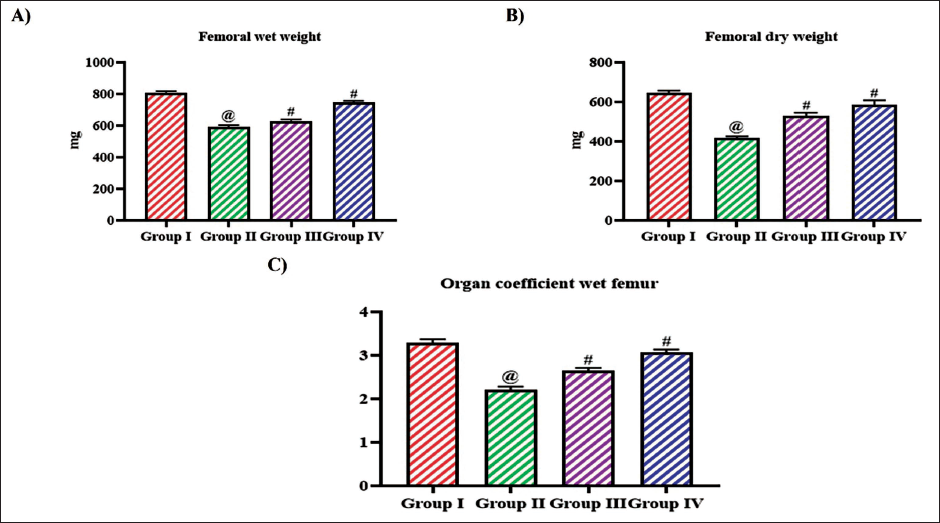
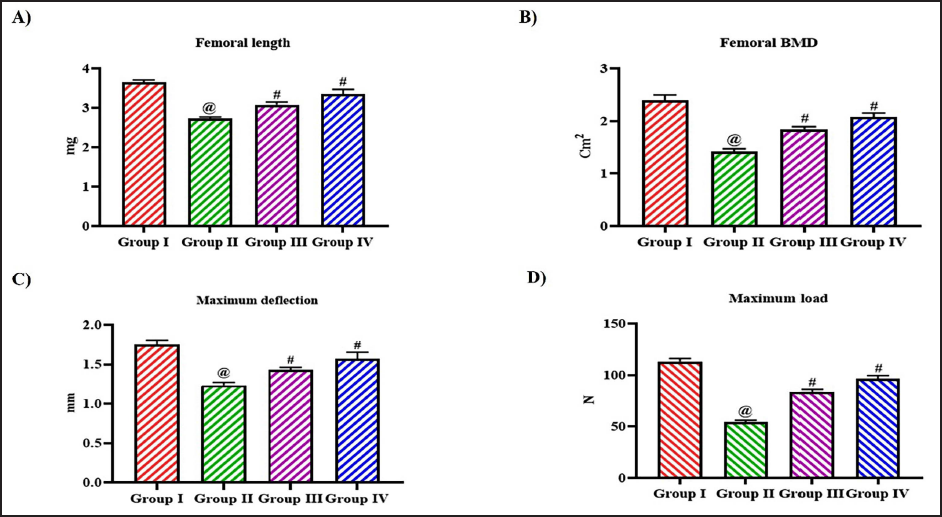
To examine the impact of tangeretin on the BMD and biomechanical property of ovariectomized rats, the femur bone was assessed. Figure 2 illustrates the femoral weight of ovariectomized untreated and tangeretin-treated rats. The organ coefficient of the wet femur was decreased in the ovariectomy untreated rats 2.1 ± 0.003 than the control and 10, 20 mg/kg tangeretin-treated rats which exhibited 3.3 ± 0.004, 2.6 ± 0.004, and 2.9 ± 0.003, respectively. The femoral length and the BMD were also decreased to 2.5 ± 0.0005 and 1.5 ± 0.03, respectively, in the untreated ovariectomized rats. Tangeretin treatment increased both the femoral length and BMD to 2.8 ± 0.002, 1.8 ± 0.04, respectively, with 10 mg/kg treatment and 3.3 ± 0.005, 2.1 ± 0.06, respectively, with 20 mg/kg treatment. Control rats exhibited increased femoral length of 3.8 ± 0.009 and BMD 2.4 ± 0.07 compared to the other experimental group rats (Figure 3).
Ameliorative Effect of Bioflavonoid Tangeretin on Femur in Ovariectomized Rats. (A) Femoral Wet Weight, (B) Femoral Dry Weight, (C) Organ Coefficient of Femoral of Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
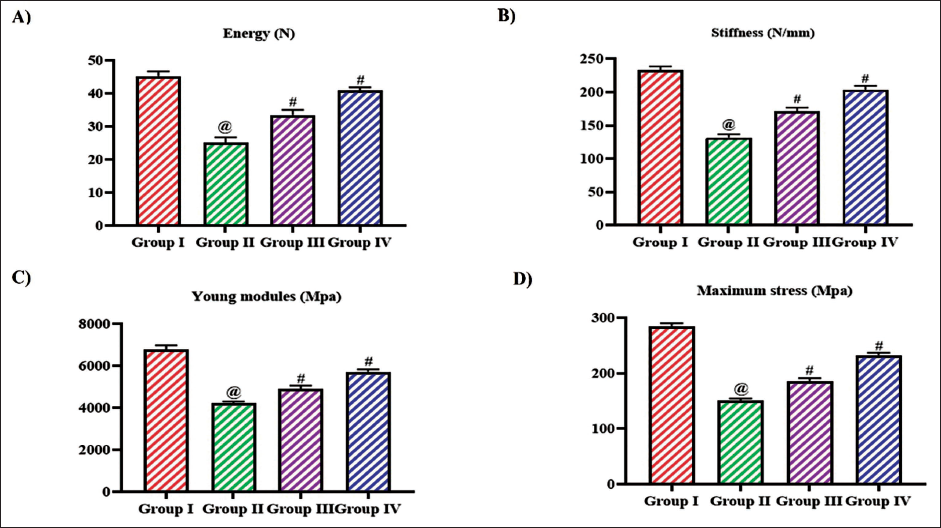
The maximum deflection and the maximum load exhibited by the control and the experimental rats were assessed, and the results were depicted in Figure 3C and D. Tangeretin treatment considerably increased the maximum deflection and maximum load to 1.3 ± 0.0002 mm and 82 ± 0.04 N, respectively, in 10 mg/kg treatment and 1.5 ± 0.0006 mm and 94 ± 0.05 N, respectively, in 20 mg/kg treatment compared to the untreated ovariectomized rats which exhibited 1.2 ± 0.0003 mm of maximum deflection and 49 ± 0.03 N maximum load. The energy, stiffness, young modules, and the maximum stress were decreased in the untreated ovariectomy rats than the other experimental group. Tangeretin treatment significantly increased the energy, stiffness, young modules, and maximum stress in a dose-dependent manner (Figure 4).
Ameliorative Effect of Bioflavonoid Tangeretin on Femur BMD in Ovariectomized Rats. (A) Femoral Length, (B) Femoral BMD, (C) Maximum Deflection, (D) Maximum Load Observed in Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
Effect of Bioflavonoid Tangeretin on Femur Biomechanics in Ovariectomized Rats. (A) Energy, (B) Stiffness, (C) Young Modules, (D) Maximum Stress Observed in Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
Impact of Bioflavonoid Tangeretin on Bone Remodeling in Ovariectomized Rats
Estradiol is an essential hormone for maintaining bone density and preventing osteoporosis by regulating bone remodeling, calcium homeostasis, and the activity of bone cells. Ovariectomy in rats significantly reduced the estradiol levels to 4.1 ± 0.0003 pg/mL, whereas the treatment with tangeretin increased the estradiol levels to 5.9 ± 0.0001, 7.6 ± 0.0002 pg/mL with 10 and 20 mg/kg treatment, respectively. Tangeretin also decreased the levels of bone GLA protein and acid phosphatase to 3.4 ± 0.0005 µg/L, 78 ± 0.006 U/L, respectively, with 10 mg/kg treatment and 62.9 ± 0.0008 µg/L, 66 0.1 U/L, respectively, with 20 mg/kg treatment. 8.4 ± 0.0006 pg/mL of estradiol, 2.1 ± 0.0002 µg/L of BGP and 48 ± 0.08 U/L of acid phosphatase (ACP) were quantified in sham-operated rats (Figure 5).
Impact of Bioflavonoid Tangeretin on Bone Remodeling in Ovariectomized Rats. Estradiol, (B) Bone GLA Protein, (C) Acid Phosphatase Quantified in Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
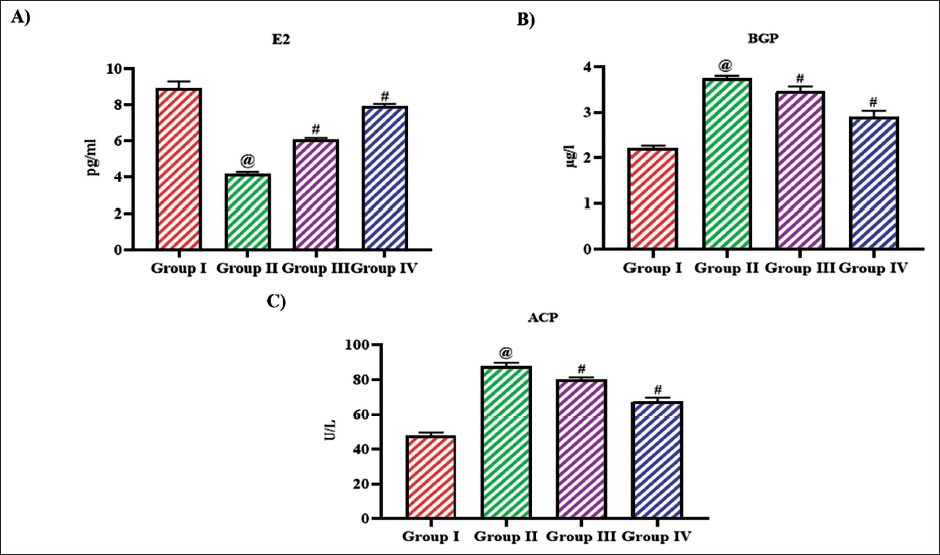
Effect of Bioflavonoid Tangeretin on Osteoporosis Risk Markers in Ovariectomized Rats
Figure 6 illustrates the levels of biochemical markers to measure the osteoporosis risk in ovariectomized rats. The levels of serum calcium, phosphorous, and creatinine were significantly decreased to 1.9 ± 0.0001 mmol/L, 0.6 ± 0.0004 mmol/L, 1.6 ± 0.001 mmol/L, respectively, in untreated ovariectomized rats compared to the control rats which exhibited 2.9 ± 0.0002 mmol/L, 2.1 ± 0.0001 mmol/L, 2.5 ± 0.0007 mmol/L of serum calcium, phosphorous, and creatinine, respectively. Both the low- and high-dose tangeretin treatment increase the levels of serum calcium, phosphorous, and creatinine to 2.1 ± 0.005 mmol/L, 1.2 ± 0.0003 mmol/L, 1.9 ± 0.0009 mmol/L, respectively, with 10 mg/kg treatment and 2.5 ± 0.004 mmol/L, 1.6 ± 0.0003 mmol/L, 2.2 ± 0.0001 mmol/L, respectively, with 20 mg/kg treatment. Tangeretin treatment decreases the TRAP levels in ovariectomized rats to 4.1 ± 0.0005 mµ/mL and 3.9 ± 0.0004 mµ/mL, respectively, with 10 and 20 mg/kg treatment, whereas the TRAP was significantly increased to 6 ± 0.0001 mµ/mL in untreated ovariectomized rats.
Effect of Bioflavonoid Tangeretin on Osteoporosis Risk Markers in Ovariectomized Rats. (A) Serum Calcium, (B) Serum Phosphorous, (C) Serum Creatinine, (D) Serum TRAP Quantified in Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
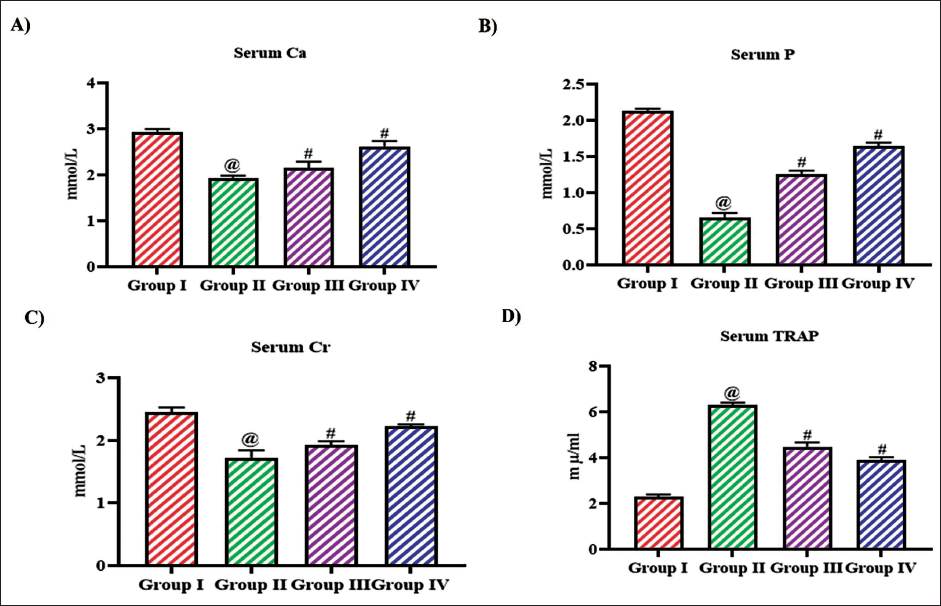
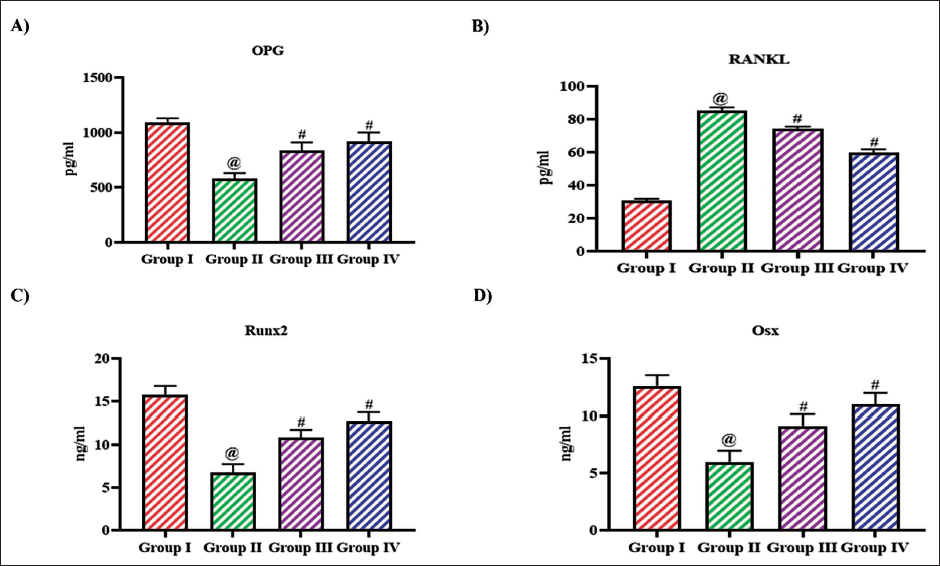
Regulating Effect of Bioflavonoid Tangeretin on RANK/RANKL/OPG Signaling in Ovariectomized Rats
Figure 7 illustrates the impact of tangeretin on the levels of significant proteins involved in bone remodeling signaling pathway in ovariectomized rats. Ovariectomy in rats significantly increased the levels of RANKL and decreased the levels of OPG, Runx2, and Osx, whereas the treatment with tangeretin in ovariectomized rats increased the levels of OPG, Runx2, Osx and decreased the RANKL levels. Both the reduction RANKL and increase in OPG, Runx2, Osx levels were observed in a dose-dependent manner.
Regulating Effect of Bioflavonoid Tangeretin on RANK/RANKL/OPG Signaling in Ovariectomized Rats. Osteoprotegerin, (B) RANKL, (C) Runx2, (D) Osx Levels Quantified in Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated Using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
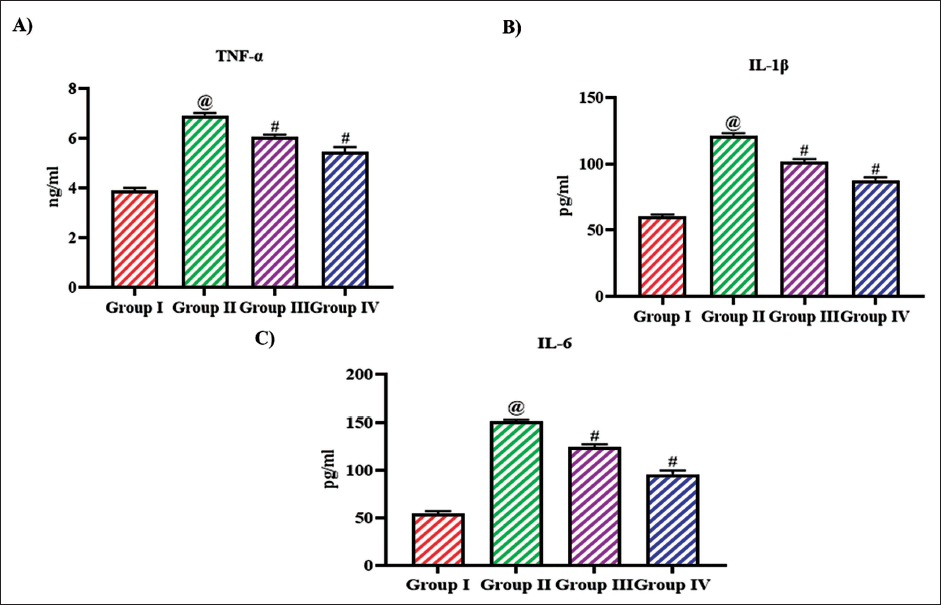
Attenuating Effect of Bioflavonoid Tangeretin against Inflammatory-Inducing Cytokine in Ovariectomized Rats
Figure 8 depicts the levels of inflammatory triggering cytokines in untreated and tangeretin-treated ovariectomized rats. Ovariectomy increased the levels of inflammatory triggering proteins TNF-α, IL-1β, and IL-6 levels to 6.8 ± 0.0004 ng/mL, 138 ± 0.08 pg/mL, 148 ± 0.02 pg/mL, respectively, compared to the control rats which exhibited 3.9 ± 0.0004 ng/mL, 60 ± 0.04 pg/mL, 54 ± 0.05 pg/mL of TNF-α, IL-1β, and IL-6 levels, respectively. Tangeretin treatment significantly reduced the levels of TNF-α, IL-1β, and IL-6 to 5.9 ± 0.0003 ng/ml, 96 ± 0.06 pg/mL, 132 ± 0.07 pg/mL with 10 mg/kg treatment, and 5.1 ± 0.0006 ng/mL, 82 ± 0.08 pg/mL, 98 ± 0.09 pg/mL with 20 mg/kg treatment.
Attenuating Effect of Bioflavonoid Tangeretin Against Inflammatory-Inducing Cytokine in Ovariectomized Rats. (A) Tumor Necrosis Factor α (TNF-α), (B) Interleukin 1β (IL-1β), and (C) Interleukin 6 (IL-6) Levels Quantified in Sham Control, Ovariectomized Untreated Rats, Ovariectomized 10 and 20 mg/kg Tangeretin-Treated Rats. Results Illustrated as Mean ± Standard Deviation (SD), n represents the Number of Animals. Differences between Groups were Evaluated using One-Way ANOVA followed by Bonferroni Post-test, with Significance Set at @,#p < 0.05.
Discussion
Tangeretin, a significant polymethoxylated flavonoid found in citrus fruit peels like oranges and tangerines, exhibits numerous beneficial bioactivities (Ashrafizadeh et al., 2020). Tangeretin’s pharmacological properties include antiproliferative, anti-invasive, and antimetastatic activities, along with the ability to induce apoptosis in certain cancers, highlighting its potential in cancer therapy (Lin et al., 2019). Studies also emphasize its antiasthmatic, antiteratogenic, and neuroprotective effects (Braidy et al., 2017), as well as its antioxidant and anti-inflammatory capacities (Arafa et al., 2021). It has also been proven to reduce blood sugar levels (Sundaram et al., 2014), inhibit inflammation, and prevent heart diseases (Vaiyapuri et al., 2013), hepatitis, kidney dysfunction (Wu et al., 2018), and hyperlipidemia (Lin et al., 2011). Notably, tangeretin exhibits potential in treating neurological disorders, such as Alzheimer’s disease, epilepsy, and Parkinson’s disease (Wani et al., 2024) and has been recognized for its anticancer effects in traditional Chinese medicine (Alex et al., 2021). The antioxidant and anti-inflammatory properties of tangeretin make it a promising candidate for treating cancer and neurological diseases. The efficacy of tangeretin on ameliorating global morbidity of postmenopausal women, osteoporosis is not yet elucidated. Hence, we aimed to examine the potency of tangeretin alleviating osteoporosis in ovariectomized rats.
Osteoporosis is a chronic systemic skeletal disorder that debilitates BMD and degrades the bone tissue microarchitecture (Mohammed et al., 2019). Osteoporosis have a severe impact on the quality of life of postmenopausal women incapacitates the physical, psychological, and economical life of the individual (Charde et al., 2023). Osteoporosis occurs when there is a negative imbalance in bone remodeling, where bone resorption surpasses bone formation. The receptors of estrogen are present ubiquitously in osteoclast, osteoblast, and osteocytes which are protecting the bone remodeling mechanism (Cheng et al., 2022). Researches and clinical studies had confirmed estrogen deficiency has a direct impact on bone resorption and turnover which eventually leads to osteoporosis (Almeida et al., 2013). Hence to assess the antiosteoporotic effect of tangeretin ovariectomy was performed in rats and treated with tangeretin.
Research has indicated that premenopausal women with obesity tend to have lower estradiol and follicle-stimulating hormone levels compared to their nonobese counterparts (El Khoudary et al., 2019). A reduction in estradiol is linked to an increased accumulation of central body fat and the promotion of visceral adiposity (Marlatt et al., 2022). Studies on animals, including mice and rats, along with human studies involving ovarian suppression through gonadotropin-releasing hormone agonists, have shown an increase in visceral fat mass without a corresponding rise in overall fat mass. The addition of estradiol reversed the gain in visceral fat (Karvonen-Gutierrez & Kim, 2016). Our results correlate with these findings, as the untreated ovariectomized rats’ weight gain is significantly higher than that of the control and tangeretin-treated rats. Tangeretin treatment increased the estradiol levels in the ovariectomized rats, which may explain the decrease in weight gain compared to the untreated ovariectomized rats. The impact of ovariectomy was clearly evidenced by the reduction in the uterus, vagina, and femoral weights, which increased with tangeretin treatment.
Bone GLA protein, also known as osteocalcin, plays a significant role in bone metabolism and is closely related to osteoporosis. Elevated levels of osteocalcin in the blood are often considered an indicator of increased bone resorption, which is a characteristic of osteoporosis. Measuring osteocalcin levels can aid in assessing bone health and the effectiveness of treatments for osteoporosis, as changes in its levels may reflect alterations in bone turnover rates (Seibel, 2005). Another effective biomarker for osteoporosis is acid phosphatase specifically TRAP. Osteoclasts are well known for their increased levels of TRAP activity, a characteristic that has long been used to identify these cells in tissue samples. Serum TRAP has been recognized as a marker for diagnosing diseases linked to excessive bone resorption and for observing the progression and rate of metabolic bone disorders. TRAP enters the serum as it is released from actively resorbing osteoclasts, while these cells maintain their cellular integrity during the continuous release of the enzyme (Rajeshwari et al., 2014). Tangeretin treatment effectively decreased the levels of significant biomarkers: bone GLA protein, acid phosphatase, and serum TRAP levels in the ovariectomized, signifying its osteoprotective property.
Estrogen influences bone health in several ways, it reduces bone resorption by decreasing the bone’s sensitivity to parathyroid hormone; stimulates the production of calcitonin, which also inhibits bone resorption; enhances calcium absorption in the intestines, thereby increasing serum calcium levels and promoting bone mass growth; decreases calcium excretion by the kidneys, which raises serum calcium concentration (Stamenkovic et al., 2022; Tu et al., 2018). Estrogens also play a significant role in maintaining adequate levels of inorganic phosphorus, which is essential for the activity of osteoblasts and osteocytes in matrix mineralization (Magne et al., 2003; Xu et al., 2003). In early postmenopausal women, serum creatinine (sCr) levels are inversely correlated with BMD, regardless of age, duration of menopause, and serum uric acid levels. As an indicator of bone health, sCr could serve as a useful measure of skeletal muscle mass and help identify potential markers for future osteoporosis research (Shaohui et al., 2024). Tangeretin treatment significantly enhanced the levels of serum calcium, phosphorous, and creatinine in ovariectomized rats confirming its alleviating potency against estrogen deficiency prompted osteoporosis in rats.
Osteoclast maturation and bone modeling were primarily controlled by the RANKL/RANK/OPG signaling pathway. RANKL, a ligand of NF-κB receptor, is secreted by osteoblasts, osteocytes stimulate the differentiation of preosteoclast and trigger the osteoclast adherence to the bone tissue (Boyce & Xing, 2007). Wnt signaling in osteoblasts and osteocytes stimulates osteoprotegerin synthesis, a cytokine receptor for NF-κB ligand (RANKL). This process inhibits osteoclast formation (Li et al., 2000; Seibel et al., 2013), leading to reduced bone resorption (Tobeiha et al., 2020). Estrogen interacts with its receptor to enhance osteoprotegerin and suppress the action of RANKL, thereby inhibiting osteoclast formation and bone resorption activity (Cheng et al., 2022). The receptor activator of NF-κB (RANK) is present on osteoclasts and is triggered when it binds with RANKL but is inhibited when it binds with OPG (Bado et al., 2017). Estrogen regulates both RANKL and OPG, increasing OPG expression and decreasing RANKL consequently reducing bone resorption. Additionally, estrogen attenuates osteoclast differentiation and promotes osteoclast apoptosis by enhancing TGFβ synthesis. In the absence of estrogen, RANKL expression is elevated, leading to increased osteoclast formation (Eastell et al., 2016).
Regulation of RANKL/RANK/OPG system is influenced by endogenous factors such as hormones, cytokines, and mesenchymal transcription factors (Hofbauer & Heufelder, 2001; Nardone et al., 2014). The estrogen deficiency enhances the levels of IL-7 which activates T cells and thereby triggers the proinflammatory cytokines and eventually formation of osteoclast (McLean,2009; Weitzmann & Pacifici, 2006). Antiresorptive drugs such as denosumab bind and inhibit the function of RANKL and alleviate osteoporosis (Pang et al., 2020). In our study also tangeretin, a potent anti-inflammatory bioflavonoid, potentially enhances the OPG and inhibited the RANKL, thereby enhancing key transcription factors Runx2 and Osx essential for osteoblast differentiation and skeletal morphogenesis. The antiresorptive potency of tangeretin was persuasively confirmed with our BMD and biomechanical analysis of femur bone in ovariectomized rats. Tangeretin increased the BMD and improved the biomechanical property in ovariectomized rats.
Conclusion
Osteoporosis is a silent global threat for women at the menopausal stage, and it has a severe impact on their physical and economic status. Estrogen deficiency is one of the prime factors that enhances bone turnover, and most of the currently available antiresorptive drugs target estrogen supplementation to treat and prevent osteoporosis but these drugs cause severe side effects and are not economically feasible. Therefore, we examined the antiresporative potency of a bioflavonoid tangeretin since they are safe and cost-effective. Tangeretin treatment prevented excess weight gain and improved the BMD, and biomechanical properties in ovariectomized rats. Tangeretin effectively stimulated the estrogen levels, increased the serum calcium, phosphorous, creatinine levels, and attenuated the serum TRAP levels. It significantly attenuated inflammatory stimulating cytokines, RANKL, and increased the OPG levels, thereby increased the transcription factors essential for osteoblast differentiation in ovariectomized rats. Our results confirm tangeretin may be a potent alternative to the currently available antiresorptive drugs.
Footnotes
Abbreviations
ACP: Acid phosphatase; BGP: Bone GLA protein; BMD: Bone mineral density; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; OPG: Osteoprotegerin; Osx: Osterix; RANK: Receptor activator of nuclear factor-κB; RANKL: Receptor activator of nuclear factor-κB ligand; Runx2: Runt-related transcription factor 2; sCr: Serum creatinine; TNF-α: Tumor necrosis factor-alpha; TRAP: Tartrate-resistant acid phosphatase.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work has been approved by the Institutional Animal Ethical Committee of Health Management Center, Affiliated Hospital of North Sichuan Medical College, Nanchong City, Sichuan Province, P. R. China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
