Abstract
Background
Diabetes mellitus is a complex metabolic condition defined by hyperglycemia, leading to various difficulties, including diabetic nephropathy. Diabetic nephropathy is a serious condition of diabetes, defined by progressive kidney injury and dysfunction.
Objectives
In this study, we committed to assessing the therapeutic effects of angelicin against streptozotocin (STZ)-induced diabetic nephropathy in rats.
Materials and Methods
Diabetic nephropathy was induced in the experimental rats by injecting 65 mg/kg of STZ. The rats with diabetic nephropathy were subsequently administered angelicin and/or standard drug metformin for a duration of 10 weeks. Upon conclusion of the treatments, the body weight, food and water consumption, blood glucose, and kidney index in the experimental rats were assessed. The concentrations of kidney function markers, inflammatory cytokine concentrations, and oxidative stress-related marker levels were assessed. The kidneys of the experimental rats underwent histological examinations.
Results
The administration of angelicin significantly increased body weight and subsequently decreased glucose and kidney index in rats with STZ-induced diabetic nephropathy. In addition, angelicin treatment reduced the concentrations of renal dysfunction indicators in diabetic nephropathy rats. Angelicin significantly decreased the inflammatory response and oxidative stress by enhancing antioxidant levels in rats with diabetic nephropathy. The histological examination results corroborated the therapeutic efficacy of angelicin in treating diabetic nephropathy.
Conclusion
The current results suggest that angelicin may effectively alleviate diabetic nephropathy induced by STZ in rats. Overall, our results highlight that angelicin may be a beneficial therapeutic option for diabetic nephropathy.
Introduction
Diabetes mellitus is a chronic metabolic condition marked by increased blood glucose due to the body’s inability to adequately generate or utilize insulin. This condition can be attributed to several genetic and environmental factors, leading to a spectrum of clinical presentations, from asymptomatic to life-threatening complications. The prevalence of diabetes has been escalating globally, constituting a major public health concern (NCD-RisC, 2024). The complications associated with diabetes can be categorized as either microvascular or macrovascular. Microvascular complications have a higher prevalence than macrovascular complications, including nephropathy, retinopathy, and neuropathy (Blaibel et al., 2024). Diabetic nephropathy is a severe issue of diabetes, defined by progressive kidney injury and dysfunction. Patients with diabetic nephropathy are at high risk of progressing to end-stage renal disease (ESRD), requiring dialysis or kidney transplantation, which can considerably affect the quality of life and impose a significant economic burden on healthcare systems. The pathogenesis of diabetic nephropathy comprises a complex interaction between metabolic and hemodynamic causes, resulting in a gradual decline in renal function (Li et al., 2022). An increase in the cases of diabetes, especially type-2 diabetes, has resulted in a global epidemic of this condition, with significant public health implications. Chronically elevated blood glucose levels, hypertension, and other factors can trigger a cascade of events, including glomerular and tubular injury, inflammation, and fibrosis, eventually resulting in clinical manifestations of diabetic nephropathy.
The burden of diabetic nephropathy is substantial, both for individuals and healthcare systems. Efforts to prevent and manage diabetic nephropathy, such as tight glycemic control and the use of targeted pharmacological interventions, have been the focus of extensive research and clinical practice (Pelle et al., 2022). The pathophysiology of diabetic nephropathy is multifaceted, including a complex interplay between hemodynamic and metabolic causes. It has been suggested that high glucose and altered renal hemodynamics can participate in the development of this condition. Specifically, high glucose levels can induce oxidative stress, inflammation, and extracellular matrix deposition within the glomeruli and tubulointerstitium, leading to glomerular and tubular necrosis and, ultimately, the onset of renal fibrosis and tubular atrophy. Furthermore, the high glucose environment can also trigger epithelial-to-mesenchymal transition in renal tubular epithelial cells, a process that has been implicated in the irreversible progression of tubulointerstitial fibrosis (Sugahara et al., 2021).
The current standard of care for diabetes and its associated kidney disease involves intensive glycemic control, blood pressure management, and the treatment of hyperlipidemia. However, these conventional therapies are often limited by side effects, reduced efficacy over time, and the inability to fully prevent or reverse the progression of diabetic nephropathy (Sawaf et al., 2022). Consequently, there is a growing need for alternative treatment options that can address the multifaceted pathogenesis of this condition. Plant-derived bioactive compounds have emerged as a useful avenue for the management of diabetes and its problems, offering the potential for improved efficacy, better tolerability, and novel mechanisms of action (Josa et al., 2024). Angelicin is a well-known bioactive compound, which belongs to the furanocoumarin class and is mostly found in various medicinal plants such as Angelica archangelica, Angelica dahurica, and Angelica sinensis. Angelicin was already suggested to have several biological properties, such as anti-cancer (Zhang et al., 2023), anti-inflammatory (Liu et al., 2013), anti-viral (Cho et al., 2013), anti-periodontitis (Li et al., 2018), anti-asthma (Wei et al., 2016), and anti-arthritic (Tian et al., 2021) effects. Nevertheless, the salutary roles of angelicin against diabetic nephropathy have not been explored yet. Therefore, in this study, we committed to assessing the beneficial effects of angelicin against streptozotocin (STZ)-induced diabetic nephropathy in rats.
Materials and Methods
Chemicals
Angelicin and STZ were procured commercially from Sigma–Aldrich, USA. The test kits utilized in this study for the evaluation of biochemical markers were obtained from Abcam and Elabscience, USA, respectively.
Animals
Sprague–Dawley rats were utilized in the current work. The rats were caged in sterilized polypropylene enclosures inside controlled laboratory conditions, maintaining a temperature of 22°C–26°C and a humidity of 40%–60%. A 12-h cycle of alternating light and darkness was instituted. Throughout the study duration, all rats were given free access to a standard diet and purified drinking water. Before initiating the study, all rats were allotted a 7-day acclimatization period in a laboratory setting.
Treatment Groups
After a 1-week acclimation, the experimental rats were categorized into four groups (groups I–IV), each comprising six rats. The normal control group (group I) received only standard food and was provided along with a buffered saline solution without any drugs. The rats in group II were administered a single dose of STZ at a dosage of 65 mg/kg to induce diabetic nephropathy. The rats were administered glucose (0.5%) to avert mortality due to sudden hypoglycemia. Following three days of STZ administration, blood glucose was assessed. Rats exhibiting glucose levels over 250 mg/dL were marked as diabetic and designated for additional testing. The treatment group (group III) consisted of rats with STZ-induced diabetic nephropathy that received a dose of 50 mg/kg of angelicin for a duration of 10 weeks. In the standard control group (group IV), rats were induced with diabetic nephropathy and received a dosage of 350 mg/kg of metformin, a standard drug, for a duration of 10 weeks. The body weight of each rat was determined by using an electronic weighing balance. During the study period, the food and water intake of the rats was assessed accurately. The urine sample was collected using metabolic cages. The glucose level was tested using a commercial glucometer (Roche, Germany).
At the completion of treatments, the rats were subsequently sacrificed under pentobarbital sodium (60 mg/kg) anesthesia. Serum was prepared from the blood using centrifugation (5,000 rpm/min for 10 min) and subsequently frozen for further analyses. The kidneys were promptly excised and weighed; one portion was stored in paraformaldehyde (4%) for histological study, while the remaining kidneys were rapidly frozen for additional analyses. The kidney index was computed using the following formulas: kidney index = kidney weight (mg)/body weight (g).
Analysis of Biochemical Markers
The glycated hemoglobin (HbA1c) concentrations were assessed utilizing kits from Abcam, USA. The HbA1c levels were evaluated by assessing the absorbance at 450 nm. The concentrations of urinary albumin, urinary glucose, N-acetyl-beta-
Analysis of Oxidative Stress Marker Levels
The excised kidneys were homogenized with a saline solution and the suspension was centrifuged at 6,000 rpm for 15 min. The resultant supernatant was employed to measure the lipid peroxidation and antioxidant biomarker levels utilizing the commercial assay kits. The concentrations of catalase (CAT), glutathione (GSH), superoxide dismutase (SOD), glutathione peroxidase (GPx), and malondialdehyde (MDA) were evaluated utilizing the kits. The tests were carried out in triplicate as per the manufacturer’s indicated protocols (Elabscience, USA).
Analysis of Pro-inflammatory Cytokines
The quantities of interleukin (IL)-1β, IL-6, and tumor necrosis factor alpha (TNF-α) in the renal tissue homogenates were evaluated using standard diagnostic kits (Elabscience, USA). The tests were carried out as per the manufacturer’s specifications for the kits.
Histopathological Analysis
Tissue samples were collected and treated with a 10% neutral formalin solution to evaluate the histopathological changes in kidney structure. Subsequently, the tissues were paraffinized, sectioned to a 5 µm, and stained with eosin-hematoxylin. Lastly, the kidney tissues were assessed microscopically at 200× magnification to assess the histological alterations.
Statistical Analysis
The results were evaluated utilizing GraphPad Prism, and the results are shown as a mean ± SD of three replicates. The data are assessed using one-way ANOVA and Duncan’s multiple range test (DMRT) analyses, with a significance set at p < .005.
Results
Effect of Angelicin on Food Intake, Water Intake, and Body Weight of Experimental Rats
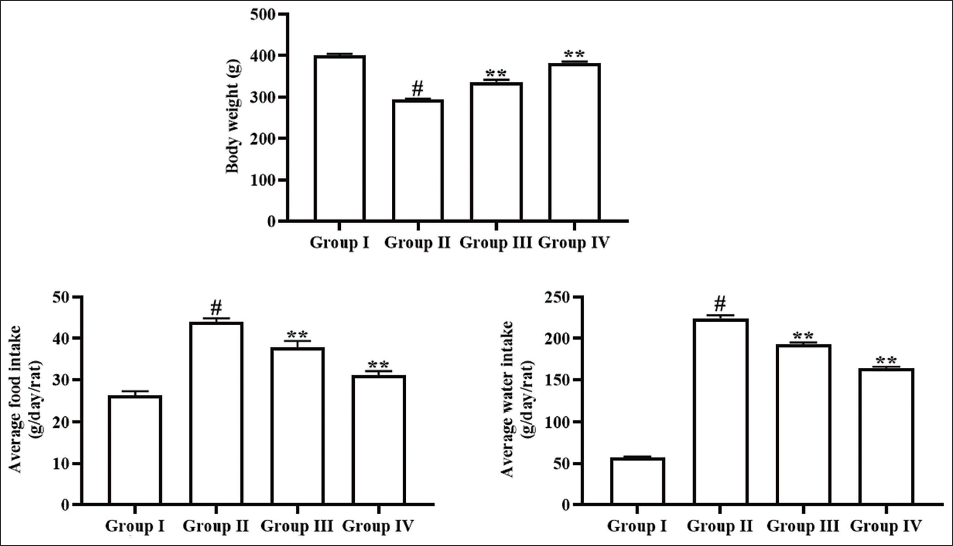
The effects of angelicin on food intake, water intake, and body weight levels in experimental rats were evaluated, as illustrated in Figure 1. The rats with STZ-induced diabetic nephropathy exhibited a considerable reduction in body weight and a subsequent elevation in both food and water consumption. Whereas the angelicin treatment at a dosage of 50 mg/kg markedly elevated the body weight and diminished the food and water consumption of the rats with STZ-induced diabetic nephropathy. These effects of angelicin treatment are further corroborated by the findings related to metformin, which also increase body weight and reduce both food and water intakes in STZ-induced diabetic nephropathy rats (Figure 1).
Effect of Angelicin on Blood Glucose, HbA1c, and Kidney Index in Experimental Rats
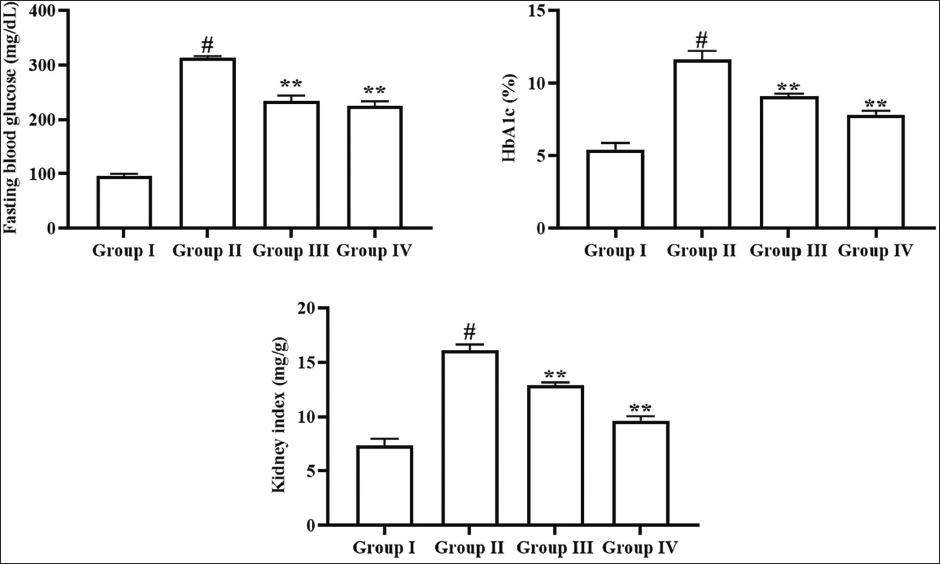
The concentrations of blood glucose, HbA1c, and kidney index of experimental rats were presented in Figure 2. The diabetic nephropathy rats exhibited a remarkable elevation in blood glucose, HbA1c, and kidney index in comparison to the control. However, the angelicin treatment at a dosage of 50 mg/kg showed a considerable diminution in the blood glucose, HbA1c, and kidney index in the rats with STZ-induced diabetic nephropathy. Furthermore, the treatment of metformin also significantly diminished these markers, subsequently further corroborating the efficacy of angelicin.
Effect of Angelicin on Kidney Function Markers in Experimental Rats
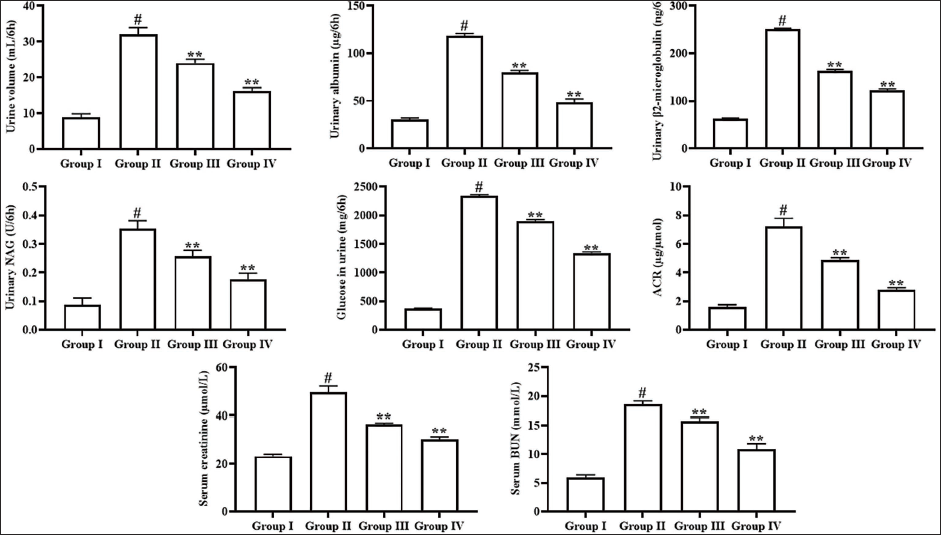
The impact of angelicin on the concentrations of both urinary and serum biomarkers of kidney function was assessed, and the outcomes are shown in Figure 3. The diabetic nephropathy rats illustrated a significant elevation in urine volume, urinary albumin, urinary B2M, urinary NAG, urinary glucose, urinary ACR, and serum creatinine and BUN concentrations in relation to the control. Captivatingly, the treatment of 50 mg/kg of angelicin to the STZ-induced diabetic nephropathy rats resulted in a significant reduction in all these kidney function biomarker levels. The aforementioned results were similarly recorded in the metformin-treated diabetic nephropathy rats, highlighting the efficacy of angelicin.
Effect of Angelicin on Oxidative Stress in Kidney Tissues of Experimental Rats
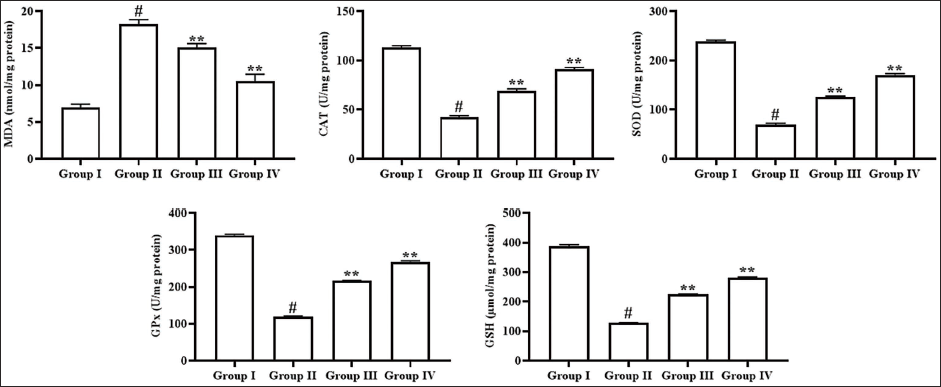
The oxidative stress levels in the kidney tissues of rats were evaluated, and the outcomes are illustrated in Figure 4. The diabetic nephropathy rats exhibited elevated lipid peroxidation marker MDA, alongside diminished CAT, GSH, SOD, and GPx concentrations in their kidneys relative to the control group. However, the treatment with 50 mg/kg of angelicin to the diabetic nephropathy-induced rats revealed a significant increase in the levels of antioxidants, alongside a decrease in MDA in their renal tissues. Moreover, metformin administration also diminished MDA levels and elevated antioxidants in the kidneys of diabetic nephropathy rats, thereby highlighting the antioxidant activity of angelicin.
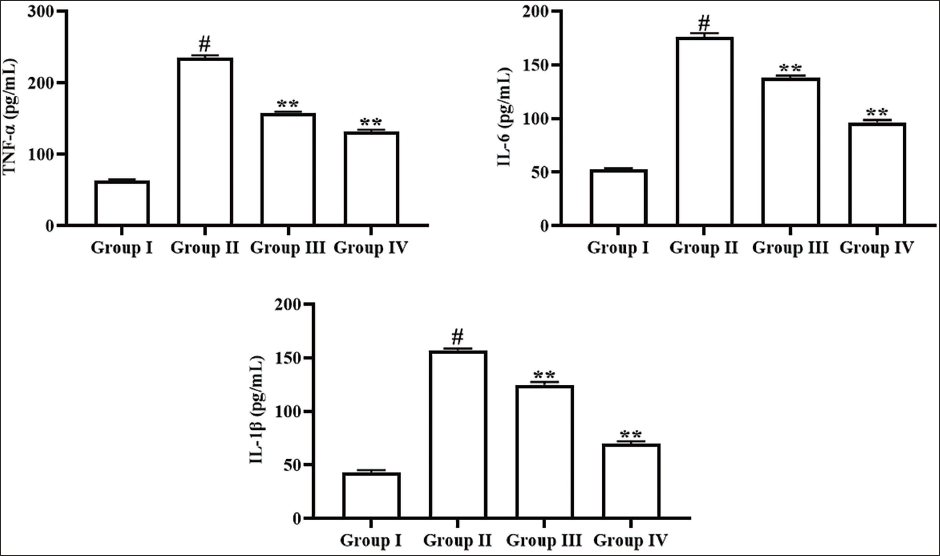
Effect of Angelicin on Inflammatory Cytokines in Kidney Tissues of Experimental Rats
The effects of angelicin treatment on the inflammatory cytokines were assessed in the kidney tissues, and the outcomes are shown in Figure 5. The rats with diabetic nephropathy exhibited substantial elevation in the IL-1β, IL-6, and TNF-α concentrations in their kidneys compared with controls. Remarkably, the treatment of 50 mg/kg of angelicin considerably decreased these cytokine concentrations in the kidney tissue of diabetic nephropathy rats. These effects of angelicin are further corroborated by the results of metformin treatment, which likewise reduced the cytokine levels in the diabetic nephropathy rats.
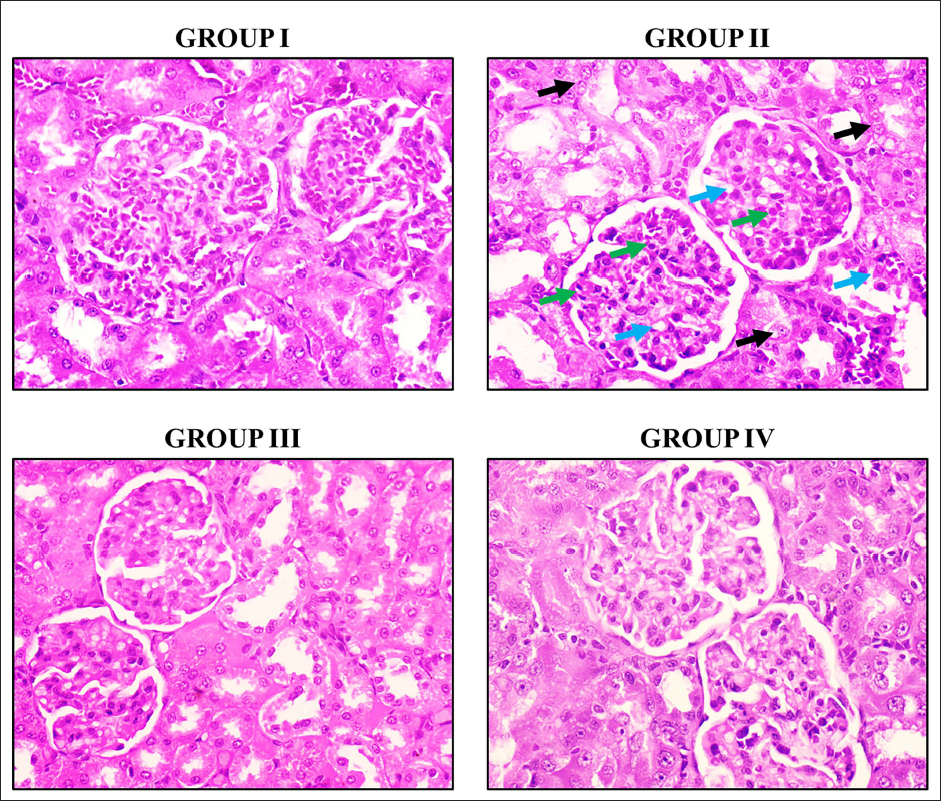
Effect of Angelicin on Kidney Histopathology of Experimental Rats
The histopathological analysis was conducted on the kidneys of the rats, and the outcomes are depicted in Figure 6. The kidneys of control rats displayed a typical morphology with distinct renal histoarchitectures. However, the kidneys of the diabetic nephropathy rats displayed glomerular atrophy, renal tubular epithelial cell injury, and heightened infiltration of inflammatory cells. Captivatingly, the angelicin treatment at a concentration of 50 mg/kg successfully alleviated histological abnormalities in the kidneys of the diabetic nephropathy-induced rats. Furthermore, metformin also diminished the histological abnormalities in the kidneys of the diabetic nephropathy-induced rats.
Discussion
Diabetes mellitus is a multifaceted metabolic illness marked by hyperglycemia, resulting in numerous consequences, including diabetic nephropathy, a major contributor to ESRD. Understanding physiological alterations can offer useful insights into the pathophysiology of diabetic nephropathy (Block et al., 2022). The STZ-induced diabetic rat model is a widely used experimental approach to study diabetic nephropathy. This model mimics several hallmarks of the human condition, including hyperglycemia, polyuria, proteinuria, and renal dysfunction. Studying the alterations in body weight, food and water consumption in this model can offer critical information about the metabolic derangements associated with the disease and uncover the underlying mechanisms driving the onset of diabetic nephropathy (Lennikov et al., 2024). Previous studies have already explored the significance of these parameters in the diabetic nephropathy model. A work by Zhou et al. (2024) demonstrated that STZ-injected rats displayed a considerable decrease in body weight and elevated food and water consumption than the control. Another study by Lv et al. (2024) further corroborated these findings, reporting that STZ-administered diabetic rats illustrated reduced body weight and elevated food and water intakes. The findings from these studies highlight the importance of closely monitoring these metabolic parameters in the management of diabetic nephropathy. Therefore, the analysis of body weight, food and water consumption in the diabetic nephropathy model can offer useful insights into the onset of the disease and provide important readouts to assess the efficacy of therapies. The present findings evidenced that the STZ-induced rats with diabetic nephropathy exhibited reduced body weight and a subsequent elevation in both food and water consumption than the control. Fascinatingly, the angelicin treatment significantly increased the body weight and reduced the food and water consumption of the rats with diabetic nephropathy, which is further highlighted by the findings of metformin treatment.
The pathogenesis of this condition involves a complex interplay between several factors, including glomerular and tubulointerstitial changes. Early diagnosis, monitoring of the condition, and interventions are crucial, as timely intervention can help slow disease progression and mitigate adverse outcomes. Hyperglycemia is an essential driver of the onset of diabetic nephropathy, resulting in structural and functional alterations in the kidneys, like glomerular hypertrophy, mesangial expansion, and tubulointerstitial fibrosis (Młynarska et al., 2024). The numerous urinary and serum biomarkers can serve as a sensitive and specific indicator of diabetic nephropathy. HbA1c, urinary albumin, urinary glucose, urinary NAG, urinary B2M, urinary ACR, serum creatinine, and BUN are among the crucial indicators that have been extensively utilized in the context of diabetic nephropathy. HbA1c is a broadly utilized indicator for studying long-term glycemic control in individuals with diabetes. In diabetic nephropathy, HbA1c levels can provide insights into the degree of chronic hyperglycemia and its impact on the kidneys (Butler et al., 2024). Urinary albumin is a well-established marker of glomerular dysfunction in diabetic nephropathy. Increased urinary albumin excretion, or albuminuria, is an early indicator of kidney damage and correlates with the severity of the disease. Urinary glucose is another marker that can be elevated in diabetic nephropathy, reflecting impaired tubular reabsorption of glucose (Lalić et al., 2024). NAG is a lysosomal enzyme found in the proximal renal tubules. Increased urinary NAG levels were noted in diabetic patients, even in the absence of overt nephropathy, and have been shown to parallel the degree of kidney injury (Kim et al., 2016). Similarly, urinary B2M, a marker of tubular dysfunction, is elevated in diabetic nephropathy. NAG and B2M are more sensitive and specific biomarkers of tubular injury, which may provide valuable information about the tubulo-interstitial changes that precede and accompany the onset of diabetic nephropathy (Monteiro et al., 2016). The urinary ACR is a valuable tool for early diagnosis and monitoring of diabetic nephropathy. This ratio provides a more accurate assessment of albumin excretion than spot urine albumin alone, as it adjusts for variations in urine dilution (Zhou et al., 2024). In addition to these urinary biomarkers, serum creatinine and BUN are commonly used to evaluate renal function in diabetic patients. Elevated levels of these parameters indicate a decline in glomerular filtration rate, which is a hallmark of advanced diabetic nephropathy. By analyzing the changes in these biomarker levels, researchers can gain a better understanding of the connection between glycemic control, renal function, and the onset of diabetic nephropathy. Such insights can inform the onset of novel diagnostic biomarkers and targeted therapeutic strategies to enhance the treatment and prognosis of patients with this debilitating complication of diabetes (Butt et al., 2024). In this study, we found that the diabetic nephropathy rats displayed elevated urinary albumin, urinary B2M, urinary NAG, urinary glucose, urinary ACR, and serum creatinine and BUN concentrations in comparison to the control. Interestingly, the treatment with angelicin successfully reduced all these kidney function biomarker levels. Therefore, it was evident that angelicin can improve kidney function in diabetic conditions by its renoprotective properties.
Oxidative stress plays an essential role in the onset of diabetic nephropathy. The disruption in the ROS accumulation and antioxidant mechanisms results in an overabundance of free radicals, leading to oxidative damage to cellular macromolecules. The instability and high reactivity of free radical species drive them to seek stability by obtaining electrons from nearby molecules, activating a cascade of chain reactions that ultimately cause cellular injury and contribute to disease development. In the context of diabetic nephropathy, this oxidative damage affects various renal cell types, including glomerular, tubular, and endothelial cells, resulting in impaired renal function and structural abnormalities (Wang & Zhang, 2024). The mechanisms by which oxidative stress contributes to diabetic nephropathy are multifaceted and involve a complex interplay of biochemical pathways. Notably, hyperglycemia, a hallmark of diabetes, promotes the generation of ROS through several pathways and increased mitochondrial ROS production. In addition to increasing ROS production, hyperglycemia impairs antioxidant defense mechanisms by promoting glycation of antioxidant enzymes, reducing their activity. The resulting oxidative stress initiates a cascade of downstream events, including activation of pro-inflammatory signaling pathways, increased extracellular matrix production, podocyte dysfunction, and tubular damage, all of which participate in the onset of diabetic nephropathy (Caturano et al., 2023).
The reduction of antioxidant protection systems in the kidneys under diabetes conditions has been widely documented. This results in the ROS generation, leading to increased oxidative damage to renal cells, participating in the onset of diabetic nephropathy. The reduction in antioxidant enzyme activity is thought to be due to the glycation of the enzymes, a process in which glucose molecules attach to proteins, altering their structure and function (Adeshara et al., 2024). The role of GSH in protecting cells against oxidative stress has been well reported. GPx and CAT are key antioxidants that facilitate the conversion of hydrogen peroxide into water. The GSH system is critical for protecting cells from oxidative damage and plays an essential role in maintaining redox homeostasis (Dilworth et al., 2024). MDA is a byproduct of lipid peroxidation, a chain reaction initiated by ROS that damages lipids in cell membranes and lipoproteins. Elevated MDA in the blood and urine are indicative of increased oxidative stress and lipid damage. In diabetic nephropathy, increased MDA levels have been observed in the kidneys, reflecting the extent of oxidative damage to renal tissues (Chang et al., 2005). Understanding the precise molecular mechanisms by which oxidative stress contributes to renal damage may lead to the development of targeted therapies that can prevent or delay diabetic nephropathy, thereby reducing the burden of this devastating complication of diabetes (Weinberg et al., 2024). Furthermore, the modulation of oxidative stress is recognized as a critical target for addressing both metabolic and hemodynamic derangements associated with diabetic nephropathy (Dawi et al., 2024). Therapeutic interventions aimed at reducing oxidative stress have demonstrated efficacy in mitigating diabetic nephropathy. The present findings clearly indicate that angelicin treatment of rats with diabetic nephropathy demonstrates a considerable elevation in antioxidant concentrations, concurrently reducing the MDA level in their kidney tissues. These findings support the antioxidant effects of angelicin.
Inflammation is a crucial player in the advancement of diabetic nephropathy. The onset of diabetic nephropathy comprises a complex interplay of various causes, including hyperglycemia, hemodynamic changes, and local growth factors, culminating in extracellular matrix accumulation, glomerular sclerosis, tubular atrophy, and ultimately, kidney fibrosis (Hu et al., 2024). Among the various proposed theories concerning the pathogenesis of diabetic nephropathy, inflammation has emerged as a central pathway driving its development and progression. The diabetic milieu, characterized by increased glucose levels, triggers a cascade of inflammatory responses within the kidney. These inflammatory cascades participate in the structural and functional deterioration of the kidney, ultimately leading to ESRD. Activation of the immune system has been implicated in the onset of diabetic nephropathy, with immune cells infiltrating the kidney and releasing several inflammatory mediators. The increase of inflammatory cytokines in kidneys and urine indicates that macrophages play an essential role in the onset of kidney disease progression (Duran-Salgado & Rubio-Guerra, 2014). IL-1β, IL-6, and TNF-α are pivotal pro-inflammatory cytokines that have been implicated in the pathogenesis of diabetic nephropathy, contributing significantly to the inflammatory cascade within the kidney.
Dysregulation of the inflammatory response, with an increased inflammatory cytokines and a lack of anti-inflammatory mechanisms, contributes to the chronic inflammation observed in diabetic nephropathy (Donate-Correa et al., 2015). Specifically, IL-1β, an imperative inflammatory cytokine, plays an essential role in the commencement and amplification of the inflammatory reaction in diabetic nephropathy. It is primarily produced by activated macrophages and other immune cells and shows its potential by binding to the IL-1 receptor on target cells, triggering intracellular pathways that result in the accumulation of other pro-inflammatory cytokines. This cytokine promotes the influx of immune cells into the kidney, exacerbating inflammation and tissue injury. IL-6, a pleiotropic cytokine, also participates in the onset of diabetic nephropathy. Elevated IL-6 was noted in the circulation and kidney tissue of individuals with diabetic nephropathy, and it has been shown to contribute to various aspects of kidney damage, including mesangial cell proliferation, extracellular matrix accumulation, and podocyte dysfunction (Reddy et al., 2024). TNF-α, another key pro-inflammatory cytokine, participates in the onset of diabetic nephropathy. It is produced by a variety of cells within the kidney, including mesangial, tubular epithelial, and infiltrating immune cells. TNF-α demonstrates its effects by binding to its receptors on target cells, activating pathways that result in the accumulation of other inflammatory mediators. Understanding the intricate roles of IL-1β, IL-6, and TNF-α in the inflammatory processes underlying diabetic nephropathy may lead to the advancement of targeted therapies focused on regulating inflammation and protecting kidney function (Joumaa et al., 2025). The present findings evidenced that the angelicin treatment successfully diminished the IL-1β, IL-6, and TNF-α concentrations in the kidneys of diabetic nephropathy-induced rats. These findings highlight that angelicin has potent anti-inflammatory activity.
Conclusion
The current results suggest that angelicin may effectively alleviate diabetic nephropathy in rats. The treatment with angelicin results in a marked decrease in glucose level and an elevation in body weight in rats with diabetic nephropathy. Moreover, the angelicin administration showed a reduction in renal dysfunction markers, inflammatory responses, and oxidative stress by enhancing antioxidant levels in rats with STZ-induced diabetic nephropathy. Overall, our findings highlight that angelicin may offer a beneficial therapeutic option for diabetic nephropathy.
Footnotes
Abbreviations
ACR: Albumin-to-creatinine ratio; B2M: Beta-2 microglobulin; BUN: Blood urea nitrogen; CAT: Catalase; GSH: Glutathione; HbA1c: Glycated hemoglobin; IL: Interleukin; MDA: Malondialdehyde; NAG: N-Acetyl-beta-
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The present study was approved by the institutional animal ethical committee, Affiliated Hospital of Hebei University, Baoding, Hebei, 071000, China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project is supported by Baoding Science and Technology Plan Project (Grant No. 2341ZF140).
Informed Consent
Not applicable.
