Abstract
Background
Alcoholic liver disease, a consequence of excessive alcohol consumption, is a major factor contributing to global morbidity and mortality. Salvianolic acid B (Sal B) has demonstrated potential in alleviating ethanol-induced hepatic damage; however, the precise mechanisms, particularly those involving the modulation of gut microbiota, remain unexplored.
Objectives
This study aimed to evaluate the efficacy of Sal B in mitigating acute alcoholic liver injury and rectifying dysbiosis of intestinal microbiota in mice.
Materials and Methods
Alcohol was used to induce liver injury, and Sal B was used for prevention in mice. Serum biochemical parameters were tested using an automatic hematology analyzer. Pathological alterations in hepatic tissue were assessed using hematoxylin–eosin staining. The mRNA expression levels of specific genes were quantified via reverse transcription polymerase chain reaction, while protein expression was evaluated through Western blot analysis. The composition of the intestinal microbiota in mice was characterized by sequencing the V3–V4 region of the 16S rRNA gene.
Results
Administration of Sal B resulted in decreased levels of aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase, as well as increased levels of total protein and albumin, in mice with acute alcohol-induced liver injury. Furthermore, Sal B effectively reduced hepatic cell degeneration and necrosis, suppressed the expression of toll-like receptor 4, nuclear transcription factor-κB, and phosphorylated nuclear transcription factor-κB proteins in liver tissue. Additionally, Sal B maintained intestinal function in the host.
Conclusion
Sal B may ameliorate ethanol-induced hepatic injury in mice by modulating the gut microbiota and influencing the TLR4/NF-κB signaling pathway.
Keywords
Introduction
The pivotal role of the gut microbiota in human health and disease represents a captivating and rapidly evolving area of research (Kalkan et al., 2023). Clinical evidence has demonstrated a strong association between gut microbiota dysbiosis and the onset of Parkinson’s disease, allergies, cardiovascular disease, liver disease, diabetes mellitus, and obesity (Yang et al., 2022). This growing interest has catalyzed further research and the development of therapeutic strategies aimed at regulating the gut microbiome (Aar et al., 2023).
Researchers have found that dysbiosis resulting from ethanol consumption plays a significant role in both the early and advanced stages of alcoholic liver disease (ALD) (Jew & Hsu, 2023). Alcohol intake can disrupt intestinal integrity and barrier function, allowing an influx of Gram-negative bacteria into the bloodstream, leading to the accumulation of endotoxins (Shang et al., 2022). Endotoxins are subsequently transported to the liver via portal vein circulation, where they activate downstream signaling pathways by binding to toll-like receptor 4 (TLR4) on Kupffer cells, thereby exacerbating liver damage (He et al., 2024). Probiotic treatment, transferring fecal microbiota, and adding dietary fiber have shown effectiveness in ameliorating liver damage in animal studies of ALD by enhancing intestinal barrier function (Li et al., 2021). Strategies targeting the intestinal flora may hold promise in mitigating alcohol-induced hepatic damage by modulating the gut microbiota and hepatic environment (Rattan & Shah, 2022).
Currently, a growing number of researchers are dedicated to developing and utilizing effective natural and bioactive substances for the prevention and treatment of ALD and its associated conditions. Moreover, with the rapid advancements in modern natural products research, natural single compounds, natural extracts, and natural compound formulations, particularly traditional Chinese medicine formulations, are playing an increasingly significant role in the treatment of ALD (Sardar et al., 2024).
To reveal this hypothesis, the current research study was designed to investigate the effects of Sal B on alcohol-induced liver injury in mice and the inner protective mechanism. The results obtained would yield a promising candidate among natural products for the remediation of alcohol-induced liver injury, further enhancing our comprehension of how SalB and gut microbiota modulation impact alcohol-induced liver injury in mice.
Materials and Methods
Chemicals and Reagent
Sal B was purchased from Maidesheng Technology Co., Ltd. (Chengdu, China), while ethanol and other necessary chemicals and materials were obtained from Sangon Biotech (Shanghai, China).
Animals and Experimental Protocols
The male ICR mice were acquired from HFK Bioscience Co. Ltd. (Beijing, China). In the experimental setup, mice were individually housed in cages maintained at a temperature of 18ºC–22ºC and relative humidity of 40%–60%, under a 12-h light/dark cycle, with unrestricted access to water. Following a week acclimatization period, 60 mice were randomly and evenly divided into 6 groups: normal control (NC), model control (MC), positive control (PC), low-, middle- and high-dosage Sal B (SBL, SBM, SBH). Mice in the NC and MC categories were given 0.2 mL of saline solution by gavage, whereas mice in the PC group were given 100 mg/kg of silymarin once a day. The mice in the three experimental groups were given Sal B at 100 (SBL), 150 (SBM), and 200 mg/kg (SBH) doses. After 15 days, except for the NC group, which received equal volumes of distilled water, the groups were subsequently administered 50% ethanol at a dose of 10 mg/kg (Duan et al., 2024). Following an 18-h fasting period, fecal samples were collected for analysis of intestinal flora. Subsequently, the mice were euthanized via cervical dislocation with 2% amobarbital sodium, and blood samples were collected. The liver was partially fixed in formalin for histological analysis, and the remaining part was kept at −80ºC for later use.
Biochemical Analysis
Concentrations of serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP), as well as increased levels of total protein (TP) and albumin (ALB), were measured with kits (Leadman Biochemical Co., Ltd., Beijing, China).
Histopathological Analysis
The hematoxylin–eosin (H&E) staining process was conducted following an established methodology (Xu et al., 2021). Liver samples were fixed in 10% formalin, embedded in paraffin, sectioned into 4 µM slices, and stained with H&E for histological examination under a light microscope.
Analysis of Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from the liver tissue using the TransZol Up Plus RNA Kit and subsequently reverse transcribed employing the TransScript One-Step RT-PCR SuperMix (TransGen Biotechnology Co., Ltd., Beijing, China). The β-actin gene was used as an internal control, and mRNA expression levels were quantified using the 2−∇∇CT method. Primers were employed as previously described (Wen et al., 2024).
Western Blotting (WB) Analysis
WB analysis was conducted using standardized procedures (Fang et al., 2023). Liver samples were processed for protein extraction and separated on a 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gel, then transferred to a polyvinylidene fluoride (PVDF) membrane. After blocking with skimmed milk, membranes were incubated with primary antibodies, followed by washing and incubation with secondary antibodies for 2 h. Protein bands were analyzed using ImageJ software.
Analysis of Intestinal Microbiota
The genomic DNA of mouse fecal flora was extracted using the Stool Genomic DNA Kit G (TransGen Biotechnology Co., Ltd., Beijing, China), followed by 16S rRNA gene sequencing conducted by LC-Bio Technologies Co., Ltd. (Hangzhou, China). The V3-V4 segment of bacterial 16S ribosomal RNA was repeatedly amplified, measured, and sequenced with general primers (341F and 806R). The Illumina NovaSeq platform was used for sequencing, and DADA2 assembled reads into tags by identifying read overlap. More diagrams were created with the R package (v3.5.2) (Li et al., 2018).
Statistical Analysis
Data are presented as the mean ± SEM. Statistical analyses were conducted using one-way analysis of variance (ANOVA) followed by Tukey’s test for multiple comparisons.
Results
Effect of Sal B on Body Weight and Serum Biochemistry
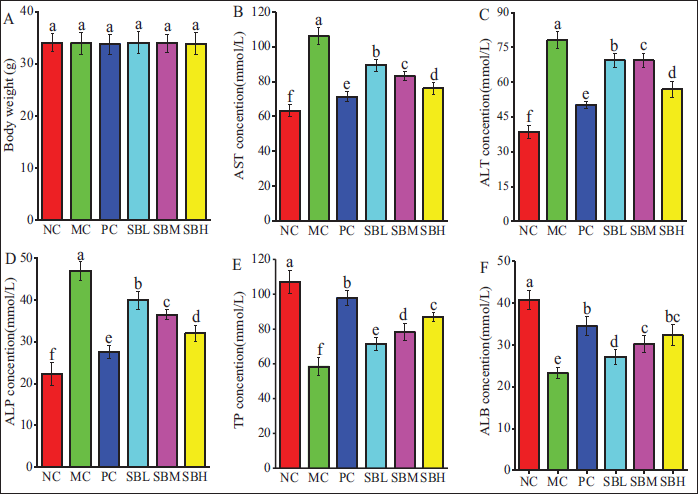
As illustrated in Figure 2A, no significant differences in body weight were observed among the groups after 15 days of intragastric administration. Compared to the NC group, the MC group mice showed a notable increase in serum ALT, AST, and ALP activities, while the concentrations of TP and ALB were notably lower (
Effects of Salvianolic Acid B (Sal B) on Body Weight (A), Serum Aspartate Aminotransferase (AST) (B), Alanine Aminotransferase (ALT) (C), Alkaline Phosphatase (ALP) (D), Total Protein (TP) (E), and Albumin (ALB) (F) in Mice.
Effect of Sal B on Histopathological Variations of Liver
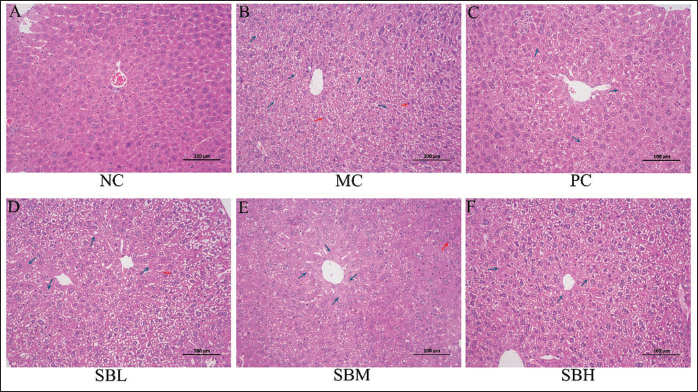
The hepatic lobule structure in the NC group appeared normal, with liver cells exhibiting complete morphology (Figure 3A). Differently, the hepatocyte structure in the MC group displayed disorganization, with pyknotic liver cell nuclei (highlighted in red) and necrotic liver cells (highlighted in blue), leading to extensive liver tissue damage (Figure 3B). Remarkably, pretreatment with three doses of Sal B mitigated the acute ethanol-induced morphological alterations (Figures 3C–3E), particularly in the high-dose Sal B group, which displayed similarities to the PC group (Figure 3F).
Effects of Salvianolic Acid B (Sal B) on Hepatic Morphological Changes of Mice (200× Magnification).
Effect of Sal B on Expression of TLR4, MyD88, NF-κB, and pNF-κB Genes
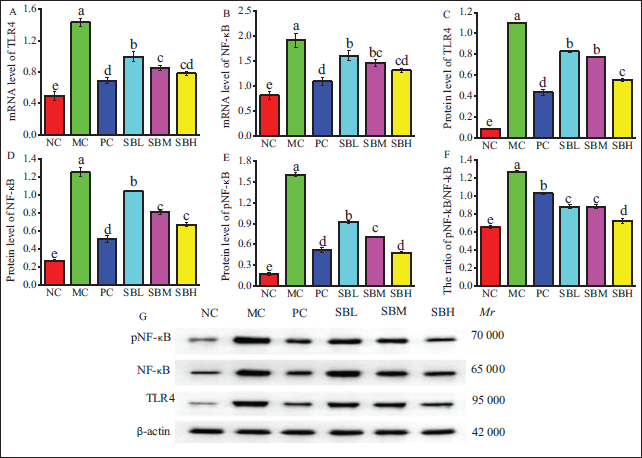
As depicted in Figure 4, the mRNA levels of TLR4, MyD88, and NF-κB p65, as well as the protein levels of TLR4, MyD88, NF-κB p65, and pNF-κB p65, were significantly elevated in the MC group compared to the NC group (
Effects of Salvianolic Acid B (Sal B) on mRNA Expression of Toll-like Receptor 4 (TLR4) (A) and Nuclear Transcription Factor-κB (NF-κB) (B), and Protein Expression of TLR4 (C), NF-κB (D), and Phosphorylated Nuclear Transcription Factor-κB (pNF-κB) (E), the Ratio of pNF-κB/NF-κB (F).
Effect of Sal B on Intestinal Bacteria
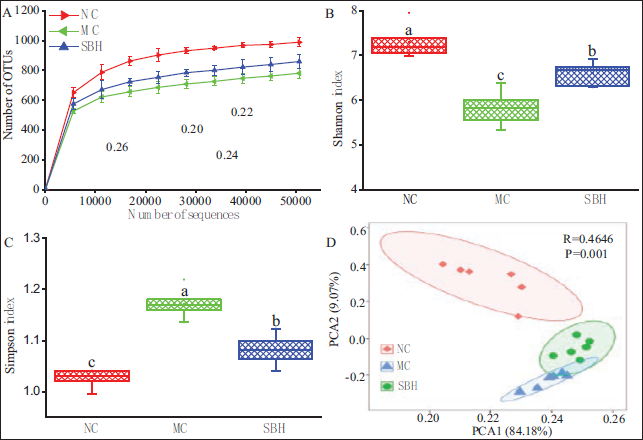
The findings depicted in Figures 2–4 suggest that high-dose Sal B had a significant protective impact on acute ALD mice. Subsequently, fecal samples from the SBH, NC, and MC groups of mice were subjected to high-throughput sequencing to analyze bacterial diversity. Rarefaction curves suggest that the sequencing data collected for this study were sufficient, covering all species in the samples (Figure 5A). Moreover, the Shannon and Simpson indices showed a significant reduction in community diversity and richness in the MC group compared to the NC group (
The Rarefaction Curves (A), Shannon Index (B), Simpson Index (C), and Principal Component Analysis (PCA) Analysis (D) of Intestinal Bacteria in Mice.
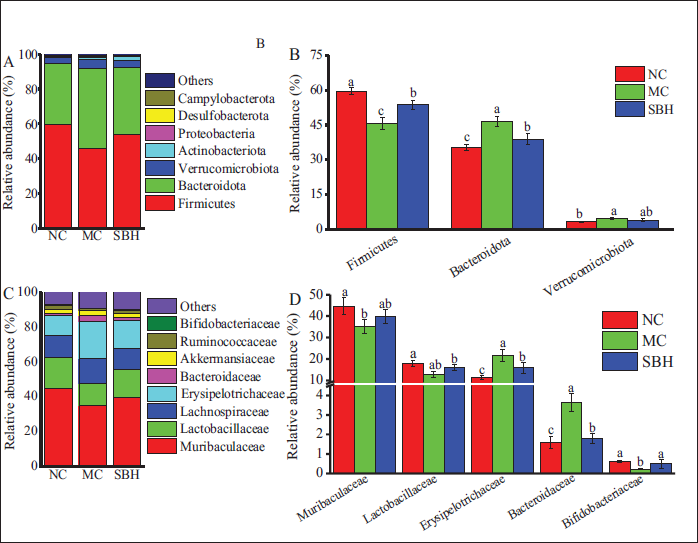
Examination at the phylum level showed that mice in the MC group had increased proportions of Bacteroidetes and
The Abundance of Bacteria at the Phylum (A, B) and Family Levels (C, D).
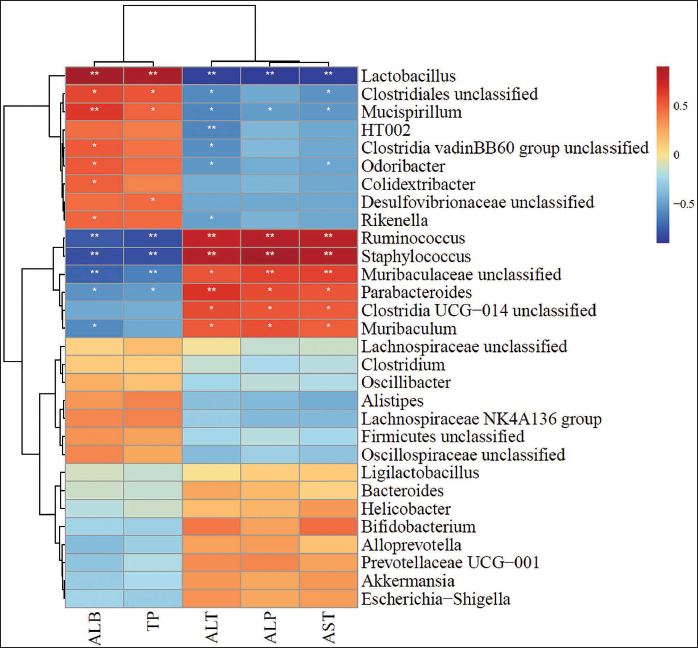
Correlation Analysis of Intestinal Microbiota and Serum Parameters
Serum parameters showed both positive and negative correlations with individual bacterial colonies (Figure 7). In particular,
Spearman Correlation Analysis Between the Abundance of Specific Genera in Fecal Intestinal Bacteria and Serum Parameters.
Discussion
ALD is a condition resulting from prolonged excessive alcohol consumption, which can advance to alcoholic hepatitis, alcoholic liver fibrosis, and ultimately, alcoholic liver cirrhosis and hepatocellular carcinoma (Zou et al., 2019). Serum levels of AST, ALT, ALP, TP, and ALB are commonly utilized as reliable biomarkers for evaluating liver function (Zhao et al., 2024). Moreover, histopathological examination serves as a direct means of assessing the efficacy of potential protective interventions (Chang et al., 2023). Previous studies demonstrated ethanol-induced hepatotoxicity in rats, as evidenced by elevated serum ALT and AST levels. In contrast, pretreatment with Sal B resulted in a significant reduction in serum ALT and AST levels (Hu et al., 2024). Furthermore, rats exposed to ethanol exhibited nuclear pleomorphism, increased cellular size, hepatocyte degeneration, hepatic cord degeneration, and cytoplasmic dissolution. However, the hepatic lesions induced by ethanol were significantly alleviated by Sal B pretreatment (Li et al., 2014). Consistent with previous research, the findings of this study suggest that pretreatment with Sal B effectively mitigated abnormal levels of serum AST, ALT, ALP, TP, and ALB, resulting in a significant reduction in liver cell damage. The results suggest that Sal B may possess a potent protective effect against acute alcohol-induced injury in mice.
Alcohol and its metabolites exert toxic effects on hepatic and intestinal epithelial cells, leading to dysfunction of the liver and gut barrier. This dysfunction facilitates the proliferation of pathogenic bacteria and disrupts the homeostasis of the intestinal flora (Zhang et al., 2021). The dysregulation of the intestinal microbiota has emerged as a significant factor in the advancement of ALD and is now recognized as a potential target for therapeutic intervention (Ohtani & Kawada, 2019). Alcohol consumption has been shown to alter the composition of the intestinal flora and diminish its diversity. The findings from the alpha diversity index indicated a reduction in microbial diversity following alcohol exposure, consistent with previous research (Fan et al., 2019). Furthermore, the findings related to PCA indicated that alcohol exerts a substantial influence on the microbial communities in mice with ALD. Notably, the administration of Sal B was observed to preserve the diversity of intestinal flora in these mice and demonstrated potential effects in modifying gut bacterial diversity. Previous studies have also demonstrated that an alcohol-containing diet in mice leads to significant changes in the structure and composition of intestinal microbiota (Warner et al., 2020).
An increasing body of research has demonstrated that intestinal microbiota is crucial in the onset and progression of various liver diseases via the gut-liver axis (Ağagündüz et al., 2023). Excessive alcohol consumption can lead to an altered composition of intestinal microbiota and dysbiosis, thereby accelerating the progression of ALD (Ma et al., 2021). Clinical studies have demonstrated that in the small intestines of patients with alcoholic liver cirrhosis, intestinal bacteria can proliferate excessively, resulting in an increased abundance of abnormal microbiota (Zhao et al., 2022). Our research indicated that a high dosage of Sal B effectively restored the structural changes in gut microbiota induced by alcohol. These findings suggest that Sal B successfully reversed alcohol-induced alterations in the community structure of gut microbiota. The gastrointestinal microbiota is extensively acknowledged for its substantial contributions to human physiology and metabolism, owing to its diverse functional capabilities. Moreover, it is well-established that the intestinal microbiota plays a crucial role in the progression of liver injury. Probiotics such as
TLR4 and nuclear transcription factor-κB (NF-κB) expression levels in this pathway are important markers of inflammation. Therefore, the TLR4 and NF-κB levels in the inflammatory pathway are important indicators of inflammation (D’Avino et al., 2023). Prior studies have suggested that Sal B may inhibit liver angiogenesis and reduce liver fibrosis by modulating the NF-κB/IkBa signaling pathway (Wang et al., 2012). Additionally, Sal B exhibits anti-inflammatory properties in neonatal cardiomyocytes exposed to LPS, potentially through inhibition of the TLR4-NFκB-TNFα pathway (Wang et al., 2011). Our findings indicate that the pre-administration of Sal B resulted in decreased protein levels of TLR4, NF-κB, and phosphorylated nuclear transcription factor-κB (pNF-κB) in mice experiencing acute alcohol-induced liver injury. These results suggest that the prophylactic efficacy of Sal B in attenuating acute alcohol-induced liver damage is associated with the modulation of the TLR4/NF-κB inflammatory pathway.
Conclusion
Our results demonstrated that Sal B could rectify the deviations of serum biochemistry and decrease hepatic cell degeneration and necrosis induced by alcohol in mice. Moreover, Sal B significantly inhibited the activation of TLR4, NF-κB, and pNF-κB in the liver and maintained intestinal function in the host. The results indicate that Sal B could potentially improve ethanol-induced liver damage in mice by influencing gut microbiota and controlling the TLR4/NF-κB pathway.
Footnotes
Abbreviations
ALB: Albumin; ALD: Alcoholic liver disease; ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; H&E: Hematoxylin–eosin; LPS: Lipopolysaccharide; MC: Model control; NC: Normal control; NF-κb: Nuclear transcription factor-κB; pNF-κB: Phosphorylated nuclear transcription factor-κB; PCA: Principal component analysis; PC: Positive control; RT-PCR: Reverse transcription polymerase chain reaction; Sal B: Salvianolic acid B; SBL: Low-dosage Sal B; SBH: High-dosage Sal B; SBM: Middle-dosage Sal B; TLR4: Toll-like receptor 4; TP: Total protein; WB: Western blotting.
Acknowledgments
The authors are thankful to the Experimental Animal Center, Quanzhou Medical College, Quanzhou, China, for providing all laboratory facilities.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All animal experiments were carried out strictly in compliance with the guidelines set forth by the Institutional Animal Care and Use Committee (IACUC) of Quanzhou Medical College (Approval No. 2021026). To minimize animal distress, anesthesia and analgesia were administered during surgical interventions, and every endeavor was made to limit the number of animals utilized in the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by education and research projects for young and middle-aged teachers of Fujian province (JAT210896).
