Abstract
Background and Objectives
Parkinson’s disease (PD) is a common age-related neurodegenerative disorder. Mitochondrial dysfunction is considered an important initiating factor for the lack of dopaminergic (DA) neurons. Therefore, identifying drugs that effectively protect DA neurons from mitochondrial damage represents a promising therapeutic strategy.
Materials and Methods
In this study, an anti-atherosclerosis (AS) drug, Ziziphora clinopodioides flavonoids (ZCF), was shown to inhibit 1-methyl-4-phenylpyridinium (MPP+)-induced DA neuron damage in vitro and in vivo and this was verified by a variety of experimental methods. Cell viability assays, cell fluorescence staining, Western blot experiments, flow cytometry analysis, and quantitative real-time polymerase chain reaction (qPCR) experiments were conducted to assess the protective effect and mechanism of ZCF on SH-SY5Y cells. Additionally, behavioral tests and immunohistochemistry (IHC) were performed to validate the protective effect of ZCF in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced PD animal model.
Results
ZCF significantly ameliorated MPP+-induced cell viability loss and elevated lactate dehydrogenase (LDH) in SH-SY5Y cells. Moreover, ZCF treatment improved mitochondrial morphological abnormalities, reduced adenosine triphosphate (ATP) content, attenuated mitochondrial membrane potential loss, cell apoptosis, mitoROS levels, and increased the level of tyrosine hydroxylase (TH). Further results indicate that ZCF treatment significantly stimulated the phosphorylation of AMP-activated protein kinase (AMPK) and its downstream target miR-181b. Additionally, the blockage of the level of AMPK with Compound C or knocking down the expression of miR-181b significantly attenuated the protective effect of ZCF. These results indicate that the AMPK/miR-181b axis plays a vital role in the protective effect of ZCF. Moreover, ZCF treatment alleviated MPTP-induced motor dysfunction and loss of TH-positive neurons in the PD mice model.
Conclusion
In brief, our findings suggest that ZCF may have potential applications for the treatment and prevention of PD.
Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disease. It is projected that its prevalence will double in the next 30 years (Tolosa et al., 2021). Research has shown that mitochondrial dysfunction triggers a cascade involving energy deficits, ultimately leading to neuronal damage in PD (Moradi Vastegani et al., 2023). For example, in PD cases and animal models, the activity of mitochondrial-associated protein complex I was significantly reduced, leading to the increase of reactive oxygen species (ROS) and subsequent inhibition of complex I. This vicious cycle leads to excessive oxidative stress and adenosine triphosphate (ATP) depletion, ultimately resulting in the loss of dopaminergic (DA) neurons in the substantia nigra (González-Rodríguez et al., 2021; He et al., 2020). Further proof of mitochondrial dysfunction linked with dopamine cell injury comes from the fact that the activation of the mitochondrial-dependent apoptotic pathway is involved in the neurodegeneration of DA neurons in the substantia nigra of PD (Liu, Liu, et al., 2019). Therefore, the preclusion of mitochondrial dysfunction is a capable strategy for PD therapies.
Ziziphora lopodioides Lam. (ZCL) is widely distributed in Xinjiang, China, Turkey, Mongolia, and Kazakhstan. ZCL has pharmacological results, such as antioxidant and anti-inflammatory, and can treat a variety of diseases, including atherosclerosis (Liu et al., 2023; Wu et al., 2020). Ziziphora clinopodioides flavonoids (ZCF) is a main bioactive total flavonoid compound separated from Z. clinopodioides Lam. It was reported that ZCF may secure myocardial tissue from ischemia–reperfusion damage by reducing ROS-related injury (Li et al., 2018). Moreover, ZCF could weaken the production of plaques in aortic tissue by regulating the PI3K/Akt/mTOR signaling pathway (Ma et al., 2023). However, the effect of ZCF on PD and its underlying mechanism remain elusive.
AMP-activated protein kinase (AMPK) is a conserved serine/threonine protein kinase that plays an energy sensor in many cells and plays a critical role in energy metabolism (Herzig & Shaw, 2018; Steinberg & Carling, 2019). In addition to its effect on energy metabolism, phosphorylation of AMPK at threonine-172 is often thought to have a protective effect on the cardiovascular system (Gélinas et al., 2018). Antecedently, we provided evidence that metformin-mediated AMPK activation defends PC12 cells from H2O2-induced oxidative injury (Zhao et al., 2019). Additionally, AMPK activation has been reported to be an upriver event in miRNA biogenesis in which AMPK can phosphorylate nucleolin in the nucleus to regulate miRNA expression (Gongol et al., 2019). Cheng et al. (2022) found that activation of AMPK/miR-181b relieves endothelial dysfunction and vascular inflammation in mice that have diabetes. Even so, it remains unclear whether the AMPK/miR-181b axis is implicated in the development of PD. In this study, we explored the protective effect of ZCF on SH-SY5Y cells and the PD mice model.
Materials and Methods
Dulbecco’s modified Eagle’s medium (DMEM), DMEM/F12, trypsin-ethylenediaminetetraacetic acid (EDTA), bovine serum albumin (BSA), and fetal bovine serum were all obtained from GIBCO (Grand Island, NY, USA). CCK-8 was obtained from APE × BIO (Shanghai, China). MitoSOX Green Mitochondrial Superoxide Indicator was obtained from Invitrogen (Carlsbad, CA); Annexin V Apoptosis Detection Kit, lactate dehydrogenase (LDH) Cytotoxicity Assay Kit, JC-1 Probes, Hoechst 33342 dye, and Enhanced ATP Assay Kit were purchased from Abcam (Shanghai, China). A phosphatase inhibitor cocktail was obtained from Thermo Scientific (Shanghai, China). The AMPK pathway inhibitor Compound C was purchased from Selleck (Texas, USA). miR-181b inhibitors were purchased from RIBOBIO (Guangzhou, China). Lipofectamine 3000 reagents were purchased from Invitrogen (Guangzhou, China).
Extraction and Preparation of ZCF
ZCL was obtained from Xin Jiang. After drying in the shade with ZCL, it was crushed through a 100-mesh sieve and extracted by ultrasound with 50 times the amount of 70% ethanol. The ultrasound time was 50 min, and the ultrasound temperature was 60°C, and the microwave power was 50 W. The filtrate was combined and concentrated under reduced pressure to form an extract and then vacuum-dried to obtain the crude total flavonoids of ZCL. Dissolve the crude flavonoid powder in water, filter, separate the filtrate using HP-20 macroporous adsorption resin, wash and collect the eluent, and distill under reduced pressure to obtain a concentrated solution, which is then obtained after freeze–drying, with a content of 30% ZCF. Take 100 mg of total flavonoids and dissolve them in 10 mL of sterilized physiological saline by ultrasound for 30 min to obtain 100 mg/kg. Dilute the remaining concentrations in pairs and store them at 4°C for later use.
Measurement of Cell Viability
The survival rate of SH-SY5Y cells was measured using the CCK-8 assay kit. Take out a 96-well plate and evenly spread SH-SY5Y cells in the wells. After 1 day of cell culture, SH-SY5Y cells were treated with different concentrations of 1-methyl-4-phenylpyridinium (MPP+), and the cell survival rate was measured using a CCK-8 assay kit 24 h later. For ZCF treatment, cells were pretreated with ZCF (12.5–400 µg/mL) for 1 h, followed by MPP+ for 24 h. CCK-8 method is used to detect cell viability. Finally, measure the absorption at 450 nm.
Measurement of LDH Release
Evaluate the release of LDH in each group. In short, remove the six-well plate and inoculate SH-SY5Y cells into the plate at a density of 2 × 105 cells per well. According to the manufacturer’s instructions for the LDH assay kit (C0017, Beyotime, Shanghai), LDH released into the culture medium was detected.
Immunofluorescence
In short, sliced samples were added to specific primary antibodies and incubated overnight at 4°C. The next day, take out the sliced samples and wash them with phosphate-buffered saline (PBS) three times, each time for 5 min. Subsequently, the secondary antibody was added at room temperature and incubated for 1.5 h. It was then incubated in the dark with an installation medium containing 4′,6-diamidino-2-phenylindole (DAPI) for 10 min. The results of immunofluorescence were collected using a Nikon A1 confocal microscope.
Measurement of ATP
According to the manufacturer’s instructions (S0027, Beyotime, Shanghai), the ATP content in SH-SY5Y cells is measured using an enhanced ATP detection kit. In short, the cell lysate is prepared by adding 200 µL of lysate per well. The cell lysate was centrifuged at 12,000 g for 5 min at 4°C, and the supernatant was taken for subsequent measurement. Measure ATP concentration using a photometer.
Western Blot Analysis
First, SH-SY5Y cells are collected and lysed using a pre-cooled radioimmunoprecipitation assay buffer (RIPA) lysis buffer. Then, measure the protein content. Separate protein samples through sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transfer the protein samples onto polyvinylidene difluoride (PVDF) membranes. Then, incubate with BSA for 1 h. Incubate the samples overnight at 4°C with primary antibodies targeting tyrosine hydroxylase (TH), P-AMPK (Thr127), AMPK, and GAPDH to detect different proteins. The next day, wash the primary antibody three times with tris-buffered saline with Tween 20 (TBST) and incubate with the secondary antibody for another hour. Finally, the protein bands were visualized using enhanced chemiluminescence (ECL) substrate and Bio-Rad file system. Quantify the intensity of the stripes using ImageJ software.
Flow Cytometry Analysis
Inoculate SH-SY5Y cells evenly into a six-well culture dish. After treatment with different concentrations of ZCF, SH-SY5Y cells were collected using trypsin and washed twice with pre-cooled PBS. Then, the cell samples were stained with different substances: JC-1 fluorescent probe was used for mitochondrial membrane potential detection; MitoSOX was used for detecting mitochondrial ROS, and Green fluorescent dye. Use Annexin-V-FITC solution and propidium iodide (PI) solution, respectively, for apoptosis detection. Finally, use the relevant software for data analysis.
Measurement of JC-1 and MitoSOX
Inoculate cells evenly into a 12-well cell culture dish, with approximately 1 × 105 cells per well. Collect drug-treated cells and use the JC-1 fluorescent probe and MitoSOX™ stained with Green fluorescent dye. Then, wash the cell culture three times with PBS and detect the fluorescence intensity using a fluorescence microplate reader.
Animal Model Establishment
The experimental animals were male C57BL/6J mice purchased from the Experimental Animal Center of Guangdong Medical University. All C57 mice were subjected to a 12-h light/dark cycle at a temperature of 25°C. Water and food are freely accessible. All animal experiments were conducted under the “Guidelines for the Care and Use of Experimental Animals” and were approved by the Longhua District Central Hospital in Shenzhen. The welfare of experimental animals strictly follows the guidelines for the review of Experimental Animal Welfare in the People’s Republic of China (GB/T 35892-2018). The experimental animals were randomly divided into a control group, a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) group, and a ZCF treatment group. The construction method of the animal model is as follows: C57 mice are intraperitoneally injected with MPTP (30 mg/kg, dissolved in 0.9% physiological saline) once a day for 7 consecutive days. ZCF treatment group mice were intraperitoneally injected with ZCF [25, 50, and 100 mg/kg dissolved in dimethyl sulfoxide (DMSO): 0.9% physiological saline = 1:9] once a day after MPTP injection. The control group mice were given an equal amount of physiological saline.
Behavioral Test
Wire-hanging Test
The wire-hanging test is used to detect the limb motor coordination of mice. The mouse was placed on a metal wire about 30 cm from the ground with the two forelimbs in the middle of the wire. Both forelimbs can be hung to record three points, one forelimb can be hung to record two points, and both forelimbs cannot be hung to record one point. Each mouse was tested three times.
Immunohistochemistry (IHC)
Perform immunohistochemical treatment on brain paraffin sections. In short, the first step is to deparaffinize the paraffin sample and rehydrate it, followed by incubation overnight with the specific primary antibody TH (1:100) at 4°C. Subsequently, wash the paraffin sections with PBS for 5 min each time. Then, add the secondary antibody at room temperature and incubate for 1.5 h. Finally, 3,3′-diaminobenzidine (DAB) staining was used.
Statistical Analysis
All the results are presented as Mean ± Standard Error of the Mean (SEM). Data were analyzed using GraphPad Prism 8.0 statistical software. Differences between groups were evaluated using Student’s t-tests when only two groups were analyzed, or by one-way analysis of variance (ANOVA) when more than two groups were compared. p < .05 was considered statistically significant.
Results
ZCF Improves Cell Survival and LDH Release in MPP+-induced Cell Damage in SH-SY5Y Cells
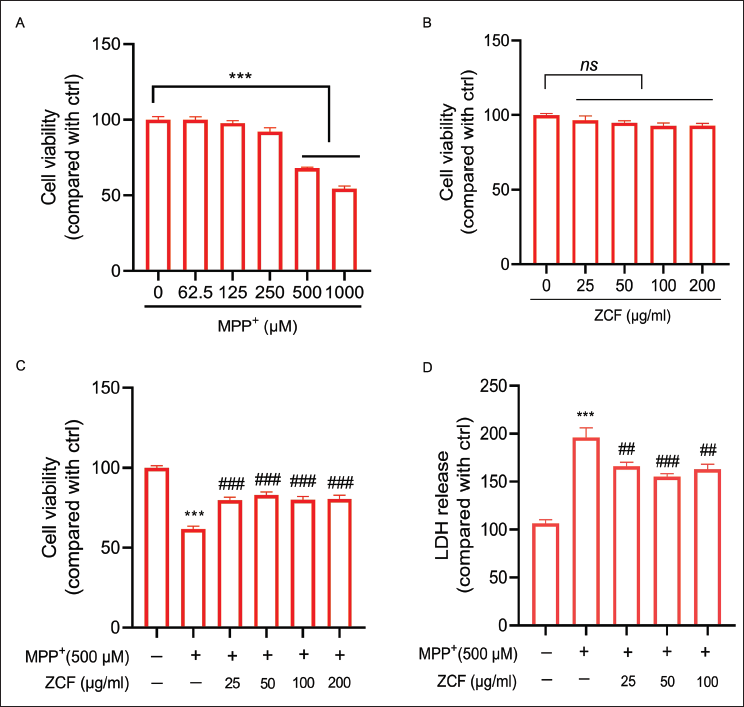
MPP+, the active metabolite of MPTP, is commonly used as an in vitro agent for modeling PD. To evaluate MPP+ cytotoxicity, SH-SY5Y cells were treated with different concentrations of MPP+ (62.5–1,000 µM) for 24 h. As shown in Figure 1A, MPP+ concentrations of 500 and 1,000 µM induced a significant reduction in cell viability, and 500 µM was chosen for the subsequent experiments. To further evaluate ZCF cytotoxicity, SH-SY5Y cells were treated with different concentrations of ZCF (25–100 µg/mL) for 24 h. As shown in Figure 1B, ZCF treatment alone did not have any obvious cytotoxic effect. To evaluate the protective effect of ZCF against MPP+-induced cell viability, SH-SY5Y cells were pretreated with ZCF for 1 h before being exposed to 500 µM MPP+ for 24 h. As shown in Figure 1C, ZCF pretreatment significantly attenuated MPP+-induced cell viability loss. Further assessment of the effects of ZCF on LDH release, the result indicated that pretreatment with ZCF significantly reduced the increase in LDH release caused by MPP+ in SH-SY5Y cells (Figure 1D).
Ziziphora clinopodioides Flavonoids (ZCF) Improves Cell Survival and Lactate Dehydrogenase (LDH) Release in 1-Methyl-4-Phenylpyridinium (MPP+)-induced Cell Damage in SH-SY5Y Cells. (A) Cell Viability was Detected in SH-SY5Y Cells Treated with Different Concentrations of MPP+. (B) Cell Viability was Detected in SH-SY5Y Cells Treated with Different Concentrations of ZCF. (C) Cell Viability was Detected in SH-SY5Y Cells Treated with Different Doses of ZCF With or Without MPP+ Treatment. (D) LDH Release was Detected in SH-SY5Y Cells Treated with Different Concentrations of ZCF With or Without MPP+ Treatment.
ZCF Improves MPP+-induced Mitochondrial Morphological Abnormalities, Reduces ATP Content, and Decreases TH Expression in SH-SY5Y Cells
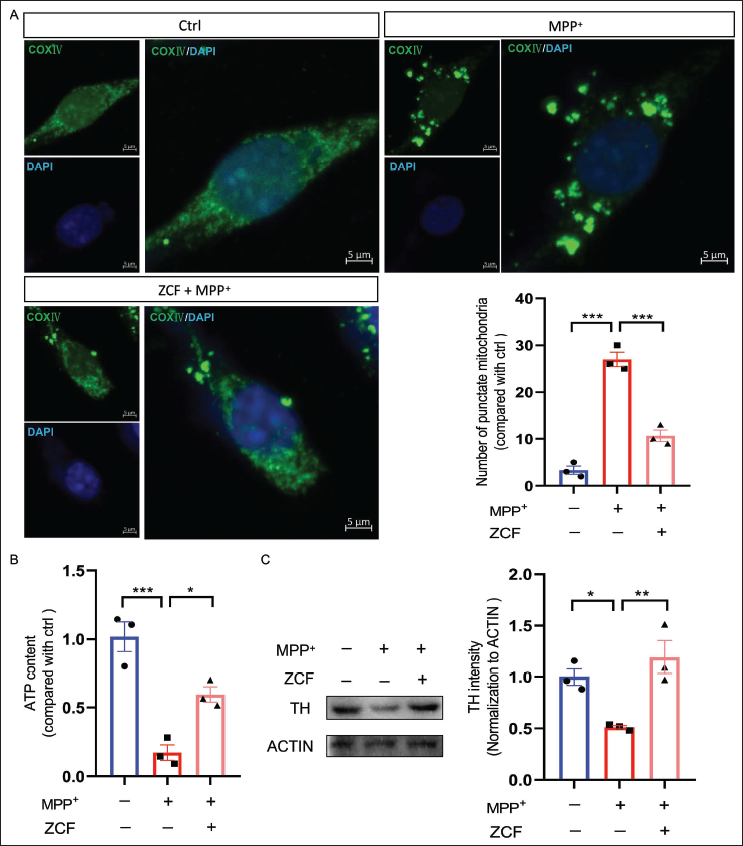
Targeting mitochondria may provide us with new therapeutic interventions in the fight against PD. Therefore, we postulated that ZCF could confer protection against MPP+-induced cell damage by enhancing mitochondrial function. To test this hypothesis, the effect of ZCF on mitochondrial morphology was observed by labeling COXIV, a widely used mitochondria marker. Obtained results revealed that MPP+ induced an increase in the number of punctate mitochondria per cell, an effect that could be improved by co-treatment with 50 µg/mL ZCF (Figure 2A). In the further study of the effect of ZCF on the ATP content in MPP+-induced cell damage in SH-SY5Y cells was detected. As shown in Figure 2B, treatment of SH-SY5Y cells with MPP+ significantly reduced ATP content, and this effect was mitigated by co-treatment with 50 µg/mL ZCF. The changes in TH are positively correlated with the changes in dopamine in the brain of PD disease. We further detected the changes of TH in SH-SY5Y cells. Exposure of cells to 500 µM MPP+ for 24 h resulted in a marked decrease in TH expression levels, and ZCF treatment upregulated TH expression levels in SH-SY5Y cells (Figure 2C).
Ziziphora clinopodioides Flavonoids (ZCF) Improves 1-Methyl-4-Phenylpyridinium (MPP+)-induced Mitochondrial Morphological Abnormalities, Reduced Adenosine Triphosphate (ATP) Content, and Decreased Tyrosine Hydroxylase (TH) Expression in SH-SY5Y Cells. (A) Immunofluorescence and Quantification of COXIV Protein Expression in SH-SY5Y Cells Treated with 500 µM MPP+, 50 µg/mL of ZCF, and 500 µM MPP+. COXIV in Green and 4′,6-Diamidino-2-Phenylindole (DAPI) in Blue. Scale Bar, 5 µm. (B) ATP Content was Detected in SH-SY5Y Cells Treated with 500 µM MPP+, 50 µg/mL of ZCF, and 500 µM MPP+. (C) Western Blot Analysis and Quantification of TH Protein Expression in SH-SY5Y Cells Treated with 500 µM MPP+, 50 µg/mL of ZCF, and 500 µM MPP+.
ZCF Improves MPP+-induced Mitochondrial Membrane Potential Loss and the Increase of Mitochondrial ROS, Apoptotic Cells in SH-SY5Y Cells
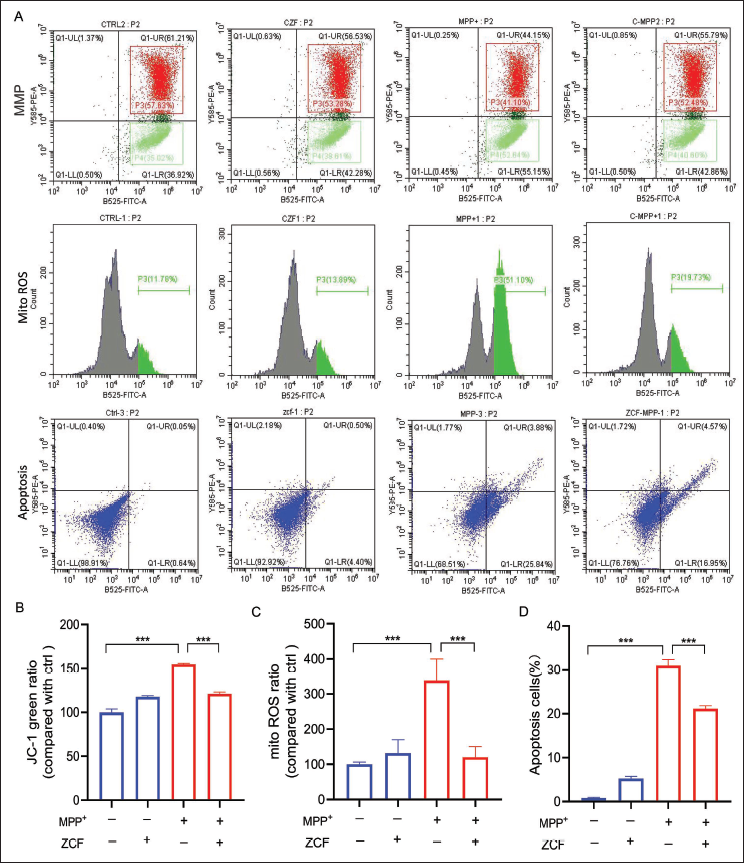
To determine whether ZCF could reduce MPP+-induced mitochondrial membrane potential loss, JC-1 dye was used to monitor mitochondrial membrane potential in SH-SY5Y cells. In brief, exposure of SH-SY5Y cells to 500 µM MPP+ increased green-fluorescence ratio, while pretreatment with 50 µg/mL ZCF for 1 h significantly attenuated MPP+-induced increase of green-fluorescence ratio and mitochondrial membrane potential loss. This indicated that pretreatment with ZCF significantly prevented the mitochondrial membrane potential loss induced by MPP+ (Figure 3A and 3B). Mitochondrial ROS was evaluated to investigate the effect of ZCF on MPP+-induced oxidative damage. SH-SY5Y cells were incubated with 50 µg/mL ZCF for 1 h before being exposed to 500 µM MPP+ for 24 h. As shown in Figure 3A and 3C, exposure of SH-SY5Y cells to 500 µM MPP+ increased mitoROS levels. Pretreatment of cells with ZCF at 50 µg/mL inhibited the MPP+-induced increase of mitoROS in SH-SY5Y cells. Cell apoptosis was further measured by Annexin V-FITC/PI staining, and the result in Figure 3A-Apoptosis and D indicated that 500 µM MPP+ exposure significantly increased the number of apoptotic cells. At the same time, pretreatment with 50 µg/mL ZCF significantly reduced the number of apoptotic cells caused compared with MPP+ treatment. In addition, 50 µg/mL ZCF treatment alone did not have any obvious effect on mitochondrial membrane potential, mitochondrial ROS, and apoptotic cells.
Ziziphora clinopodioides Flavonoids (ZCF) Improves 1-Methyl-4-Phenylpyridinium (MPP+)-induced Mitochondrial Membrane Potential Loss, Mitochondrial Reactive Oxygen Species (ROS) Elevation, and Cell Apoptosis in SH-SY5Y Cells. (A and B) Fluorescence-activated Cell Sorting (FACS) Analysis and Quantification of JC-1 Green Ratio in SH-SY5Y Cells After Treatment with ZCF, MPP+, and ZCF Plus MPP+. (A and C) FACS Analysis and Quantification of mitoROS Levels in SH-SY5Y Cells After Being Treated with ZCF, MPP+, and ZCF Plus MPP+. (A-Apoptosis and D) FACS Analysis and Quantification of Cell Apoptosis Ratio in SH-SY5Y Cells After Treatment with ZCF, MPP+, and ZCF Plus MPP+.
ZCF Improves MPP+-induced Cell Damage by Activating the AMPK/miR-181b Axis
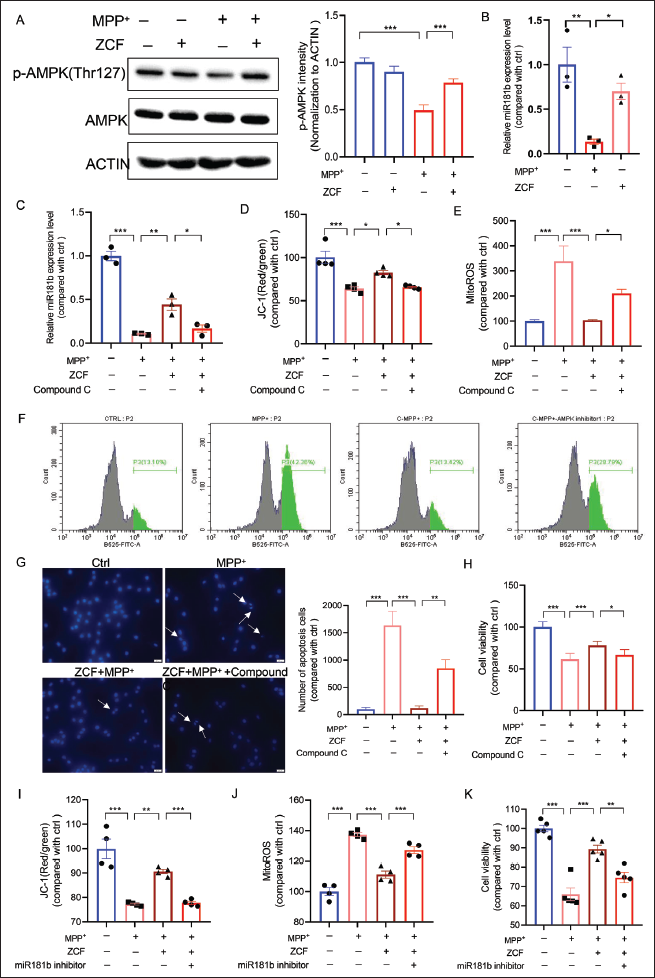
SH-SY5Y cells were pretreated with 50 µg/mL ZCF for 1 h before being exposed to 500 µM MPP+ for 24 h, and the phosphorylation of AMPK was tested by Western blot. As shown in Figure 4A, MPP+ induced a significant downregulation in the phosphorylation of AMPK, and ZCF treatment stimulated the phosphorylation of AMPK when compared with the MPP+ treatment group. 50 µg/mL ZCF treatment alone did not have any obvious effect. Moreover, MPP+ induced a significant decrease in the expression level of miR-181b, which was reversed after pretreatment with 50 µg/mL ZCF (Figure 4B). To investigate whether AMPK is required for the protective effect of ZCF, we tested the mitochondrial membrane potential, mitochondrial ROS production, and cell apoptosis of ZCF in SH-SY5Y cells when AMPK is inhibited by AMPK inhibitor Compound C. As shown in Figure S1 (available online as Supplementary Material), Compound C treatment alone for 30 min at doses of 2.5 and 5 µM did not have any toxic effects. Therefore, we used 5 µM of Compound C for 30 min in our follow-up experiments. The results of the real-time polymerase chain reaction (PCR) demonstrate that ZCF failed to increase the expression level of miR-181b when AMPK is inhibited by Compound C (Figure 4C). Moreover, the results of the JC-1 staining and flow cytometry showed that the pretreatment of Compound C blocked the effect of ZCF on improving mitochondrial membrane potential loss and mitochondrial ROS increase (Figure 4D–4F). Hoechst 33342 staining analysis further showed that pretreatment of Compound C blocked the anti-apoptotic effect and promoted the cell survival effect of ZCF (Figure 4G and 4H). Furthermore, transfection of miR-181b inhibitors (50 nmol/L) eliminates the improvement effect of ZCF towards MPP+-induced mitochondrial dysfunction and cell viability loss (Figure S2A available online as Supplementary Material and Figure 4I–4K). The transfection of miR-181b inhibitors treatment alone did not have any toxic effects (Figure S2B–S2D available online as Supplementary Material).
Ziziphora clinopodioides Flavonoids (ZCF) Improves 1-Methyl-4-Phenylpyridinium (MPP+)-induced Cell Damage in SH-SY5Y Cells by Activating the AMP-Activated Protein Kinase (AMPK)/miR-181b Axis. (A) Western Blot and Quantification of Phosphorylated AMPK, Total AMPK Expression Levels in SH-SY5Y Cells After Treatment with ZCF, MPP+, and ZCF Plus MPP+. (B) The Expression Levels of miR-181b were Detected by Real-time Polymerase Chain Reaction (PCR) in SH-SY5Y Cells After Being Treated with ZCF, MPP+, and ZCF Plus MPP+. (C) The Expression Level of miR-181b was Detected by Real-time PCR in SH-SY5Y Cells After Being Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + AMPK Inhibitor Compound C. (D) Red and Green Fluorescence Intensity Analysis of JC-1 Staining Assay in SH-SY5Y Cells After Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + AMPK Inhibitor Compound C. (E and F) mitoROS Fluorescence Intensity Analysis of MitoSOX™ Green Fluorogenic Dye in SH-SY5Y Cells After Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + AMPK Inhibitor Compound C. (G) Representative Pictures and Quantification of Hoechst 33342 Staining in SH-SY5Y Cells After Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + AMPK Inhibitor Compound C. (H) Cell Viability was Detected in SH-SY5Y Cells Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + AMPK Inhibitor Compound C. (I) Red and Green Fluorescence Intensity Analysis of JC-1 Staining Assay in SH-SY5Y Cells After Treatment with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + miR-181b Inhibitor. (J) mitoROS Fluorescence Intensity Analysis of MitoSOX™ Green Fluorogenic Dye in SH-SY5Y Cells After Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + miR-181b Inhibitor. (K) Cell Viability was Detected in SH-SY5Y Cells Treated with ZCF, MPP+, ZCF Plus MPP+, and ZCF + MPP+ + miR-181b Inhibitor.
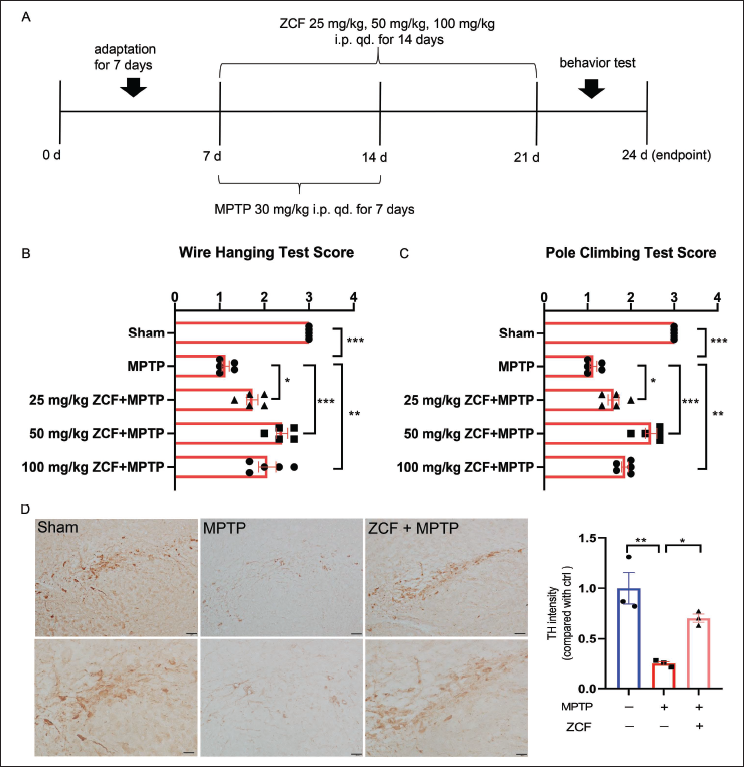
ZCF Improves Motor Dysfunction and Loss of TH-positive Neurons in Parkinson’s Mice Model
To evaluate the protective effect of ZCF in a PD mice model. The animal experimental dosing protocol is shown in Figure 5A. The pole-climbing test and wire hanging test were performed for motor function assessment. 30 mg/kg MPTP treatment for 7 days significantly decreased the scores of mice in the wire-hanging test (Figure 5B) and pole-climbing test (Figure 5C). ZCF administration (25, 50, and 100 mg/kg) notably increased the scores of mice in the behavioral test (Figure 5B and 5C). In addition, ZCF administration alone did not affect the motor dysfunction of mice (Figure S3A and S3B, available online as Supplementary Material). it indicates that this dose had no significant toxicity to mice. Since the animals receiving 50 mg/kg ZCF displayed better functional outcomes when compared with the MPTP treatment groups, this dose was selected for the following studies. Assessment of TH in substantia nigra sections revealed that TH-positive neurons from the MPTP group had decreased significantly when compared with the Ctrl group. TH-positive neurons were increased by 50 mg/kg ZCF treatment (Figure 5D).
Ziziphora clinopodioides Flavonoids (ZCF) Improves Motor Dysfunction and Loss of Tyrosine Hydroxylase (TH)-positive Neurons in Parkinson’s Model Mice. (A) Schedule for Animal Experiments. (B) Result of Wire-hanging Test. (C) Results of Pole-climbing Test. (D) Representative Pictures and Quantification of Immunohistochemistry (IHC) were Marked with Parkinson’s-related Landmark Dopamine Neurons TH.
Discussion
In this study, we revealed the potential protective effects of ZCF against the toxic effects of MPP+/MPTP. ZCF improved motor dysfunction and the loss of TH-positive neurons in an MPTP-induced PD mice model. We also found that ZCF protected SH-SY5Y cells from MPP+-induced toxicity. At the molecular level, ZCF treatment significantly stimulated the phosphorylation of AMPK and its downstream target miR-181b. Further experiments with kinase inhibitors attenuated the protective effect of ZCF. Our results indicate that ZCF was able to protect neuronal cells against MPP+-induced cell injury via the activation of the AMPK/miR-181b axis.
As DA neuronal degeneration is a main pathological feature of PD, many studies have focused on developing drugs to ameliorate dopamine neuron damage. A recent study revealed that Sigma-1 receptors regulate mitophagy to protect DA neurons (Wang et al., 2021). Consistent with these reports, we found that ZCF upregulated TH expression levels in SH-SY5Y cells and improved the loss of DA neurons in the MPTP-induced PD mice model.
Injection of MPTP is mostly used in mouse models of PD (Mustapha & Mat Taib, 2021). MPTP itself is non-toxic and acts by producing the toxic forms of MPP+ in the brain (Gerlach et al., 1991). Hence, mitochondrial abnormalities are the main significant factor contributing to injury in PD. Moreover, there is ample evidence to suggest that mitochondria are the main source of intracellular free radicals (Bertero & Maack, 2018; Okoye et al., 2023; Turrens, 2003). Therefore, improving mitochondrial function is a feasible strategy for preventing or delaying oxidative stress and neurodegeneration in PD. Here, for the first time, we found that ZCF treatment improved MPP+-induced mitochondrial morphological abnormalities, reduced ATP and mitoROS levels, and attenuated mitochondrial membrane potential loss, ultimately decreasing cell apoptosis in SH-SY5Y cells. It has been reported that microRNAs (miRNAs) in the blood serve as one of the biomarkers for the early diagnosis of PD (Liu, Chen, et al., 2019). Our data showed that miR-181b level decreased in the SH-SY5Y cells after MPP+ treatment. After ZCF treatment, the expression level of miR-181b was increased. Moreover, transfection of miR-181 inhibitors ameliorated the neuroprotective effect of ZCF. It has been reported that AMPK activation is an upstream event of miR-181b upregulation in endothelial cells (Cheng et al., 2022); hence, the phosphorylation of AMPK was tested in our study. Our result showed that MPP+-induced a significant downregulation in the phosphorylation of AMPK, and ZCF treatment stimulated the phosphorylation of AMPK in SH-SY5Y cells. Therefore, our findings also link miRNA in neuronal cells to AMPK, which is a key protein involved in energy metabolism.
Conclusion
Our study demonstrated that ZCF protects neuronal cells against MPP+-induced cell injury via the activation of the AMPK/miR-181b axis. These results support the potential application of ZCF in preventing neuronal cell damage in neurodegenerative diseases such as PD.
Footnotes
Abbreviations
AMPK: AMP-activated protein kinase; AS: Anti-atherosclerosis; ATP: Adenosine triphosphate; DA: Dopaminergic; LDH: Lactate dehydrogenase; PD: Parkinson’s disease; ROS: Reactive oxygen species; TH: Tyrosine hydroxylase; ZCF: Ziziphora clinopodioides flavonoids.
Authors Contribution
XBC and JC performed, analyzed experiments, and co-wrote the manuscript. BL, XFW, ZQW, HL, ZXZ, and YZC performed part of the experiments. LG and SL supervised the study and conceptualized the project. XBC, JC, LG, and SL revised the manuscript. SL and LG provided equal contributions in designing and overseeing the overall study. All authors approved and reviewed the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal experiments were approved by the Animal Experimental Ethics Committee of Longhua District Central Hospital in Shenzhen (Approval number: 20220301).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by project grants from the district-level scientific research project for medical and health institutions in Longhua District, Shenzhen (No. 2021080), National Natural Science Foundation of China (Grant No. 82201620), and the China Postdoctoral Science Foundation (No. 2023M734024).
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
