Abstract
Background
Rectal cancer has a high morbidity and mortality rate in China, endangering public health. Sterol regulatory element-binding protein cleavage-activating protein/sterol regulatory element-binding protein 1 (SCAP/SREBP1) signaling pathway is essential in tumor cell proliferation and apoptosis. Dihydroartemisinic (DHA) can exert anti-tumor effects by regulating tumor cell iron death.
Objectives
This study aimed to investigate the specific role and mechanism of DHA in inhibiting the proliferation of rectal cancer by inducing iron death through the SCAP/SREBP1 pathway.
Materials and Methods
Six groups were set: model group, SCAP/SREBP1 agonist group, SCAP/SREBP1 inhibitor group, 10%, 30%, and 50% DHA group, and randomly selected 10 SD rats with successful modeling in each group male rat. The pathological changes of the rectal tissue of rats were observed by staining, and cell viability, proliferation, and apoptosis were detected using cell counting kit-8 (CCK-8), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution, and flow cytometry. The levels of SCAP, SREBP, and glutathione peroxidase 4 (GPX4) were detected by quantitative reverse transcription polymerase chain reaction (qRT-PCR), and protein expressions of SCAP, SREBP, and GPX4 were detected by Western blot.
Results
The normal mice in the control group had a complete tissue structure, while the rectal tissue epithelial cells in the model group were irregular and deformed. Compared with the model group, the SCAP/SREBP1 agonist and 30% DHA group showed improvement, but/SREBP1 inhibitor group was more serious than the model group. DHA can be found to be negatively correlated with the proliferative activity of rectal cancer cell lines, and SCAP/SREBP1 can affect cell proliferation. The effect of DHA on rectal cancer cells was correlated with concentration and time dependence. When the concentration reaches 50%, the apoptosis rate reaches 20% and 25%, respectively, in 2 and 3 days. GSH decreased with the increase in DHA concentration. DHA can promote the expression of SCAP/SREBP1 and reduce the expression of GPX4, and SCAP/SREBP1 can also affect the expression of GPX4, and the decrease of SCAP/SREBP1 expression will lead to the increase of GPX4 expression.
Conclusion
DHA can inhibit the viability and promote apoptosis of rectal cancer cells by increasing the concentration of GSH and SCAP/SREBP1 signaling pathways. Therefore, DHA may become a new therapeutic method to inhibit rectal cancer cells.
Keywords
Introduction
Rectal cancer has a relatively high prevalence in developed countries and developed regions and is the third most common tumor (Baidoun et al., 2021). Relevant data show that the incidence and mortality rates of colorectal cancer in China have risen over the past decade, ranking fourth and fifth, respectively, among the diseases with the highest incidence and mortality rates among all malignant tumors in 2018 (Patel et al., 2022). This phenomenon is due to the changes in the living habits and food culture of Chinese residents, along with the development of society and economy, as well as the ageing of the population caused by increasing levels. Rectal cancer not only endangers life and health but also seriously affects the continuous development of the social economy (Patel et al., 2022).
In the process of fatty acid synthase expression, sterol regulatory element-binding proteins (SREBPs), as a typical transcription factor that can control fat metabolism, play a key role. The proliferation of cancer cells is caused by the increase in fatty acid synthase, and its mechanism is to induce cancer cells to enter the division phase (Kawamura et al., 2022). During the intracellular synthesis of SREBP, the Golgi apparatus is the main activity site of the substance, and it needs to be effectively synthesized in the endoplasmic reticulum first, so this also determines to a certain extent that the activation of SREBP needs to be transferred from the endoplasmic reticulum to the Golgi apparatus. In addition, this process requires the participation of SREBP cleavage-activating protein (SCAP) (Cheng et al., 2022). SCAP is a membrane protein composed of 1,276 amino acid residues. It binds to a protein called insulin-inducible gene (INSING) in the endoplasmic reticulum membrane, blocking Golgi transport and activating SREBP to upregulate the expression of adipogenic enzymes, promoting tumor cell proliferation (Liu et al., 2020). Many studies have shown that (Aldaalis et al., 2022; Ang et al., 2021), the SCAP/SREBP pathway can be involved in the formation of fat, and if this pathway is abnormal, it may lead to the formation and expansion of tumors. There are two proteases (S1P and S2P) at enzyme sites 1 and 2 in the body that can participate in the activation reaction of SREBPs in the Golgi apparatus. The enzymes can cleave SREBPs in order to release their N-terminal structures domain; the released domain enters the nucleus, leading to the overexpression of SREBPs targeting lipase, which causes abnormal lipid metabolism in which it participates, resulting in abnormal lipid production, so that lipid consumption occurs continuously and abnormal cell proliferation activities (Tiong et al., 2022).
At present, the medical treatment methods for cancer not only have limited curative effects but also have many side effects, so it is urgent to find and develop alternative drugs with fewer side effects and high efficiency. In the research and development of methods for treating cancer, Chinese medicine is one of the most important branches. In recent years, an active ingredient of traditional Chinese medicine containing peroxy groups has been found in Artemisia annua, which is a sesquiterpene lactone called artemisinin (Chen et al., 2020). The discovery of artemisinin has led to research on the physical and chemical properties of artemisinin, and it was discovered that a derivative of artemisinin not only has anti-malarial activity but also may play an important role in anti-viral and anti-bacterial aspects (Çapcı et al., 2021; Mancuso et al., 2021). This derivative is dihydroartemisinin (DHA). Due to the increase in the incidence of cancer, the current research focus on DHA is mainly to explore its anti-tumor effect (Efferth & Oesch, 2021). At present, through the research on the mechanism of DHA-induced autophagy, cell cycle arrest and apoptosis, it is found that it can inhibit the proliferation of most cancer cells, such as liver cancer, pancreatic cancer and leukemia, thus leading to the restriction of tumor cell development (Kiani et al., 2020). Through relevant experimental studies, it was found that DHA caused the imbalance of iron homeostasis due to abnormal iron metabolism in cancer cells and even caused ferroptosis (Battaglia et al., 2020; Peng et al., 2020; von Schacky, 2021). Compared with common apoptotic cell death methods, it is found that the mechanism of ferroptosis is lipid peroxidation, and this reaction requires a large amount of available iron in the cell, which is also accompanied by an increase in the level of reduced glutathione (GSH). Therefore, ferroptosis, as a new type of cell death, has been gradually included in the anti-tumor experimental research (Bordini et al., 2020). From the above, both ferroptosis and SCAP/SREBP1 are related to lipid metabolism. Therefore, this study will further reveal the relationship between dihydroartemisinin and SCAP/SREBP1 and its mechanism of inducing cell death from the perspective of ferroptosis.
Materials and Methods
Experimental Reagents, Instruments, and Animals
Reagents and Instruments
Dihydroartemisinin (Shanxi Yuning Biotechnology); dimethyl sulfoxide (DMSO) (Beijing Golden Clones Biotechnology); fetal bovine serum (FBS) (Huankai Microbiology, Ltd., Guangdong); phosphate-buffered saline (PBS) (Nanjing Senbega Biotechnology); Pancreatin-ethylenediaminetetraacetic acid (EDTA) Digestion Solution (Hunan Fenghui Biotechnology Co., Ltd.); propidium iodide (PI) staining solution (Wuxi Puhe Biomedical Technology Co., Ltd.); hematoxylin–eosin (HE) dyeing solution (Yingxin Laboratory Equipment Ltd., Shanghai); MTT solution (Isejiu, Lianyungang, Jiangsu Ltd.); Dulbecco’s modified Eagle medium (DMEM) medium (Wengjiang Chemical Reagent Ltd., Guangdong); paraffin slicer (Wuxi Shuangsheng Petrochemical Equipment Co., Ltd.); enzyme-linked immunoassay instrument (Jinan Haolaibao Medical Equipment); flow cytometer (Jinan Haolaibao medical equipment); gel imaging system (Shandong Kangjian Biotechnology Co., Ltd.); complementary deoxyribonucleic acid (cDNA) reverse transcription kit (Yizhi Bio, Ltd., Jiangsu).
Animals
Seventy male Sprague-Dawley (SD) rats with a weight of about 160–170 g were placed in suitable cages, set in controllable conditions, and fed with solid food that meets market regulations. Under this condition, the rats were acclimated to the environment, but the timing of eating and drinking by the rats was not specified. Randomly select 10 as the control group and the rest as the experimental group.
Experimental Method
Rectal Cancer Model and Group Drug Treatment
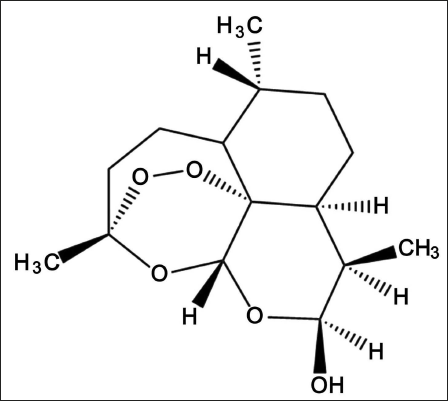
All 60 rats were subcutaneously injected with dimethylhydrazine (30 mg/kg) every Friday morning, and a successful rectal cancer model could be obtained by continuous injection for 3 months. After the successful model building, continue to feed for 16 weeks. The rats after successful modeling were randomly divided into six groups, namely, the model group, SCAP/SREBP1 agonist group, SCAP/SREBP1 inhibitor group, and 10%, 30%, and 50% DHA groups, with 10 rats in each group. For the rats in the control group and the model group, 0.2 mL of DMSO and 1 mL of normal saline were injected every day; the remaining three groups were injected according to the concentration of the substance they needed, and 1 mL of physiological saline was also injected. Saline, specifically the SCAP/SREBP1 agonist group, was injected with 0.2 mL of SCAP/SREBP1 agonist, the SCAP/SREBP1 inhibitor group was injected with 0.2 mL of SCAP/SREBP1 inhibitor, and the 10%, 30%, and 50% DHA groups were each injected with 0.2 mL of DHA. The molecular structural formula of DHA is shown in Figure 1.
The Molecular Structure of Dihydroartemisinic (DHA).
Rectal Tissue Extraction
The rats, 3 months after the injection, were anesthetized and then killed. After the death, the rectum was dissected and removed, and the rectal tissue was found in the rectum and then stripped out. The excised rectal tissues were fixed with 4% paraformaldehyde for 1 day. Equipped with 70%, 85%, 95%, and 100% ethanol, respectively, and then soaked the tissue according to the gradient to make it dehydrated, transparent, embedded, and finally made into paraffin for the experiment.
HE Staining
The prepared paraffin was cut into thin slices using a paraffin microtome according to the specification of 5 µm. The HE-stained slices were observed at random angles of view under a microscope, and the pathological morphology of the tissue was recorded.
Cell Viability Detection
Inoculate rectal cancer cells extracted from rectal cancer tissue into DMEM high-glucose medium containing 10% FBS and then culture the medium in a sterile incubator at 37°C for 5% CO2. Observe daily until the concentration of cells in the medium is 70%. Then add trypsin-EDTA digestion solution with a concentration of 0.25%, wait until the cell suspension can be obtained after digestion, and then inoculate the cell suspension with a density of 1 × 105 cells/mL into a 96-well plate and inoculate 100 µL per well. Wash once with PBS solution. After washing, 10% CCK-8 culture solution was added to each well for a 30 min incubation, and the temperature was set at 37°C. Put the cultured cell liquid at 450 nm for detection and record its optical density value; the optical density value under this light wave increases with the increase of cell viability, and set the cell viability represented by the optical density value of the control group as 1. On this basis, the relative cell viability is calculated, and a curve is drawn according to the data.
Detection of Cell Proliferation and Inhibition
Rectal cancer cells in the logarithmic growth phase were taken and cultured in a medium; 2 × 103 cells per well were inoculated in 96-well culture plates, and five replicate wells were set for each group of samples. After 4 days, add 20 µL of MTT solution with a concentration of 5 g/L to each well and continue to incubate for 240 min. Take out and suck off the supernatant in the wells, remove the supernatant, add 150 µL of DMSO to each well, mix thoroughly for 10 min and compare the colors. Set the wavelength of the ELISA to 490 nm, then detect and record the light absorbance value OD in each well. Inhibition rate = (1 – OD of drug-dosing group/OD of control group) ×100%.
Detection of Apoptosis Rate
PBS solution was added to the medium of three groups of DHA-intervened logarithmically growing rectal cancer cells. Then the three groups were placed in a CO2 incubator for continuous culture for 1 and 2 days. After culture, collect the two groups of cells according to the number of cells in each group higher than 1 × 106 and then add 500 µL of 70% ethanol to fix the cells for 12 h. The fixed cells were washed with PBS solution. After washing, use a centrifuge to remove the liquid. Then add 0.5 mL of PI staining solution with a concentration of 10 mg/L, store it at room temperature in the dark for 60 min for staining, and use flow cytometry to detect cell cycle and apoptosis after staining.
Detection of GSH Level
Proteins in each group of cells were extracted and then quantified. Then, take 100 µg of the total protein and add it to 2 mL of the reagent prepared according to the instructions, shake it well, keep the mixture for 10 min, and centrifuge it fully (4,000 r/min). After centrifugation, 1 mL of the supernatant was absorbed to carry out the color reaction according to the reaction system in the manual, and it was left at room temperature for 5 min. Then set a wavelength of 420 nm and record the absorbance, compare it with the absorbance value of the cells in the control group, calculate the relative GSH content of the cells in the other groups except the control group, and then count the data and draw a graph.
Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
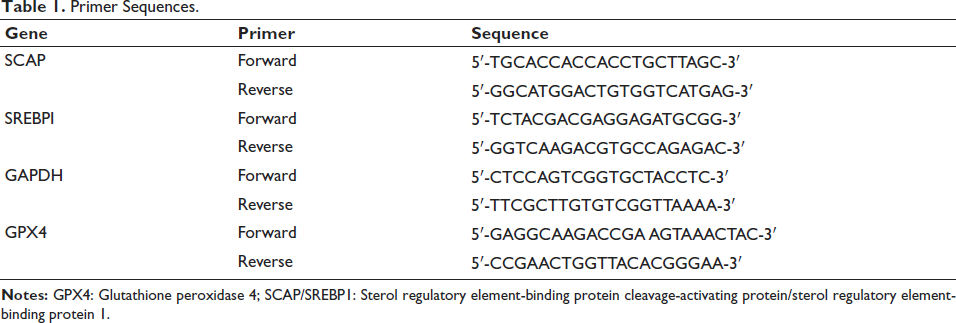
The total ribonucleic acid (RNA) extracted with TRIzol reagent was reverse transcribed into cDNA using a cDNA reverse transcription kit. SCAP, SREBP1, and GPX4 were detected by a PCR instrument to detect their expression in tissues. 2−△△CT, calculate the relative expression levels of SCAP, SREBP1, and GPX4 mRNA. Primers are listed in Table 1.
Primer Sequences.
Western Blot
The cells were washed twice with PBS and lysed to obtain the total protein of the cells required for this study. SDS-PAGE electrophoresis supernatant containing 50 µg protein, the gel obtained in the previous step was slowly moved onto PVDF. Then, 5% bovine serum albumin was effectively added to PVDFA. It was effectively blocked in a constant temperature environment of 25°C. After storage for 1 h, the operator used the primary antibody (1:1,000 dilution of SCAP, SREBP1, GPX4 rabbit polyclonal antibody), secondary antibody (diluted 1:2,000, goat anti-rabbit secondary antibody) to incubate the samples obtained in the above steps. After culturing, ImageJ14.0 software was used to quantitatively analyze the images developed by the ECL reagent and gel imaging system to detect the protein levels of SCAP, SREBP1, and GPX4.
Statistical Analysis
SPSS 21.0 and GraphPad Prism were used for statistical analysis, and the calculated data were represented by Mean ± SD, which conformed to the normal distribution and the homogeneity of variance; all data calculations were performed using the F test, and the comparison of data between groups was performed using the LSD method. p <.05 was considered a difference.
Results
HE Staining Results
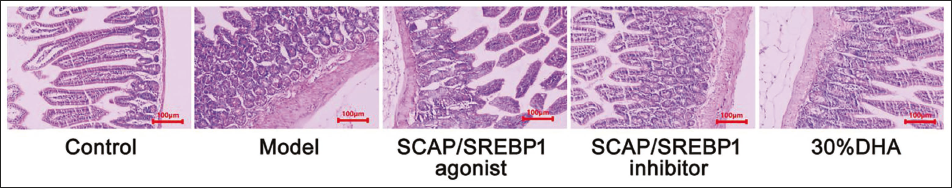
The tissue structure of the control group was intact, and the epithelial cells of the rectal tissue in the model group were irregular and deformed, and the SCAP/SREBP1 agonist and 30% DHA group were improved compared with the model group, but the SCAP/SREBP1 inhibitor group was better than the model group; and the group is more severe (Figure 2).
Histopathological Changes of the Rectum. ×100. Scale Bar = 100 µm.
Cell Viability Test Results
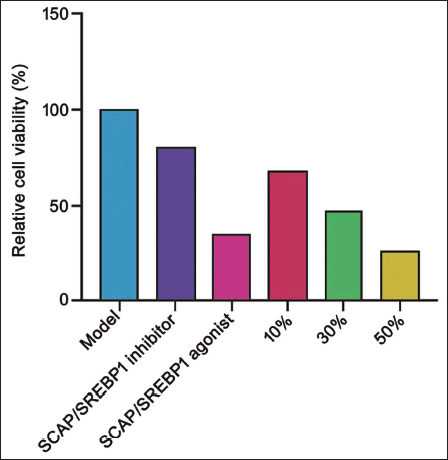
Most studies have shown that DHA has an inhibitory effect on tumor cell viability. In order to detect the effect of DHA on the viability of rectal cancer cells and determine the concentration of the drug, a CCK-8 reagent was used to detect the viability of the cells. It can be seen that the relative cell viability decreases with the increase in DHA concentration (Figure 3).
Cell Viability of Each Group.
Cell Proliferation and Inhibition
According to the research data, compared with the control group and the model group, the inhibition rate of the inhibitor group will be greater than that of the agonist group. For the DHA group, the higher the concentration of DHA and the longer the time, the slower the proliferation rate of rectal cancer cells, as shown in Figure 4.
The Effect of Dihydroartemisinic (DHA) Concentration on the Inhibition Rate. **p <.01.
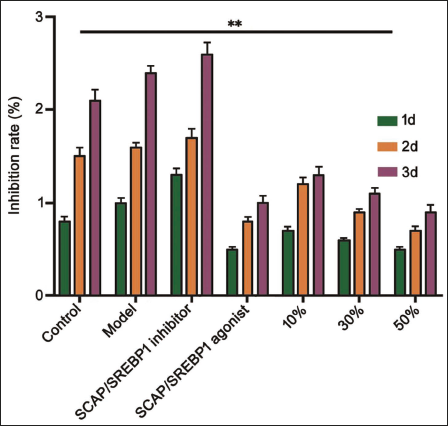
DHA Can Regulate the Apoptosis of Colorectal Cancer Cells
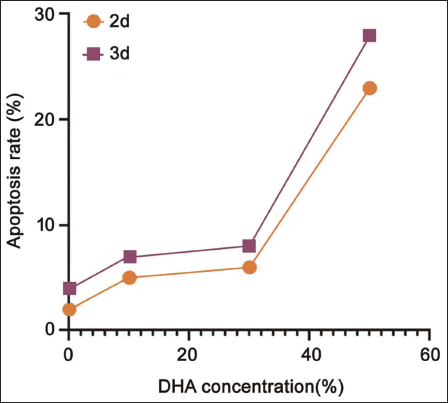
As the concentration of DHA increased, the apoptosis of colorectal cancer cells also showed a positive correlation trend. This phenomenon is not affected by the culture time (Figure 5).
Effect of Dihydroartemisinic (DHA) Concentration on Apoptosis Rate.
GSH Content Test Results
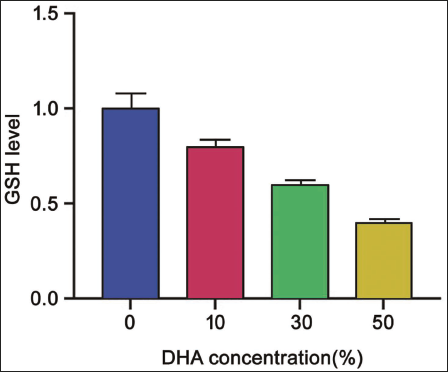
The GSH level is inversely proportional to the DHA concentration. When the DHA concentration is 0, the GSH level is the highest at 1, and when the DHA concentration is 50%, the GSH level drops below 0.5 (Figure 6).
Effect of Dihydroartemisinic (DAH) on Glutathione (GSH) Content.
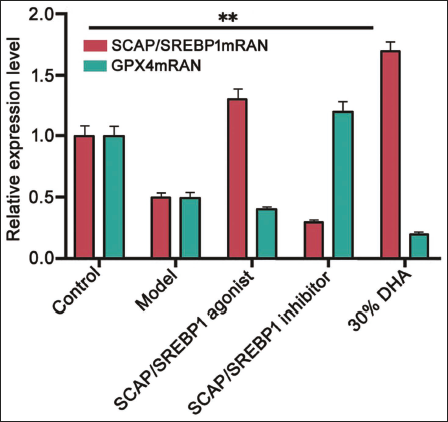
Relative Levels of SCAP, SREBP1, and GPX4 mRNA
The results of the relative levels of SCAP, SREBP1, and GPX4 mRNA were higher in the agonist group and the 30% dihydroartemisinin group than in the inhibitor group; and for GPX4 mRNA, the agonist group and the 30% dihydroartemisinin group had significantly lower levels than the inhibitor group (Figure 7).
Relative Levels of Sterol Regulatory Element-binding Protein Cleavage-activating Protein (SCAP), Sterol Regulatory Element-binding Protein 1 (SREBP1), and Glutathione Peroxidase 4 (GPX4) mRNA. **p <.01.
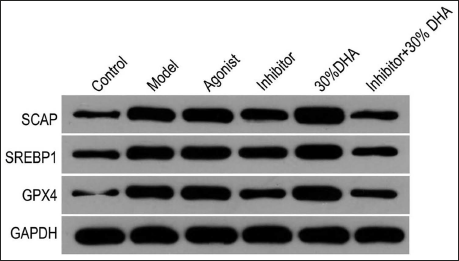
Relative Protein Expression of SCAP, SREBP1, and GPX4
Under the condition of 30% dihydroartemisinin, the expressions of SCAP, SREBP1, and GPX4 are vigorous. In order to clarify whether this phenomenon is related to the SCAP/SREBP1 pathway, we added a pathway agonist to the culture of rectal cancer cells. We found that the expression of the three proteins was significantly increased. In order to verify the inhibitory effect of dihydroartemisinin on this pathway, we respectively gave pathway inhibitors, inhibitors + 30% dihydroartemisinin to model rats and found that consistent results and the expressions of the three proteins were significantly lower (the inhibitor group was lower than the model group), but the levels of inhibitor + 30% dihydroartemisinin group were significantly lower than those of 30% dihydroartemisinin group (p <.05) (Figure 8).
Relative Protein Expression of Sterol Regulatory Element-binding Protein Cleavage-activating Protein (SCAP), Sterol Regulatory Element-binding Protein 1 (SREBP1), and Glutathione Peroxidase 4 (GPX4).
Discussion
Dixon et al. (2012) discovered in 2012 that ferroptosis, which includes apoptosis, necrosis, and autophagy, requires the participation of iron in its death process, which is also the characteristic that distinguishes it from other cell deaths (Du et al., 2021; Yi et al., 2020). When ferroptosis occurs, it will be accompanied by the increase of intracellular ROS, a decrease of GSH level and the loss of GPX4 activity (Wang et al., 2021). Yu et al. (2015) showed that the curative effect of ferroptosis on tumor cells is mainly to reduce the durability of tumor cells to chemotherapy, and drugs and activators related to ferroptosis can induce ferroptosis in tumor cells.
Ferroptosis is related to lipid peroxidation, and SREBP1 controls lipid synthesis and decomposition in vivo (Chen et al., 2021). SREBP1 can bind the important regulatory protein SCAP, which can activate SREBP1 through the endoplasmic reticulum, forming the SCAP/SREBP1 pathway, which can ensure the balance of glucose metabolism and lipid metabolism in the body (Oishi et al., 2017; Zeng et al., 2022). The bound complex is transported to the Golgi membrane through vesicles and then finds and binds to the receptor on the Golgi membrane, and the product, after hydrolysis, is transported to the nucleus, where the reaction takes place and the corresponding action is completed (Jin et al., 2021).
This study found that with the increase of dihydroartemisinin concentration, the viability of rectal cancer cells decreased continuously, which indicated that dihydroartemisinin could effectively inhibit the viability of rectal cancer cells. According to the results of HE staining, compared with the intact tissue structure of the control group, the rectal tissue epithelial cells of the model group were irregular and deformed, which were all improved in the SCAP/SREBP1 agonist and dihydroartemisinin group. However, the SCAP/SREBP1 inhibitor group was more serious than the model group, so it can be found that both SCAP/SREBP1 and dihydroartemisinin are related to rectal cancer cells and even inhibit rectal cancer cells. It can be seen that the inhibition rate of the inhibitor group is greater than that of the agonist group compared with the control group and the model group by measuring the cell proliferation with the MTT solution. As the concentration of DHA increased, the apoptosis of colorectal cancer cells also showed a positive correlation trend. This phenomenon was not affected by culture time (p <.05). In addition, from the results of the apoptosis rate of each group, it can be found that the effect of dihydroartemisinin on rectal cancer cells is related to the concentration and time. When the concentration reaches 50%, the apoptosis at 2 and 3 days reaches 20% and 25%, respectively. GSH decreased with the increase of the concentration of dihydroartemisinin, and the level of GSH was related to ferroptosis, indicating that dihydroartemisinin can promote the occurrence of ferroptosis and then promote apoptosis of cancer cells. Dihydroartemisinin can encourage the expression of SCAP/SREBP1 and reduce the expression of GPX4, and SCAP/SREBP1 can also affect the expression of GPX4. This shows that dihydroartemisinin can affect SCAP/SREBP1, and both dihydroartemisinin and SCAP/SREBP1 can affect ferroptosis, which can indirectly indicate that dihydroartemisinin can be detoxified through the SCAP/SREBP1 signaling pathway.
Conclusion
In summary, dihydroartemisinin can inhibit the viability and promote apoptosis of rectal cancer cells by increasing the concentration of dihydroartemisinin and SCAP/SREBP1 signaling, therefore inducing ferroptosis to cause cancer cell apoptosis. Dihydroartemisinin can be a therapeutic means to inhibit colorectal cancer cells. However, this study still has deficiencies. For example, the effect of dihydroartemisinin on rectal cancer through other signaling pathways is still unclear. Therefore, in-depth research will continue to explore its effects and mechanisms in the future.
Footnotes
Abbreviations
CCK-8: Cell counting kit-8; DHA: Dihydroartemisinic; GPX4: Glutathione peroxidase 4; GSH: Glutathione; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; qRT-PCR: Quantitative reverse transcription polymerase chain reaction; SCAP/SREBP1: Sterol regulatory element-binding protein cleavage-activating protein/sterol regulatory element-binding protein 1.
Acknowledgments
The authors gratefully acknowledge the Second Affiliated Hospital of Nanjing University of Chinese Medicine for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethics committee of the Second Affiliated Hospital of Nanjing University of Chinese Medicine.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
