Abstract
Background
Diabetic retinopathy (DR) is a severe microvascular complication of diabetes and a leading cause of vision impairment worldwide, with inflammation and apoptosis playing crucial roles in its pathogenesis.
Objectives
Based on a rat model of DR, the therapeutic effects of melatonin and a specific inhibitor on retinal lesions were compared, and their molecular mechanisms were explored.
Materials and Methods
We carefully selected 48 rats for the experiment. They will be evenly divided into four groups for the experiment: normal, inhibitor, model, and melatonin. The model, inhibitor, and melatonin groups underwent retinal lesion induction. After modeling, the model group received normal saline, the inhibitor group received SB 203580, and the melatonin group received melatonin. After 7 days, samples were collected for analysis. Western blotting was used to measure the protein expression levels of caspase-3, P38 mitogen-activated protein kinase (p38 MAPK), and phosphorylated P38 mitogen-activated protein kinase (p-p38 MAPK). The normal group served as an untreated control. Quantitative polymerase chain reaction (qPCR) was performed to detect Bcl-2-associated X protein (Bax) and B-cell lymphoma 2 (Bcl-2) messenger ribonucleic acid (mRNA) expression, and enzyme-linked immunosorbent assay (ELISA) was used to measure serum interleukin-1 beta (IL-1β) and interleukin-18 (IL-18) levels.
Results
Western blot analysis revealed no significant differences in the expression levels of p38 MAPK protein across all experimental groups (p > .05). However, the levels of phosphorylated p38 MAPK (P-p38 MAPK) and caspase-3 were significantly higher in the model, melatonin, and inhibitor groups compared to the normal group (p < .05). In contrast, these levels were reduced considerably in the melatonin and inhibitor groups relative to the model group (p < .05). qPCR analysis showed significant upregulation of Bax mRNA and downregulation of Bcl-2 mRNA in the model, melatonin, and inhibitor groups compared to the normal group (p < .05). After treatment, both the melatonin and inhibitor groups exhibited significant decreases in Bax expression and increases in Bcl-2 expression compared to the model group (p < .05). ELISA results indicated significantly higher levels of IL-1β and IL-18 in the model, melatonin, and inhibitor groups compared to the normal group (p < .05). Compared to the model group, both the melatonin and inhibitor groups showed significant reductions in IL-1β and IL-18 levels (p < .05).
Conclusion
Melatonin can suppress inflammation and apoptosis in DR rats by modulating the MAPK signaling pathway.
Keywords
Introduction
Diabetic patients often experience multiple complications, with diabetic retinopathy (DR) being one of the most severe, significantly impacting both health and daily activities. DR is the foremost cause of vision loss in individuals with diabetes. It is also the leading cause of bilateral blindness among patients seeking ophthalmic care (Griffith et al., 2012). After an in-depth study, it was found that the early pathological change of DR was inflammation, which could lead to necrosis and apoptosis of optic nerve cells, resulting in vision decline in patients with DR and blindness in severe cases (Li et al., 2017). Therefore, regulating the inflammatory response pathway of DR and inhibiting cell necrosis and apoptosis have become new ideas for the treatment of early DR.
Under hyperglycemic conditions, several interconnected signaling pathways are activated, leading to inflammatory responses, with the mitogen-activated protein kinase (MAPK) signaling pathway being one of them. Once activated, this pathway can exert its effects through multiple mechanisms. It can directly influence the expression of genes encoding inflammatory factors, thereby promoting their release. Additionally, the MAPK pathway can indirectly contribute to inflammation by modulating the activity of other signaling pathways or transcription factors that are involved in the inflammatory response. This dual role of the MAPK pathway underscores its significance in the regulation of inflammation and highlights its potential as a therapeutic target for inflammatory diseases. These inflammatory factors can reactivate the MAPK signaling pathway again, thus forming a vicious cycle and accelerating the progression and deterioration of DR. Therefore, the production of inflammatory factors can be inhibited. By reducing inflammatory factors, the progression of DR can be effectively slowed down, which is a new research direction at present.
Melatonin is an endogenous indoleamine synthesized by the pineal gland and part of the retina. It has functions such as inhibiting oxidation, preventing apoptosis, and reducing inflammation, and has been linked to various metabolic diseases. It has a certain protective effect on the heart, brain, liver, and other important organs. Some studies have found that melatonin is involved in regulating DR and has played a partial therapeutic role (D’Angelo et al., 2020).
Exploring the therapeutic mechanism of melatonin is an interesting and significant task. Melatonin can regulate the MAPK signaling pathway in the body, affect the release of inflammatory factors, and ultimately inhibit inflammation and apoptosis in DR rats. This research discovery offers novel insights into the clinical management of DR. The purpose of this article was to explore the therapeutic effect of melatonin on DR, especially its influence mechanism of inflammation and apoptosis, in order to provide a valuable reference for researchers in related fields.
Materials and Methods
Laboratory Animals and Grouping
In the current investigation, a cohort of 48 Sprague–Dawley rats was utilized, with an equal distribution of 24 male and 24 female subjects. These rats were sourced from a reputable supplier, Shanghai SLAC Laboratory Animal Co. Ltd., which holds a valid license. The rats exhibited a consistent body weight range, averaging 220 g with a permissible deviation of ±20 g. Divide them into groups of four rats each: the normal control group, which served as the baseline for comparison; the melatonin treatment group, which received melatonin supplementation; the disease model group, which was subjected to specific conditions to mimic the target pathology; and the inhibitor intervention group, which received a pharmacological inhibitor relevant to the study objectives. Prior to the commencement of the experiment, the study protocol underwent rigorous review and was granted approval by the Animal Ethical Committee of Dalian Medical University. This approval ensured that all procedures adhered to the highest ethical standards for animal research. Inclusion criteria included: all rats underwent a comprehensive health examination before the start of the experiment to ensure that there were no signs of disease and their weight was within the prescribed range. Rats showing signs of disease, substandard weight, or abnormal behavior before the experiment were excluded.
Laboratory Reagents and Instruments
This experiment employed a comprehensive set of reagents and equipment to ensure accurate and reliable results. The fluorescence quantitative polymerase chain reaction (qPCR) instrument used was Applied Biosystems 7500 (Foster City, CA, USA), which is widely recognized for its precision in nucleic acid amplification and detection. For microscopic analysis, an optical microscope (Leica DMI 4000B/DFC425C) from Wetzlar, Germany, was utilized to provide high-resolution imaging of cellular structures and tissues. An apoptosis detection kit from Sigma–Aldrich (St. Louis, MO, USA) was employed to assess cell death pathways, while an Image-Pro image analysis system from Bio-Rad (Hercules, CA, USA) was used for quantitative image analysis. The P38 mitogen-activated protein kinase (p38 MAPK) inhibitor SB 203580, obtained from MedChem express (MCE) (Monmouth Junction, NJ, USA), was used to specifically inhibit the p38 MAPK signaling pathway. Anti-Bax (Bcl-2-associated X protein), anti-caspase-3 (cysteinyl aspartate-specific proteinase-3), anti-p38 MAPK, and anti-phosphorylated (p)-p38 MAPK antibodies were all sourced from Abcam (Cambridge, MA, USA). These antibodies were essential for the detection and quantification of key proteins involved in apoptosis and stress signaling pathways. Additional reagents included an enzyme-linked immunosorbent assay (ELISA) kit from Boster (Wuhan, China) for cytokine detection, an AceQ QPCR SYBR Green Master Mix kit, and HiScript II Q RT SuperMix for qPCR (+gDNA wiper) kit from Vazyme (Nanjing, China) for qPCR applications. Streptozotocin (STZ), a widely used agent for inducing diabetes models, was purchased from Sigma–Aldrich. Finally, an Image Lab image analysis system was used for further analysis and documentation of experimental results.
Modeling
STZ was dissolved in 0.1M citric acid buffer (pH 4.5) and given to rats by intraperitoneal injection at a dose of 55 mg/kg. After injection, rats were placed in standard feeding conditions and freely ingested food and water. Through the tail vein blood sampling method, on the third day after injection of STZ, the fasting blood glucose level of rats was measured by a blood glucose meter, and the rats with significantly increased fasting blood glucose levels were regarded as successful establishments of the diabetes model.
Processing of Rats in Each Group
First of all, the basic living conditions and diets of the four groups of mice were all consistent. No additional artificial intervention was made to the rats in the normal group; only the environment and food necessary for growth were provided. Model group rats were constructed with DR rats, and the same amount of normal saline was given by intraperitoneal injection, and then they were killed. The rat model of DR was prepared in the melatonin group, and 10 mg/kg·d of melatonin was injected intraperitoneally. After the DR rat model was prepared in the inhibitor group, the inhibitor SB 203580 100 mg/kg·d was injected intraperitoneally. One hour later, intraperitoneal injection of melatonin 10 mg/kg·d was performed. The intervention lasted 7 days. Materials were collected.
Collection of Materials
Once the experimental models were successfully established, the corresponding treatments were initiated based on the assigned groups. After a 7-day treatment period, biological samples were collected from the mice for further analysis. The rats were anesthetized using an appropriate anesthetic protocol to ensure humane handling. The researchers precisely located the abdominal aorta of the mice (a major blood vessel that can provide sufficient blood volume for subsequent analysis) and collected blood samples from it. These blood samples were then centrifuged to separate the serum, which contains various proteins and other soluble factors that can be analyzed to assess the biological response to the treatments. The serum obtained through centrifugation was subjected to ELISA. This method can be an optimal solution for protein detection. In addition to blood sampling, retinal tissues were collected from a total of 12 rats for further molecular analysis. These tissues were processed for Western blotting, a technique used to detect specific proteins based on their molecular weight and antibody reactivity, and qPCR, a method for accurately measuring the expression levels of specific genes. The retinal tissues were stored and transported in Eppendorf (EP) tubes (Eppendorf, Hamburg, Germany), which are known for their reliability and compatibility with various laboratory procedures. These containers ensured the integrity and stability of the samples during processing and storage.
Western Blotting
To begin the Western blotting procedure, the lysis buffer was prepared, and previously frozen retinal tissue samples were carefully added to the buffer. We placed the ice on the outside of the mixture solution to store the obtained samples at a low temperature. This step was to prevent the proteins in the mixture from degrading at room temperature. Then, we incubated for 1 h. The time must be precisely controlled to ensure that the tissue is completely lysed within the incubation time to minimize experimental errors as much as possible. After incubation, we needed to obtain soluble protein. Therefore, we centrifuged the sample at 14,000 g for 10 min. After comprehensive consideration, we chose the bicinchoninic acid (BCA) assay to determine the protein content. This method involves measuring the optical density of the sample and generating a standard curve to determine the protein concentration using a microplate reader.
Next, the proteins were denatured to ensure they were in a suitable state for electrophoresis. We need to separate proteins of different qualities in advance for the subsequent experimental steps. The entire electrophoresis process is closely monitored. We need to observe whether the marked protein can be seen at the bottom of the gel. If so, it indicates that the separation is complete. Before the experiment starts, other experimental equipment also needs to be prepared. A polyvinylidene fluoride (PVDF) membrane needs to be prepared in advance, and then the separated proteins are transferred to this membrane according to the standard transfer protocol. After ensuring that the proteins are completely transferred to the membrane, we add the pre-prepared blocking solution. Keep it for one and a half hours to prevent the non-specific binding of antibodies.
Primary antibodies specific to the target proteins [p38 MAPK (1:1,000), phosphorylated P38 mitogen-activated protein kinase (p-p38 MAPK) (1:1,000), and caspase-3 (1:1,000)] were applied to the membrane. The primary antibody on the membrane will specifically bind to the corresponding antigen in a short while. This was followed by the application of a secondary antibody (1:1,000) conjugated to an enzyme or fluorophore to facilitate detection. The membrane was thoroughly washed to remove any unbound antibodies and then developed in the dark to prevent light-induced degradation of the signal. Finally, add the chemiluminescent reagent to the membrane. The staining time should last approximately 1 min. After the signal is fully developed, we can clearly observe the protein bands.
QPCR Assay
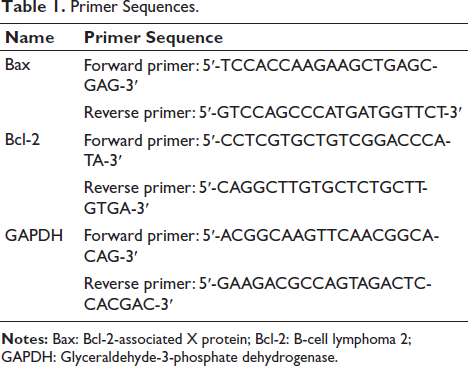
In the previous step, we obtained total ribonucleic acid (RNA). However, further experiments are needed. We still need to obtain the corresponding complementary deoxyribonucleic acid (cDNA). Therefore, cDNA can be obtained from RNA. Use a reverse transcription kit. Obtain cDNA for subsequent analysis as the template for further analysis. The reverse transcription process was carried out under the following conditions: First, we needed to heat the instrument. 51°C is a suitable temperature. Heat it initially to 51°C. Heat for 2 min. Then, further heating is required. 96°C is our next target temperature. Heat for 10 min. Immediately after, the temperature was lowered to 60°C for annealing, which lasted for 30 s. This completed one full reaction cycle. The above reaction was repeated 40 times. After the reaction was completed, we obtained a large amount of messenger ribonucleic acid (mRNA). These mRNAs need to be evaluated using additional experimental methods. Next, it is necessary to select an appropriate reference gene for subsequent assays. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) is a reference gene that meets our requirements. Table 1 presents the key materials needed for this experiment. These primer sequences were carefully designed to ensure specificity and efficiency in the amplification process, thereby enabling accurate quantification of the mRNA levels.
Primer Sequences.
ELISA
Abdominal aorta blood was collected and centrifuged, and the serum was taken. The ELISA kit was used for the following procedures: sample loading, addition of standard, biotinylated antibody working solution, enzyme conjugate working solution, and plate washing. The absorbance was determined using a microplate reader at 450 nm.
Statistical Analysis
The data analysis, statistics, and visualization of this study were completed by Statistical Package for the Social Sciences (SPSS) 20.0 software. Counting data are expressed by the mean and standard deviation to clearly show the centralized trend and variability of measured values. One-way analysis of variance (ANOVA) was used for multi-group comparison. When significant differences were found, the least significant difference (LSD) method was used to determine the specific differences. For normal distribution data with equal variance, the independent sample t-test is used to compare the two groups of mean values. When the variance is unequal, Welch’s t-test is used to explain. Non-normal or unequal variance data are tested by the non-parametric method. Rank data are analyzed by the rank sum test, which is suitable for ordered or non-normal data. Classification analysis uses the chi-square test to evaluate the correlation or differences between variables.
Results
p38 MAPK and p-p38 MAPK Detected via Western Blotting
The results presented in Figure 1 demonstrate that p38 MAPK protein expression was detectable in all experimental groups, indicating the presence of this protein kinase in both normal and disease model conditions. The levels of caspase-3 and phosphorylated p38 MAPK (p-p38 MAPK) were notably lower in the untreated normal group, suggesting a baseline level of activity for these proteins under physiological conditions. In contrast, the model group exhibited higher levels of caspase-3 and p-p38 MAPK, indicating increased activation of these proteins in the disease state.
Relevant Protein Expression Detected Through Western Blotting.
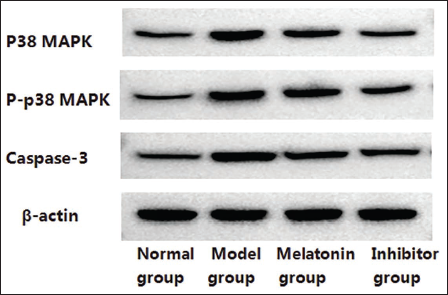
Statistical analysis, as shown in Figure 2, indicated no significant difference in the relative expression levels of p38 MAPK between the normal and model groups (p > .05), suggesting that the total protein levels of p38 MAPK were not significantly altered by the disease model. However, the levels of caspase-3 and p-p38 MAPK were significantly higher in the model group compared to the normal group (p < .05), highlighting the increased activity of these proteins in the disease condition.
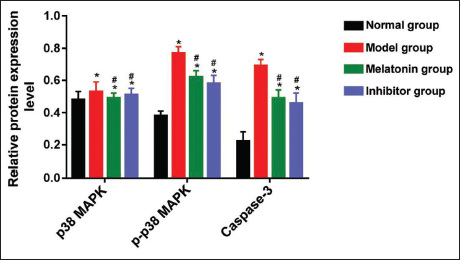
In rats treated with melatonin or inhibitors, the relative protein expression levels of p-p38 MAPK and caspase-3 were significantly reduced compared to the untreated model group (p < .05). This reduction suggests that both melatonin and inhibitors effectively attenuated the activation of these proteins, potentially mitigating the disease-related signaling pathways. Notably, the levels of p-p38 MAPK and caspase-3 in the treated groups were lower than those observed in the model group, indicating the therapeutic potential of these interventions. Additionally, no significant differences were observed between the melatonin-treated and inhibitor-treated groups in terms of p-p38 MAPK and caspase-3 expression (p > .05).
Related MRNA Expressions Detected by QPCR
As depicted in Figure 3, the relative mRNA expression levels of Bax and B-cell lymphoma 2 (Bcl-2) were analyzed in rats with DR modeling and compared to an untreated normal group. The results showed that rats with DR had a significantly higher relative mRNA expression level of Bax, a pro-apoptotic protein, and a considerably lower relative mRNA expression level of Bcl-2, an anti-apoptotic protein, compared to the normal group (p < .05). This suggests an imbalance in the apoptosis-related gene expression, favoring cell death in the disease model.
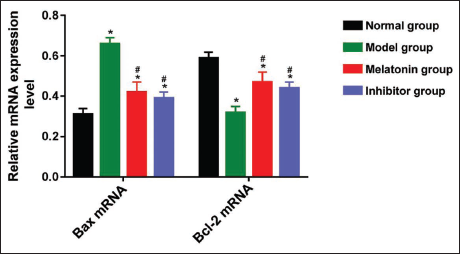
In rats treated with melatonin or the inhibitor, the relative mRNA expression level of Bax was significantly reduced compared to the model group, indicating a potential anti-apoptotic effect of these treatments. Additionally, the relatively low Bcl-2 expression observed in the model group was significantly increased after treatment with either melatonin or the inhibitor (p < .05), further supporting the notion that these interventions may restore the balance between pro- and anti-apoptotic factors. Importantly, no significant differences were found in the relative mRNA expression levels of Bax and Bcl-2 between the melatonin-treated and inhibitor-treated groups (p > .05), suggesting that both treatments had comparable effects on modulating apoptosis-related gene expression.
Results of ELISA
As shown in Figure 4, the ELISA was used to measure the levels of interleukin-1 beta (IL-1β) and interleukin-18 (IL-18) in the serum samples collected from the rats. The results indicated that, after the induction of the disease model, the levels of both IL-1β and IL-18 were significantly higher in the model, melatonin-treated, and inhibitor-treated groups compared to the untreated normal group (p < .05). This suggests an inflammatory response associated with the disease state.
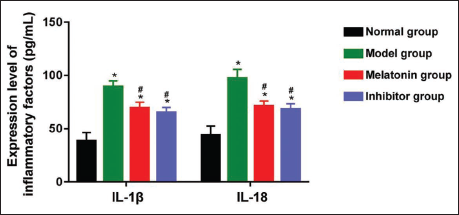
However, rats treated with melatonin or the inhibitor exhibited significantly lower levels of IL-1β and IL-18 compared to the model group (p < .05), indicating that both treatments effectively reduced the inflammatory response. The trends in IL-1β and IL-18 expression were similar between the melatonin-treated and inhibitor-treated groups, suggesting that both interventions had comparable anti-inflammatory effects. Statistical analysis revealed no significant differences in the levels of IL-1β and IL-18 between the melatonin-treated and inhibitor-treated groups (p > .05), further supporting the notion that both treatments were equally effective in modulating the inflammatory response.
Discussion
Existing studies on DR have shown that the visual function of patients with DR has already changed before the appearance of microvascular lesions, so early neuroprotection is of great importance. Retinal nerve cell apoptosis represents a fundamental pathological change that occurs in the early stages of diabetes mellitus. This process is characterized by programmed cell death of retinal neurons, which is triggered by various factors associated with hyperglycemia and metabolic disturbances. As diabetes progresses, the extent of apoptosis increases, leading to cumulative damage to the retinal structure and function. This progressive increase in apoptosis is a key contributor to the development of DR, a common and severe complication of diabetes that affects vision. Understanding the mechanisms underlying retinal nerve cell apoptosis is essential for developing therapeutic strategies to mitigate DR and preserve visual function.
DR can be classified according to the course of the disease. It can be further divided into mild, moderate, and severe stages. As the disease progresses, non-proliferative diabetic retinopathy (NPDR) may lead to proliferative diabetic retinopathy (PDR), the more advanced stage where new, fragile blood vessels form in the retina. These new vessels are prone to leaking and can cause severe vision impairment, including retinal detachment and neovascular glaucoma. Long-term hyperglycemia stimulation can lead to hypoxia and ischemia in retinal tissues and trigger inflammatory responses. This process leads to an increase in the expression of inflammatory factors, accompanied by the accumulation of white blood cells, disruption of the blood-retinal barrier, and the formation of new blood vessels (Barber, 2003). Studies have shown that during the NPDR stage, a large number of inflammatory factors are released, such as IL-1β and IL-18, and so on (Barber et al., 1998).
P38 MAPK is a key protein in the occurrence and development of DR (Blum et al., 2018). The main function of P38 MAPK is to produce inflammatory factors by regulating transcription factors such as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). Liu et al. (2017) found that the phosphorylation levels of reactive oxygen species (ROS), malondialdehyde (MDA), p38MAPK, and JNK increased in retinal ganglion cells under a high glucose environment. El-Remessy et al. (2013) found that the expression of tumor necrosis factor alpha (TNF-α), vascular endothelial growth factor (VEGF), ICAM1, and p38MAPK in the retina of DR rats increased, leading to retinal cell apoptosis and increased vascular permeability. Zhu et al. (2017) found that high glucose stimulation increased the levels of p38MAPK and NF-κB in BV2 cells, damaged the integrity of the blood-retina barrier, and caused DR.
Bcl-2 is a key protein in the process of apoptosis mediated by oxidative stress. Bcl-2 is an important molecule that regulates mitochondrial apoptosis pathway and can inhibit apoptosis (Wang et al., 2019). The expression level of Bax directly reflects the degree of apoptosis (Ye et al., 2019). The expression of Bcl-2 protein in the retinal tissue of diabetic rats induced by STZ decreased, while the expression of Bax protein was increased. This indicates that STZ can induce apoptosis in retinal tissue. Bcl-2 expression increased, and Bax expression decreased after melatonin intervention, indicating that melatonin can inhibit STZ-induced retinal apoptosis in diabetic rats.
Melatonin has good anti-oxidative stress, anti-inflammatory, and anti-apoptosis activities (Ostjen et al., 2019). Li et al. (2013) showed that melatonin had neuroprotective effects on diabetic rats and was associated with inhibiting the apoptosis of retinal nerve cells. The pineal gland secretes melatonin and has non-toxic properties, making it relatively safe to use in the eyes (Meng et al., 2017); while melatonin is fat-soluble and water-soluble, both the cell membrane and mitochondrial membrane are composed of lipids, and this double solubility allows the drug to effectively cross the blood-eye barrier and the blood-aqueous barrier to play its full role in the eyes (Hu & Li, 2019). All the above important research results further prove the feasibility of melatonin in the treatment of early DR.
In the rat model of DR, the pathological development affected the expression of key proteins related to apoptosis. Specifically, the pro-apoptotic signal-related caspase-3 and Bax proteins were further activated and expressed in the retinal tissues of the diseased rats. The alteration in Bcl-2 expression indicates an increase in cell death in retinal tissue.
In addition to these changes in protein expression, the study also examined the inflammatory response in the DR model. During the development of diabetes, the expression of two inflammatory factors is activated. This increase in inflammatory markers suggests that inflammation may promote tissue damage and disease progression in the pathological process of DR.
The molecular mechanism of DR still needs further exploration. More evidence is needed to guide the potential therapeutic effect of melatonin. The results of this study provide some insights. After treatment with melatonin, significant reductions in the expression levels of caspase-3 and Bax were observed, indicating a potential anti-apoptotic effect. After treatment, the low expression of Bcl-2 caused by diabetic lesions was also significantly improved. This indicates that melatonin may help to adjust the balance of these two key proteins. Additionally, the levels of IL-1β and IL-18 were significantly reduced after melatonin treatment, highlighting its anti-inflammatory properties.
In order to clarify the potential mechanism of melatonin’s therapeutic effect, it is mainly reflected in its ability to inhibit inflammatory reactions and apoptosis. Studies have shown that melatonin can significantly reduce the levels of inflammatory factors IL-1β and IL-18, which is helpful in alleviating the inflammatory reaction of retinal tissue, which may slow down the progression of DR. In addition, melatonin also has an anti-apoptosis effect; by reducing the expression level of caspase-3 and Bax while increasing the expression of Bcl-2, it can adjust the balance between pro-apoptosis and anti-apoptosis proteins and protect retinal cells from apoptosis. This study discusses the function of the MAPK signaling pathway. This upregulation may promote inflammation and apoptosis. Importantly, melatonin treatment significantly reduced p-p38 MAPK expression, demonstrating an effect similar to that of p38 MAPK inhibitors. The results suggest that melatonin may be able to regulate the p38 MAPK pathway, thereby regulating inflammation and apoptosis in retinal tissue. Thus, melatonin has shown remarkable anti-inflammatory and anti-apoptosis effects in the treatment of DR. It can effectively reduce the expression of inflammatory factors and the level of apoptosis in the retina by regulating the p38 MAPK signaling pathway. Although some meaningful results have been achieved in this study, there are still some shortcomings. For example, this study only focuses on the effect of melatonin on the p38 MAPK signaling pathway but does not discuss other possible signaling pathways, such as NF-κB or ERK1/2, which may also play an important role in the inflammation and apoptosis of DR.
Conclusion
Melatonin is an important hormone that can effectively inhibit the inflammatory reaction in the retina of DR rats induced by STZ by regulating the MAPK signaling pathway. This regulation further leads to the reduction of apoptosis, thus protecting retinal cells to a certain extent and delaying the process of DR.
Footnotes
Abbreviations
ANOVA: Analysis of variance; Bax: Bcl-2-associated X protein; BCA: Bicinchoninic acid; Bcl-2: B-cell lymphoma 2; CAT: Catalase; cDNA: Complementary deoxyribonucleic acid; CRP: C-reactive protein; DR: Diabetic retinopathy; ELISA: Enzyme-linked immunosorbent assay; EP tubes: Eppendorf tubes; GSH: Glutathione; ICAM-1: Intercellular adhesion molecule 1; IL-1β: Interleukin-1 beta; IL-18: Interleukin-18; JNK: c-Jun N-terminal kinase; LSD: Least significant difference; MAPK: Mitogen-activated protein kinase; MCE: MedChem express; MDA: Malondialdehyde; mRNA: Messenger ribonucleic acid; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; NPDR: Non-proliferative diabetic retinopathy; PDR: Proliferative diabetic retinopathy; P38 MAPK: P38 mitogen-activated protein kinase; p-p38 MAPK: Phosphorylated P38 mitogen-activated protein kinase; PVDF: Polyvinylidene fluoride; qPCR: Quantitative polymerase chain reaction; RNA: Ribonucleic acid; ROS: Reactive oxygen species; SOD: Superoxide dismutase; SPSS: Statistical Package for the Social Sciences; STZ: Streptozotocin; TCA: Tricarboxylic acid cycle; T lymphocytes: T-cell lymphocytes; TNF-α: Tumor necrosis factor alpha; VEGF: Vascular endothelial growth factor.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
