Abstract
Thyroid hormone deficiency can impair testicular function. However, knowledge of the effects of mitogen-activated protein kinase (MAPK) pathways on testicular mitochondrial oxidative damage induced by hypothyroidism is still rudimentary. This study aims to explore the possible mechanisms of testicular mitochondrial oxidative damage in hypothyroidism rats. Wistar male rats were randomly divided into control (C), low- (L), and high-hypothyroidism (H) groups (1 ml/100 g body weights (BWs)/day 0, 0.001% and 0.1% propylthiouracil, respectively) by intragastric gavage for 60 days. Blood samples were collected to measure the levels of serum triiodothyronine (T3), thyroxine (T4), and thyroid stimulating hormone (TSH). Testicular mitochondrial homogenates were used to measure the activities of superoxide dismutase (SOD), catalase (CAT), and Ca2+-ATPase as well as protein and mRNA expression of androgen receptor (AR), p38 MAPK, and c-Jun NH2-terminal kinase (JNK). Results showed that the BWs, testes weights, and levels of T3 and T4 were all significantly decreased and the testes coefficient and level of TSH were significantly increased in the H group. There were significant decreases in SOD activity in the H group as well as decreases in CAT and Ca2+-ATPase activities in the L and H groups. Additionally, protein expression of AR decreased significantly and protein expression of phosphorylated p38MAPK and JNK increased significantly in the H group. Therefore, the study suggests that hypothyroidism could affect male reproductive function by disturbing expression of AR, changing the activity of Ca2+-ATPase, inducing oxidative stress and then leading to activation of p38MAPK and JNK signaling in the testicular mitochondria.
Keywords
Introduction
Iodine deficiency leads to a spectrum of disorders that are collectively called iodine deficiency disorder and hypothyroidism. Iodine is a crucial factor in the synthesis of thyroid hormone (TH). TH is essential for the growth of animals and is a major regulator of oxygen consumption, mitochondrial energy metabolism, and many biological processes. 1,2 TH regulates the maturation and growth of testis during testicular development in mammals, and testicular function can be impaired under TH dysfunction. 3
The androgen receptor (AR) is a single-copy gene on the X chromosome and a ligand-dependent transcription factor that belongs to the steroid hormone receptor family and plays a crucial role in the pathogenesis of infertility. 4,5 The level of AR is regulated by steroid ligand endogenous testosterone (T), which is based on the transcriptional regulatory function of AR. 6 It has been demonstrated that AR expression is significantly decreased in stress-induced rats, which may result in reduction of T, disrupt AR signaling, and induce cell apoptosis. 7 In addition, AR plays a vital function in regulating T synthetized, which is related to the regulation of cell proliferation and apoptosis. The classical genomic mechanism of T signaling occurs when T diffuses into the cell and binds to AR, then the ligand receptor complex translocates to the nucleus, where it binds to androgen response elements (AREs) in the regulatory regions of genes to modify their translation. Nonclassical signaling occurs when the ligand receptor complex or T itself activates non-genomic cytoplasmic signaling pathways, or when in certain circumstances, the AR binds to AREs in the absence of T. 8 Calcium is an intracellular signaling that is associated with regulating various cellular processes, including proliferation, differentiation, development, and cell death. Alteration of the Ca2+ concentration in the cytoplasm and mitochondrial damage is involved in cadmium-induced apoptosis. 9,10 Mitochondrial Ca2+-ATPase is a regulator that maintains the low level of free Ca2+ in cells, and T can restrict Ca2+ flux associated with AR, which leads to calcium overload and mitochondrial dysfunction. 11,12 Several molecular events, including overproduction of reactive oxygen species (ROS) and activation of signaling, may be involved in the process mentioned above. 13
Mitochondria are the major generators of ROS and targets of elevated ROS exposure. In a physiological state, there is a balance between the production of ROS and neutralization of ROS in the antioxidant system to avoid oxidative stress, and TH is an essential factor in this balance. 14,15 Superoxide dismutase (SOD) and catalase (CAT) play crucial roles in the antioxidant defense system. Alteration of the TH level is known to modulate testicular function by regulating antioxidation. 16 It can lead to oxidative stress when the balance between the production of ROS in the mitochondria and antioxidation of cells is disturbed. The antioxidant machineries of cells are related to several phase II detoxification enzymes, including SOD and CAT. Meanwhile, overexpression of the Ca2+-binding protein can protect against oxidative stress by reducing the production of ROS and increasing the activity of antioxidases, such as SOD and CAT. 17,18 However, the molecular mechanisms of hypothyroidism on AR, Ca2+ overload, and oxidative stress lack evidence and still require further investigation.
p38 mitogen-activated protein kinase (p38MAPK) and the c-Jun NH2-terminal kinase (JNK) belong to the MAPKs family, which are primarily activated by stress-related stimuli, and are largely associated with cell apoptosis in germ cells. 19,20 Dysregulation of mitochondrial activity was associated with excessive ROS formation, reduced mitochondrial membrane potential, and ROS-mediated activation of ASK1/p38MAPK. 21 JNK have an essential role in modulating the functions of proapoptotic and antiapoptotic proteins located in the mitochondria. Accordingly, following activation by apoptotic stimuli, JNK readily translocates to mitochondria and activates the caspase-9 cascade. 22 A previous study has reported that Ca2+ overload promotes apoptotic development by upregulating phosphorylation of p38MAPK. 10,23 p38MAPK and JNK phosphorylation were disrupted in a congenital hypothyroidism rat model, and thyroid stimulating hormone (TSH) regulated the phosphorylation of p38MAPK. 24 –26 However, the relationship between Ca2+-ATPase and p38MAPK as well as JNK signaling in hypothyroidism still needs to be researched.
Propylthiouracil (PTU) is used for treating hyperthyroid condition. 14 PTU can decrease the conversion of peripheral thyroxine (T4) to triiodothyronine (T3) and reduce the serum T3 concentration, so it is usually applied to induce chemical induction of a hypothyroid state. 2
In our previous study, changes in sex hormones, sperm motility, testicular minerals, oxidative stress, and c-Fos abnormal expression in hypothyroid male rats were determined and resulted in the alteration of the testicular cell cycle as well as apoptosis and eventually lead to testicular dysfunction. 15 In the present study, a hypothyroid rat model induced by PTU was established to explore the activities of SOD, CAT, and Ca2+-ATPase as well as protein and mRNA expression of AR, p38MAPK, and JNK in testicular mitochondria to further understand the molecular mechanism of testicular dysfunction in hypothyroid males.
Materials and methods
Chemicals and reagents
PTU was obtained from Shanghai Chaohui Pharmaceutical Co., Ltd (Shanghai, China). SOD, CAT, and Ca2+-ATPase kits were obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Radioimmunoprecipitation Assay (RIPA) buffer, phenylmethanesulfonyl fluoride, and phosphatase inhibitors were obtained from Solarbio Sciences and Technology Co., Ltd (Beijing, China). Antibodies against AR, p38MAPK, JNK, phosphorylated p38MAPK (p-p38MAPK), and phosphorylated JNK (p-JNK) were provided by Abcam Biotechnology (Cambridge, Massachusetts, USA), and β-actin was purchased from Zhongshan Golden Bridge Biotech (Beijing, China). Peroxidase-conjugated secondary antibodies were purchased from Elabscience Biotechnology Co., Ltd (Wuhan, China). Bicinchoninic acid (BCA) and enhanced chemiluminescence (ECL) were provided by Thermo Scientific Biotechnology (Massachusetts, USA). TRIzol reagent was obtained from Life Technologies (Carlsbad, California, USA).
Animals and experiments
Thirty male Wistar rats (40 days old, 27 240–270 g) were obtained from the Center for Experimental Animals at Gansu University of Chinese Medicine (Gansu, China) with a National Animal Use License number of SCXK (Gan 2015-0002), and they were acclimated in the laboratory for 1 week before the experiment. Rats were housed in plastic cages maintained in an air-conditioned animal room (temperature: 20 ± 1°C, relative humidity: 50 ± 5%) with a 12-h light/12-h dark cycle; rats received a standard chow diet and had ad libitum access to tap water.
Then, rats were randomly divided into three groups (n = 10 per group), the control (C), low dose (L), and high dose (H) groups, and received normal saline, 0.001%, and 0.1% PTU by intragastric gavage for 60 days, respectively. 15,28 Their body weights (BWs) were measured once every 3 days.
All experiments and treatments were approved by the Institutional Animal Ethical Clearance of Lanzhou University (ethical clearance number: IRB160218-1). All efforts were made to minimize the number of rats used in the study and reduce their suffering.
Sample preparation and hormone estimations
All rats were anesthetized with diethyl ether and then killed by cervical dislocation after 60 days following a standard protocol and ethical procedures. 29,30 Then, blood samples were rapidly obtained by cardiac puncture, and serum was obtained by centrifugation at 1204 × g for 10 min. After the pretreatment of serum sample according to the manufacturer’s instructions, T3, T4, and TSH were analyzed in γ-immuno counter by the method of radioimmunoassay. 15 The bilateral testes were removed, washed with cold PBS, weighed, and prepared for testicular mitochondria homogenization to measure the activities of SOD, CAT, and Ca2+-ATPase as well as protein and mRNA expression of AR, p38MAPK, and JNK.
BWs and testes coefficient
The initial and final BWs were recorded on the first day and day of killing. The testes coefficient was defined as testes weights/100g BWs.
Preparation of the mitochondrial homogenate
Briefly, rat testes were washed with ice-cold isolation buffer (100.02 μM EDTA-2Na, 9.99 mM Tris, 136.89 mM sodium chloride (NaCl), and 9.99 mM sucrose, pH 7.4), minced into small pieces, and then homogenized. The homogenate was centrifuged at 4°C and 1204 × g for 10 min. The supernatant was collected and centrifuged again at 4°C, 12,000 × g for 10 min. 31 The pellet was resuspended in lysis buffer for Western blot analysis of mitochondrial protein and in TRIzol for mitochondrial mRNA analysis. Meanwhile, the mitochondrial suspension was applied to examine the activities of SOD, CAT, and Ca2+-ATPase.
Measurements of SOD, CAT, and Ca2+-ATPase
The activities of total SOD (hydroxylamine method), CAT (visible light method), and Ca2+-ATPase (colorimetric method) were assayed using reagent kits that were completed according to the manufacturer’s instructions by the assay of ultraviolet–visible spectroscopy, and the activities were expressed as unit per milligram of protein. 15 The lowest detection limits of SOD, CAT, and Ca2+-ATPase are 5.0, 0.2, and 0.05 U/ml, respectively.
Western blotting
AR, p38MAPK, JNK, and phosphorylated protein expression were measured using Western blotting. After testicular mitochondrial preparation, protein samples were prepared by incubating them in RIPA buffer that contained a protease inhibitor and phosphatase inhibitor. The protein concentration was determined using a BCA protein assay kit. Twenty micrograms (μg) of proteins was boiled with 5× sodium dodecyl sulfate (SDS) sample buffer (250 mM Tris-hydrogen chloride (HCl), pH 6.8, 7.46 mM bromophenol blue, 346.76 mM SDS, 5% β-mercaptoethanol, 50% glycerol) for 10 min. After denaturation, the samples were subjected to 8% SDS-polyacrylamide gel electrophoresis at 80 V for 30 min and then at 120 V for 70 min in SDS running buffer (25.01 mM Tris, 191.82 mM glycine, 3.47 mM SDS); then, the samples were subsequently transferred to polyvinylidene membranes at 210 mA for 2.5 h in transfer buffer (25.01 mM Tris, 191.82 mM glycine, 20% methanol). 15,31,32
The membranes were blocked for 2 h at room temperature in Tris-buffered saline/Tween-20 (TBS-T) (10 mM Tris-HCl (pH 7.5), 150.58 mM NaCl, 0.1% Tween-20) containing 5% bovine serum albumin (BSA) and then incubated overnight at 4°C with primary antibodies. The dilution ratio of anti-AR was 1:200, and the ratios of anti-p38MAPK, anti-p-p38MAPK, anti-JNK, anti-p-JNK, and anti-β-actin were all 1:1000. The antibodies were all diluted in TBS-T containing 5% BSA. The membranes were washed and incubated with peroxidase-conjugated secondary anti-mouse or anti-rabbit IgG antibodies diluted in TBS-T for 2 h at room temperature, and the dilution ratio of the secondary antibodies was 1:4000. 15,31,32 Subsequently, the membranes were developed using ECL Western blotting detection reagents, and the images of the blots were obtained with the Bio-Rad Molecular Imaging ChemiDoc™ XRS+ System (Bio-Rad, USA). The intensity of each chemiluminescent band was quantified using Image ProPlus 6.0 software (USA).
Real-time quantitative polymerase chain reaction
The mRNA expression of AR, p38MAPK, and JNK in the testicular mitochondria was determined by real-time (RT) quantitative polymerase chain reaction (PCR). Briefly, total RNA was extracted with TRIzol reagent according to the manufacturer’s protocol. 33 Then, 1.5 μg of total RNA was reverse transcribed with a PrimeScript™ RT Master Mix cDNA Synthesis Kit. RT-PCR was performed using a SYBR® Premix Ex Tap™ II Kit on iQ™ 5 RT-PCR Detection System (Bio-Rad, USA). This protocol involved an initial denaturation at 95°C for 30 s, 50 cycles of denaturing at 95°C for 5 s, annealing at 55°C for 30 s, and extension at 72°C for 30 s, according to the manufacturer’s instructions. The primer sequences are shown in Table 1, and an analysis of relative mRNA expression was performed using a method proposed by Pfaffl. 34
Primer sequences for RT-PCR analysis.

RT-PCR: real-time polymerase chain reaction; AR: androgen receptor; JNK: c-Jun NH2-terminal kinase; p38MAPK: p38 mitogen-activated protein kinase.
Statistical analysis
All data are presented as the mean ± standard deviation. Comparisons were performed with one-way analysis of variance among multiple groups and were followed by a least significant difference test between two groups using SPSS 22.0 statistics software (Chicago, Illinois, USA). Differences were considered to be significant when p < 0.05.
Results
The influence of hypothyroidism on BWs, testes weights, testes coefficient, and serum hormone levels
Significant decreases were observed in BWs for 30 and 60 days as well as testes weights for 60 days, whereas a significant increase in the testes coefficient was found in the H group compared to the C and L groups (p < 0.05; Table 2). The T3 and T4 levels in serum significantly decreased, whereas the TSH level significantly increased in the H group compared to the C and L groups (p < 0.05; Table 3).
The influence of hypothyroidism on BWs, testes weights, and organ coefficient.a
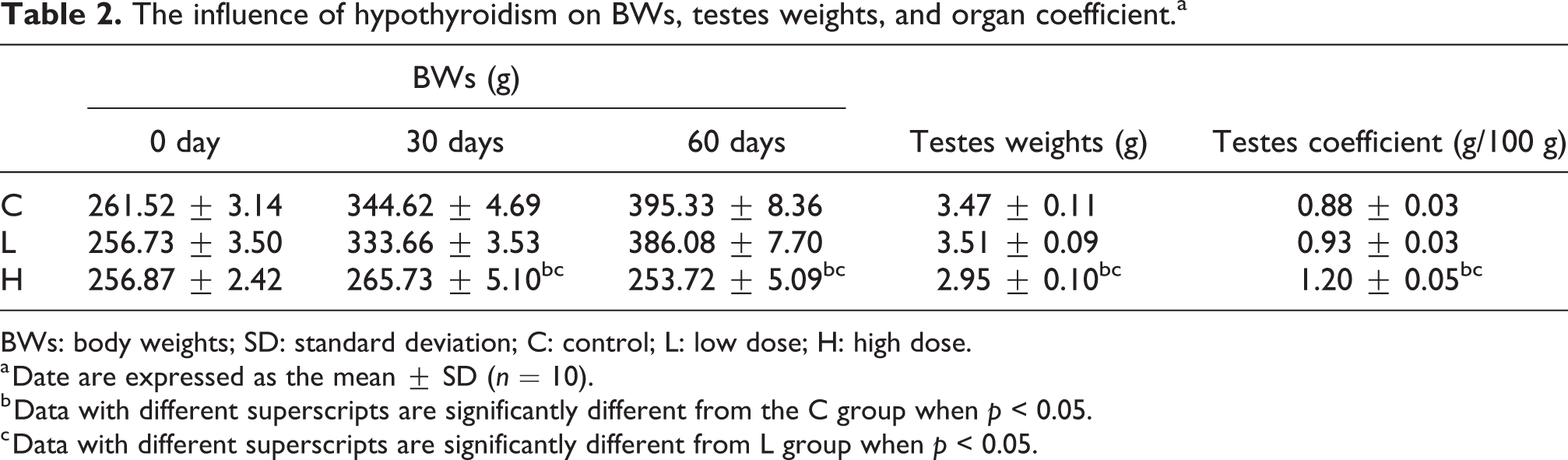
BWs: body weights; SD: standard deviation; C: control; L: low dose; H: high dose.
a Date are expressed as the mean ± SD (n = 10).
b Data with different superscripts are significantly different from the C group when p < 0.05.
c Data with different superscripts are significantly different from L group when p < 0.05.
The influence of hypothyroidism on the serum hormone level.a

T3: triiodothyronine; T4: thyroxine; TSH: thyroid stimulating hormone; SD: standard deviation; C: control; L: low dose; H: high dose.
a Date are expressed as the mean ± SD (n = 5).
b Data with different superscripts are significantly different from the C group when p < 0.05.
c Data with different superscripts are significantly different from the L group when p < 0.05.
Influence of hypothyroidism on SOD, CAT, and Ca2+-ATPase activities
As seen in Figure 1, there was a significant decrease in SOD activity found in the H group compared to the C group (p < 0.05). The CAT and Ca2+-ATPase activities were significantly decreased in the H (p < 0.001, p < 0.05) and L (p < 0.01, p < 0.05) groups compared to the C group.
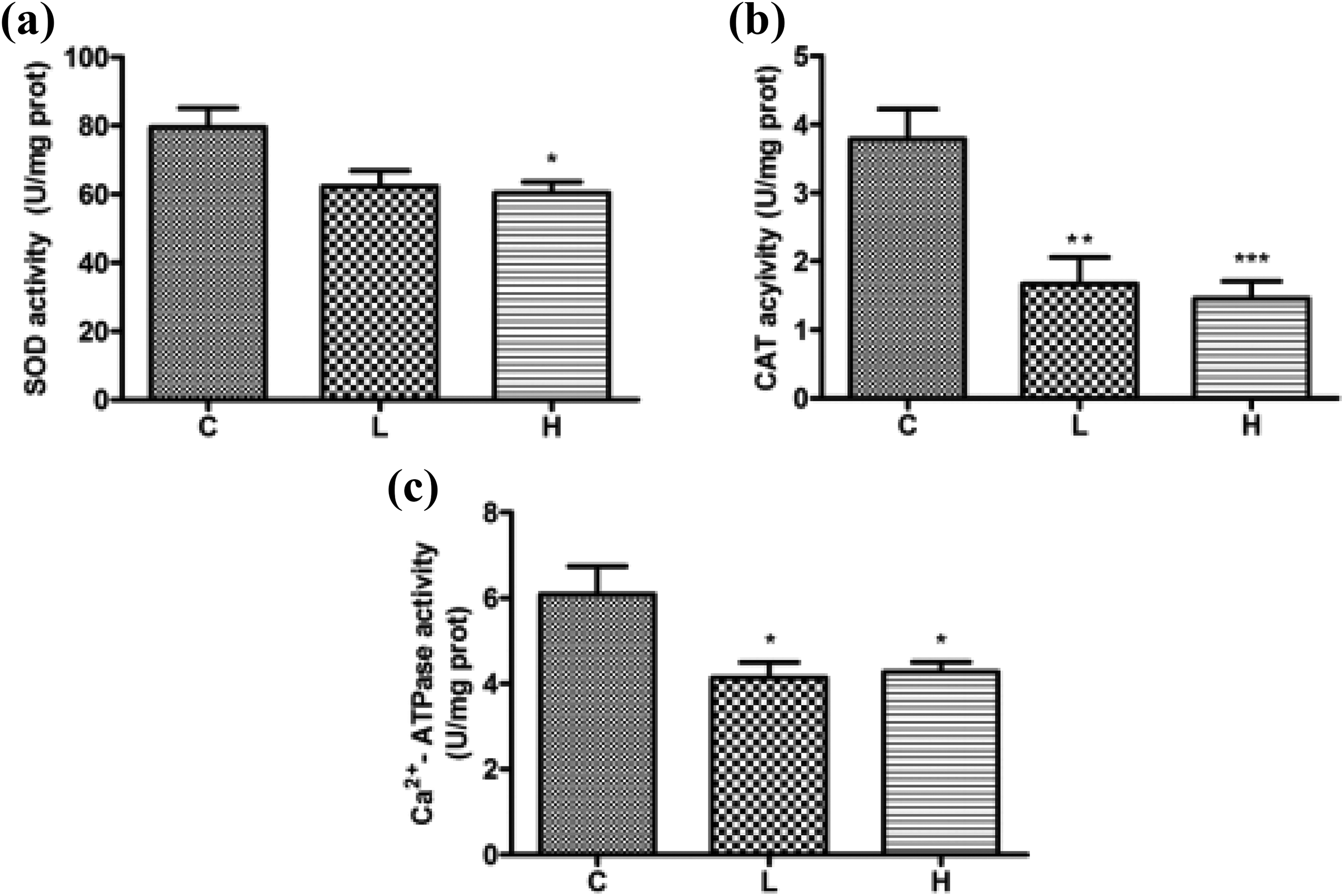
The influence of hypothyroidism on the activities of SOD (a), CAT (b), and Ca2+-ATPase (c) in testicular mitochondria. Data are expressed as the mean ± SD (n = 5). Data with different superscripts are significantly different from C group when *p < 0.05, **p < 0.01, and ***p < 0.001. SOD: superoxide dismutase; CAT: catalase; SD: standard deviation; C: control.
Influence of hypothyroidism on AR, p38MAPK, and JNK protein expression
Protein expression of AR, p38MAPK, and JNK was determined with a Western blotting analysis, and the results are shown in Figure 2. Protein expression of AR was significantly decreased in the H group compared to the C and L groups (p < 0.05). Phosphorylation of p38MAPK and JNK was significantly increased in the H group compared to the C (p < 0.001, p < 0.01) and L (p < 0.01, p < 0.05) groups.

Influence of hypothyroidism on protein expression of AR (a), p38MAPK (b), and JNK (c). The density of each band of AR was measured and compared to that of β-actin and p-p38; p-JNK was normalized to the respective total form of the protein. The results are expressed as the mean ± SD (n = 5). Data that have different superscripts are significantly different from the C group when *p < 0.05, **p < 0.01, and ***p < 0.001 and from L group when #p < 0.05 and ##p < 0.01. AR: androgen receptor; JNK: c-Jun NH2-terminal kinase; p38MAPK: p38 mitogen-activated protein kinase; SD: standard deviation; C: control: L: low dose.
Influence of hypothyroidism on AR, p38MAPK, and JNK mRNA expression
To determine whether the mRNA expression of AR, P38 MAPK, and JNK could be found in mitochondria, the mRNA expression of above three genes was measured by RT-PCR, and the results are shown in Figure 3. There was a significant increase in AR mRNA expression in the H group compared to the C group (p < 0.05). The mRNA expression of p38MAPK was significantly increased in the H group compared to the C group (p < 0.05), whereas there was no significant change in JNK mRNA expression among the different groups (p > 0.05).

Influence of hypothyroidism on mRNA expression of AR (a), p38MAPK (b), and JNK (c). Each mRNA was normalized to the β-actin mRNA expression level in the same preparation, and the mean of each experimental control was assigned a value of 1.0. The values are expressed as the mean ± SD (n = 5). Data with H group are significantly different from the C group when *p < 0.05. AR: androgen receptor; JNK: c-Jun NH2-terminal kinase; p38MAPK: p38 mitogen-activated protein kinase; SD: standard deviation; C: control; H: high dose.
Discussion
The results of this study showed that significant decreases in T3 and T4 and an increase in the TSH levels were detected in the H group compared to the C and L groups, which established a PTU-induced hypothyroidism model. This finding is consistent with previous evidence from Sahoo et al. and our research team. 2,15
As we know, THs are crucial factors for the growth of animals, and there were significantly decreases in BWs and testes weights in the H group of PTU-treated rats compared to the C and L groups in this study, which suggest that there is an association between decreased BWs and hypothyroidism. The current results were also consistent with previous studies. 2,35,36 The testes coefficient was increased in the H group compared with C and L groups, which might be owing to the fluid accumulation. 37 The different trends of it between the present and previous studies could be related to the great differences in the ratio between the BWs and the testes weights. The testes coefficient was defined as testes weights/100g BWs. In the previous study, the BWs of rats in the hypothyroid group changed from 100% to 68% and the testes weights changed from 100% to 70% compared with the C group, and the changed ranges of them were of no great difference. However, in this study, the BWs of rats in the H group changed from 100% to 64% and the testes weights changed from 100% to 85% compared with the C group, and the changed range in the BWs, maybe due to the individual difference of rats during their grow process, was larger than it in the testes weights.
Male reproduction is governed by the classical hypothalamo-hypophyseal testicular axis, and TH has been shown to exert a modulatory influence on this axis. 38 A previous study found that T is associated with the transcriptional regulatory function of AR. 6 Our previous study showed that hypothyroidism can decrease the level of T and increase the level of E2, 15 and our present study showed that hypothyroidism can decrease protein expression of AR and increase mRNA expression of AR. These findings demonstrated the relationship between the T level and AR in hypothyroidism rats. From genes to proteins, there are posttranslational mechanisms that control gene expression, including control of translation and protein half-life. A previous study suggested that decreased AR protein may be associated with the reduction in the level of aromatase induced by TH. 39,40 On the other hand, the T3 and T4 levels are related to the expression of AR mRNA in testicular tissues. 41 –43 At the same time, there was no specific trend between mRNA and protein expression in mammalian cells, 44 and microRNAs (miRNAs) regulate the expression of protein-coding genes. 45 The miRNAs are small (18–25 nucleotides), endogenous, noncoding RNA molecules involved in the posttranscriptional modulation of gene expression and the genes for these small noncoding RNAs are transcribed by RNA polymerase II and III to form mature miRNAs. 46,47 The function of miRNAs in posttranscriptional gene regulation can result in translational repression or mRNA degradation via binding to complementary sequences in the 3′-UTR of mRNAs. 48 Approximately, 700 miRNAs have been found in mammals and they play important roles in biological functions. 46,47 Among these miRNAs, miR-124a was a potent inhibitor of AR and it was found to bind the 3′-UTR of glucocorticoid and mineralocorticoid receptors directly, thereby reducing the protein levels and interfering with their translational activity. Stanley et al. also reported that an increased expression of miR-124a and a negative trend of AR protein expression were revealed in thyroid tumor. 48 The result of our study is consistent with the reports mentioned above. In this study, there was a significant decrease in AR protein expression, but not in AR mRNA expression, which might be related to the function of miRNAs in posttranscriptional gene regulation. The result of our study is consistent with the reports mentioned above. Of course, the exact mechanism warrants further study.
Ca2+-ATPase maintains the balance of the intracellular Ca2+ concentration, and intracellular Ca2+ can become overloaded when the balance is disrupted. Previous studies have confirmed that a disruption of the intracellular Ca2+ level can lead to oxidative stress and is associated with mitochondria dysfunction, which results in the opening of the high-conductance channel in the mitochondrial membrane, leading to intracellular Ca2+ overload. 13,49,50 In this study, decreased activity for Ca2+-ATPase was found. The Ca2+ mentioned above suggests that hypothyroidism may lead to an imbalance of Ca2+ homeostasis by changing the activity of Ca2+-ATPase. This observation agrees with the results of some previous studies. 49,50 Meanwhile, some researchers found that the endogenous T levels could restrict intracellular Ca2+ flux, which is involved in the expression of AR. 11,12 Therefore, it is plausible that the changes of AR expression can induce the unbalance of Ca2+ homeostasis that occurs in hypothyroidism.
It has also been shown that antioxidation is suppressed by iodine deficiency in hypothyroidism, which results in a state of cellular oxidative stress under excessive generation of ROS. 1,51 Oxidative stress is a critical pathogenic mechanism in various diseases, and ROS are also known to play a role in the pathogenesis of thyroid dysfunctions. 52 TH is essential endocrine regulator of the metabolic status and has critical effects on antioxidation. Studies have reported that T3 and T4 regulate oxygen consumption, affect spermatogenesis, and mediate the differentiation and maturation of germ cells. 53,54 In the present study, the decreases in the SOD and CAT activities were consisted with the decline in the T3 and T4 levels, which suggested an impairment of antioxidation in hypothyroidism. These results also agree with the studies of Sahoo et al. 2 and Bhanja and Chainy. 51 In addition, the altered T level and AR expression associated with the unbalance of Ca2+ homeostasis can regulate the activities of SOD and CAT. 6,50 Therefore, oxidative stress could also be induced by changing AR expression and disturbing the balance of Ca2+ homeostasis in hypothyroidism, consequently affecting reproductive function.
Furthermore, it has been shown that p38MAPK signaling is commonly related to cell death and germ cell apoptosis in hypothyroid rats. 24,55 In addition, there may be many effects, such as the activity of antioxidant defense enzymes, and the disruption of the phosphorylation/dephosphorylation balance on p38MAPK. 2,24 At the same time, the p38MAPK cascade is also activated by extracellular stimuli, such as oxidative stress, differentiation, and growth factors, which converge to activate MKK3/6, then phosphorylates and activates p38MAPK. 56 On the other hand, JNK signaling can be initiated by cytokines and stress stimuli factor, leading to activation of MKK4 or MKK7. Then, the Thr and Tyr residues of JNK are phosphorylated. It has been confirmed that JNK plays an essential role in modulating the functions of proapoptotic and antiapoptotic proteins located in the mitochondria as well as regulating the mitochondrial function and mitochondria-mediated apoptosis by increasing the expression of proapoptotic genes through transactivation of c-Jun/AP1-dependent mechanisms. 22,31 In addition, some studies have found that the intracellular Ca2+ overload and increase in cytosolic Ca2+ are caused by activation of p38MAPK and JNK signaling in adult rats. 10,23 The current study showed that the p38MAPK and JNK phosphorylation levels and p38MAPK mRNA expression levels were increased, while there was no significant change in JNK mRNA expression, which could be related to posttranslational mechanisms controlling gene expression, including translational control and control of protein half-life. Thus, there is no predictive correlation between steady-state levels of mRNA and those of protein in mammalian cells. 44 However, these findings suggest that hypothyroidism may affect reproduction by activating p38MAPK and JNK signaling, which is mediated by Ca2+ to some extent.
Conclusions
In summary, the results of this study indicate that hypothyroidism can lead to dysfunction of the testicular mitochondria, and the possible molecular mechanisms are changes in AR expression, and Ca2+-APTase activity, induction of oxidative stress and leading to alterations of p38MAPK and JNK signaling. Of course, the additional molecular mechanisms involved in these processes still need to be determined.
Footnotes
Author contribution
X-R Chang and Y-L Yao contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (8157120143) and the Fundamental Research Funds for the Central Universities (lzujbky-2017-it34).
