Abstract
Background
Diabetic nephropathy (DN) is a serious complication of both type 1 and type 2 diabetes defined by progressive kidney damage and, ultimately, kidney failure. The growing prevalence of diabetes worldwide, coupled with lifestyle changes, has led to a rise in the incidence of DN, posing a significant public health and financial burden.
Objectives
This work aims to assess the therapeutic significance of yangonin on DN in an experimental rat model.
Materials and Methods
The experimental rats received 65 mg/kg of streptozotocin (STZ) to induce DN. The rats with DN were then treated with yangonin for 12 weeks. After the treatments had been completed, the body weight changes and blood glucose levels in the experimental rats were determined. The kidney dysfunction biomarkers, including creatinine, urea, uric acid, blood urea nitrogen (BUN), and marker enzymes, were assessed utilizing commercial assay kits. The concentrations of inflammatory cytokines and oxidative stress-related biomarkers were evaluated using assay kits. The renal tissues of experimental rats underwent histological study.
Results
The treatment of yangonin considerably elevated the body weight and subsequent reduction in glucose levels in the DN rats. Furthermore, the concentrations of renal dysfunction markers and marker enzymes were decreased by yangonin in the DN rats. The yangonin effectively reduced inflammatory response and oxidative stress by boosting the anti-oxidant levels in rats with DN. The histological analysis results further confirmed the therapeutic efficacy of yangonin against DN.
Conclusion
The current findings indicate that yangonin may effectively mitigate DN in rats, which highlights that yangonin possesses the potential to serve as an advantageous treatment option for DN.
Introduction
Diabetic nephropathy (DN), a serious complication of a diabetic condition, is a progressive kidney disease defined by the gradual deterioration of the kidneys’ filtration ability, resulting in the accumulation of waste products in the body. This condition affects up to 50% of individuals living with both type 1 and type 2 diabetes. It is a pivotal cause of end-stage renal disease (ESRD), which often results in dialysis or kidney transplantation (Swaminathan et al., 2024). The pathophysiology of DN is multifaceted, involving a complex interplay of metabolic, hemodynamic, and inflammatory factors. Sustained hyperglycemia, a hallmark of diabetes, triggers a cascade of events, including the activation of various biochemical pathways, which contribute to structural and functional modifications in the kidneys. Additionally, glomerular hyperfiltration and increased intraglomerular pressure, driven by hemodynamic alterations, play a crucial role in the onset of DN (Jung & Yoo, 2022). The burden of DN is significant, both for the individual and the healthcare system. The economic impact of DN is also substantial, with the disease accounting for a significant portion of healthcare expenditures related to diabetes (Zhao et al., 2024).
The pathophysiology of DN is closely linked to the interplay between oxidative stress and inflammation. Oxidative stress, a condition of disproportion between free radical production and the body’s anti-oxidant defense mechanisms, is a hallmark of the diabetic state (Darenskaya et al., 2021). Inflammation, another key player in the onset of DN, is closely intertwined with oxidative stress. The production of proinflammatory cytokines can result in nerve tissue damage and the development of debilitating neuropathies (Liang et al., 2024). The interplay between oxidative stress and inflammation in the onset of DN is a complex and multifaceted process. Emerging strategies aimed at neutralizing or targeting the underlying mechanisms of oxidative stress and inflammation may hold promise in the future management of this debilitating complication of diabetes (Jin et al., 2023).
Despite the availability of various treatment modalities, managing DN remains a significant challenge. The current mainstay of treatment for DN focuses on tight glycemic control, blood pressure management, and the use of renin–angiotensin–aldosterone system inhibitors. However, even with these interventions, a significant proportion of patients with DN continue to experience disease progression and the development of ESRD (Psyllaki & Tziomalos, 2024). One of the key challenges in the management of DN is the altered pharmacokinetics and pharmacodynamics of glucose-lowering medications in patients with renal impairment. Further research is needed to develop novel therapeutic strategies that target the underlying pathogenic mechanisms of DN. Current treatment options, while moderately effective, face significant challenges in preventing the development of DN (Karalliedde et al., 2022). One promising avenue for addressing these challenges is the exploration of plant-derived bioactive compounds and their applications in managing DN. These naturally occurring molecules have shown promising results in targeting the various pathways involved in the pathogenesis of the disease (Zac-Varghese et al., 2024). Yangonin is one of the major kavalactones, which extensively occurs in the kava plant (Piper methysticum) roots. Numerous previous studies already highlighted the pharmacological properties of yangonin, including anti-inflammation and anti-nociception (Chow et al., 2024), anti-osteoporosis (Lu et al., 2022), hepatoprotective (Kong et al., 2021), anti-cholestatic (Gao et al., 2018), and anti-cancer (Liu et al., 2017) properties. Nonetheless, its therapeutic functions in relation to diabetes-associated complications, particularly DN, have not been documented yet. Hence, this work aims to study the therapeutic significance of yangonin on DN in experimental rats.
Materials and Methods
Chemicals
The major chemicals utilized in the present work, including yangonin, streptozotocin (STZ), and so forth, were purchased from Sigma–Aldrich, USA. The assay kits for biochemical markers were procured from Abcam, USA.
Animals
Male Wistar rats aged 8 weeks and weighing 180–210 g were utilized in the current work. The rats were caged in sterilized confines inside controlled laboratory conditions, maintaining a temperature of 22°C–26°C and humidity of 40%–60%. A 12-h cycle of alternating light and dark was established. Throughout the entire trial period, all rodents were granted unrestricted access to standard rodent chow (Bio-Serv, USA) and filtered drinking water. The institutional animal ethics committee approved the experimental procedures for the animals. Prior to the commencement of the trial, all rats were allotted a 7-day acclimatization period in a laboratory setting.
Experimental Groups
Following a 1-week acclimatization period, the experimental rats were divided into four groups, with six rats in each group. The normal control (NC) group received only rodent food and was administered merely a buffered saline solution without any drugs/compounds (Group I). The rats from the disease group were administered a single dose of STZ at a concentration of 65 mg/kg (dissolved in 0.01 M sodium citrate buffer; pH 4.5) to initiate DN (Group II). The rats were given glucose (0.5%) to prevent mortality resulting from abrupt hypoglycemia. After a 3-day administration of STZ, the blood glucose was evaluated. Rats demonstrating glucose levels over 250 mg/dL were fixed as diabetic and chosen for subsequent testing. In the treatment group, the rats with STZ-induced DN were subsequently administered a dosage of 50 mg/kg of yangonin (Kong et al., 2021) for 10 weeks (Group III). In the standard group, animals were induced with DN and administered a dosage of 350 mg/kg of metformin for 10 weeks (Group IV). The body weight of each rat was meticulously measured throughout the study. The blood glucose concentrations of the rats were determined in tail tip samples using a commercial glucometer (Roche).
Analysis of Renal Dysfunction Markers
The serum samples were utilized to examine the renal dysfunction markers, including creatinine (ab65340), uric acid (ab65344), urea (ab83362), blood urea nitrogen (BUN; ab83362), aspartate transaminase (AST; #ab263883), and alanine transaminase (ALT; #ab285264) using commercial diagnostic kits. The tests were conducted using three replicates in accordance with the directions provided by the assay kit manufacturer (Abcam, USA).
Oxidative Stress Markers
The levels of the oxidative stress marker and anti-oxidants in the renal tissues of the experimental rats were assessed using an assay kit. The concentrations of glutathione (GSH), catalase (CAT), superoxide dismutase (SOD), and malondialdehyde (MDA) were assessed using the kits. The tests were conducted in triplicate and executed in accordance with the manufacturer’s specified protocols (Abcam, USA).
Analysis of Inflammatory Cytokines
The inflammatory cytokine concentrations, including interleukin (IL)-6 and tumor necrosis factor-alpha (TNF-α) in the renal tissue homogenates, were assessed using standard diagnostic kits (Abcam, USA). The tests were conducted according to the manufacturer’s specifications for the kits.
Histopathological Analysis
The kidney tissue samples were collected and treated with a formalin solution (10%) to evaluate the changes in kidney histopathology. Subsequently, the kidney tissues were paraffinized, cut into 5 µm size, and stained with eosin-hematoxylin. Finally, the kidney tissues were examined microscopically to assess the histological alterations (Chtourou et al., 2022).
Statistical Analysis
The results are shown as a mean ± SD from three replicate assays. A one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT) were employed to study the statistical significance using GraphPad software (version 9), with p < .05 denoting significance.
Results
Effect of Yangonin on Body Weight and Glucose Levels in Experimental Rats
The results of yangonin on body weight and glucose level in the rats were assessed, as depicted in Figure 1. The diabetes nephropathy-induced rats showed a significant (p < .01) diminution in body weight and subsequent elevation in their glucose concentration in comparison to the control. Interestingly, the yangonin at 50 mg/kg of concentration significantly (p < .05) reduced the glucose and increased the body weight in rats with diabetes nephropathy. This activity of yangonin is also supported by the results of metformin, which also reduces blood glucose and elevates body weight in DN rats (Figure 1).
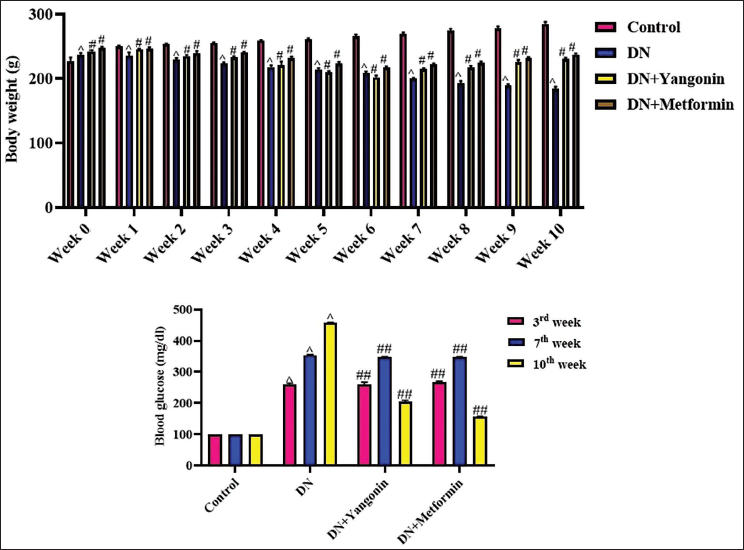
Effect of Yangonin on the Renal Dysfunction Markers in Experimental Rats
Figure 2 illustrates the concentrations of renal dysfunction markers in the rats. The rats with DN demonstrated a significant (p < .01) increase in serum concentrations of creatinine, uric acid, urea, and BUN. Furthermore, the increased AST and ALT activities were also noted in the DN rats (Figure 3). Captivatingly, the yangonin at 50 mg/kg concentration exhibited a significant (p < .05) reduction in the concentrations of these renal dysfunction markers. The treatment of standard drug metformin also effectively reduced these renal dysfunction marker levels, which further supports the activity of yangonin.
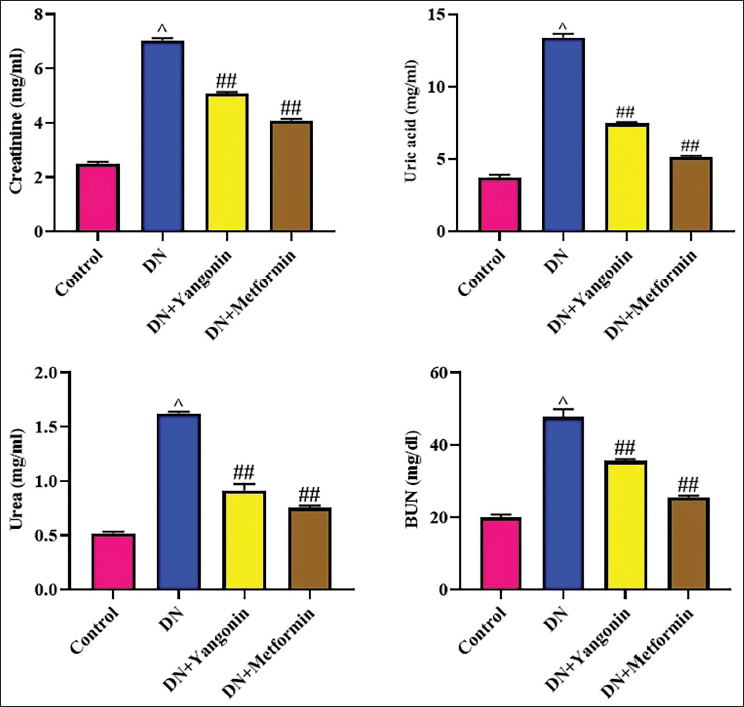
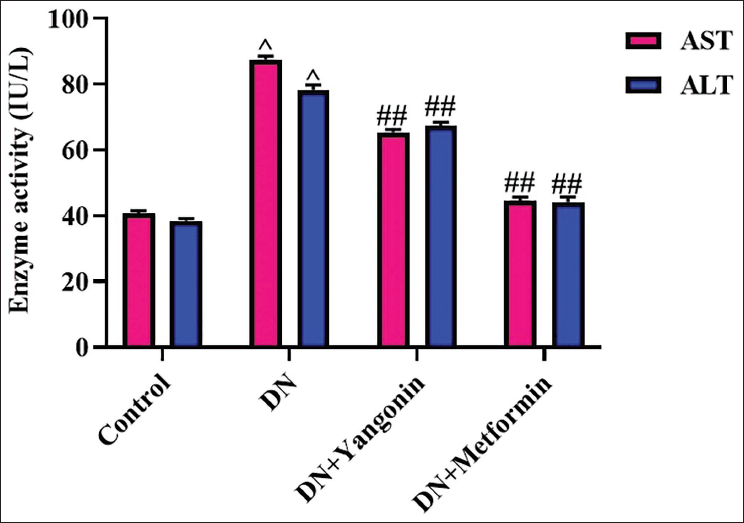
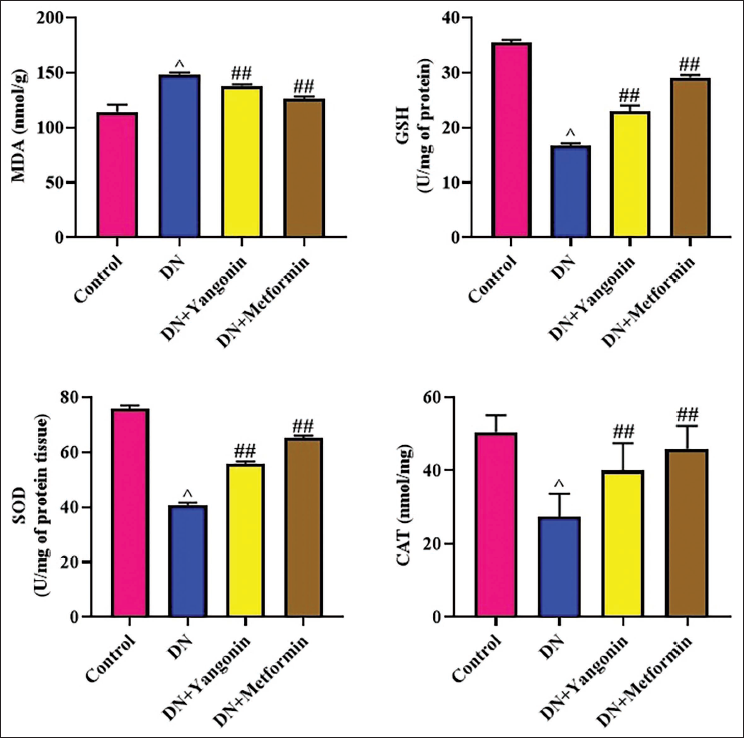
Effect of Yangonin on Oxidative Stress Level in Experimental Rats
Oxidative stress levels were assessed in the renal tissues of the experimental rats (Figure 4). DN rats demonstrated reduced anti-oxidants (p < .01), including CAT, SOD, and GSH, and a subsequent increase in MDA levels in their kidney tissues when compared with control. Nevertheless, the rats with DN demonstrated a significant (p < .05) elevation in the concentrations of SOD, GSH, and CAT and a reduction in MDA in their kidney tissues. Furthermore, the treatment of metformin also reduced the MDA and increased the anti-oxidants in the kidneys of DN rats, which further supports the anti-oxidant properties of the yangonin (Figure 4).
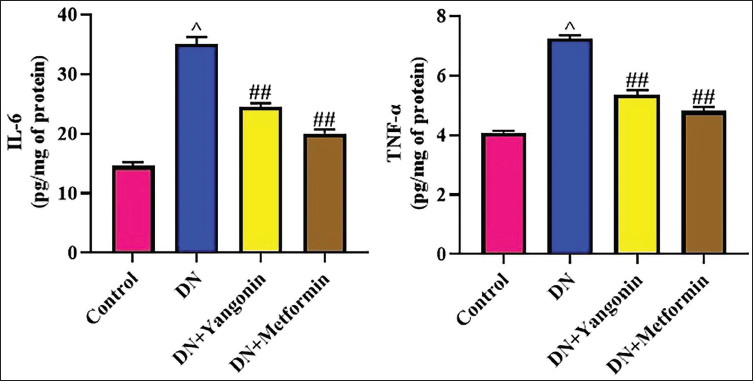
Effect of Yangonin on Inflammatory Cytokines in Experimental Rats
The anti-inflammatory effects of yangonin in the DN rats were evaluated by measuring the concentrations of inflammatory cytokines (Figure 5). The rats with STZ-induced DN demonstrated significant (p < .01) elevations in the IL-6 and TNF-α concentrations relative to the control. In contrast, 50 mg/kg of yangonin significantly (p < .05) reduced the IL-6 and TNF-α levels in the rats with DN (Figure 5). The anti-inflammatory properties of yangonin are further supported by the results of metformin, which also diminished these cytokine levels in DN rats.
Effect of Yangonin on Renal Tissue Histopathology of Experimental Rats
Figure 6 displays the histological studies of the renal tissues. The kidney tissues from the control rats exhibited a normal appearance with characteristic renal histoarchitectures. Contrastingly, the renal tissues of the DN rats exhibited glomerular shrinkage, cellular apoptosis, compromised renal tubular lining cells, tissue degradation, edema, and an elevated infiltration of inflammatory cells. Interestingly, the yangonin at 50 mg/kg concentration effectively mitigated these histological abnormalities in the kidney tissues of rats with DN. The standard drug metformin also reduced the histological changes in the kidney tissues of the DN rats (Figure 6).
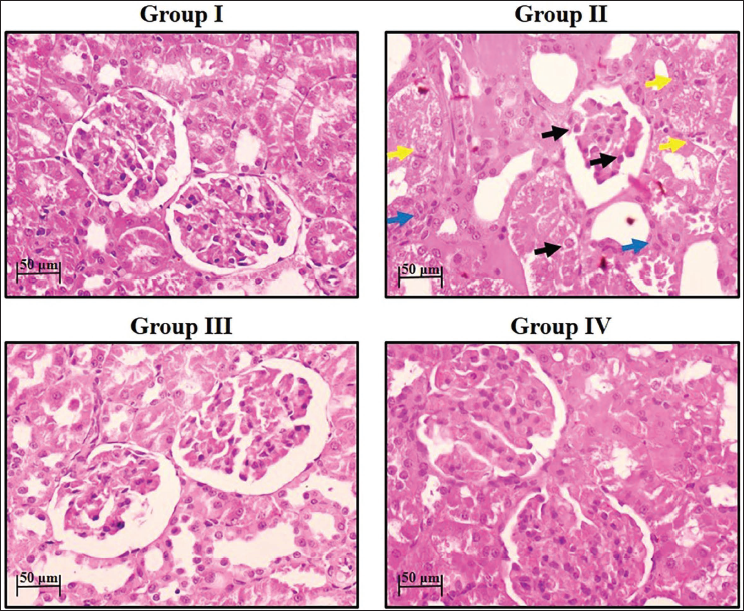
Group I: The Kidney Tissues from the Control Rats Exhibited a Normal Appearance with Characteristic Renal Histoarchitectures.
Discussion
DN is a severe problem associated with diabetes, characterized by a gradual decline in kidney function, and can result in kidney failure. The growing prevalence of diabetes worldwide, coupled with lifestyle changes, has led to a rise in the incidence of DN, posing a significant public health and financial burden. Comprehensive care, with a focus on early detection and treatment, is essential to mitigate the devastating effects of DN (Bilen et al., 2023). The pathogenesis of DN is multifaceted, involving renal damage due to prolonged exposure to high blood glucose levels (Kumar et al., 2023). The analysis of body weight and blood glucose is crucial in the context of DN. Elevated blood glucose levels over time can result in numerous comorbidities, including chronic kidney disease. Diabetic patients with renal impairment often experience weight fluctuations and difficulties in maintaining optimal glycemic control, which can exacerbate the progression of kidney disease (Li et al., 2024). Understanding the connection between these factors and the onset of DN is essential for the implementation of targeted interventions and the improvement of patient outcomes. Early detection of renal disease in diabetic patients is an opportunity to prevent the onset of DN and its associated comorbidities. Optimal glycemic control remains the mainstay of therapy for DN, as it has been shown to delay the onset and slow the advancement of the disease (Choi et al., 2023). In this study, the rats with DN demonstrated a marked reduction in body weight and subsequent elevation in glucose levels. However, the yangonin treatment considerably reduced the glucose level and increased the body weight in DN-induced rats.
The pathophysiology of DN is complex, involving an intricate interplay of metabolic and hemodynamic factors. One of the key biochemical markers used to assess and monitor kidney function in DN is the assessment of renal dysfunction markers (Silva et al., 2021). Creatinine, a byproduct produced from the degradation of creatine phosphate in muscle, and its clearance is a widely used measure of glomerular filtration rate. In DN, glomerular hyperfiltration and subsequent glomerular injury can lead to a progressive decline in creatinine clearance, indicating a worsening of kidney function (Han et al., 2023). Urea, on the other hand, is the primary nitrogenous waste product of protein metabolism. In DN, the generation of urea can occur due to decreased renal clearance, reflecting the progressive deterioration of kidney function. Uric acid is another important biomarker in DN. Hyperuricemia, a condition characterized by elevated serum uric acid concentrations, is commonly observed in DN patients and is believed to participate in the onset of the disease (Ephraim et al., 2021). BUN is a measurement of the amount of nitrogen urea in the blood. In DN, an increase in BUN levels can indicate a reduction in the kidney’s ability to filter and excrete urea, a hallmark of declining renal function. The assessment of these biomarkers is crucial for early diagnosis, monitoring, and management of DN (Yin et al., 2024). In this work, the results showed that the rats with DN demonstrated a considerable increase in serum concentrations of creatinine, uric acid, urea, and BUN in comparison to the control. However, the yangonin treatment exhibited a notable reduction in the concentrations of these renal dysfunction markers in the rats with DN.
The need for novel biomarkers that can improve risk assessment and enable early detection of DN has emerged in recent times. One potential avenue for exploration is the analysis of AST and ALT in individuals with DN. These enzymes are released into the bloodstream because of cellular injury, and their elevated levels may reflect the underlying inflammatory, fibrotic, and oxidative stress processes that participate in the onset of diabetic kidney disease. Plasma tubular markers, such as these transaminases, have been said to be related to a reduction in kidney function and the onset of diabetic kidney disease (Luo et al., 2024). The advantages of using AST and ALT as biomarkers for DN are that they can potentially detect renal tubular injury before the onset of microalbuminuria, which is a traditional marker of glomerular damage. Moreover, these biomarkers have been shown to be useful indicators of disease development and long-term prognosis (Li et al., 2024). The analysis of AST and ALT may provide valuable insights into the pathological processes underlying DN and could serve as promising biomarkers for early detection and treatment (Xu et al., 2021). This study revealed that rats with DN exhibited a significant elevation in serum activities of both AST and ALT enzymes relative to the control. Nonetheless, the yangonin treatment demonstrated a significant diminution in the activities of these marker enzymes in rats with DN.
It is well known that oxidative stress is integral to the pathophysiology of DN. The analysis of oxidative stress markers in the kidney tissues of individuals with DN can offer useful insights into the underlying mechanisms and potential therapeutic methods (Goycheva et al., 2023; Mansoor et al., 2022). GSH is a critical anti-oxidant that helps protect cells from oxidative damage. In DN, decreased GSH has been noted, indicating an imbalance in the redox system and increased susceptibility to oxidative stress (Singh et al., 2024). CAT and SOD are two key enzymes that play a vital role in neutralizing reactive oxygen species (ROS) and the maintenance of cellular homeostasis. It has been shown that the activities of these enzymes are often impaired in the kidneys of individuals with DN, resulting in the generation of harmful oxidants and subsequent injury to kidney tissues (Sun et al., 2023). MDA, a key biomarker of lipid peroxidation, is commonly elevated in renal tissues of individuals with DN, reflecting the extent of oxidative damage to cellular membranes and lipids (Zhang et al., 2024). Understanding the role of oxidative stress in DN can inform the development of targeted therapies aimed at diminishing oxidative damage and enhancing kidney function in individuals with diabetes (Bohlouli et al., 2021). The current findings exhibited that the DN rats demonstrated reduced anti-oxidants (CAT, SOD, and GSH) and subsequent increase in MDA levels in their kidney tissues when compared with control. Nevertheless, the rats with DN demonstrated a remarkable elevation in their anti-oxidant concentrations and reduction in MDA levels in their kidney tissues in response to the yangonin treatment, which supports its anti-oxidant properties.
Increasing evidence suggests that inflammation plays an essential role in the onset of DN. Specifically, the proinflammatory cytokines IL-6 and TNF-α play an essential role in the pathogenesis of this condition. The causal influence of these cytokines depends on body composition, with excess fatty tissue contributing to chronic inflammation in both hypertensive and diabetic elderly individuals (Donate-Correa et al., 2021; Zhao et al., 2020). Inflammation is defined by an elevation in macrophages in various tissues, accompanied by increased cytokine production. This inflammatory response is believed to be a driver of insulin resistance (Liu et al., 2022). Beyond insulin resistance, the inflammatory process appears to play a central role in the onset of diabetic complications, including nephropathy. It has been shown that high TNF-α in adipose tissue is associated with insulin resistance and that neutralization of this cytokine can restore glucose uptake by peripheral tissues. Similarly, the level of TNF-α correlates with insulin resistance and decreases with weight loss (Hu et al., 2024). In the context of DN, inflammation is a major contributor to the development of the disease. Elevated levels of IL-6 and TNF-α have been observed in the renal tissues of individuals with DN. These cytokines can disrupt insulin signaling pathways, leading to impaired kidney function and the development of proteinuria, a hallmark of DN (Zheng et al., 2021). A deep understanding of the underlying mechanisms by which these inflammatory mediators contribute to the onset of this debilitating condition may inform the development of targeted therapeutic interventions to prevent or slow the progression of DN (Song et al., 2024). The present studies indicated that rats with DN demonstrated significant elevations in the IL-6 and TNF-α concentrations relative to the control. Nevertheless, the yangonin successfully reduced the IL-6 and TNF-α in the DN rats, which highlights its anti-inflammatory activity.
Despite the promising findings in this study, several limitations should be considered. First, the current study is conducted only in an animal model, which may not fully reflect the complexity of DN in humans. Second, the sample size of rats used may limit the generalizability of the results. Additionally, the duration of the treatment was relatively short, and the long-term effects of yangonin on DN remain unclear. Furthermore, while the study demonstrates the potential of yangonin in mitigating oxidative stress and inflammation, the precise molecular mechanisms underlying its effects were not explored in depth. Future studies should focus on investigating the specific molecular pathways involved in yangonin’s action, particularly through in-depth molecular studies and human clinical trials.
Conclusion
The present findings indicate that yangonin may effectively mitigate DN in rats. The treatment of yangonin led to a significant reduction in glucose and an elevation in body weight of the DN rats. Furthermore, the yangonin treatment also demonstrated a decrease in renal dysfunction markers, inflammatory response, and oxidative stress via boosting the anti-oxidant levels in rats with DN. In conclusion, these findings suggest that yangonin possesses the potential to serve as an advantageous treatment option for DN. Despite the promising findings in this study, several limitations should be considered. The current study is conducted only in animal models, which may not fully reflect the complexity of DN in humans. The sample size and the duration of the treatment were relatively short, and the long-term effects of yangonin on DN remain unclear. Furthermore, while the study demonstrates the potential of yangonin in mitigating oxidative stress and inflammation, the precise molecular mechanisms underlying its effects were not explored in depth. Future studies should focus on investigating the specific molecular pathways involved in yangonin’s action, particularly through in-depth molecular studies and human clinical trials.
Footnotes
Abbreviations
ALT: Alanine transaminase; ANOVA: Analysis of variance; AST: Aspartate transaminase; BSA: Bovine serum albumin; BUN: Blood urea nitrogen; CAT: Catalase; DMRT: Duncan’s multiple range test; DN: Diabetic nephropathy; ELISA: Enzyme-linked immunosorbent assay; ESRD: End-stage renal disease; GSH: Glutathione; IL-6: Interleukin-6; MAPK: Mitogen-activated protein kinase; MDA: Malondialdehyde; NC: Normal control; RIPA: Radioimmunoprecipitation assay; ROS: Reactive oxygen species; SD: Standard deviation; SOD: Superoxide dismutase; STAT3: Signal transduction and activator of transcription 3; STZ: Streptozotocin; TCID50: 50% tissue culture infective dose; TBST: Tris-buffered saline with Tween; TNF-α: Tumor necrosis factor-alpha; NF-κB: Nuclear factor kappa B; IκB: Inhibitory κB; p-P65: Phosphorylated P65.
Author’s Note
Zhenzhen Hao and Lin Li contributed equally to the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethics Committee of Hebei University Affiliated Hospital China (Hbuah20220815).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project is supported by the mechanism of AGE activating EGFR-mediated fibrosis in diabetes nephropathy by regulating the balance of tyrosine phosphatase SHP1/SHP2 (Grant No. 2441ZF089).
