Abstract
Background
Squamous cell carcinoma is a skin carcinoma and can be fatal if not cured on time. Overexposure to ultraviolet (UV) light is the most common causative agent responsible for such a condition. Genetic and immunologic factors may contribute to its underlying pathology. Current therapeutic approaches towards this carcinoma seem effective, but chances of remittance and therapy-associated side effects have also been observed in many cases. Natural products offer a safe and alternative line of treatment, affording minimal side effects. Ethnomedicinally, the aerial parts of Persicaria capitata are known to be utilized in treating illnesses connected to the urinary system and skin ailments. The current study focuses on in vitro anti-cancer activity of P. capitata involving A-431 cell lines.
Objectives
Using the epidermoid skin cancer cell line (A-431), the current study attempts to ascertain the in vitro anti-cancer efficacy of ethanolic extract and its purified fraction derived from P. capitata flowers.
Materials and Methods
We studied the pharmacognostic and phytochemical profile of P. capitata and evaluated its properties, including its antioxidant and cytotoxic activity in A-431 cell lines.
Results
Pharmacognostic standardization confirms the identity of the plant, and phytochemical data revealed the presence of flavonoids as possible constituents in the purified fraction. Antioxidant data of the extract showed an absorbance of 0.415 ± 0.017, which confirms its potential in scavenging free radicals. The cytotoxic potential of purified fraction has been determined using A-431 lines via MTT assay in relevance to its ethnomedicinal usage in skin disorders. It showed promising results with an IC50 value of 475.22 µg/mL.
Conclusion
The purified fraction showed considerable cytotoxicity against A-431, as demonstrated by the MTT assay and fluorescence microscopy.
Introduction
Squamous cells are epithelial cells that form on the skin’s surface and the lining of organs, digestive tracts, and respiratory tracts. In addition to their protective roles for the skin, these cells are involved in selective permeability, diffusion, cytokeratin, keratin production, and the gaseous exchange between blood and air in the lungs (Alam & Ratner, 2001). Uncontrolled and abnormal growth of these cells resulted in the formation of one of the most devastating types of skin cancer, which affects individuals regardless of their race, age, or sex (Linares et al., 2015). Overexposure to ultraviolet (UV) radiation (UVA, UVB) is the most common outcome of squamous cell carcinoma (Schwartz, 1988). Usually, this cancer affects the hands, arms, neck, ears, face, or other parts of the body and can become fatal if not detected or cared timely. Surgical procedures along with photodynamic therapy are among the contemporary treatment approaches to curb this metastatic carcinoma (Combalia & Carrera, 2020), but the chances of reoccurrence (Wang et al., 2013) and therapy-associated side effects have also been observed (Corchado-Cobos et al., 2020; Lefèvre et al., 2023). Nature blessed humans by providing numerous medicinal and aromatic plants containing metabolites to prevent or alleviate various diseases and disorders (Abdallah et al., 2023). They seem to be the distinguished sources of complex phytoconstituents capable of exerting a wide array of therapeutic applications (Jamshidi-Kia et al., 2018). Their usage in treating and preventing diseases has been exploited by humans from the ancient era to the contemporary world (Petrovska, 2012). Genus Persicaria encompasses several medicinally important species having substantial pharmacological activities as evident from the literature. A few common therapeutically active species from the genus Persicaria include Persicaria capitata, Persicaria amphibia, Persicaria acuminata, Persicaria chinensis, Persicaria glabra, Persicaria hydropiper, Persicaria hydropiperoides, Persicaria lapathifolia, and so on (Seimandi et al., 2021). P. capitata Buch.-Ham. ex D. Don (H. Gross) (syn. Polygonum capitatum), Polygonaceae, commonly known as the pink knotweed, is an important species from this genus having pharmacological and industrial applications. The scientific classification of P. capitata has been mentioned in Table 1 (EPPO global database).
Taxonomic Classification of Persicaria capitata as per EPPO Global Database.
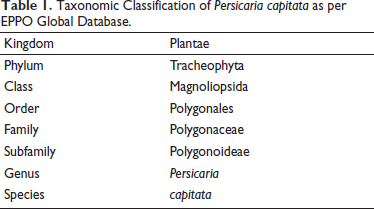
The plant is around 10–15 cm tall and native to the Himalayas in Northern India. Leaves are alternately oval, base, and wedge-shaped, sometimes with “V”-shaped markings on the leaf surface with stolon, rooting at its nodes, annual branches ascending upward, and a red surface (Hsu et al., 2004). Ethnobotanically, the plant is used by people in China for dysentery, skin ulcers, and inflammation (Gu & Xiao, 2015), whereas in India majorly aerial parts are extensively used in folk practices, particularly for skin diseases (Radha et al., 2021; Saha et al., 2011; Singh et al., 2017). The plant also contains diverse phytoconstituents, namely, flavonoids, lignanoids, phenols (Yang et al., 2022), and various extracts obtained from aerial parts/whole plant are reported to be pharmacologically active (Lin et al., 2022). In a study involving P. capitata, 80% methanolic extract obtained from the leaf and stems showed strong antioxidant activities in 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) and hydroxyl radicals, chelating activity against Fe2+, and xanthine oxidase inhibition assay (Huang et al., 2008). Several flavonoids (quercitrin, protocatechuic acid, quercetin, and kaempferol) isolated from this plant possessed strong scavenging capacity when evaluated against hydrogen peroxide assay (Liu et al., 2008). In an anti-cancer study, davidiin, isolated from this plant, was tested against four hepatocellular carcinomas (HCC) cell lines, and the compound significantly inhibited HCC cell growth (Wang et al., 2014). Quercetin, isolated from P. capitatum was tested against human gastric cancer cells GES-1 and proved to protect against gastric inflammation and apoptosis associated with Helicobacter pylori infection (Zhang et al., 2017). Although the whole plant has been harnessed for its therapeutic potential significantly, however, fewer studies have been reported on its flowers. So, this underlined research on flower heads of P. capitata will add more insights into the phytochemical profile of this species and its cytotoxic potential against skin cancer cell lines.
Materials and Methods
Reagent
A-431 cell lines were procured from NCCS, Pune, India. Major chemicals like a minimum essential medium (MEM), non-essential amino acid (NEAA), fetal bovine serum (FBS), Dulbecco’s phosphate buffered saline (DPBS), trypsin-EDTA solution, and MTT reagent were purchased from MP Biomedicals, Germany.
Plant Material
Flower heads of P. capitata were collected from the Khanyara region of District Kangra (32.2259°N, 76.3666°E), Himachal Pradesh, India, and its adjoining areas. Plant inhabited on rocky substrates was selected for the study and subjected to authentication from NIScPR, New Delhi (authentication number NIScPR/RHMD/Consult/2023/4429-30-1).
Pharmacognostic Studies
To ensure the quality and purity of selected plant material apart from macroscopy and microscopic characteristics, various pharmacognostic parameters were also taken into consideration, namely, ash value, moisture content determination, extractive value, swelling index, foaming index, and preliminary phytochemical screening as per the prescribed methodology of Acharyya et al. (2018) & Sohi and Shri (2018).
Fractionation and Liquid Chromatography–Mass Spectrometry (LC–MS)
The collected flower heads were subjected to continuous hot extraction (solution) using ethanol and extract was obtained. The extract was further fractionated via column chromatography using gradient elution consisting of chloroform:methanol: water (8:0.5:0.05 to 8:1:0.1), and purified fractions were collected. The purified fraction (PF3–PF5) was subjected to LC–MS for phytochemical analysis. The separation of the constituent was achieved on the C18 column via gradient elution consisting of 0.1% formic acid in water and acetonitrile delivered at a 1.2 mL/min flow rate, and eluent entered to MS through an ESI source.
Pharmacology
Total Antioxidant Activity and Reducing Power Assay
Using the phosphomolybdenum method, the total antioxidant capacity of the extract was assessed following the protocol outlined by Ponnamma et al. (2017), and a reducing power assay was conducted as per the methodology of Subramanian et al. (2013).
MTT Assay and Fluorescence Microscopy
In vitro, the anti-cancer activity of purified fraction (PF3–PF5) of P. capitata was evaluated on skin cancer cell lines (A-431) as per protocol illustrated in Figure 1. MTT assay was performed on the PFE (ethanolic extract of P. capitata flowers) as well as on the PF3–PF5 (Gaikwad & Shinde, 2022; Gerlier & Thomasset, 1986). Acridine orange and propidium iodide fluorescent labeling was also applied to the purified fraction to distinguish between live and dead cells (Bank, 1988; Liu et al., 2015).
Protocol of MTT and Fluorescence Microscopy.

Data Analysis
Three replicates were used for each experiment, and the results were displayed as Mean ± SD. Using GraphPad Prism 7’s t-test, non-linear regression analysis/one-way analysis of variance (ANOVA), the anti-cancer activity of the flower was ascertained using statistical analysis; a statistically significant difference was denoted by p < .05.
Results
Morphology
This perennial prostrate trailing herb’s leaves (elliptic ovate to elliptic) are about 1–3 cm long and 0.7–2 cm wide with distinct V-shaped markings. Stem is branched, having short internodes. Numerous fine hairs are present throughout the leaves and stems. Adventitious roots often arise from proximal nodes. Pink flowerheads (6–10 mm in diameter), usually arranged in capitate clusters, have short pedicels and also serve as potential sources of food for pollinators.
Microscopy
Anisocytic stomata and a wavy epidermis are among the leaf’s histological features, as shown in Figure 2B(a–c). Non-glandular multicellular (covering) trichomes were widely dispersed throughout the leaf in addition to fibers, as marked in Figure 2B(d–g). Druse crystals (star-like) were also found to be present in leaves when observed under high magnifications (SEM) as shown in Figure 2B(h–i). The transverse section (TS) of leaves showed the presence of a polygonal layer of epidermal cells, a parenchymatous layer of cells, collenchyma, and druse crystals, as indicated in Figure 2B(j–m). A distinct layer of cortex, endodermis, vascular bundles, pith, and multicellular covering trichomes was visible in the stem anatomy, Figure 2C(a–c). Root showed the presence of cork cells, endodermis, phloem, and medullary rays as evident from Figure 2D(a–c). The fine powder of the flower was examined under a light microscope, and as Figure 2E(a–f) illustrates, trichomes, crystals, pollen grains, and fibers were prominent. The region of the seed’s outer layer where fragmented test was visible as indicated in Figure 2E(g). In Figure 2F(b,c), vesicles were apparent, showing a favorable association between plant roots and arbuscular mycorrhizal fungi (AMF).
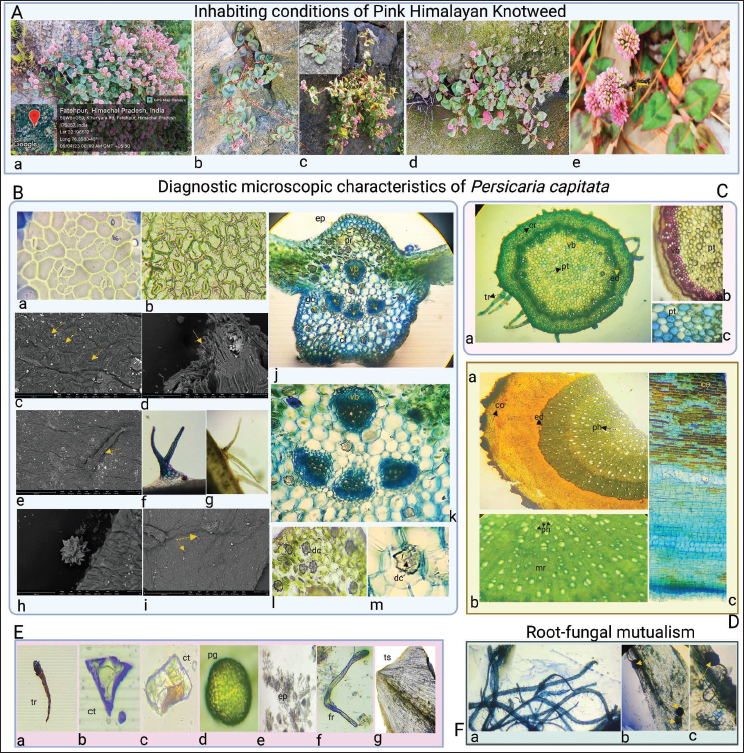
Standardization Parameters
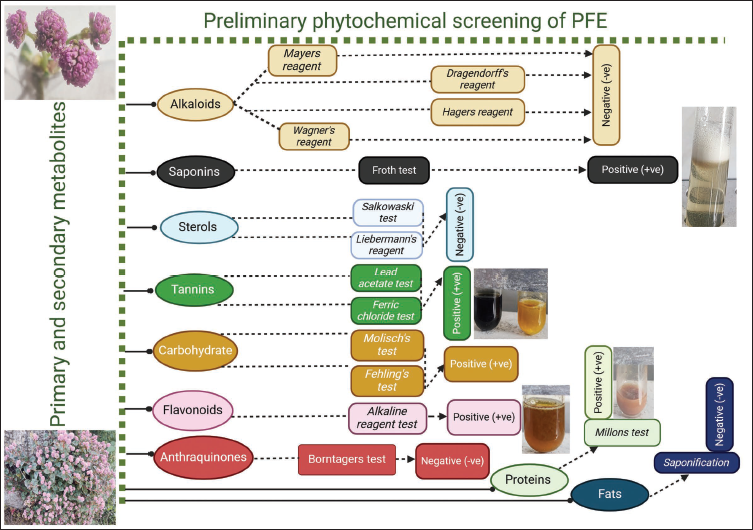
For the determination of inorganic impurities in the sample, the ash value was calculated and found to be 1.5%w/w. The values of moisture content, swelling index, and foaming index were found to be 8%v/v, 0.0 mL, and >100, respectively. The water and alcohol-soluble extractive values were measured as 13%w/w and 10%w/w, indicative of more polar constituents in the sample. Preliminary phytochemical screening was conducted on the ethanolic extract of P. capitata, which revealed the presence of primary (carbohydrates, proteins) and secondary metabolites (saponins, tannins, flavonoids) as illustrated in Figure 3.
Preliminary Phytochemical Screening of Primary and Secondary Metabolites.
LC–MS Analysis
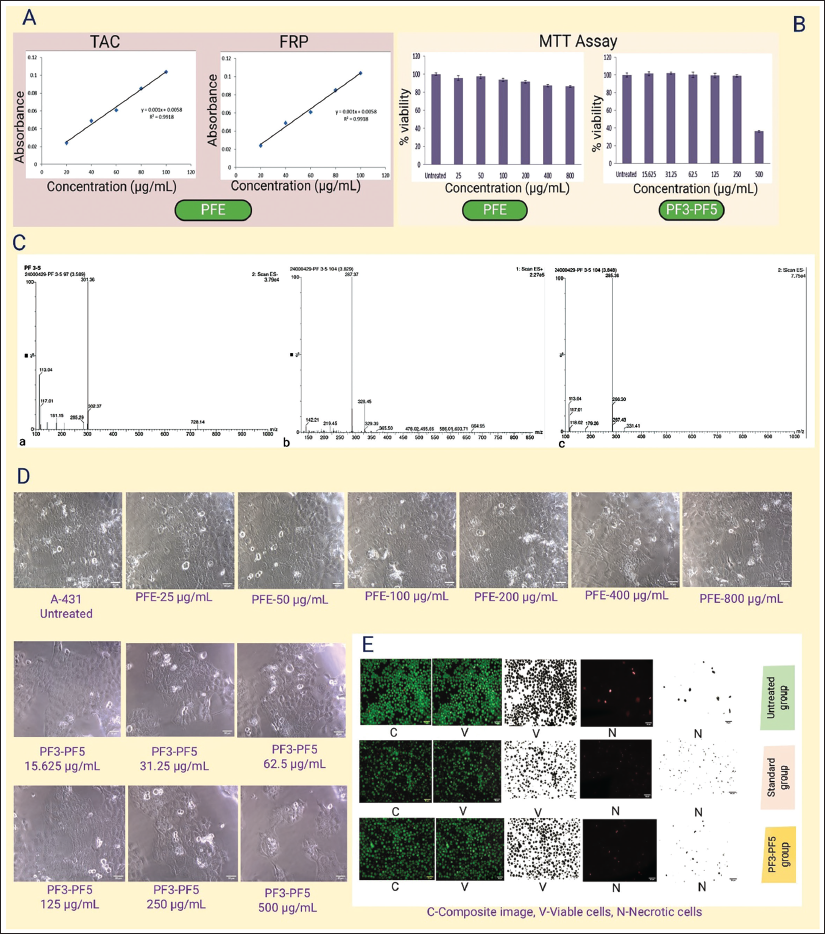
LC–MS profiling confirms that the purified fraction encompasses two major compounds eluted at Rt. 3.58 and 3.83, and detected in ESI positive and negative ion modes. Compound 1, eluted at Rt. 3.58, molecular weight 302.3 Da was confirmed by full scan spectra that show m/z 301.3 [M − H]+ adduct formed in ESI negative ion mode (Figure 4C(a)) and utilized for molecular weight confirmation. According to a literature survey, compound 1 was identified as quercetin and was previously reported from P. capitata (syn. P. capitatum) aerial parts (Liao et al., 2011; Liu et al., 2008; Zhang et al., 2017). Quercetin has been reported to induce melanoma suppression (Peng et al., 2020) and has shown a promising role as an anti-proliferative agent (Vafadar et al., 2020), particularly in melanoma cell lines (Soll et al., 2020). Moreover, compound 2, eluted at Rt. 3.83, molecular weight 286.3 Da was confirmed by full scan spectra that showed m/z 287.3 [M + H]+, m/z 328.4 [M + ACN + H]+, and m/z 285.3 [M − H]− adducts formed in ESI positive and negative ion modes (Figure 4C(b,c)), respectively, and utilized it for molecular weight determination. According to a literature survey, compound 2 was identified as luteolin which was previously reported from this species (Yang et al., 2022). Luteolin has been reported to inhibit human melanoma cell lines (Juszczak et al., 2022).
Pharmacology
Total Antioxidant Capacity and Ferrous Reducing Power Assay
The phosphomolybdenum test was used to evaluate the total antioxidant capacity (TAC) of ethanolic extract of P. capitata. The extract showed absorbance of 0.104 ± 0.012 at 100 µg/mL when compared with standard (0.472 ± 0.007 ascorbic acid) at the same concentration. For the ferrous reducing power (FRP) assay, the crude extract of the flower showed a maximum absorbance of 0.415 ± 0.017 compared with ascorbic acid (0.467 ± 0.010), which proved its prominent reducing power capacity. The results of TAC and FRP have been marked in Figure 4A.
MTT Assay and Fluorescence Microscopy
Ethanolic extract of P. capitata (PFE) was tested against A-431 cell lines at various concentrations range at 25, 50, 100, 200, 400, and 800 µg/mL, but IC50 value was not achieved even at the highest dose of 800 µg/mL. Purified fraction (PF3–PF5) was evaluated for its cytotoxic potential following the same procedure at concentrations ranging from 15.625 to 500 µg/mL using MTT assay. PF3–PF5 inhibited the proliferation of skin cancer cell lines (A-431) at IC50 at 475.22 µg/mL. The percentage viability of PFE and PF3–PF5 have been represented in Figure 4B and D, fluorescence microscopy (acridine orange/propidium iodide staining) of PF3–PF5 confirms the necrosis by measuring cell viability (live/dead assay) when compared with untreated, standard (docetaxel) groups as illustrated in Figure 4E. Cells were counted from images taken with a 20× objective.
Discussion
Natural products offer a safer alternative adjuvant therapy to prevent several carcinomas and serve as an inspiration for the drug discovery process (Butler, 2004). Genus Persicaria encompasses various therapeutically active species having cytotoxic properties. Several anti-cancer species including P. maculosa (Vasas et al., 2020), P. orientalis (Ansari et al., 2017), and P. salicifolia (Mohamed Mohamed Youssef & El-Swaify, 2018), were therapeutically active. Advancements in traditional or Indigenous knowledge helped in finding targets for specific diseases (Lone et al., 2015). P. capitata (Buch.-Ham. ex D. Don) H. Gross (syn. P. capitatum Buch.-Ham. ex D. Don) belonging to smartweed/buckwheat family (Polygonaceae) is a perennial creeping herb that grows 10–15 cm tall and is endemic to Northern India and the Himalayas (Hsu et al., 2004). The plant can adapt to multiple life forms, including terrestrial and lithophytic ones, and has also been employed as a ground cover in garden landscaping and as a substrate in green roofing (Dhaliwal & Sharma, 1999; Pêgo et al., 2011). Ethnomedicinal records about P. capitata revealed that the aerial parts of this species have been used to treat several conditions related to skin (Gu & Xiao, 2015; Singh et al., 2017). The aerial parts of the plant have been used externally as well as internally in ancient practices. In the Traditional Chinese System of Medicine (TCM) the plant has been used by Miao people to alleviate symptoms associated with the urinary tract (Han et al., 2018). The plant is a reservoir of complex secondary metabolites capable of exhibiting pharmacological activities, including cytotoxic, antioxidant, anti-microbial, and anti-inflammatory (Lin et al., 2022). Pink-headed Persicaria is a source of aliphatic hydrocarbons/aliphatic alcohols (tricosane, octacosa-1,27-diene, pentacosanol), aliphatic acids (acetic acid, succinic acid, linoleic acid), glycerides (hexadecanoic-2,3-dihydroxypropylester), phenols, phenolic acids and their esters, phenolic glycosides, alcoholic glycosides, alkyl glycosides, volatile oils (trans-β-farnesene, 1-hexanol, 3-octanol, hyacinthin) (Yang et al., 2022). In a recent study, several compounds (stigmast-5-en-3-O-β-
Conclusion
The present study aimed to construct the standardization profile of Pink Himalayan Knotweed and investigate its potential as a cytotoxic agent in A-431 cell lines. Microscopic studies of various plant parts confirm the diagnostic characteristics of P. capitata. Phytochemical analysis via LC–MS confirmed the presence of secondary metabolites in flower. In vitro, screening in A-431 cell lines revealed that purified fraction isolated from the plant exhibited promising cytotoxic activity and was found to attenuate proliferation in selected cell lines.
Footnotes
Abbreviations
ABTS: 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); AMF: Arbuscular mycorrhizal fungi; AO/PI: Acridine orange and propidium iodide; DPBS: Dulbecco’s phosphate buffered saline; FBS: Fetal bovine serum; FRP: Ferrous reducing power; HCC: Hepatocellular carcinomas; LC–MS: Liquid chromatography–mass spectrometry; MEM: Minimum essential medium; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; NEAA: Non-essential amino acid; PFE: Ethanolic extract of P. capitata flowers; PF3–PF5: Purified fraction of P. capitata; SEM: Scanning electron microscopy; TAC: Total antioxidant capacity.
Acknowledgments
The authors of this research work are grateful to Professor Vivek Sharma, Government College of Pharmacy Rohru; Professor Vinay Thakur, Government Pharmacy College Kangra at Nagrota Bagwan, Himachal Pradesh, India, and the Department of Pharmaceutical Sciences, CT University Ludhiana, Punjab, India, are responsible for providing technical support. The authors also acknowledge the LC–MS and SEM facilities provided by Poornayu Research Labs and Dextrose Laboratories in Bangalore, India.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
