Abstract
Background
Gestational diabetes, defined by increased blood glucose levels during pregnancy, impacts a considerable proportion of pregnant women globally. The proper management of gestational diabetes is crucial, as it is connected with short- and long-term health consequences for both the mother and fetus.
Objectives
This research work was devoted to addressing the beneficial roles of pimpinellin against streptozotocin (STZ)-induced gestational diabetic complications in the experimental rat model.
Materials and Methods
Pregnant rats were treated with STZ to induce gestational diabetes, after which the gestational diabetes rats received pimpinellin treatment for 14 days. Upon completion of treatments, measurements were taken for body weight, blood glucose, and the survival and body weight of the pups. The levels of lipid profile markers, anti-oxidant concentrations, and proinflammatory cytokine levels were assessed utilizing commercial diagnostic kits. A histological examination was performed on the pancreas collected from the treated rats.
Results
The findings of this work demonstrated that pimpinellin at 50 mg/kg concentration effectively lessened the glucose level and body weight of the gestational diabetes rats. The pimpinellin treatment also increased the survival of the pups. The treatment with pimpinellin successfully decreased inflammatory cytokines, regulated lipid profile biomarkers, and mitigated oxidative stress by increasing anti-oxidants in rats with gestational diabetes. The histopathological examination of the pancreas also validated the salutary properties of pimpinellin.
Conclusion
The results of this work highlight that pimpinellin positively influences the reduction of gestational diabetes conditions and its associated complications in rats. Consequently, it possesses the potential to serve as an effective treatment alternative for managing gestational diabetes.
Introduction
Gestational diabetes is a form of diabetes that arises during pregnancy, marked by elevated blood glucose levels that can adversely affect the health of both the mother and the developing fetus. This condition is becoming increasingly prevalent worldwide, with empirical studies indicating a rise in its incidence parallel to the growing prevalence of overweight and obesity among pregnant women (Gitlin et al., 2024). Gestational diabetes has been connected with numerous adverse pregnancy outcomes, such as a high risk of congenital anomalies, stillbirth, and the infant being large for gestational age. The complications of gestational diabetes surpass the duration of pregnancy, as affected women have an elevated risk of subsequently developing type 2 diabetes (Deitch et al., 2023). The correlation between maternal body mass index and weight gain during pregnancy with the prevalence of large-for-gestational-age newborns in women with gestational diabetes has been a subject of continuous research. Maternal prepregnancy body mass index is recognized as an independent predictor of newborn birth weight; nevertheless, several research indicates that maternal adiposity may serve as a more robust predictor of macrosomia than hyperglycemia alone (Lyu et al., 2024). Additionally, there is evidence to suggest that children born to mothers with gestational diabetes are at an elevated risk of progressing into childhood obesity and glucose intolerance in early adulthood, potentially perpetuating a transgenerational cycle of diabetes (Eades et al., 2024). Gestational diabetes is linked to several detrimental short- and long-term health effects for both the mother and the child. Gestational diabetes women have a high risk of progressing gestational hypertension, preeclampsia, and type 2 diabetes. Children born to women with gestational diabetes are more prone to macrosomia, have a heightened risk of congenital anomalies, and face an elevated likelihood of juvenile obesity and glucose intolerance (Makasheva et al., 2024).
The pathophysiology of gestational diabetes is complex, involving a combination of insulin resistance and dysregulated insulin production, which can lead to adverse maternal and fetal outcomes if left untreated. Existing treatment options, such as lifestyle modifications, insulin, and oral hypoglycemic agents, can effectively control blood glucose levels, but they are not without their drawbacks. For instance, insulin therapy can increase the risk of hypoglycemia, while oral hypoglycemic agents may have contraindications or be less effective in certain cases (Nakshine & Jogdand, 2023). The adverse effects include an elevated risk of hypoglycemia, weight gain, and injection site reactions. Additionally, the use of oral hypoglycemic agents during pregnancy was connected with a high risk of neonatal complications, including hypoglycemia and respiratory distress syndrome. Consequently, the exploration of plant-based therapies has gained increasing attention as a potential alternative approach to address the challenges associated with gestational diabetes (Rosu et al., 2024).
Many plant-based bioactive compounds have demonstrated promising anti-diabetic properties in both in vitro and in vivo studies. These compounds have been shown to elevate insulin sensitivity, increase glucose uptake, and modulate key metabolic pathways involved in the pathogenesis of diabetes. In addition to their potential therapeutic effects, plant-based therapies may offer a more natural and potentially safer alternative to conventional treatments, with a lower risk of adverse side effects (Jaworsky et al., 2023). Pimpinellin is a primary coumarin-like bioactive compound that is widely present in the roots of Toddalia asiatica, Angelica dahurica, and Zosima absinthifolia. It has been well reported that pimpinellin has anti-allergic (Li & Wu, 2017) and hepatoprotective (Oh et al., 2002) properties. Recent studies have highlighted that pimpinellin has mitigated ulcerative colitis (Lv et al., 2025), ameliorated macrophage inflammation (Yang et al., 2024), and demonstrated anti-convulsant (Łuszczki et al., 2023) and anti-seizure properties. However, the therapeutic effects of pimpinellin against gestational diabetic complications have not been studied yet. This research work was devoted to addressing the beneficial roles of pimpinellin against streptozotocin (STZ)-induced gestational diabetic complications in an experimental rat model.
Materials and Methods
Chemicals
All chemicals and reagents utilized in this work were procured from Sigma–Aldrich, USA. To estimate the biochemical parameters, commercial diagnostic kits were purchased from Abcam, USA and Elabscience, USA, respectively.
Animals
The 8-week-aged Wistar rats of both genders, weighing around 210 ± 30 g, were procured from an institutional animal facility and utilized in this study. Animals were then housed in infection-free confines and maintained underregulated laboratory circumstances, which included a temperature of 23°C–25°C, humidity of 40%–70%, and a 12-h light/dark cycle. Animals were fed a commercial diet, which comprised total protein: 14 g, total fat: 4 g, fiber: 5 g, total carbohydrate: 42 g, and caloric value: 3.85 kcal/g (Bio-Serv, USA). The methodologies used in the experimental animals were verified and approved by the Institutional Animal Ethics Committee.
Establishment of Gestational Diabetes
To establish gestational diabetes, the rats were continually administered with a high-fat diet (HFD) for 8 consequent weeks, and body weight increase was continuously observed. Thereafter, the rats were fasted overnight, succeeded by daily vaginal swabs to ascertain the estrous stage. Subsequently, the estrous stage rats were placed with healthy male rats (2 females: 1 male) for mating. Following a 24-h interval, the presence of sperm was assessed to verify the pregnancy on day 0. Thereafter, pregnant rats were distinguished and segregated from other rats. The non-pregnant rats were omitted from further experiments. The pregnant animals received an administration of STZ (40 mg/kg) to induce gestational diabetes, whereas the control animals were given an equivalent volume of buffered saline devoid of STZ (Yang et al., 2022).
Tereatment Groups
Control rats were administered only saline devoid of STZ and/or pimpinellin (Group I). The treatment rats with gestational diabetes were categorized into two groups as follows: the STZ-induced rats with gestational diabetes (Group II) and gestational diabetes rats treated with 50 mg/kg of pimpinellin (Group III) (Liu et al., 2021). The pimpinellin treatment was administered orally for 14 days. Upon completion of treatment, rats were fasted overnight, sacrificed under anesthesia, and samples were obtained for subsequent studies. The live pups and their body weight, survival index, and anogenital distance (AGD) of the pups were evaluated.
Fasting Blood Glucose (FBG) and Body Weight Analysis
The FBG and body blood glucose levels of the experimental rats were evaluated on the final gestation day. FBG levels were estimated by using an Accu-Chek glucometer. The body weight was precisely determined using a sophisticated electronic weighing balance.
Lipid Marker Analysis
The serum lipid biomarkers, including low-density lipoprotein (LDL), high-density lipoprotein (HDL), triglycerides, and total cholesterol, in the experimental rats were assessed using commercial diagnostic kits in accordance with the manufacturer’s instructions (Elabscience, USA).
Analysis of Biochemical Marker Levels
The tissue homogenate of the pancreas was prepared by homogenizing the tissues with buffer and centrifuging at 10,000 rpm for 15 min. The concentrations of malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), glutathione S-transferase (GST), and glutathione peroxidase (GPx) in the supernatants of the pancreas tissue homogenate were analyzed using commercial diagnostic kits (Elabscience, USA). The amounts of inflammatory cytokines, including interleukin (IL)-6 and tumor necrosis factor (TNF)-α, in the serum were analyzed using kits. The manufacturer’s instructions (Abcam, USA) were adhered to for these assays.
Histopathological Analysis
Subsequent to surgical extraction from the experimental rats, the pancreas was rinsed with saline, dehydrated through a sequence of ethanol, and subsequently embedded in paraffin. The pancreatic tissues were then subsequently sectioned into 5 µm slices. The pancreas was stained with hematoxylin and eosin and analyzed with a microscope for histological changes.
Statistical Analysis
All the statistical assays were executed using GraphPad software (version 9). The values are depicted as the mean ± standard deviation (SD) (n = 6) of three replicates. The data are scrutinized utilizing one-way analysis of variance (ANOVA) and Tukey’s post hoc test, with p < .05 as significant.
Results
Effect of Pimpinellin on FBG and Body Weight of Experimental Rats
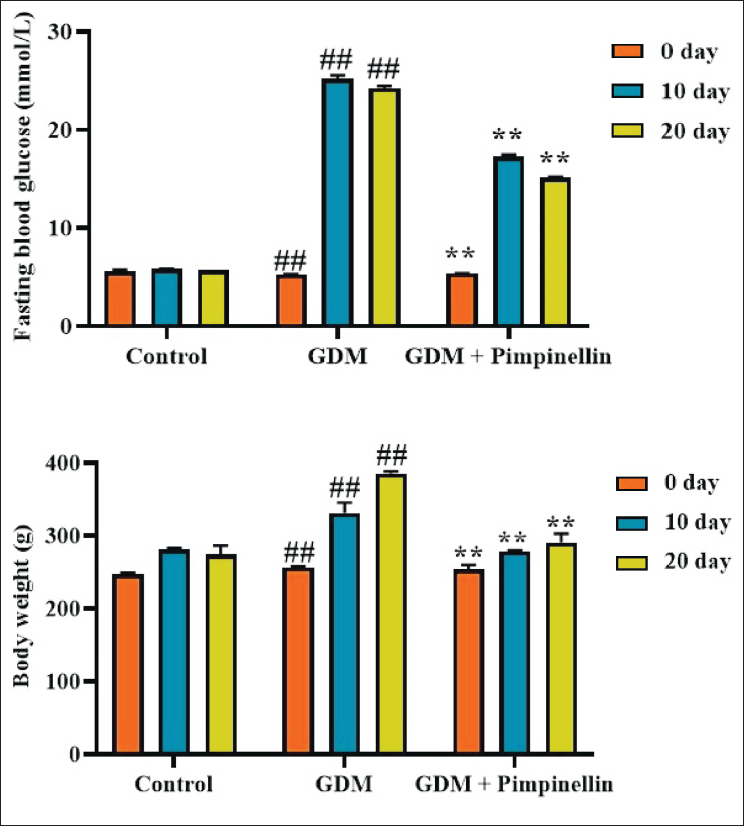
Figure 1 illustrates the impact of pimpinellin on the FBG and body weight of the gestational diabetes-induced rats. In comparison to the control group, the gestational diabetes rats exhibited increased FBG and body weight levels. Nonetheless, the treatment with pimpinellin at 50 mg/kg concentration resulted in a significant diminution in both FBG and body weight in the gestational diabetes rats.
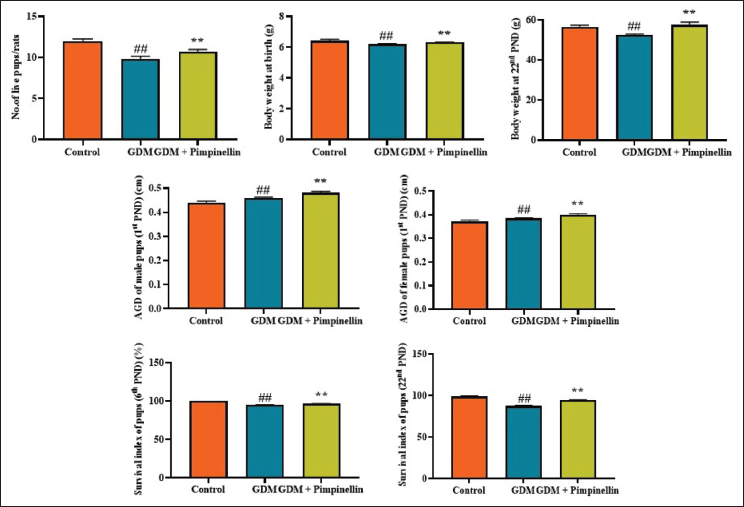
Effect of Pimpinellin on the Number, Body Weight, Survival, and AGD Level of Pups
The effects of pimpinellin on the pup’s number, body weight, and AGD of pups are presented in Figure 2. The gestational diabetes rats exhibited a reduced number of live pups in comparison to the control group. A reduction in body weight and a minor decrease in the AGD of male and female pups were noted in the gestational diabetes rats. Nonetheless, pimpinellin treatment at a 50 mg/kg concentration markedly enhanced the live pup’s number and the survival index. Furthermore, pimpinellin also resulted in elevated body weight and AGD of both female and male pups.
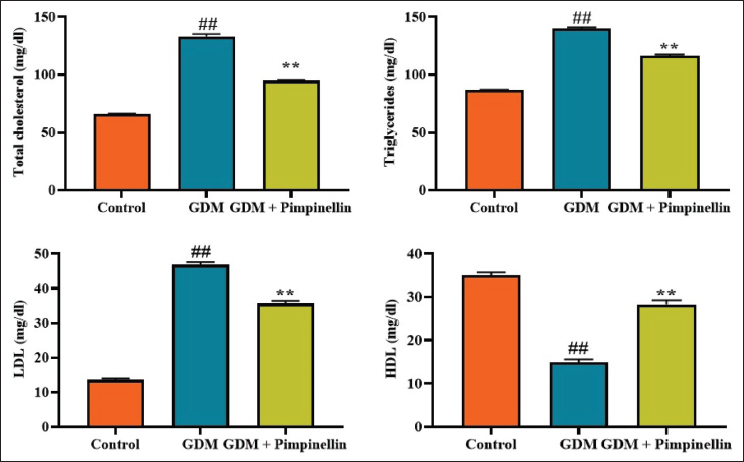
Effect of Pimpinellin on Lipid Biomarkers in Experimental Rats
Figure 3 exhibits the concentrations of lipid markers in both control and treatment rats. The rats with gestational diabetes had increased LDL, triglycerides, and total cholesterol while displaying a decrease in HDL levels compared to the control group. In contrast, pimpinellin at a 50 mg/kg concentration significantly decreased LDL, total cholesterol, and triglycerides contents in the gestational diabetes-induced rats. Furthermore, pimpinellin also elevated HDL concentration in rats with gestational diabetes, demonstrating its anti-hyperlipidemic efficacy.
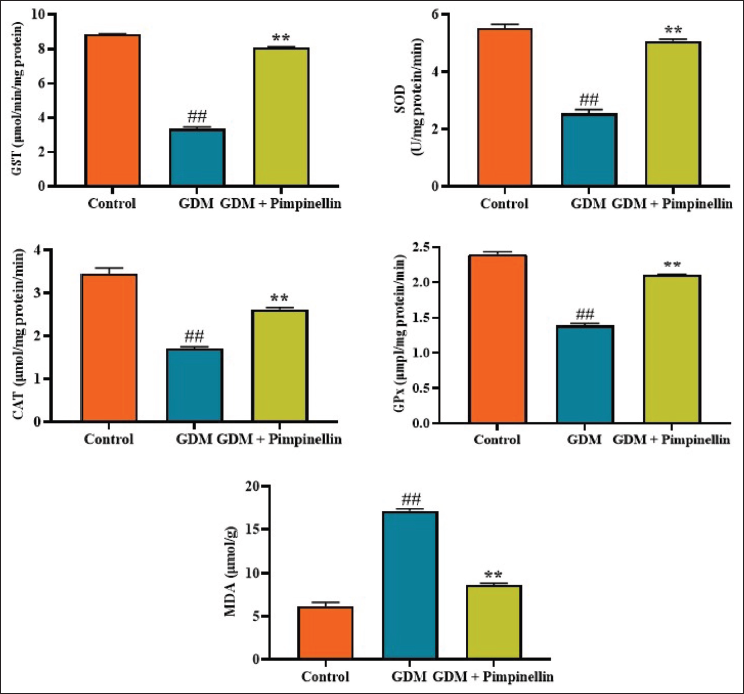
Effect of Pimpinellin on Oxidative Stress in Experimental Rats
The oxidative stress marker levels, comprising MDA, SOD, GPx, CAT, and GST, were quantified in experimental animals, with the findings illustrated in Figure 4. The gestational diabetes-induced rats exhibited a significant reduction in their SOD, GPx, CAT, and GST concentrations when compared with control. Nonetheless, the pimpinellin treatment at a dosage of 50 mg/kg significantly increased their anti-oxidant contents in the gestational diabetes rats. These findings illustrate the anti-oxidant efficacy of pimpinellin.
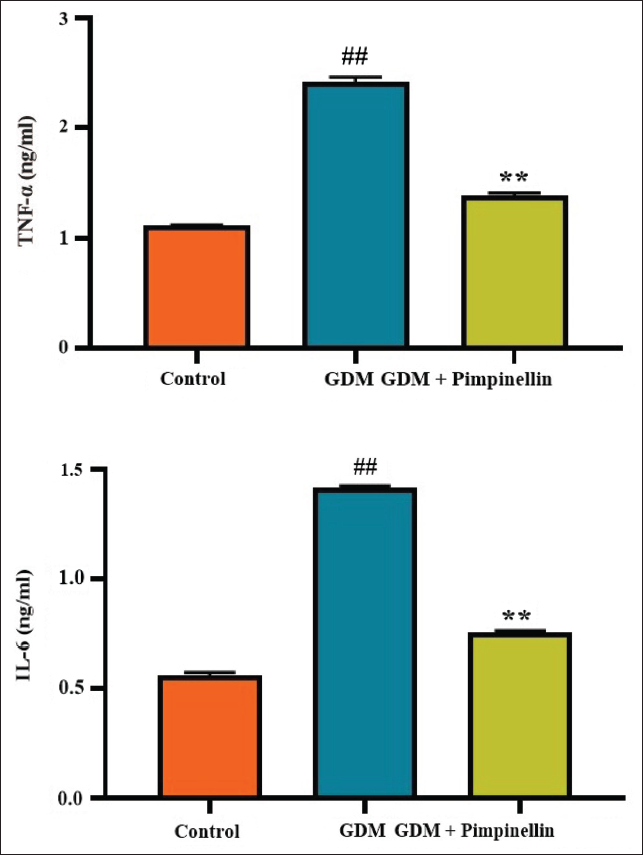
Effect of Pimpinellin on Inflammatory Cytokines in Experimental Rats
Cytokine levels were assessed in the serum of rats, with results depicted in Figure 5. The rats with gestational diabetes exhibited a substantial elevation in both IL-6 and TNF-α concentration when compared with the control. In contrast, the pimpinellin treatment significantly diminished the IL-6 and TNF-α content in the gestational diabetes rats, which proves its anti-inflammatory properties.
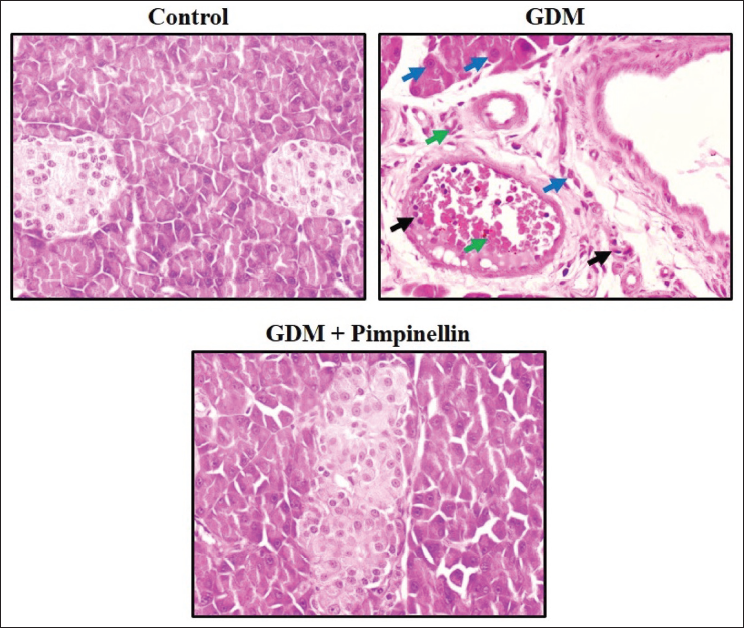
Effect of Pimpinellin on Histopathology of Pancreas Tissue of Experimental Rats
The outcomes of a histopathological evaluation of the pancreatic tissues of rats are depicted in Figure 6. The pancreatic tissues derived from control rats displayed no indications of inflammation and demonstrated a typical cellular architecture. Captivatingly, the pancreas of the rats with gestational diabetes exhibited heightened inflammatory cell influx, atrophy of islet cells, and hypertrophy of adipose tissue. Interestingly, the histopathological alterations in the pancreas of gestational diabetes rats were significantly reduced by the pimpinellin treatment.
Discussion
Gestational diabetes poses a considerable health risk during pregnancy, with potentially severe implications for both the mother and the growing fetus. Gestational diabetes is defined by increased blood glucose levels identified for the first time during pregnancy (Le et al., 2023). This syndrome is linked to a heightened risk of problems, including macrosomia, preeclampsia, and an increased likelihood of developing type 2 diabetes in both the mother and the child later in life (Minschart et al., 2023). Maternal obesity is an established risk factor for gestational diabetes, as it enhances insulin resistance and diminishes glucose tolerance. Moreover, research has shown that FBG levels during pregnancy can be a strong predictor of adverse outcomes, even in women who do not meet the criteria for gestational diabetes (Rodrigo et al., 2023). The current results demonstrated that the gestational diabetes rats exhibited increased FBG and body weight levels. However, the pimpinellin treatment led to a substantial diminution in both FBG and body weight in the gestational diabetes rats.
The proper management of gestational diabetes is crucial, as it is connected to several health complications. One important aspect of managing gestational diabetes is the analysis of the mother’s lipid profile levels. Gestational diabetes is characterized by increased peripheral insulin resistance, which leads to elevated glucose and fatty acid transfer from the mother to the fetus (Dalfrà et al., 2024). This exaggerated metabolic response can have far-reaching consequences, as gestational diabetes has been linked to a high risk of developing cardiovascular disease. Moreover, the offspring of women with prenatal diabetes have an increased likelihood of developing juvenile obesity and glucose intolerance in early adulthood, establishing a transgenerational cycle of diabetes perpetuation (Xu et al., 2024). The analysis of the maternal lipid profile is crucial in understanding the metabolic changes associated with gestational diabetes and their potential long-term implications. Preconceptional obesity, a known risk factor for gestational diabetes, has been shown to influence the trajectory of maternal lipid metabolism during pregnancy. Specifically, women with preconceptional overweight or obesity may exhibit more pronounced changes in lipid metabolism, potentially contributing to the high risk of maternal complications (Zhang et al., 2024). Furthermore, the presence of dyslipidemia, characterized by an imbalance in lipid levels in gestational diabetes women, is considered a surrogate marker for developing type 2 diabetes. Therefore, the careful monitoring and management of maternal dyslipidemia during and after gestational diabetes can provide valuable insights into the underlying metabolic derangements and help in the implementation of targeted interventions to abrogate the long-term cardiovascular and metabolic risks (Wu et al., 2023). The present results demonstrated that rats with gestational diabetes had increased LDL, triglycerides, and total cholesterol while decreasing HDL levels. Contrastingly, the pimpinellin treatment significantly decreased LDL, total cholesterol, and triglyceride levels, with a subsequent elevation in HDL concentration in gestational diabetes-induced rats. These findings demonstrated the anti-hyperlipidemic properties of the pimpinellin.
One of the key pathophysiological mechanisms underlying gestational diabetes is the role of oxidative stress and its related markers. Oxidative stress, characterized by an imbalance between ROS production and the body’s antioxidant defenses, has been thoroughly investigated in relation to gestational diabetes. Increased oxidative stress can lead to cellular damage, inflammation, and impaired insulin signaling, all of which participate in the onset of gestational diabetes (Zheng et al., 2024). MDA, a marker of lipid peroxidation, is elevated in women with gestational diabetes, indicating increased oxidative damage to cellular lipids. Similarly, reports have highlighted that antioxidants, like SOD, CAT, GPx, and GST, are often dysregulated in gestational diabetes, suggesting an impaired cellular defense against oxidative insults (Wang et al., 2024). The relationship between oxidative stress and gestational diabetes is complex and multifaceted. Hyperglycemia, a hallmark of gestational diabetes, can further exacerbate oxidative stress through the generation of advanced glycation end products (AGEPs) and the activation of various signaling pathways. Conversely, increased oxidative stress can disrupt insulin sensitivity and participate in the onset of insulin resistance, a key feature of gestational diabetes (Fernández-Valero et al., 2023). Comprehending the complex interplay between oxidative stress and gestational diabetes is crucial for developing targeted interventions and improving maternal and fetal outcomes (Diniz et al., 2023). The findings of this work proved that the rats with gestational diabetes exhibited a significant reduction in their antioxidants and an elevation in MDA levels. Interestingly, the pimpinellin treatment significantly increased their antioxidant levels in the gestational diabetes rats, which evidences the antioxidant efficacy of pimpinellin.
The pathophysiological mechanisms underlying the onset of gestational diabetes are still not well studied, but increasing evidence suggests that inflammation and inflammatory cytokines play a significant role (Jiménez-Osorio et al., 2023). Inflammation, as indicated by elevated concentrations of inflammatory markers like TNF-α and IL-6, has been consistently related to the onset of type 2 diabetes, and this association appears to extend to gestational diabetes as well. Inflammatory cytokines were shown to implicate in insulin resistance, a hallmark of gestational diabetes (Xuan Nguyen et al., 2023). The increased levels of inflammatory markers observed in women with gestational diabetes may be indicative of an underlying activated innate immune response, which has been proposed as a common antecedent for both type 2 diabetes and associated conditions, such as dyslipidemia and atherosclerosis. This chronic inflammatory state may also participate in the increased risk of adverse health outcomes in gestational diabetes women (Visiedo et al., 2023). Moreover, the long-term complications of gestational diabetes on the offspring, like the high risk of childhood obesity and glucose intolerance, may also be influenced by the inflammatory environment in the uterus. Understanding the role of inflammation in the pathophysiology of gestational diabetes is crucial for the development of targeted interventions to prevent and manage this condition (Torres-Torres et al., 2024). The current results demonstrated a drastic elevation in IL-6 and TNF-α concentrations in the rats with gestational diabetes. Captivatingly, the pimpinellin treatment significantly diminished the IL-6 and TNF-α concentrations in the gestational diabetes rats, which evidences its anti-inflammatory effects.
Conclusion
The present study reveals that pimpinellin exerts a positive impact on reducing the severity of gestational diabetes and its associated complications in rats. Notably, treatment with pimpinellin significantly lowered FBG levels and body weight in gestational diabetes-induced rats, indicating its potential therapeutic benefits. Moreover, pimpinellin treatment was found to mitigate oxidative stress and inflammation by enhancing antioxidant defenses and reducing inflammatory cytokine levels in these rats. These findings suggest that pimpinellin may serve as a promising treatment alternative for managing gestational diabetes. However, to fully elucidate the therapeutic effects of pimpinellin on gestational diabetes, further studies are warranted to provide a more comprehensive understanding of its mechanisms of action and potential clinical applications.
Footnotes
Abbreviations
AGD: Anogenital distance; CAT: Catalase; FBG: Fasting blood glucose; GPx: Glutathione peroxidase; GST: Glutathione S-transferase; HDL: High-density lipoprotein; HFD: High-fat diet; IL-6: Interleukin 6; LDL: Low-density lipoprotein; MDA: Malondialdehyde; SOD: Superoxide dismutase; STZ: Streptozotocin; TNF-α: Tumor necrosis factor alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Ethical Committee of The Affiliated Hospital of Hebei University (Approval No. 2024081501).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
