Abstract
Background
Schisandra chinensis is a traditional Chinese medicine and is widely consumed as a health food due to its immune-enhancing properties. However, limited research has been done on its nutrient composition.
Objectives
In this study, liquid chromatography-tandem triple quadrupole mass spectrometry (LC-QTRAP-MS/MS) was developed for quantitative analysis of nucleosides, nucleobases (NNs), and amino acids (AAs) in 16 batches of S. chinensis samples collected from Liaoning, Jilin, and Heilongjiang provinces.
Materials and Methods
A Waters X-Bridge Amide column (2.1 × 100 mm, 3.5 µm) was used for analysis. The mobile phase consisted of A (water with 0.2% formic acid) and B (acetonitrile with 0.2% formic acid) with a flow rate of 0.6 mL/min. The column temperature was maintained at 30°C. The mass spectrometry was equipped with an electrospray ionization source and operated in positive ion multiple reaction monitoring (MRM) modes.
Results
The method demonstrated acceptable linearity within the tested concentration range. The average recovery of the 30 components ranged from 96.55% to 103.85%, with relative standard deviations (RSD) ranging from 1.02% to 2.88%. The quantitative data indicated that differences in the contents of NNs and AAs in S. chinensis from different regions were evident. The analysis of clustering heat maps revealed that the 16 batches of S. chinensis samples could be grouped into five categories. Stacked bar chart analysis showed that the levels of arginine and uridine were higher in most regions. Correlation analysis indicated strong correlations among the 14 AAs, with 42 groups showing significant relationships. Additionally, seven robust correlations were observed among the 16 NNs.
Conclusion
This established method provides essential data for the quality assessment and utilization of S. chinensis resources.
Introduction
Schisandra chinensis (Turcz.) Baill., a kind of traditional Chinese medicine, is suitable for the treatment of symptoms caused by deficiency of both the heart and kidney energies, such as agrypnia, dreaminess, and dizziness (National Pharmacopoeia Committee, 2020; Song et al., 2021; Su et al., 2022; Yan et al., 2016). Schisandra is also utilized in the production of health foods, including Schisandra drinks (Juan et al., 2023), Schisandra herb tea, and Schisandra wine. Scientific studies have shown that S. chinensis has many activities such as enhancing immunity (Cheng et al., 2024; Wang et al., 2023; Zhao et al., 2021), protecting the liver (Feng et al., 2021; Wu & Wang, 2020; Zhao et al., 2022; Zhu, Li, et al., 2023), anti-tumor (Huang et al., 2023; Sun et al., 2024; Wang et al., 2024; Yang et al., 2024), and regulating blood sugar levels (Liu et al., 2024; Zhao et al., 2024; Zhu, Zhao, et al., 2023).
Previous studies on active ingredients of S. chinensis primarily concentrated on lignans and terpenes, such as Schisandrol A (Wu et al., 2022) and Wuweizidilactone A (Zhang et al., 2023). However, amino acids (AAs) and nucleosides have been infrequently reported. As nutrient constituents, nucleosides, and AAs play an important role in promoting growth, maintaining normal metabolism, and ensuring life activities. AAs serve as the primary constituents of proteins and possess significant nutritional and medicinal properties (Lian et al., 2019). For example, serine is recognized for its effective hypoglycemic activity, while arginine has demonstrated notable immunomodulatory effects (Albaugh et al., 2017; Holm et al., 2019). Nucleosides are fundamental components of biological cells, crucial for sustaining life processes, and act as bio-active agents that enhance immune responses and exhibit anti-viral capabilities (Ludek et al., 2018). These compounds are significant in herbal medicines and have beneficial effects. They are often utilized as quality control ingredients in products like donkey-hide gelatin, Ganoderma lucidum, and Cordyceps. Understanding the composition and content of AAs and nucleosides in S. chinensis is important for evaluating its nutritional value and beneficial effect.
So far, various analytical methods have been developed to analyze AAs and nucleobases (NNs), such as high-performance liquid chromatography (HPLC) and capillary electrophoresis (CE). Ultra-performance liquid chromatography-tandem triple quadrupole mass spectrometry (UPLC-QTRAP-MS/MS) could avoid the miss of target ions caused by unresolved background interferences in complex matrices (Bisht et al., 2021; Luo et al., 2022). In this study, an LC-QTRAP-MS/MS method was developed for the simultaneous identification and quantification of 14 AAs and 16 NNs in S. chinensis from 16 different regions, which was beneficial for evaluating the quality and nutritional value of S. chinensis.
Materials and Methods
Materials and Reagents
Acetonitrile (HPLC grade) was obtained from Tedia Company, Inc. (Fairfield, USA). Deionized water was prepared by a Milli-Q water purification system (Millipore, USA). Fourteen kinds of AAs mixed standard (No. B2007072) were purchased from Tanmo Quality Inspection Technology Co., Ltd. (Changzhou, China), including glycine (1), serine (2), aspartic acid (3), valine (4), glutamic acid (5), isoleucine (6), methionine (7), arginine (8), histidine (9), threonine (10), leucine (11), cystine (12), phenylalanine (13), and lysine (14). The NNs were purchased from Shanghai Yuanye Bio-Technology Co., Ltd. (Shanghai, China), including guanine (15), adenine (16), uracil (17), hypoxanthine (18), thymidine (19), guanosine (20), inosine (21), uridine (22), adenosine (23), cytidine (24), xanthine (25), thymine (26), 2′-deoxyuridine (27), 2′-deoxyadenosine (28), 2′-deoxycytidine (29), and allopurinol (30). The purities of all the standard components were more than 98%.
Sample Preparation
Separately, 16 batches of samples were collected from Panjin in Liaoning (Z1), Benxi in Liaoning (Z2), Suihua in Heilongjiang (Z3), Chaoyang in Liaoning (Z4), Yanbian in Jilin (Z5), Anshan in Liaoning (Z6), Tonghua in Jilin (Z7), Dandong in Liaoning (Z8), Liaoyang in Liaoning (Z9), Hegang in Heilongjiang (Z10), Qiqihaer in Heilongjiang (Z11), Yichun in Heilongjiang (Z12), Jilin in Jilin (Z13), Baishan in Jilin (Z14), Mudanjiang in Heilongjiang (Z15), and Fushun in Liaoning (Z16). They were validated to be S. chinensis (Turcz.) Baill. by Professor Shengjin Liu from the Department of Pharmacy, Nanjing University of Chinese Medicine, Nanjing, China. They were freeze–dried by the vacuum freeze–drying equipment (FD-1-50, Boyikang Biotech, China) and stored in drying vessels. The voucher specimen (No. 20211026zy) was deposited in the Pharmaceutical College in a dark, dry environment.
The dried samples were ground up and sieved through a 40-mesh stainless-steel sieve before extraction. One gram of each sample powder was accurately weighed and redissolved in 20 mL of distilled water into a conical flask. All mixtures were placed into an ultrasonic bath (40 kHz, KeDao, China) for 60 min at room temperature. The same amount of solvent was added to the flask to compensate for the weight lost during extraction. After centrifugation at 12,000 rpm for 10 min, each supernatant was filtered through a 0.22 µm polytetrafluoroethylene filter membrane and stored in a glass vial at 4℃.
Standard Preparation
The standard stock solutions were diluted with water to appropriate concentrations and stored at 4℃. Calibration curves were established for all standards.
Chromatographic and Mass Conditions
The analysis was conducted using a Shimadzu Nexera LC-30AD UPLC system (Shimadzu, Kyoto, Japan) comprising an online degasser, a double solvent delivery pump, an auto-sampler, and a column temperature controller. A Waters X-Bridge Amide column (2.1 × 100 mm, 3.5 µm) was used for analysis, and the column temperature was maintained at 30℃. The mobile phase consisted of A (water with 0.2% formic acid) and B (acetonitrile with 0.2% formic acid) with a flow rate of 0.6 mL/min. The gradient elution was as follows: 0–2.5 min, 15% A; 2.5–5 min, 15%–50% A; 5–7 min, 50% A; 7–8 min, 50%–15% A, and 8–11 min, 15% A. The injection volume was 1 µL.
The mass spectrometry was equipped with an electrospray ionization source and operated in positive ion multiple reaction monitoring (MRM) modes. The parameters were optimized with an ion spray voltage of 4,500 V, source temperature of 150℃, desolvation temperature of 550℃, collision gas pressure of 9 psi, curtain-gas pressure of 40 psi, ion source gas-1 pressure of 50 psi, ion source gas-2 pressure of 50 psi, entrance potential (EP) of 10 V, and collision cell exit potential (CXP) of 10 V.
Multivariate Statistical Analysis
To find out the similarities and differences of the analytes in S. chinensis samples, various chemometric analysis methods, such as principal component analysis (PCA), were performed using the OECloud tools at
Results
A reliable LC–MS/MS method was developed after an assessment of the linearity, limits of detection (LOD), limits of quantitation (LOQ), precision, repeatability, stability, and recovery.
Method Validation
Calibration Curves, Liner Range, LOD, and LOQ
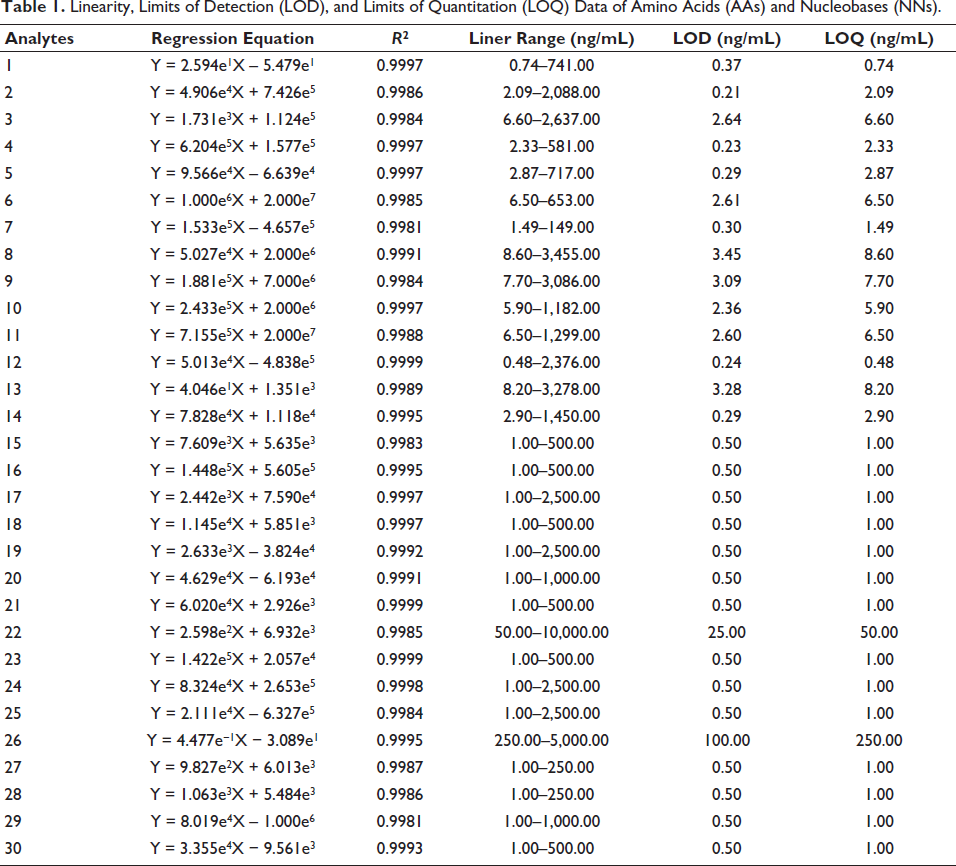
LC–MS was used to analyze different concentrations under specified chromatographic and mass conditions. The peak areas were plotted against the concentrations to create calibration curves and linear ranges. The LOD and LOQ for each analyte were determined at signal-to-noise ratios (S/N) of 3 and 10, respectively. As shown in Table 1, each standard exhibited good linearity across a wide concentration range (R2 > 0.998). The LOD and LOQ values ranged from 0.21 to 100.00 ng/mL and 0.48 to 250.00 ng/mL, respectively.
Linearity, Limits of Detection (LOD), and Limits of Quantitation (LOQ) Data of Amino Acids (AAs) and Nucleobases (NNs).
Precision, Repeatability, and Stability
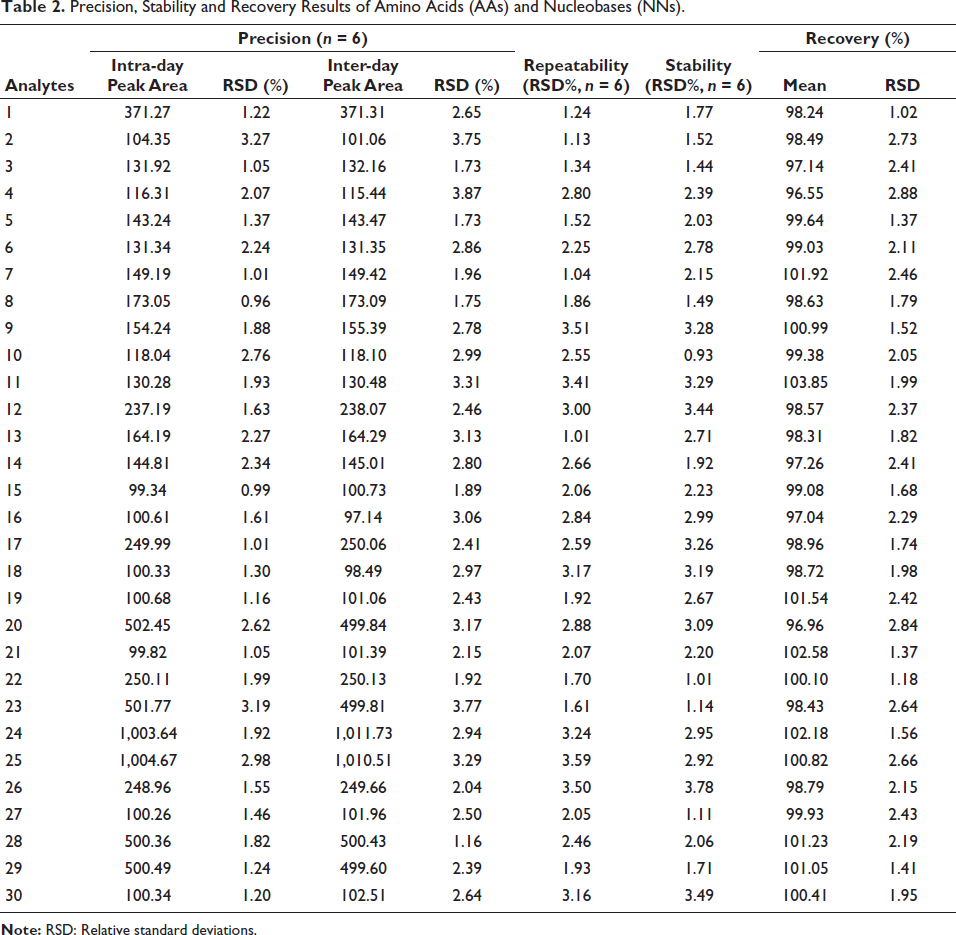
The precision of the assay was evaluated by determining the peak area of each component in the standard solution over one and three consecutive days. The experiments were repeated six times, and the peak area’s relative standard deviations (RSD) were calculated. In addition, the repeatability of the assay was assessed by analyzing sample Z1 in six replicates using the same method. The variations were presented as RSD. To test stability, the Z1 solution was injected into the LC–MS system at 0, 2, 4, 8, 12, and 24 h, respectively. The data in Table 2 showed that the RSD values for both intra-day and inter-day precision of the 30 analytes were less than 3.87%. Similarly, the RSD values for the repeatability and stability of these compounds were less than 3.59% and 3.78%, respectively. These results suggested that the established method provided good accuracy and precision.
Precision, Stability and Recovery Results of Amino Acids (AAs) and Nucleobases (NNs).
Recovery
A recovery assay was carried out to assess the method’s accuracy. Thirty known standard compositions were added to Z1, and the spiked samples were then extracted and analyzed as previously described. The test was replicated three times. The results, summarized in Table 2, showed that the recovery ranged from 96.55% to 103.85%, and the RSD ranged from 1.02% to 2.88%. These results further indicated that the established method was sufficiently accurate for determining the 30 compounds.
Sample Analysis
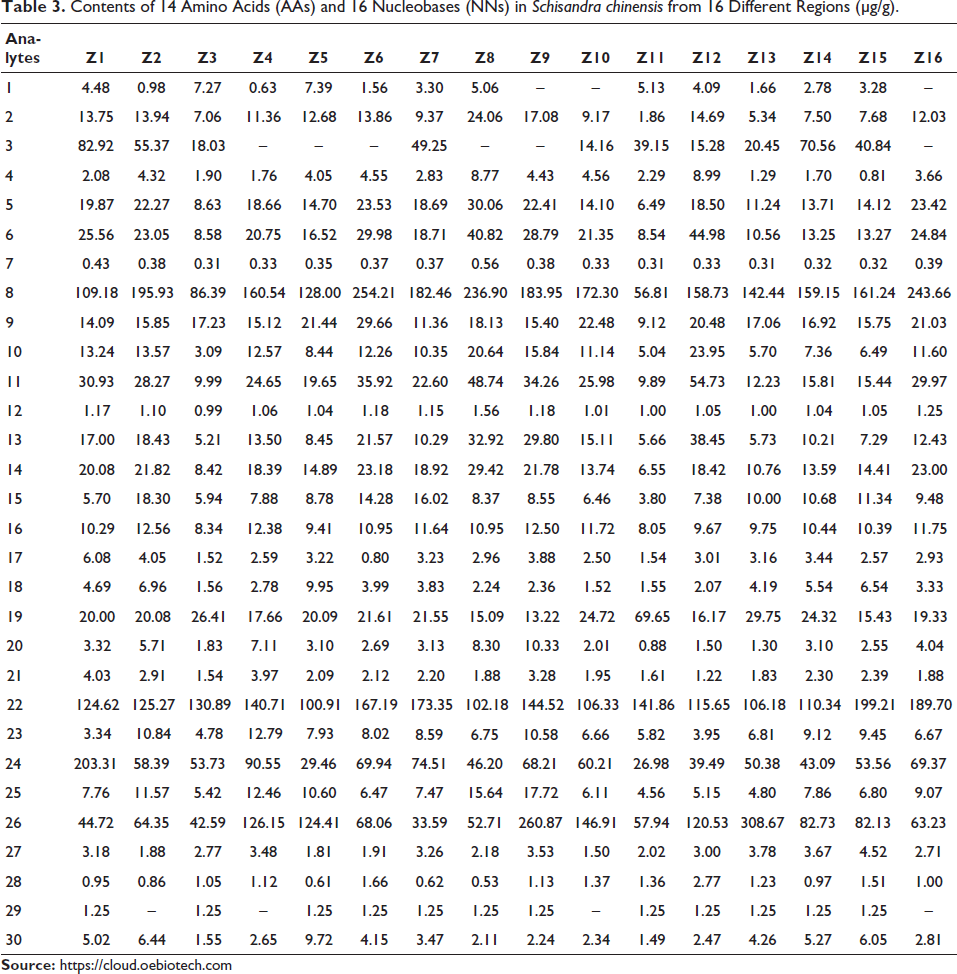
The developed LC–MS/MS method was applied to analyze the 30 compositions (consisting of 14 AAs and 16 NNs) in 16 samples collected from different regions in China. All the contents are displayed in Table 3. The results showed that the contents of AAs and NNs in S. chinensis from various regions were different. The total AAs were most abundant in S. chinensis from the Z8 region with a value of 506.01 µg/g. The S. chinensis from the Z9 region had the largest amount of total NNs, reaching as much as 555.60 µg/g.
Contents of 14 Amino Acids (AAs) and 16 Nucleobases (NNs) in Schisandra chinensis from 16 Different Regions (µg/g).
Cluster Heatmap Analysis
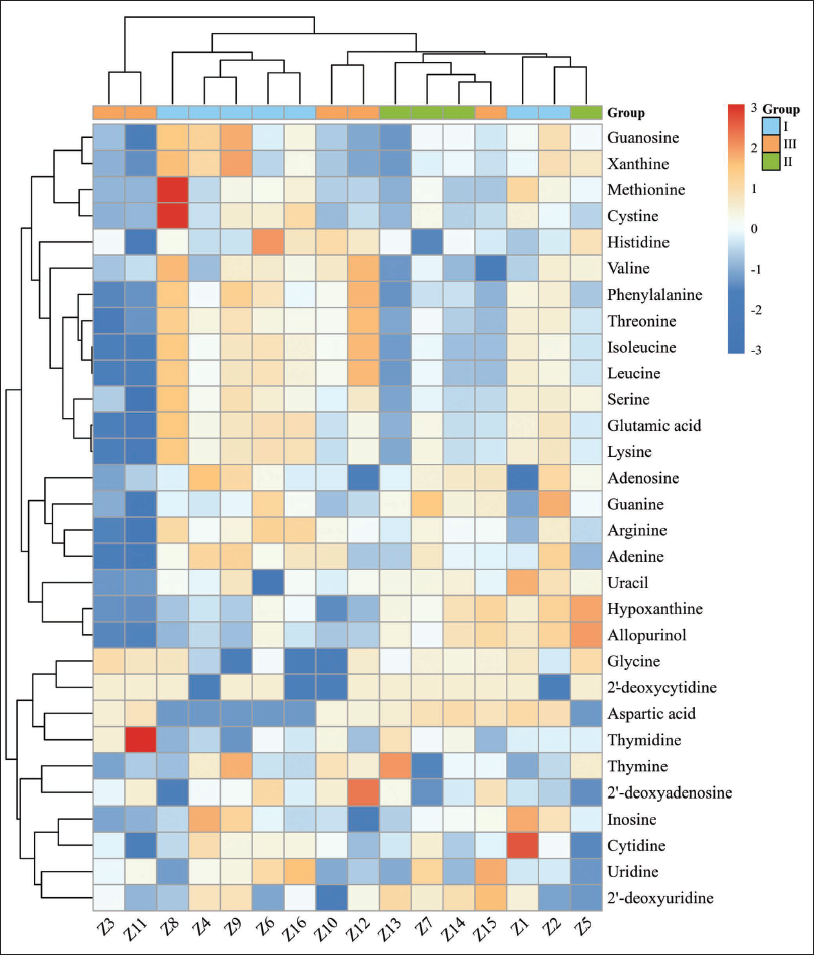
Clustering heat maps, to visually analyze experimental data distribution, are achieved by assigning different colors to different clusters in order to facilitate the interpretation and understanding of patterns. The analysis results indicated that the 16 batches of S. chinensis samples were classified into five categories (Figure 1). Samples Z1, Z2, and Z5 belong to the same class, which is characterized by relatively high levels of serine, uracil, hypoxanthine, and allopurinol content. Samples Z7, Z13, Z14, and Z15 fell into another category with relatively high levels of 2′-deoxycytidine, aspartic acid, and 2′-deoxyuridine content. Additionally, samples Z10 and Z12 belonged to a distinct category with relatively high levels of histidine and aspartic acid content. Samples Z8, Z4, Z9, Z6, and Z16 were grouped due to their relatively high contents of threonine, leucine, isoleucine, serine, glutamic acid, and lysine but low aspartic acid content. Lastly, samples Z3 and Z11 were in a category of their own due to their relatively low levels of major analytes, with the exceptions of glycine, 2′-deoxycytidine, aspartic acid, and uridine.
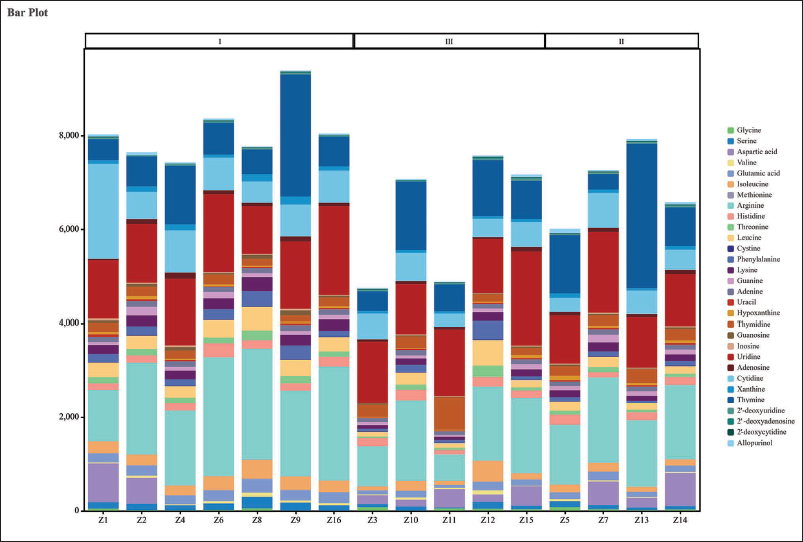
Stacked Bar Chart Analysis
The stacked bar chart can be used to visualize the distribution of various AAs and NNs in S. chinensis across different regions. According to the stacking diagram (Figure 2), Schisandra from the Liaoning region (Z1, Z2, Z4, Z6, Z8, Z9, and Z16) contained higher levels of AAs and NNs. The contents of arginine and uridine were high in S. chinensis from most regions. S. chinensis from the Z9 and Z13 regions had a higher content of thymine, which was consistent with the findings of the cluster heat map analysis. The disparities in the composition of AAs and nucleosides among these samples might be attributed to natural factors such as temperature and light conditions at the respective collection sites. Such variations pose challenges for subsequent studies aimed at evaluating the quality of S. chinensis based on a single index component. Therefore, a comprehensive analysis should consider either the total concentration of AAs or the proportion of each specific component. Samples Z3 and Z11, originating from Suihua and Qiqihaer, exhibited similarities in their AA and nucleoside compositions, which might be linked to the comparable latitudes and landforms of these two locations.
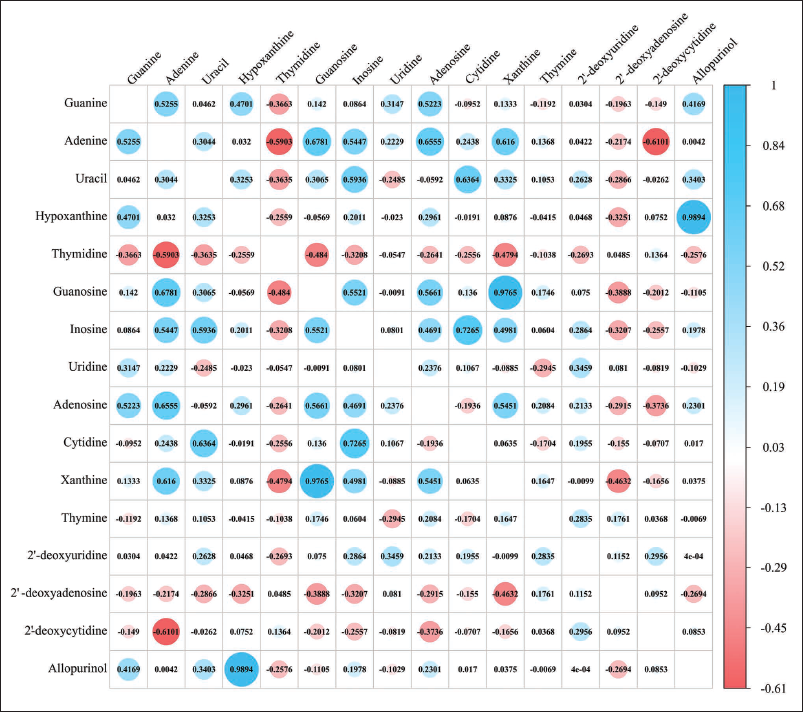
Correlation Analysis
To investigate the correlation in ingredient accumulation, a correlation analysis of AAs was conducted, in which darker shades of blue indicated stronger positive correlations, while darker shades of red indicated stronger negative correlations. The absolute values of the correlation coefficients (r) ranged from 0.8 to 1.0, indicating very strong correlations, and from 0.6 to 0.8, indicating strong correlations. As depicted in Figure 3, 42 groups of strong correlations were found among the 14 AAs studied. For instance, serine, glutamic acid, and lysine displayed highly significant positive associations with correlation coefficients exceeding 0.9. Similarly, among the 16 NNs depicted in Figure 4, a total of seven robust correlations were observed. Notably, a strong positive correlation (r > 0.95) was identified between hypoxanthine and allopurinol.

Discussion
According to the data in Table 3, arginine was the highest component among the 30 analytes in S. chinensis from Z6 (254.21 µg/g), which was much more than glycine (1.56 µg/g). Noteworthily, arginine plays a critical role in relieving angina pectoris (Egashira et al., 1996; Sha et al., 2024) and regulating the host immune response (Agidigbi et al., 2023; Li et al., 2023; Menezes-Garcia et al., 2020; Xie et al.,2021). Arginine is metabolized in the body to produce nitric oxide (NO), which promotes the relaxation of vascular smooth muscle and subsequently reduces vascular resistance. Additionally, arginine may inhibit the oxidation of low-density lipoprotein (LDL) and diminish the formation of lipid deposits within the inner layer of blood vessels. This dual action contributes to a lower workload and diminished heart-required effort during blood circulation. Moreover, the NO generated from arginine enhances the phagocytic activity of macrophages and regulates T lymphocyte proliferation and the antibody-mediated immune response. Thymine was the most abundant nucleoside in S. chinensis from the Z13 region. Besides, uridine was consistently present in high levels across all batches of S. chinensis samples and is considered a potential treatment option for anemia (Russo et al., 2020).
Conclusion
In this study, an LC-QTRAP-MS/MS method was developed to simultaneously measure 14 AAs and 16 NNs in S. chinensis from 16 different regions. This method ensured accurate results quickly by avoiding interference from complex matrices, taking only 9 min. The method’s precision was confirmed with intra-day and inter-day precision values of less than 3.27% and 3.87%, respectively. The recovery rate was between 96.55% and 103.85%, with an RSD of 1.02% to 2.88%.
The comprehensive evaluation included cluster heat map analysis, stacked bar chart analysis, and correlation analysis. The quantitative data revealed significant differences in the contents of AAs and NNs in S. chinensis from various regions, which posed challenges for future studies assessing S. chinensis quality using a single index component. Therefore, it might be more effective to assess quality by considering the total content or the ratio of each component. This established method provided essential data for the further quality assessment and utilization of S. chinensis resources.
Footnotes
Acknowledgments
The authors are particularly grateful to Professor Hong-Jiang Chen for his team’s constant support and encouragement.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Visiting Scholars Program of Jiangsu Province, China (2024GRFX090); the “Qinglan Project” of Jiangsu Province, China; and the 2023 Excellent Teaching Team in the Blue Project of Jiangsu Province Universities, China.
