Abstract
Background
Colon cancer is a significant public health issue, known as a major cause of cancer-associated mortalities globally. Understanding the underlying molecular mechanisms that direct the onset of colon cancer is necessary to develop novel therapeutic targets.
Objectives
This study was dedicated to studying the anti-cancer properties of the reserpine against 1,2-dimethylhydrazine (DMH)-treated colon cancer in rats.
Materials and Methods
In the present work, colon cancer was triggered in rats by the administration of DMH and thereafter treated with reserpine prior to and throughout the DMH administration. After completing the treatments, alterations in body weight were assessed. The levels of inflammatory cytokines, oxidative markers, xenobiotic-metabolizing enzymes, tumor markers, and apoptotic proteins in the rats were carefully analyzed using commercial kits. The colon mucosa of the rats was subjected to histopathological studies.
Results
The current results proved that the administration of reserpine significantly increased body weight in DMH-administered rats. The administration of reserpine substantially diminished oxidative stress, regulated inflammatory cytokine levels, and inhibited the AKT/mTOR axis in DMH-induced rats. Furthermore, reserpine also regulated the activities of xenobiotic-metabolizing enzymes and apoptotic protein levels and reduced the tumor markers in DMH-administered rats. The results of the histopathological analysis also witnessed the anti-cancer roles of reserpine.
Conclusion
The present work shows that reserpine exhibits significant anti-cancer effects against DMH-induced colon cancer in rats. These findings demonstrate that reserpine has the capacity as an anti-cancer agent to treat colon cancer.
Introduction
Colon cancer, a malignant growth originating in the large intestine, has become a significant public health issue worldwide, with its prevalence and death rates posing a substantial burden on healthcare systems globally. The prevalence of colon cancer is particularly alarming, as it ranks third in frequency among men and second among women, making it a prominent cause of cancer-associated deaths (Siegel et al., 2023). Despite developments in early detection and therapy, colon cancer remains a significant cause of mortality globally. Ongoing efforts to improve cancer screening, promote healthy lifestyle choices, and enhance access to quality healthcare are crucial in mitigating the global burden of this disease. The genetic and molecular mechanisms underlying colon cancer have been extensively studied, and researchers have identified several key factors that contribute to its development and progression. Understanding these complex pathways is essential to develop more effective prevention and treatment methods (Marcellinaro et al., 2023).
Despite developments in therapy, including chemotherapy, radiotherapy, and surgery, the survival rates for patients with colon cancer have not improved significantly in recent years. Understanding the primary causes and underlying pathophysiology of colon cancer is essential in developing effective prevention and early detection methods (Roshandel et al., 2024). The primary causes of colon cancer are multifactorial, involving both genetic and environmental factors. Genetic predisposition, particularly in the form of hereditary syndromes, can significantly elevate an individual’s risk of progressing colon cancer. Additionally, lifestyle factors, such as being overweight or obese, smoking, heavy alcohol use, and having diabetes, have been strongly connected with the risk of colon cancer (Adigun et al., 2023). Dietary and nutritional factors also play a critical role in the progression of colon cancer. Up to 80% of all colon cancer incidences and mortalities are referable to diet, indicating that many cases may be preventable through dietary modifications. Specifically, more consumption of red meat, low consumption of vegetables and fruits, and a lack of certain nutrients, like vitamin D, have been connected with a high risk of colon cancer (Alsheridah & Akhtar, 2018; Oliveira et al., 2024).
While conventional cancer therapies have had some success, they often come with several drawbacks, such as drug resistance, non-targeted delivery, and adverse effects. In this context, exploring plant-derived bioactive compounds has gained increasing attention as a potential avenue for more effective and safer cancer treatment (Aiello et al., 2019). Plant-based bioactive compounds, also known as phytochemicals, have been recognized for their diverse range of beneficial health effects, including anti-cancer, radiosensitizing, and chemosensitizing properties against several cancers. One of the primary advantages of plant-derived bioactive compounds is their relatively low toxicity to normal cells, making them an attractive alternative or complement to conventional cancer therapies (Esmeeta et al., 2022). Reserpine is a major alkaloid compound found extensively in the roots of the Rauwolfia serpentina plant. Previous studies have reported the pharmacological properties of the reserpine, such as blood pressure lowering effect (Siddiqui et al., 2020), nephroprotective (Xu et al., 2024), anti-retinal ciliopathy (Chen et al., 2023), and anti-tumor (Ramamoorthy et al., 2018) properties. Apart from these biological effects, the anti-cancer effects of reserpine against colon cancer have not been studied yet. Consequently, the current work was dedicated to studying the anti-cancer property of reserpine against 1,2-dimethylhydrazine (DMH)-induced colon cancer in rats.
Materials and Methods
Experimental Animals
The present study included 8-week-aged Wistar rats, each weighing over 210–230 g. The rats were maintained at a temperature of 20°C–23°C, with a humidity of 50%–60%, and 12-h light and dark series. Additionally, rats received a constant supply of food and drinking water for the whole length of the experiment. The protocols conducted on the experimental rats were approved by the institutional animal ethics committee.
Experimental Design and Treatment Protocols
The rats were allocated into four experimental groups (n = 6). The rats from group I were administered only with regular diet. Group II rats were administered with DMH (20 mg/kg) weekly for 10 subsequent weeks to initiate colon cancer, after which they were observed to finish 18 consecutive weeks. Groups III and IV rats received low and high doses of reserpine (25 and 50 mg/kg, respectively) for 4 weeks prior to and throughout DMH induction, followed by an observation period of 18 weeks. Upon concluding the therapies, the alterations in body weight were assessed, and the findings were documented.
Polyps Number Analysis
The colon specimen was removed after the sacrification of the animals. The excised tissue was subsequently rinsed with saline and longitudinally sectioned without disrupting the existing polyps. The polyp numbers were subsequently enumerated utilizing a macroscopic observer.
Analysis of Oxidative Stress Biomarkers
The concentration of oxidative stress marker TBARS in liver and colon specimens was evaluated using diagnostic kits. The superoxide dismutase (SOD) and catalase (CAT) activities were evaluated utilizing the kits. The tests were done in triplicates and performed according to the manufacturer’s (Abcam, USA) prescribed guidelines.
Analysis of Xenobiotic Enzyme Activities
The liver tissue homogenates of experimental rats were analyzed for xenobiotic enzymes, including cytochrome-P450 (cyt-P450), cytochrome-4502E1 (cyt-4502E1), cytochrome-b5 (cyt-b5), NADPH-cytochrome-b5 reductase (NADPH-cyt-b5R), NADPH-cytochrome-P450 reductase (NADPH-cyt-P450R), and glutathione S-transferase (GST) with commercial assay kits. The tests were done with three replicates according to the procedures specified by the assay kit’s manufacturer (Abcam, USA).
Analysis of Inflammatory Cytokines
The serum inflammatory cytokine concentrations, including interleukin (IL)-1α, IL-1β, IL-2, IL-4, IL-10, IL-12, and granulocyte-macrophage colony-stimulating factor (GM-CSF) in the rats were analyzed utilizing diagnostic kits (Elabscience, USA). The experiments were done according to the manufacturer’s specifications for the kits.
AKT/mTOR, Apoptotic, and Tumor Biomarker Levels
The AKT and mTOR concentrations in the colon specimens were evaluated using commercial diagnostic kits (LSBio, USA). The concentrations of apoptotic proteins, including Bax, Bcl-2, caspase-3, and -9 in the colon tissues of rats were analyzed utilizing commercially available assay kits, following the manufacturer’s instructions (ThermoFisher, USA). The alpha-fetoprotein (AFP) and carcinoembryonic antigen (CEA) concentrations were studied using the designated assay kits using manufacturer’s procedures (Abcam, USA).
Histological Analysis
The histopathological evaluation of colon tissues in the experimental rats was conducted. The removed tissues were paraffin-embedded and sliced into 5 µm thickness. After the wax removal and rehydration, the slides were stained with hematoxylin-eosin. The sections were ultimately analyzed using a light microscope.
Statistical Analysis
The findings are portrayed as mean ± SD of triplicates. One-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT) were performed to examine statistical significance, with p < .05 indicating significance.
Results
Effect of Reserpine on Body Weight of the Experimental Rats
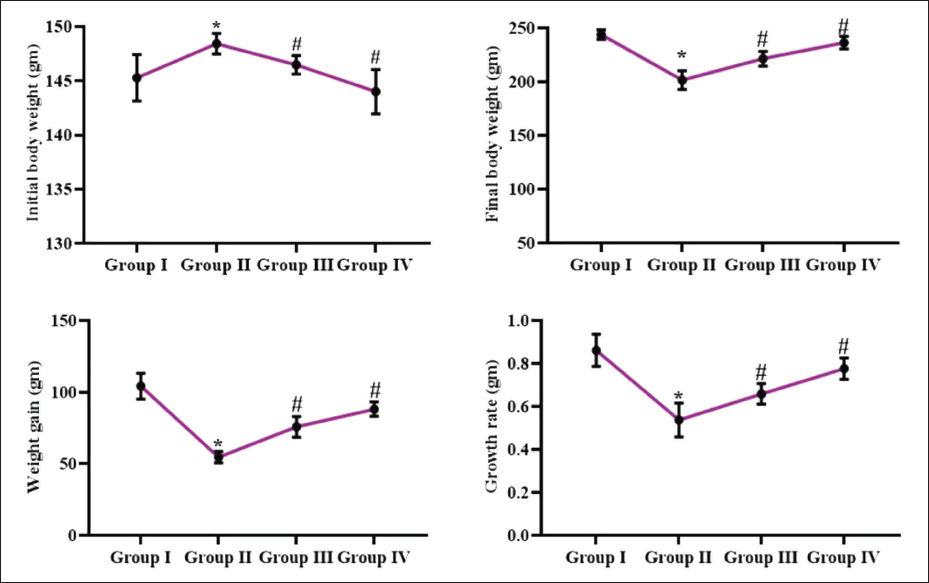
The effects of reserpine on body weight in the DMH-administered rats were evaluated, with the results illustrated in Figure 1. The DMH induction demonstrated a substantial reduction in body weight gain in rats compared to the control. Captivatingly, the reserpine administration resulted in significant weight increase and weight gain in DMH-administered rats. In comparison to the 25 mg/kg, the 50 mg/kg of reserpine markedly increased weight gain of DMH-treated rats.
Effect of Reserpine on the Polyp Numbers in the Experimental Rats
Table 1 illustrates the occurrence of colon polyps in the experimental rats. The control rats displayed no polyp development. However, DMH-challenged rats had a 100% incidence of colon polyps. The administration of reserpine at dosages of 25 and 50 mg/kg demonstrated a significant decrease in the development of colon polyps in rats induced with DMH (Table 1).
Effect of Reserpine on the Polyp Numbers in the Experimental Rats.
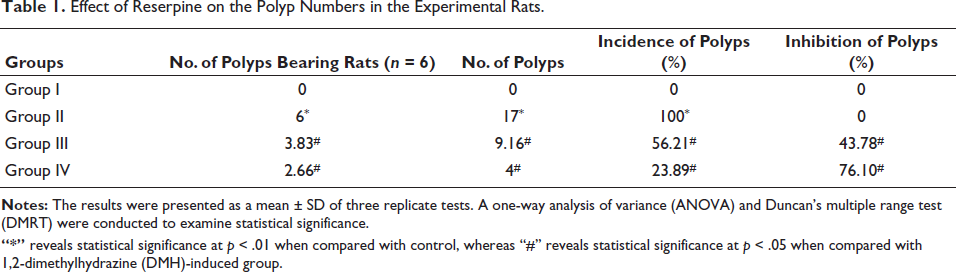
“*” reveals statistical significance at p < .01 when compared with control, whereas “#” reveals statistical significance at p < .05 when compared with 1,2-dimethylhydrazine (DMH)-induced group.
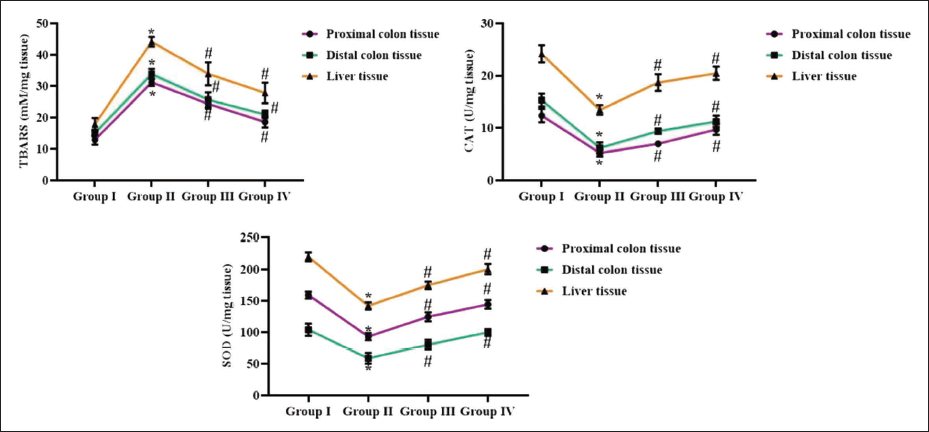
Effect of Reserpine on the Oxidative Stress Level in Experimental Rats
The oxidative stress levels were evaluated in the colon and liver tissues of the experimental rats. The DMH-challenged rats exhibited elevated TBARS levels in the liver tissue as well as distal and proximal colon tissues. Furthermore, the DMH-induced rats also illustrated a significant diminution in SOD and CAT concentrations in the liver and colon tissues (Figure 2). Notably, treatment of DMH-administered rats with reserpine at concentrations of 25 and 50 mg/kg remarkably reduced the TBARS in the liver and colon tissues (Figure 2). Moreover, the reserpine at both dosages effectively enhanced SOD and CAT activities in the colon and liver tissues of DMH-induced rats.
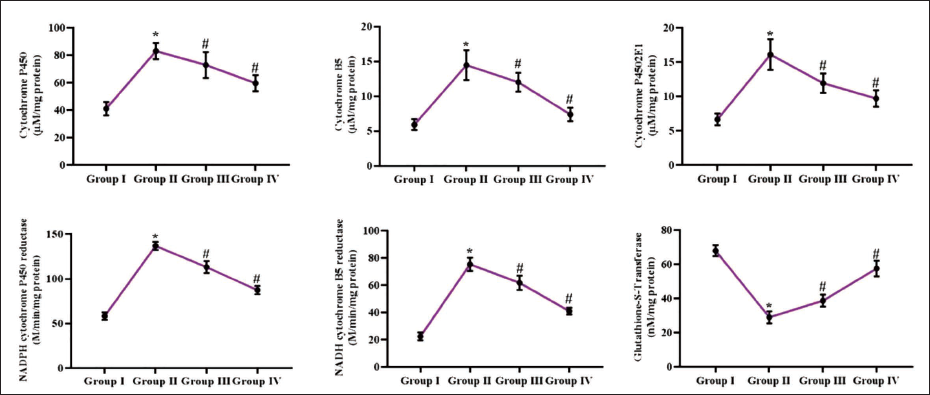
Effect of Reserpine on the Xenobiotic Enzyme Activities in the Experimental Rats
The xenobiotic enzymes, including cyt-P450, cyt-b5, cyt-4502E1, NADPH-cyt-b5R, NADPH-cyt-P450R, and GST, were examined in liver tissues of experimental rats (Figure 3). The concentrations of cyt-P450, cyt-b5, cyt-4502E1, NADPH-cyt-b5R, and NADPH-cyt-P450R enzymes were substantially increased in the hepatic tissues of DMH-administered rats. Furthermore, the DMH exposure also diminished the GST concentration in their hepatic tissues. Captivatingly, the reserpine at dosages of 25 and 50 mg/kg remarkably reduced the activities of cyt-P450, cyt-b5, cyt-4502E1, NADPH-cyt-b5R, NADPH-cyt-P450R enzymes (Figure 3) and eventually increased the GST activity in the DMH-treated rats.
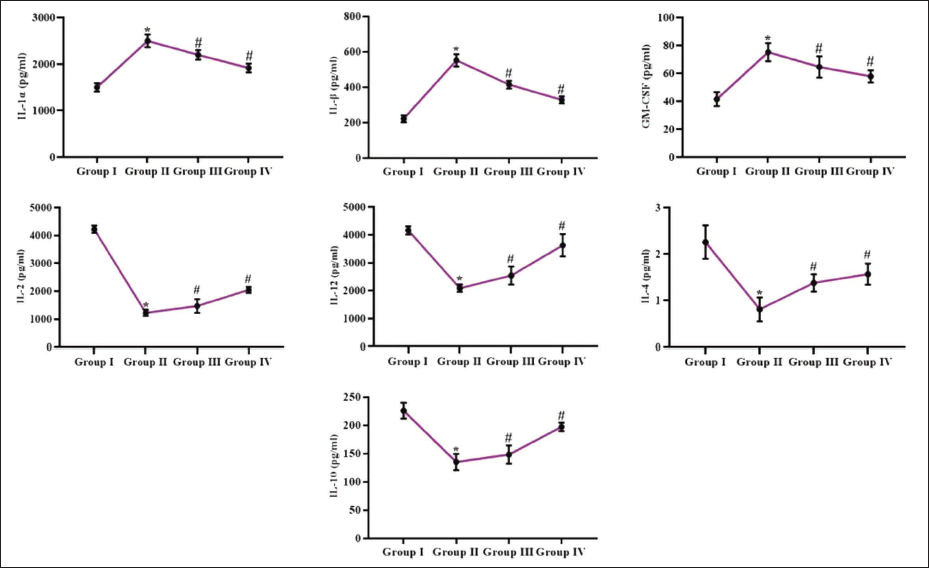
Effect of Reserpine on Inflammatory Cytokines in the Experimental Rats
The serum IL-1α, IL-1β, IL-2, IL-4, IL-10, IL-12, and GM-CSF concentrations in the rats were assessed, with the results displayed in Figure 4. The concentrations of IL-1α, IL-1β, and GM-CSF were significantly increased, and IL-2, IL-4, IL-10, and IL-12 cytokines were reduced in the serum of DMH-challenged rats. Notably, the reserpine treatment at 25 and 50 mg/kg dosages successfully regulated the concentrations of these inflammatory cytokines in DMH-induced rats.
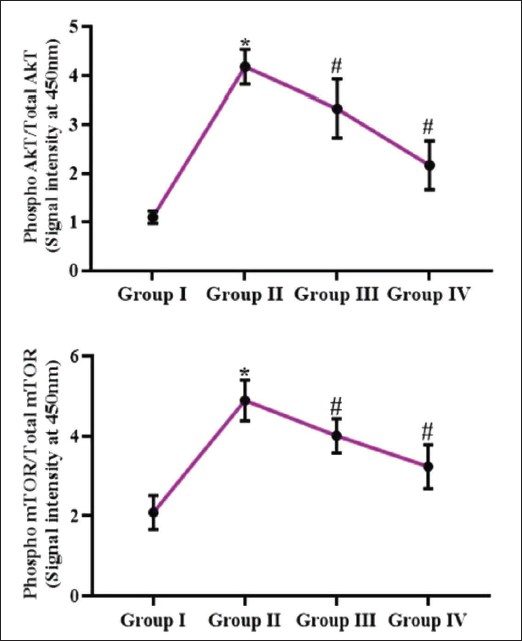
Effect of Reserpine on AKT and mTOR Levels in Experimental Rats
Figure 5 presents the effects of reserpine on the AKT and mTOR expressions in the colon tissues of rats. The DMH-challenged rats exhibited increased mTOR and AKT levels. Nevertheless, the 25 and 50 mg/kg of reserpine considerably reduced the mTOR and AKT concentrations in the colon tissues of the DMH-treated rats.
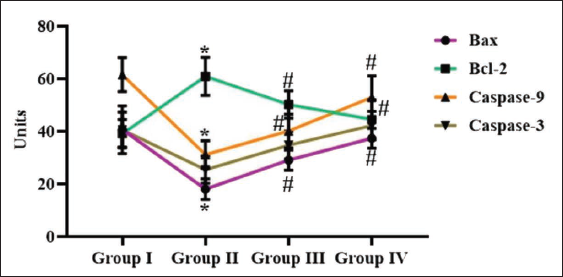
Effect of Reserpine on the Apoptotic Proteins in Experimental Rats
The impact of reserpine on the apoptotic protein expressions were evaluated, and results depicted in Figure 6. The significant diminution in Bax, caspase-3, and -9 levels and subsequent increase in Bcl-2 level were seen in the DMH-treated rats. Nonetheless, the 25 and 50 mg/kg of reserpine remarkably enhanced the Bax, caspase-3, and -9 levels and eventually diminished Bcl-2 levels in the DMH-treated rats (Figure 6).
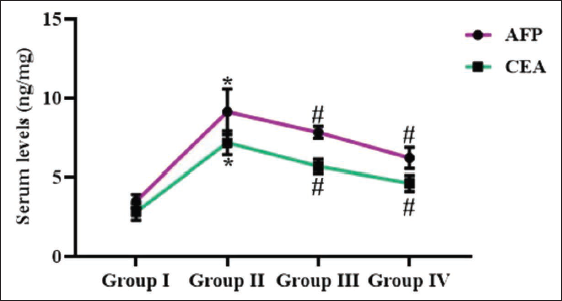
Effect of Reserpine on the Tumor Marker Levels in Experimental Rats
Figure 7 illustrates the serum tumor markers, such as CEA and AFP in the experimental rats. In the DMH-administered rats, the increased CEA and AFP were observed. Fascinatingly, the 25 and 50 mg/kg of reserpine remarkably suppressed both CEA and AFP concentrations in the DMH-treated rats.
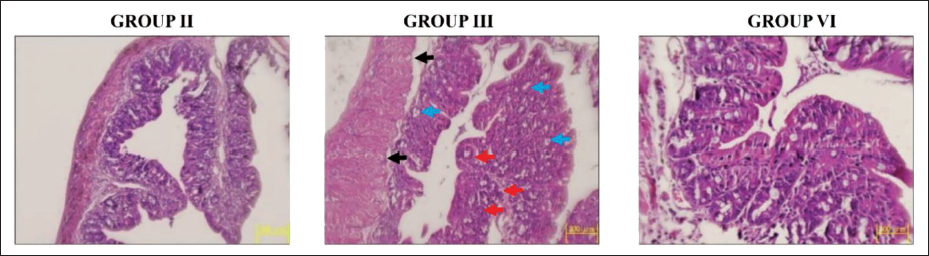
Effect of Reserpine on Colon Histology of Experimental Rats
The histology of the colonic mucosa of rats was assessed, and the corresponding images are given in Figure 8. Control rats displayed typical cellular configurations devoid of any anomalies. Contrarily, notable histological alterations, such as epithelial lining destruction, the occurrence of aberrant crypt foci, and dysplastic cells inside the glands, were seen in the colon specimens of DMH-administered rats when compared with control. Interestingly, the reserpine at 25 and 50 mg/kg dosages effectively ameliorated the histological alterations triggered by DMH and diminished the quantity of proliferating cells in the colonic region of DMH-treated rats.
Discussion
Colon cancer is a significant public health concern, ranking among the major causes of cancer-associated mortalities globally. Understanding the underlying mechanisms that lead to the onset of colon cancer is essential for the identification of novel therapies (Ionescu et al., 2023). Maintaining a healthy body weight is essential for individuals undergoing cancer treatment, as weight loss can have significant impacts on their overall health and prognosis. In the case of colon cancer-induced rats, the analysis of body weight changes can provide information into the onset of disease and the effectiveness of various treatment strategies (Mandic et al., 2023). Weight loss can be a common symptom of colon cancer, and it is often connected with a poorer prognosis. However, the degree of weight loss can differ depending on the disease’s stage and the patient’s response to therapy (Bowers et al., 2022). The present findings demonstrated a significant reduction in body weight in the DMH-treated rats. Fascinatingly, the reserpine effectively augmented the body weight in DMH-treated rats.
Oxidative stress has been thought of as a driven factor in the onset of various chronic diseases, including cancer (Sawai et al., 2023). In the context of colon cancer, the chemical carcinogen DMH has been found to cause oxidative injury to the colon tissues. Oxidative stress is characterized by increased TBARS, a marker of lipid peroxidation, has been observed in DMH-induced colon cancer. Simultaneously, the antioxidants like CAT and SOD have been shown to be altered in this model, potentially contributing to the disproportion between pro-oxidant and antioxidant systems (Basak et al., 2023). The impact of oxidative stress on the pathogenesis of DMH-induced colon cancer is multifaceted. Elevated oxidative stress can result in DNA injury, genomic variability, and the activation of signaling that enhances cell growth and survival, all of which are hallmarks of carcinogenesis. Furthermore, the depletion of antioxidants can further exacerbate the oxidative insult, developing a vicious cycle that perpetuates the development of colon cancer (Bardelčíková et al., 2023). In this work, the elevated TBARS level and a subsequent reduction in the antioxidants CAT and SOD were seen in liver and colon tissues of DMH-treated rats. Captivatingly, the reserpine administration to the DMH-administered rats led to a decreased TBARS level and an eventual increase in the CAT and SOD activities, which proves its antioxidant properties.
The progression of colon cancer is a highly complex mechanism that participates the interplay of various enzymes and pathways. Among the key players in this pathophysiology are the xenobiotic-metabolizing enzymes. The cyt-P450 enzymes, particularly CYP2E1, play an essential role in the metabolism of the carcinogen DMH, which is known to induce colon cancer. These enzymes can activate the carcinogen, leading to the generation of reactive intermediates that can directly damage DNA and participate in the initiation of colon cancer (Beyerle et al., 2020). Cyt-b5 and the associated NADPH-cyt-b5R enzyme are also involved in the metabolic activation of DMH, as they can modulate the activity of the cyt-P450 enzymes. Furthermore, the NADPH-cyt-P450R enzyme is essential to the proper functioning of the cyt-P450 system, and its dysregulation can impact the overall metabolic process (Venkatachalam et al., 2020). In addition to the activation of carcinogens, the GST enzymes play an essential role in the neutralization of these reactive intermediates. The balance between the activation and detoxification pathways mediated by these enzymes is crucial in determining the susceptibility to DMH-induced colon cancer (De et al., 2023). In this work, we found that the cyt-P450, cyt-b5, cyt-4502E1, NADPH-cyt-b5R, and NADPH-cyt-P450R enzymes were significantly increased, and GST activity was diminished in the hepatic tissues of DMH-administered rats. Fascinatingly, reserpine treatment successfully diminished the activities of cyt-P450, cyt-b5, cyt-4502E1, NADPH-cyt-b5R, and NADPH-cyt-P450R enzymes and eventually increased the GST in DMH-treated rats. These findings confirm that reserpine promoted the metabolism of carcinogen in the experimental rats.
Colon cancer is a complex and multifaceted condition, with chronic inflammation playing a crucial role in its pathogenesis. Chronic inflammation can act as a predisposing factor for the malignant transformation of cells, resulting in the onset of colon cancer (Borowczak et al., 2022; Burgos-Molina et al., 2024). It has been already highlighted the connection between chronic inflammation and colon cancer. The presence of inflammatory cells and regulators in the tumor microenvironment can enhance the proliferation and survival of tumor cells (Nardone et al., 2023). One of the key inflammatory cytokines implicated in the pathophysiology of colon cancer are IL-1α and IL-1β. These cytokines can trigger the production of other inflammatory regulators and can also activate the NF-κB signaling, which is known to play a pivotal role in the immune responses, cell survival, and inflammation (Braumüller et al., 2022). Other inflammatory cytokines have also been shown to participate in the onset colon cancer. IL-2 participates in the activation and growth of T cells, which can promote anti-tumor immune responses. IL-4, on the other hand, is thought to have a protective role in colon cancer, as it can block the proliferation of tumor cells and enhance the differentiation of immune cells. IL-10 is an anti-inflammatory cytokine that can inhibit the immune reactions against cancer cells, thus promoting tumor progression. IL-12 is a cytokine that plays a crucial role in the activation of the Th1 immune response, which can block the growth and metastasis of tumor cells (Bhat et al., 2022). Another important inflammatory mediator in the pathophysiology of colon cancer is GM-CSF. This marker is implicated in the differentiation and activation of myeloid cells, like macrophages and granulocytes, which can participate in the inflammatory microenvironment of the tumor (Chen et al., 2014). The current findings evidenced the increased concentrations of IL-1α, IL-1β, and GM-CSF and reduced IL-2, IL-4, IL-10, and IL-12 cytokine concentrations in the DMH-treated rats. Surprisingly, the reserpine treatment effectively regulated these inflammatory cytokine concentrations in the DMH-administered rats.
The AKT/mTOR axis is one of the extensively studied pathways in the field of colon cancer. The AKT/mTOR axis is an essential regulator of cell growth, proliferation, survival, and metabolism. Alterations in this signaling are frequently observed in several cancers, including colon cancer. Phosphorylated and activated AKT then goes on to phosphorylate and regulate a wide range of substrates, including mTOR (Leiphrakpam & Are, 2024). The hyperactivation of the AKT pathway is a characteristic of colon cancer. The activation of AKT subsequently results in the mTOR activation, a central regulator of cell growth and metabolism. The mTOR signaling plays a pivotal role in promoting cell proliferation, survival, and angiogenesis, all of which contribute to tumor progression (Świechowski et al., 2024). Targeting the AKT/mTOR pathway has developed as a talented method to treat colon cancer. Various inhibitors of this pathway, such as AKT and mTOR inhibitors, were developed and are currently under investigation in clinical trials. These inhibitors have shown the potential to suppress tumor progression and improve the efficiency of standard chemotherapeutic agents (Popova & Jücker, 2021). The present work evidenced that the rats with DMH-administered colon cancer exhibited increased mTOR and AKT levels. However, the treatment of reserpine markedly reduced the mTOR and AKT levels in the colon tissues of the DMH-administered rats. These findings supported that reserpine inhibited the AKT/mTOR axis in colon cancer-induced rats.
Apoptosis is a tightly regulated mechanism that eliminates unnecessary or damaged cells, maintaining a crucial balance between cell growth and death. Defects in the apoptotic signaling pathways can participate in the onset of colon cancer, as they allow the genetic mutations, promote growth factor-independent cell growth, and confer resistance to immune-based and conventional cancer therapies (Orlandi et al., 2023). The Bcl-2 family of proteins, like pro-apoptotic Bax and the anti-apoptotic Bcl-2, are central regulators of the apoptosis. Bax is a crucial pro-apoptotic protein that promotes the mitochondrial membrane permeability, resulting in the release of apoptogenic proteins such as cyt-c. The activation of Bax is countered by the Bcl-2, which inhibits Bax-mediated mitochondrial outer membrane permeability and subsequent caspase activation. In colon cancer, the balance between pro- and anti-apoptotic proteins is often dysregulated, with elevated Bcl-2 and decreased Bax expressions, contributing to cancer cell growth and resistance to apoptosis (Kaloni et al., 2023). In addition to the Bcl-2 family, the caspase family of proteases, particularly caspase-3 and -9, play pivotal roles in the execution of the apoptotic program. Caspase-3 is the central effector caspase that degrades several cellular components, resulting in the characteristic morphological and biochemical alterations related with apoptosis. Caspase-9, on the other hand, is an initiator caspase that is activated by the apoptosome complex, which is developed upon the cyt-c release from the mitochondria (Yang et al., 2023). In colon cancer, the dysregulation of caspase-3 and -9 has been observed, with decreased activity or expression of these proteases implicating in the apoptosis evasion by cancer cells (D’Aguanno & Del Bufalo, 2020). The current findings exhibited a significant diminution in Bax, caspase-3, and -9 levels and a subsequent increase in Bcl-2 level in the DMH-administered rats. Though, reserpine significantly enhanced the Bax, caspase-3, and -9 levels and eventually diminished the Bcl-2 levels in the colon tissues. These results highlighted that reserpine successfully activated the apoptotic pathway in the colon cancer condition in rats.
The tumor markers CEA and AFP play a major role in the pathophysiology of colon cancer. AFP and CEA are two well-known tumor markers that have been extensively explored in the colon cancer. AFP, a glycoprotein produced during fetal development, is typically elevated in certain types of cancer, including colon cancer. CEA, on the other hand, is a cell surface glycoprotein that is often overexpressed in several adenocarcinomas, including colon cancer (Ren et al., 2023). The diagnostic and prognostic significance of these tumor markers in colon cancer has been the focus of numerous studies. Increased AFP and CEA concentrations have been observed in colon cancer patients, and these markers have been used to aid in the diagnosis, monitoring, and treatment of the disease. The presence of these tumor markers in the tumor microenvironment can have significant roles for the pathophysiology of colon cancer. The dysregulation of these tumor markers may contribute to the immunosuppressive and subsequent pro-tumor effects within the tumor microenvironment, ultimately influencing disease progression and patient outcomes (Wojtalewicz et al., 2023). The elevated CEA and AFP concentrations were seen in the DMH-administered rats. Fascinatingly, the reserpine significantly decreased the CEA and AFP concentrations in the serum of DMH-treated rats.
Conclusion
The present study showed that reserpine exhibits significant anti-cancer properties against DMH-treated colon cancer in rats. The administration of reserpine significantly increased body weight, regulated the inflammatory cytokine levels, and mitigated oxidative stress in the DMH-treated rats. The administration of reserpine markedly regulated changes in xenobiotic-metabolizing enzymes, reduced tumor markers, and regulated apoptotic proteins in the DMH-induced rats. These results highlight that reserpine has potential as a chemotherapeutic agent to treat colon cancer. Furthermore, the remaining works are still warranted in the future to fully understand the anti-cancer mechanisms of reserpine against colon cancer.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work has been approved by the Institutional Animal Ethical Committee of the Department of Gastrointestinal Burns Surgery, Affiliated Hospital of Xiangnan University, Chenzhou, Hunan 423000, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
