Abstract
Ionizing radiation is a widely used therapy for solid tumors. However, high-dose ionizing radiation causes apoptosis, transforms normal cells into tumor cells, and impairs immune functions, leading to the defects in the removal of damaged or tumor cells. In contrast, low-dose radiation has been reported to exert various beneficial effects in cells. This experimental study investigated the effect of γ rays at low dose on the development of colorectal tumor in a 1,2-dimethylhydrazine (DMH)-induced colon cancer. Colorectal tumor model was induced in Wistar rats by subcutaneous injection of DMH (20 mg/kg) once a week for 15 weeks. Starting from zero day of DMH injection, a single low dose of whole-body γ irradiation of 0.5 Gy/week was applied to the rats. A significant reduction in lipid peroxidation, nitric oxide, and elevation in the glutathione content and antioxidant enzyme activity (superoxide dismutase and catalase) were observed after γ irradiation comparing with DMH group. Moreover, γ ray reduced the expressions of multidrug resistance 1 (MDR1), β-catenin, and cytokeratin 20 (CK20) those increased in DMH-treated rats. However, survivin did not change with γ ray treatment. A histopathological examination of the DMH-injected rats revealed ulcerative colitis, dysplasia, anaplasia, and hyperchromasia. An improvement in the histopathological picture was seen in the colon of rats exposed to γ rays. In conclusion, the present results showed that low-dose γ ray significantly inhibited DMH-induced colon carcinogenesis in rats by modulating CK20, MDR1, and β-catenin expression but not survivin expression.
Introduction
Colorectal cancer is one of the major causes of morbidity and mortality worldwide. 1–2 However, the causes of this disease are not completely understood. It is the third most common form of malignancy in both men and women. 3 Colon carcinogenesis is a multistep process and is thought to arise by the genetic alterations involving a variety of oncogenes and tumor suppressor genes that transform normal colonic epithelium into an invasive carcinoma. 4 Currently, multi-marker profile is commonly applied to distinguish colorectal carcinomas from other cancers. The simple epithelial intermediate filament keratin 20 (cytokeratin; CK20) is characteristically present in non-neoplastic epithelium of the colon and rectum and is constitutively expressed in carcinomas arising from these sites. Hence, CK20 plays a crucial role in immunoprofiling of colorectal carcinomas, including both primary and metastatic cancer tissues. 5
Several environmental factors including lifestyle and perhaps hormones are similarly important in the induction and progression of these tumors. Many epidemiological data indicate an increased risk of colorectal cancer through consumption of red or processed meat but not white. 4,6–7 Among these factors, the inflammatory response has been indicated by findings that anti-inflammatory agents including aspirin reduce the risk of colorectal cancer. 8–9 1,2-Dimethylhydrazine (DMH) is a potent colon carcinogen inducing colorectal tumors in experimental animals 1,4,10 and is the most widely used model of chemically induced colon carcinogenesis. DMH-induced colon cancer is a multistep process involving a series of pathological alterations, such as formation of aberrant cryptic foci. 4 Umesalma and Sudhandiran 11 have already reported that active metabolite of DMH is excreted through bile and is responsible for its carcinogenic effect on colon while passing through the digestive tract. DMH is metabolized in liver to form azoxymethane and methylazoxymethanol, which is further transported to colon via bile or blood to generate its ultimate carcinogenic metabolite, diazonium ion that elicits an oxidative stress by methylating biomolecules of colonic epithelial cells and leads to promutagenic events as a result of inflammation and tumor promotion.
Radiotherapy can be defined as the use of ionizing radiation in the treatment of malignant disease. It can be used both as radical treatment, where the aim is to remove the cancer cells completely, and as palliative treatment, where the aim of the radiotherapy is to reduce pain and other discomfort. Radiotherapy is, after surgery, the most widely used and effective means of treating cancers, and it is estimated that over 50% of all cancer patients require radiotherapy at some time during their illness. 12,13 It is well known that ionizing radiations induce oxidative stress on target tissues, mainly through the generation of reactive oxygen species (ROS), resulting in imbalance of the prooxidant and antioxidant status/levels in the cells, attack diverse cellular macromolecules such as DNA, lipids, and proteins, eventually inducing cell death. 14
Gamma rays are form of ionizing radiation used in radiotherapy. They are produced spontaneously as certain elements (such as radium, uranium, and cobalt 60), release radiation as they decompose, or decay. Each element decays at a specific rate and gives off energy in the form of γ rays and other radiation. 12 Therefore, the purpose of this study was to investigate the effect of γ rays at low dose on the development of colorectal tumor in DMH-induced rat colon carcinogenesis.
Materials and methods
Chemicals
DMH and tris(hydroxymethyl)aminomethane–hydrochloric acid (Tris-HCl) buffer were purchased from Sigma (St Louis, Missouri, USA). Perchloric acid, thiobarbituric acid (TBA), and trichloroacetic acid (TCA) were purchased from Merck (Darmstadt, Germany). All other chemicals and reagents used in this study were of analytical grade. Double-distilled water was used as the solvent.
Animals
Adult male Wistar albino rats weighing 200–250 g (10–12 weeks) were obtained from The Holding Company for Biological Products and Vaccines (VACSERA, Cairo, Egypt). The animals were kept in wire-bottomed cages in a room under standard conditions of illumination with a 12-h light/12-h dark cycle at 25 ± 2°C for 1 week until the beginning of treatment. The rats were provided tap water and a balanced diet ad libitum. We followed the European Community Directive (86/609/EEC) and National Rules on Animal Care that was carried out in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals 8th edition.
Experimental protocols
Irradiation
Prior the irradiation, the animals were anesthetized by subcutaneous injection of 25 mg/kg ketamine hydrochloride and 5 mg/kg xylazine. The rats were subjected to γ ray total body irradiation in a chamber using a cobalt-60 teletherapy machine. Each rat was exposed to total dose of 0. Gy in one fraction (dose rate 0.02 Gy/min). The dosage was calculated at 2.5 cm depth and 80 cm source–skin distance as described previously. 15 Animals were returned to their home cages following irradiation.
Colon cancer induction
DMH was dissolved in 1 mM ethylenediaminetetraacetic acid–containing 1 mM sodium bicarbonate (pH 6.5). Animals were given a weekly subcutaneous injection of DMH in the groin at a dose of 20 mg/kg body weight for 15 weeks. 16
Experimental animals
Twenty-eight rats were used in this experiment. The animals were randomly divided into four groups with seven animals in each group and were treated as follows:
Group 1 (Con): Rats were not treated with DMH and γ radiation, but received 0.5 ml/week saline (0.9% sodium chloride) given orally for 15 weeks. Group 2 (DMH): Rats were subcutaneously injected with DMH once a week for 15 weeks in order to induce colon cancer. Group 3 (γR): Rats were given equal volume of saline as described for group 1 and were subjected to whole-body irradiation of a single dose of 0.5 Gy/week for 15 weeks together. Group 4 (DMH + γR): Rats were subcutaneously injected with DMH at a dose of 20 mg/kg, starting from zero day of DMH injection and continued for 15 weeks, and a single dose of whole-body γ-irradiation of 0.5 Gy/week was applied together.
One week following the last administration, all the animals were cervically dislocated. Colons were removed quickly, cleaned free of irrelevant material and immediately perfused with ice cold saline, and immediately homogenized to give 10% (w/v) homogenate in ice-cold medium–containing 50 mM Tris-HCl, pH, 7.4. The homogenate was centrifuged at 3000 r/min for 10 min at 4°C. The supernatant (10%) was used for the various biochemical determinations. The total protein content of the homogenized colon was determined by the method of Lowry et al. 17 using bovine serum albumin as a standard.
Histopathological examination
The colons were fixed in 10% neutral-buffered formalin for 24 h, dehydrated in ascending concentrations of ethyl alcohol, cleared in xylene, and mounted in molten paraplast at 58–62°C. Slices of 4–5 μm thickness were obtained from the prepared blocks and stained with hematoxylin–eosin (H&E). Periodic acid–Schiff (PAS) reaction for goblet cells staining was done by oxidizing the obtained sections (4–5 μm) for 5 min in 1% aqueous periodic acid, washed under running tap water for 5 min, rinsed in distilled water, and then treated with Schiff’s reagent for 15 min. Afterward the sections were washed under running tap water for 10 min and counterstained with hematoxylin. Finally, for collagen content, Masson’s trichrome was performed. Briefly, the histological sections were deparaffinized, rehydrated, washed in running water (2 min), immersed in 5% iron alum (10 min) and Regaud’s haematoxylin (3 min), and rinsed in distilled water, 95% alcohol, and picric alcohol. Then the sections were washed again in running water (10 min), rapidly immersed in xylidine ponceau aqueous solution (3 min), distilled water and 1% glacial acetic acid, and 1% phosphomolybdic acid (3 min). Next, they were rinsed again in distilled water, immersed in aniline blue for 2–5 min, dehydrated, cleared, and mounted under a coverslip. The preparations obtained were visualized using a Nikon (Eclipse E200-LED, Tokyo, Japan) microscope at a magnification of 400×.
Oxidative stress markers
Homogenates of colon were used to determine malondialdehyde (MDA) as indicator of lipid peroxidation (LPO) by reaction of TBA according to the method of Ohkawa et al. 18 Nitrite/nitrate (nitric oxide; NO) 19 and glutathione (GSH) 20 were also chemically measured.
Enzymatic antioxidant status
Homogenates of colon were used for determination of superoxide dismutase (SOD) activity according to Nishikimi et al. 21 and catalase (CAT) activity as described by Aebi. 22
Real-time PCR
Total RNA was isolated from the colon tissue using an RNeasy Plus Minikit (Qiagen, Valencia, California, USA). One microgram total RNA and random primers were used for complementary DNA (cDNA) synthesis using the RevertAid™ H Minus Reverse Transcriptase (Fermentas, Thermo Fisher Scientific Inc., Canada). For real-time polymerase chain reaction (RT-PCR) analysis, the cDNA samples were run in triplicate. For a reference gene, β-actin (sense: 5′-GGCATCCTGACCCTGAAGTA-3′; antisense: 5′-GGGGTGTTGAAGGTCTCAAA-3′) was used. RT-PCR reactions were performed using Power SYBR® Green (Life Technologies, Carlsbad, California, USA) and was conducted on the Applied Biosystems 7500 instrument (Waltham, Massachusetts, USA). The typical thermal profile is 95°C for 4 min, followed by 40 cycles of 94°C for 60 s and 55°C for 60 s. After PCR amplification, the differences (Δ) between the threshold cycle (Ct) is calculated by subtraction of the β-actin Ct from each sample Ct. For relative quantitation of gene expression, the 2−ΔΔCt. The method of Pfaffl was used for data analysis. 23 The PCR primers for β-catenin (sense: 5′-GCTGACCTGATGGAGTTGGA-3′; antisense: 5′-GCTACTTGCTCTTGCGTGAA-3′), MDR1 (sense: 5′-CTGGTTTGATGTGCACGATGTTGG-3′; antisense: 5′-TGCCAAGACCTCTTCAGCTACTG-3′) and survivin (sense: 5′-GCATGGGTGCCCCGACGTTG-3′; antisense: 5′-GCTCCGGCCAGAGGCCTCAA-3′) genes were synthesized by Jena Bioscience GmbH (Jena, Germany). Primers were designed using Primer-Blast program from National Center for Biotechnology Information.
Immunohistochemistry for detection of CK20
For immunohistochemistry, the colon section (4 μm) were deparaffinized and then boiled to unmask antigen sites, and the endogenous activity of peroxidase was quenched with 0.03% hydrogen peroxide (H2O2) in absolute methanol. The sections were then incubated overnight at 4°C with a 1:200 dilution of rabbit monoclonal CK20 antibody (ab76126; Abcam, Cambridge, UK) in phosphate-buffered saline (PBS). After removal of the unbound primary antibody by rinsing with PBS, slides were incubated with a 1:500 dilution of biotinylated anti-rabbit secondary antibody. Bound antibodies were detected with avidin biotinylated peroxidase complex (ABC-kit Vectastain) and the chromogen 3,3′-diaminobenzidine tetrachloride (DAB) was used as substrate. After appropriate washing in PBS, slides were counterstained with hematoxylin. Scoring method is based on estimating both the percentage of positively stained cells on a slide (proportion score; PS) and the strength of the staining (intensity score; IS). PS is ranked into five categories as 1 = <5%, 2 = 5–25%, 3 = 25–50%, 4 = 50–75%, 5 = 75–100%. IS is graded as 1 for very weak, 2 for week, 3 for moderate, and 4 for strong. The total score (TS) was multiplication of PS and IS, and it ranges from 1 (lowest) to 20 (highest). All sections were incubated under the same conditions with the same concentration of antibodies and at the same time, and so the immunostaining was comparable among the different experimental groups.
Statistical analysis
Results were expressed as the mean ± standard error of the mean. Data for multiple variable comparisons were analyzed by one-way analysis of variance (ANOVA). For the comparison of significance between groups, Duncan’s test was used as a post hoc test according to the Statistical Package for the Social Sciences (SPSS version 17.0).
Results
In the control group, normal histological structure of the mucosa, submucosa, and muscularis layers of colon sections were observed. An intact mucosal gland with lined epithelial cells and goblet cells was also noticed in mucosa layer (Figure 1(a)). Cancer-induced group showed abnormal structure in mucosal glands represented in dysplasia, anaplasia, and hyperchromasia, and also an acute infiltration of inflammatory cells was observed in mucosa and lamina propria (Figure 1(b)). Gamma radiation-treated rats (γR group) had normal structure of colon similar to the control one (Figure 1(c)). Colon cancer-induced rats treated with γ radiation (DMH + γR group) showed clear improvement in mucosal layer with few inflammatory cells infiltration in the periglandular and in the lamina propria layers (Figure 1(d)).

Photomicrographs of colon sections of different groups stained by H&E (×400). (a) Normal histological features of the colon with glandular structure and the lining epithelium of mucosa from the control rats. (b) Colon from the cancer-induced rats shows anaplasia, dysplasia and hyperchromasia. (c) Colon section of γ radiation treated rats shows normal histological features of the glandular structure of mucosa and lamina propria. (d) Colon from the cancer-induced rats treated with γ radiation shows clear improvement in the glandular structure and the lining epithelium of mucosa, a few inflammatory cells infiltration was also seen. H&E: hematoxylin and eosin.
Histological examination of control colon section stained with PAS reaction showed normal PAS reaction located markedly in mucous secreting goblet cells (Figure 2(a)). In cancer-induced group, faint or weak PAS reaction was located in the goblet cells of mucosal glands (Figure 2(b)). Normal PAS reaction just like the control group was seen in mucosal gland goblet cells in γR-treated group (Figure 2(c)). A marked improvement in PSA reaction was observed in cancer-induced group treated with γ radiation (Figure 2(d)).
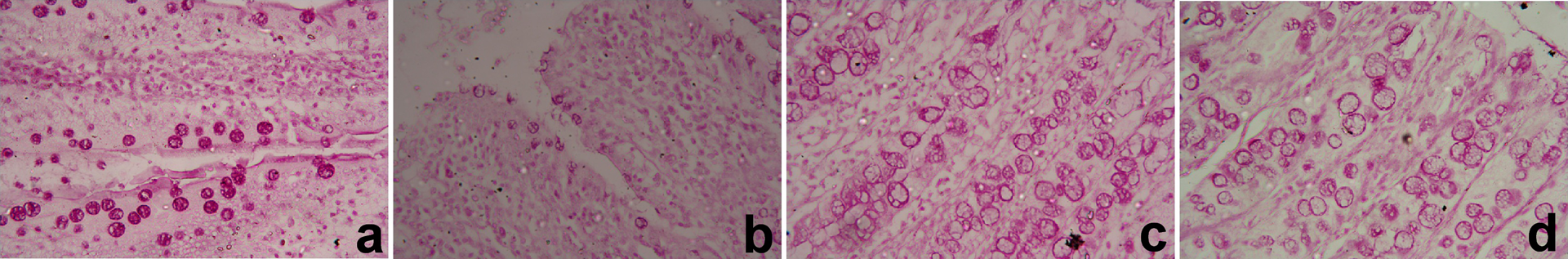
Photomicrographs of colon sections of different groups stained by PAS (x400). (a) Colon section from the control rats shows strong positive PAS reaction in the goblet cells of mucosa. (b) Colon of cancer-induced rats with weak PAS reaction in the goblet cells of mucosa. (c) Colon section of γ radiation treated rats with positive PAS reaction. (d) Colon from the cancer-induced rats treated with γ radiation showed moderate positive PAS reaction in the goblet cells of mucosa. PAS: Periodic acid–Schiff.
Histological examination of control colon sections stained with Masson’s trichrome showed normal collagenous content in mucosa submucosa and masculosa (Figure 3(a)). Cancer-induced group showed mild increase in collagenous content represented in highly compact collagen bundles in submucosa and lamina propria, also in mucosa periglandularly. Dilatation and congestion in blood vessels of submucosa was seen (Figure 3(b)). While in group treated alone with γ radiation, normal collagen content was observed, just like in the control one (Figure 3(c)). A clear improvement in collagenous content in all layers was showed in cancer-induced group treated with γ radiation (Figure 3(d)).

Photomicrographs of colon sections of different groups stained by Masson’s trichrome (×400). (a) Colon from the control rats shows normal collagenous content periglandular in the mucosa, lamina propria, and masculosa layers. (b) Colon from the cancer-induced rats shows an intensive increase in collagenous content periglandular in mucosa, lamina propria, and masculosa layers. (c) Colon from the γ radiation-treated rats shows normal collagenous content in all layers. (d) Colon from the cancer-induced rats treated with γ radiation shows clear improvement in the collagenous content in all layers.
Immunohistochemical examination of control colon showed moderate positive reaction in CK20 immunostaining located in glandular cells of mucosa layer (Figure 4(a)). Meanwhile, in cancer-induced group (DMH) marked increase in anti-CK20 reaction was clearly shown in mucosal glands (Figure 4(b)). γR group showed moderate positive reaction in CK20 like the control group (Figure 4(c)). After treatment with γ radiation to cancer-induced group (DMH + γR), moderate improvement represented by mild positive reaction in CK20 in the glandular cells of mucosa layer was seen (Figure 4(d)). In this case, the number of CK20 immunostaing cells was decreased significantly (p < 0.05; Supplementary data; Table 1S).

Photomicrographs of colon sections of different groups immunostained with CK20 (×400). (a) Colon from the control rats shows normal anti-CK20 reaction in the cells of mucosal glands. (b) Colon from the cancer-induced rats shows an intensive increase in anti-CK20 reaction in the cells of mucosal glands. (c) Colon from the γ radiation-treated rats shows normal anti-CK20 reaction in the cells of mucosal glands. (d) Colon from the cancer-induced rats treated with γ radiation shows moderate improvement in anti-CK20 reaction in the cells of mucosal glands. CK20: cytokeratin 20.
Oxidative stress markers in rat colon exposed to γR at low dose and DMH-induced colon cancera.

γR: gamma radiation; DMH: 1,2-dimethylhydrazine; MDA: malondialdehyde; NO: nitric oxide; GSH: glutathione.
aValues are the mean ± SEM (n = 7).
bp < 0.05: significant change with respect to the control group.
cp < 0.05: significant change with respect to DMH group.
Effects of γR on LPO, NO, and GSH levels during colon cancer induced by DMH are presented in Table 1. LPO and NO in colon homogenates were markedly (p < 0.05) increased in DMH group as compared to corresponding values of the control group. Meanwhile, a post hoc comparison of means in DMH + γR group indicated significant (p < 0.05) reduction in LPO and NO levels in rats treated with γR compared with the γR group. Colon cancer in rats caused overproduction of cellular oxidants and modulation of antioxidant defense system. As observed in our study, DMH injection led to modulation of several parameters of oxidative stress relative to the control animals. After 15 weeks of DMH injection, GSH content in the colon homogenates was decreased significantly (p < 0.05) compared with the controls (Table 1). Moreover, the depletion in GSH was still observed after the treatment with γR in DMH + γR group. As shown in Table 1, we found that γR alone caused oxidative stress as indicated by significant (p < 0.05) increase in LPO and NO levels in the colon tissue. While GSH content didn’t change after exposure to γR.
As shown in Table 2, DMH treatment led to modulation of antioxidant enzymes relative to the control rats. After 15 weeks of DMH administration, SOD and CAT activities in the colon homogenates decreased significantly (p < 0.05) compared with the control. Meanwhile, treatment with γ rays significantly (p < 0.05) increased the activities of SOD and CAT in DMH + γR group compared with the control group. Moreover, γR treatment alone showed significant (p < 0.05) increase in SOD and CAT activities in γR group compared with the control group (Table 2).
Antioxidant enzyme activity in rat colon exposed to γR at low dose and DMH-induced colon cancera.

γR: gamma radiation; DMH: 1,2-dimethylhydrazine; SOD: superoxide dismutase; CAT: catalase.
aValues are the mean ± SEM (n = 7).
bp < 0.05: significant change with respect to the control group.
cp < 0.05: significant change with respect to DMH group.
In order to elucidate the mechanisms of how γR exerts its antitumor effect, we examined the expression of survivin, MDR1, and β-catenin in the colon tissues. DMH was known to cause inflammation and γR was known to possess anti-inflammatory property. By RT-PCR, there was a marked (p < 0.05) increase in the expression of β-catenin, MDR1, and survivin in the colon tissue from rats injected with DMH compared with the control group. In the presence of γR (group 4), there were a marked (p < 0.05) decrease in the expression of MDR1 and β-catenin in the rats compared with the DMH group (Figure 5). Consistent with the RT-PCR, γR nonsignificantly changed the expression of MDR1 and β-catenin in the colon tissues from normal rats. However, in rats exposed to γR, the expression of survivin in colon tissues from DMH-treated and normal groups were upregulated significantly (p < 0.05) compared with the control group.

Relative quantification using RT-qPCR of mRNA expression of β-catenin, MDR1, and survivin genes in colon of the rats treated with DMH and γ radiation. Values are the mean ± SEM (n = 7). #p < 0.05: significant change with respect to the control group. $p < 0.05: significant change with respect to DMH group. RT-qPCR: real-time quantitative polymerase chain reaction; MDR1: multidrug resistance-1; mRNA: messenger RNA; SEM: standard error of mean; DMH: 1,2-dimethylhydrazine.
Discussion
The development of colorectal cancer is a complex pathological process and involves in multiple steps and stages with changes from normal crypt foci to aberrant crypt foci, adenomas formation, expansion, and eventual development to colorectal cancer. 24 In this study, the effects of γ rays at low dose on rats treated with DMH-induced colorectal cancer were examined. The results showed that γR could inhibit DMH-induced colon cancer, suggesting that γR indeed had significant inhibitory effects on the carcinogenesis formation in the colon of rats.
DMH is a procarcinogen and is metabolized to a methyl free radical and generates hydroxyl radical or H2O2 in the company of metal ions that may contribute to the initiation of cancer stages and lipid peroxidation. Lipid peroxidation and/or MDA formation are one of the important and relevant markers of oxidative damage and elevated levels of LPO product have been found after treatment with DMH. 4,25 Consistent with the previous reports, our results also showed remarkable increase in the MDA level after DMH injection.
Oxidative stress and inflammation play important roles and are documented to influence tumor initiation and promotion. 4 NO has been reported to exert dichotomous effects within the multistage model of cancer. 26 It modulates different cancer-related events including angiogenesis, apoptosis, cell cycle, invasion, and metastasis. 27 In contrast to tumor-promoting effects, NO has also been reported to have tumoricidal effects. The effects of NO in tumor biology are broad, spanning its involvement in cellular transformation, formation of neoplastic lesions, and initiation and regulation of the metastatic cascade. 28 NO may mediate DNA lesions by the formation of toxic and mutagenic species, by direct modification of DNA, or by inhibition of DNA repair mechanisms. 29 NO promotes angiogenesis by activation of COX-2, which stimulates the production of proangiogenic factors and prostaglandins. The main source of NO that reported in our study are inducible nitric oxide synthase (iNOS) and endothelial NOS expressions. 30 Samanta et al. 31 found that iNOS messenger RNA (mRNA) expression was increased in DHM-induced colon cancer.
Elimination of free radicals in biological systems is achieved through enzymatic and nonenzymatic antioxidants, which act as major defense systems against free radicals. 32 GSH and its oxidized counterpart represent a major redox buffer system of the cell. GSH can act either as a nonenzymatic antioxidant by direct interaction of –SH group with ROS or it can be implicated in the enzymatic detoxification reaction for ROS, as a cofactor or coenzyme. The GSH levels in rats injected with DMH showed significant decrease in our study and in the previous studies. 33,34 Furthermore, DMH treatment generates free radicals in colonic tissue, and their level is controlled by GSH and other enzymatic antioxidants like SOD, CAT, GSH peroxidase, and GSH reductase by scavenging the free radicals. Thus, DMH treatment decreases the activity of antioxidant enzymes. 25,33
Studies have shown varied findings with respect to the effect of radiotherapy on the oxidant–antioxidant status. Some authors have observed increased oxidative stress after radiotherapy and have suggested that radiation-induced free radicals cause oxidative damage to biomolecules. 35,36 It is believed that the extent of cellular damage by low-radiation dose is proportional to the effects observed at high radiation dose per the “linear no-threshold” hypothesis. However, this notion may not be true at low-dose radiation exposure in the living system. 37 In the present study, we observed stimulation of antioxidant status in the colon after whole-body exposure to low doses of γ radiation. This effect might be due to an increase in the expression of mRNA for γ-glutamylcysteine synthase, a rate-limiting enzyme in GSH synthesis. In addition, under mild oxidative stress, cells are able to regulate a variety of physiological mechanisms in an attempt to cope up with the oxidative stress that causes the stimulation of antioxidant enzymes. 37
The adaptive response of low γ radiation is associated with the induction of antioxidant and detoxification enzymes. For example, low-dose γ ray irradiation increases GSH and thioredoxin, leading to the prevention of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced brain damage. 38 Consistent with other reports regarding induction of antioxidant and detoxification enzymes by low γ radiation, we also found low γ radiation (0.5 Gy) increased the SOD and CAT activities. This finding is in accordance with other reports. 39,40
β-Catenin is a key regulator and an important element of the cadherin-mediated cell–cell adhesion system. 41 In the present study, β-catenin expression was increased in the colon samples of DMH-injected rats. This finding is consistent with the previous studies by Iwamoto et al. 42 and Saad et al. 43 Our finding highlighted the roles of this molecule in the maintenance of normal function and properties of colon cancer. Earlier study by Latifah et al. 44 showed that β-catenin expression in a specific carcinogen-induced colon cancer rats was reduced by germinated brown rice treatment. In the present report, γ radiation causes β-catenin down expression.
MDR is one of the major reasons for the occurrence of failure in chemotherapy-based treatment. Of the many mechanisms of MDR, the high expression of the human MDR1 gene and the P-glycoprotein (Pgp) transporter encoded by MDR1 is an important focus of research. 45 High expression of the MDR1 gene has been reported in a significant percentage of a wide variety of untreated human cancers including colorectal tumors. For example, 30–96% of human colorectal cancers at the time of diagnosis overexpress Pgp. Pgp also plays important roles in cell cycle, differentiation, and apoptosis. 46 For example, several lines of evidence indicate that Pgp confers protection from ceramide-mediated apoptosis. 47 In addition, it has also been shown that γR-induced apoptosis and/or cell cycle arrest is associated with increased levels of ceramide. 48,49 Furthermore, ceramide and its metabolites have been shown to inhibit colorectal and liver carcinogenesis. 50 Pgp may thus support tumor development by influencing one or more of these functions and γR, by virtue of its ability to inhibit Pgp, can inhibit tumor development. In the present study, low γR exposure caused significant decrease in mRNA of MDR1 expression. The present data is in accordance with the previous report of Li et al. 51 that found treating human colorectal cancer HCT-8 cells with low-radiation dose decreased MDR expression markedly and manifesting that low radiation dose can reverse MDR caused by high-radiation dose.
According to our observations in the colon after 15 weeks of DMH exposure, there was an increase in survivin expression. Our finding is in agreement with that of Jin et al. 52 and Chu et al., 53 who demonstrated that the expression levels of survivin mRNA and protein were higher in colorectal carcinoma cells than in normal cell line. The changes in proliferation and apoptosis activity in rats injected with DMH could be also due to the upward shift of survivin expression, as a clear consequence of survivin dual function as mitotic regulator and apoptosis inhibitor. 54 Furthermore, survivin’s function as a mitotic regulator and apoptosis inhibitor, 54,55 will contribute to inflammatory bowel disease (IBD) induction and the tumor progression. We report here that low dose of γ radiation induces an adaptive response by increasing the expression of mRNA of survivin in accordance with Grdina et al. 56 and Fraunholz et al. 57
CKs are group of approximately 20 proteins that consist of a type of intermediate filament and are differentially expressed in epithelia of various sites. 58 In the present report, we observed significant differences in CK20 immunostaining between DMH and normal colorectal tissues (Figure 4 and Supplementary data; Table 1S). Specifically, we demonstrated that, compared with normal tissue, colorectal cancers were more likely to be positive for CK20. The expression of CK20 has been observed to vary among different epithelial tumors, and this marker is currently used as a diagnostic tool to help determine the site of origin of metastatic carcinomas. Colon adenocarcinomas typically express CK20. 59 In the present report, low dose of γ radiation induced reduction in CK20 immunostaining.
In conclusion, we showed that γR at low dose significantly inhibited DMH-induced colon carcinogenesis in rats by modulating β-catenin, MDR1 and cytokeratin 20 expression but not survivin expression. Overall, the present data indicate that low dose of γ radiation holds great promise in the field of colon cancer radiotherapy. However, the accuracy of this hypothesis needs to be proved by further experiments.
