Abstract
Background
Allicin has anti-tumor activity and an inhibitory effect on lung cancer.
Objectives
In this experiment, lung cancer mice were used as the research subjects to explore the mechanism of allicin with cisplatin in inhibiting the growth and metastasis of Lewis cells in lung cancer mice.
Materials and Methods
By constructing a lung cancer mouse model, the mouse Lewis cells were divided into a model group, allicin group, cisplatin group, and allicin combined with cisplatin group to measure hypoxia-inducible factor 1a (HIF-1a) expression of Lewis cells in lung cancer mice and their effects on cell growth, metastasis, apoptosis, and invasion.
Results
The proliferation rate, migration, and invasion numbers of Lewis cells in the allicin combined with the cisplatin group were lower, and the apoptosis rate was higher (vs. model group) (p < .05). After the HIF-1a inhibitor was applied under the condition of allicin intervention, the HIF-1a level decreased (vs. the allicin group, HIF-1a Inhibitor group), HIF-1a level in allicin combined with HIF-1a agonist group was increased (vs. allicin group, p < .05). After the HIF-1a inhibitor was used under the intervention of allicin combined with cisplatin, HIF-1a expression decreased (vs. model group, p < .05), and the inhibitory effect of allicin and cisplatin on the growth and metastasis of mouse lung cancer Lewis cells was more significant (vs. model group), p < .05.
Conclusion
Allicin can inhibit the growth and metastasis of Lewis cells in lung cancer mice by inhibiting the expression of HIF-1a, and its combination with cisplatin is a better anti-tumor approach with less toxic and side effects, which is worthy of future clinical application.
Introduction
Lung cancer is a common malignancy that poses a serious threat to human health and safety. Its occurrence is closely related to environmental pollution, smoking, genetics, and other factors (Abou-Zeid et al., 2023). Research data showed that Bao et al. (2023), the mortality rate and incidence rate of lung cancer in China are higher than in the world on average, and the number of lung cancer patients is increasing, and the incidence rate is gradually increasing. It has been reported by Feng et al. (2023) that the majority of male lung cancer patients in my country are over 50 years old, and the majority of female patients are over 40 years old, and the increase in the number of female patients is higher than that of male patients. The currently used radiotherapy and chemotherapy drugs can achieve certain clinical effects, but some patients will have poor prognoses. According to statistics (Kong et al., 2023), the current 5-year survival in China is only 16.1%. Therefore, it is vital to find more effective treatment options.
Allicin is a compound extracted from garlic that has antioxidation, anti-inflammatory, anti-bacterial, and immune regulation effects. It can induce apoptosis of fibroblasts, block excessive proliferation, and inhibit cervical cancer, ovarian cancer, gastric cancer, and other malignant tumors’ cell proliferation and growth (Alamir & Patil, 2021; Bai et al., 2023). Studies by Bhaumik et al. (2019) have shown that allicin can inhibit behaviors of human tongue squamous cell carcinoma CAL-27 cells by down-regulating the expression of apoptosis inhibitors and other signaling pathways. Huang et al. (2020) found through in vivo experiments that allicin can inhibit tumor growth of endometrial cancer-bearing mice and can promote the apoptosis of endometrial cancer-bearing cells. Allicin also has an obvious anti-lung cancer effect. Research by Khakbaz et al. (2021) pointed out that allicin can induce DNA damage in non-small cell lung cancer cells, thereby inhibiting cell activities. Studies by Maitisha et al. (2021) also showed that allicin can inhibit the activities of mouse lung cancer A549 cells by activating Wnt/β-catenin signaling and preventing tumors from metastasizing to the lungs. Another study pointed out that (Pandey et al., 2020), allicin inhibits A549 cell proliferation and induces cell apoptosis, which may be related to hypoxia-inducible factor 1a (HIF-1a). In addition, many clinical studies and animal experiments have confirmed that combining allicin and various anti-tumor drugs can enhance the drug’s anti-tumor effect (Rosas-Gonzalez et al., 2020; Zhang & Yang, 2019). Zhou et al. (2022) found that compared with 5-fluorouracil alone, the combination of allicin and 5-fluorouracil is more effective in inducing the apoptosis of liver cancer cells. Alamir and Patil (2021) studied by constructing a mouse model of liver cancer and found that the combined application of allicin and lily polysaccharide can reduce the level of vascular endothelial growth factor (VEGF) in mice and promote the release of immune-related factors such as tumor necrosis factor α, thereby effectively inhibiting tumor growth in mice.
Cisplatin is also used in the chemotherapy of advanced lung cancer, but there is a decrease in chemotherapy sensitivity, which affects the therapeutic effect. In addition, cisplatin also has certain side effects, such as bone marrow suppression, nausea, and vomiting (Bai et al., 2023). Studies have pointed out (Bhaumik et al., 2019) the combination of allicin and cisplatin can improve the efficacy of tumor treatment and reduce the adverse reactions of cisplatin. Allicin combined with cisplatin has a prominent synergistic effect on the anti-proliferation behavior of cervical cancer HeLa cells (Bhaumik et al., 2019). The study by Borlinghaus et al. (2021) also showed that both allicin and cisplatin alone could inhibit the growth of adenoid cystic carcinoma-M cell xenografts in nude mice, but the inhibitory effect was more obvious when these two were used in combination. In addition, Cai et al. (2019) pointed out that allicin combined with cisplatin can antagonize the oxidative damage caused by cisplatin to human embryonic kidney HEK293 cells and reduce the nephrotoxicity of cisplatin.
However, whether the combination of allicin and cisplatin has an inhibitory effect on the growth and metastasis of mouse lung cancer Lewis cells has not been reported in the relevant literature, and its mechanism of action has not yet been clarified and needs to be further explored. Therefore, this article intends to establish a mouse model to investigate the effect of allicin and cisplatin on the growth and metastasis of Lewis cells in lung cancer mice and to explore its mechanism in depth. The research idea is shown in Figure 1.
Research Idea.
Materials and Methods
Experimental Reagents and Instruments
Lewis cells of lung cancer mice (Cell Resource Center, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences); Transwell Chamber (Costar); Automatic Biochemical Analyzer (Plan Medical); Flow Cytometry (Third Military Medical University); Automatic Inverted Fluorescence Microscope (product number Observer Z1, Germany); primary antibody, secondary antibody (Santa Cruz Company); HIF-1a agonist, HIF-1a antagonist (SANYO Japan); Dulbecco’s modified Eagle medium (DMEM) medium (purchased from Shanghai Sunshine); methyl thiazolyl tetrazole (MTT, purchased from Wuhan Punuo Sai); cisplatin (MedChemExpress). Allicin molecular structure diagram (Figure 2).
Allicin Molecular Structure Diagram.
Experimental Method
Establishment of Lewis Lung Cancer Mouse Model
Take 50 healthy mice and subcutaneously inoculate 0.2 mL (Lewis lung cancer viable cell concentration 1 × 106/mL) cell suspension in the left underarm of the mice on the first day of the experiment with a 1 mL syringe. The results of the imaging examination showed that if there was a tumor in the lung in the mice, the modeling was successful. If the mouse died during the modeling process, it was considered a modeling failure.
Grouping and Treatment of Mice
After successful modeling (n = 40), mice were randomly assigned into model group, allicin group, cisplatin group, and allicin combined with cisplatin group. Model group (n = 10) mice did not take intervention measures; the allicin group (n = 10) received 20 mg/kg allicin orally, once a day for three consecutive days, and the cisplatin group (n = 10) received 2 mg/kg cisplatin intraperitoneally, every 3 days. Injected once a day, allicin combined with the cisplatin group (n = 10) received the combined intervention of allicin (20 mg/kg) and cisplatin (2 mg/kg) according to the above method.
Cell Grouping and Intervention
Lewis cells were cultured in DMEM medium, digested with trypsin, and the logarithmic growth phase cells were divided into model group (Lewis cells were cultured normally), allicin group (Lewis cells + 30 µmol/L allicin), cisplatin group (Lewis cells + 5 µg/mL cisplatin) and allicin combined with cisplatin group (Lewis cells + 30 µmol/L allicin + 5 µg/mL cisplatin). In addition, cells were divided into HIF-1a inhibitor group (Lewis cells + HIF-1a inhibitor 3 µg/mL), HIF-1a agonist group (Lewis cells + HIF-1a agonist 3 µg/mL), allicin + HIF-1a inhibitor group (Lewis cell + 30 µmol/L allicin +HIF-1a inhibitor 3 µg/mL), allicin + HIF-1a agonist group (Lewis cell + 30 µmol/L allicin + HIF-1a agonist 3 µg/mL), cisplatin + HIF-1a inhibitor group (Lewis cells + 5 µg/mL cisplatin + HIF-1a inhibitor 3 µg/mL), allicin combined with cisplatin + HIF-1a inhibitor group (Lewis cells + 30 µmol/L allicin + 5 µg/mL cisplatin + HIF-1a inhibitor 3 µg/mL).
Pathological Tissue Sampling
Three weeks after the intervention, mice were sacrificed, and lung tissues were taken out. The lung tumor tissues of the mice were peeled off, weighed, and the tumor inhibition was calculated as [1 – (intervention group/model group) tumor weight].
MTT Detection of Lewis Cell Proliferation Rate
Each group of cells was seeded in a well plate and cultured for 2 days, and then 20 µL of MTT solution was poured into it. Incubate at room temperature for 4 h, discard the supernatant, add dimethyl sulfone solution to react, and finally detect the density value with a microplate reader, and repeat the measurement three times.
Flow Cytometry Detection of Lewis Cell Apoptosis
After trypsin digestion, cells were transfer to the Eppendorf (EP) tube, and start centrifugation, 1PBS solution is fully washed, 5 min × three times, after the washing is completed, give an appropriate amount of 75% ethanol solution for fixation, Overnight, add an appropriate amount of RNase solution and start incubation. Half an hour later, add propidium iodide (PI) staining solution, culture in a dark environment, and use a FACSCalibur flow cytometer for detection.
Transwell Detection of Lewis Cell Invasion and Migration
Culture medium: Matrigel gel ratio 1:5, dry, density 3 × 105 cells/mL inoculate upper chamber, a lower chamber with serum culture solution, culture for 24 h, washed with PBS, fixed with formaldehyde, permeabilized with methanol, dry, take pictures, and record.
Western Blot
Cells were lysed to take supernatant, bicinchoninic acid (BCA) was used to measure protein concentration, protein: loading buffer = 1:4 was added, protein samples were loaded, electrophoresed, transferred to a membrane, followed by blocking, washing, primary antibody incubated overnight, and then secondary antibody was incubated for 2 h, fully washed and then exposed.
Real-time Polymerase Chain Reaction
Ribonucleic acid (RNA) was extracted, followed by complementary deoxyribonucleic acid (cDNA) synthesis, which was used for qRT-PCR [the internal reference was glyceraldehyde 3-phosphate dehydrogenase (GAPDH)]. Gene levels were estimated using the 2−∆∆Ct method. Table 1 lists the primers and the sequence of the primers.
Primers and Primer Sequences.

Statistical Analysis
SPSS26.0 and GraphPad Prism were used for statistical analysis, and the calculated data were displayed as Mean ± SD and assessed by F test and LSD. p < .05 refers to a difference.
Results
Allicin Combined with Cisplatin Can Enhance Tumor Suppression Effect
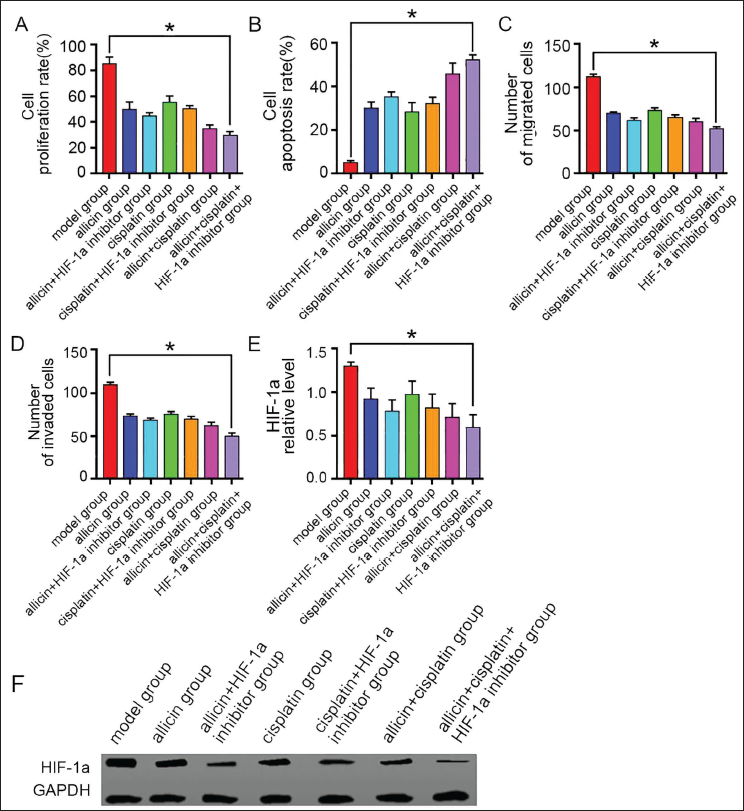
The lung cancer rat model was successfully established with a success rate of 97.5%. After intervention with allicin and cisplatin, the proliferation rate, invasion, and migration number of lung cancer Lewis cells in the allicin group and cisplatin group were significantly lower than in the model group, and the apoptosis rate was higher (Figure 3) (p < .05). In further experiments, it was found that allicin combined with cisplatin had a more significant effect on promoting apoptosis and inhibiting activities of lung cancer Lewis cells (vs. allicin group, cisplatin group) (Figure 3) (p < .05), which shows that allicin combined with cisplatin can enhance the tumor-suppressive effect.
The Growth, Metastasis and Hypoxia-inducible Factor 1a (HIF-1a) Expression Level of Mouse Lung Cancer Lewis Cells in Each Group (n = 7) (A) Quantification Diagram of Cell Proliferation Ability Detected by Methyl Thiazolyl Tetrazole (MTT) Assay; (B) Quantification Diagram of Cell Apoptosis Detected by Flow Cytometry; (C, D) Quantification Diagram of Cell Invasion and Migration Detected by Transwell; (E) Real-time Polymerase Chain Reaction (PCR) Experimental Detection of HIF-1a Expression; and (F) Western Blot Detection of HIF-1a Expression, Compared with the Model Group, *p < .05.
Allicin Can Inhibit HIF-1a Expression
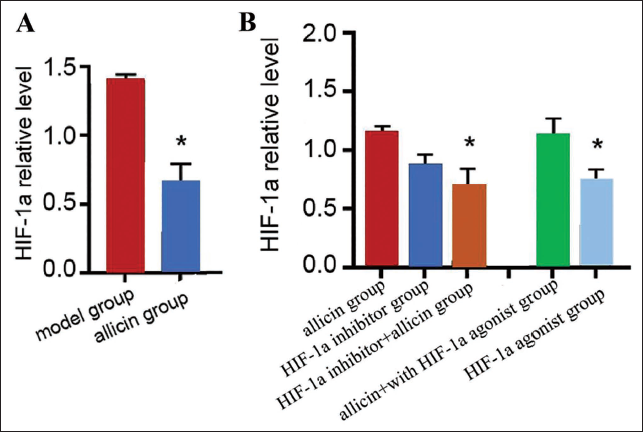
Detection of HIF-1a level by polymerase chain reaction (PCR) found that its level in the allicin group was lower (vs. the model group) (p < .05) (Figure 4A), suggesting that allicin may have an inhibitory effect on the expression of HIF-1a. In order to find out this issue, an HIF-1a inhibitor was applied under the condition of allicin intervention, and the level of HIF-1a was found to be decreased (vs. the allicin group, HIF-1a inhibitor group). The level of HIF-1a in allicin combined with the HIF-1a agonist group increased (vs. the allicin group), but it was lower than HIF-1a agonist group, p < .05, which indicated that allicin had an inhibitory effect on HIF-1a (Figure 4B).
Allicin Can Inhibit Hypoxia-inducible Factor 1a (HIF-1a) Expression (A, B) Real-time Polymerase Chain Reaction (PCR) Experimental Detection of HIF-1a Expression; Compared with the Model Group, *p < .05.
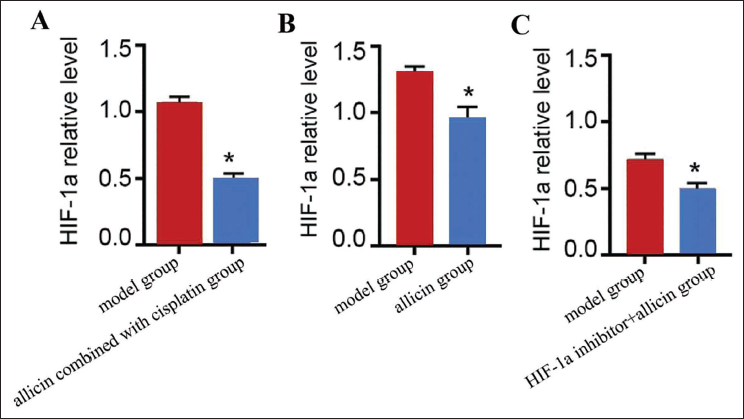
Allicin Combined with Cisplatin Inhibits the Growth and Metastasis of Lewis Cells in Lung Cancer Mice by Inhibiting the Expression of HIF-1a
This study confirmed that allicin has an effect on inhibiting HIF-1a, and the use of HI under the intervention conditions of allicin combined with cisplatin F-1a inhibitor, HIF-1a expression decreased (vs. model group) (Figure 5B and C) (p < .05), allicin combined with cisplatin had a more inhibitory effect on the growth and metastasis of mouse lung cancer Lewis cells (Figure 5A), which fully demonstrated that allicin combined with cisplatin inhibited the growth and metastasis of mouse lung cancer Lewis cells by inhibiting the expression of HIF-1a.
Allicin Combined with Cisplatin Inhibits the Growth and Metastasis of Lewis Cells in Lung Cancer Mice by Inhibiting the Expression of Hypoxia-inducible Factor 1a (HIF-1a) (A–C) Real-time Polymerase Chain Reaction (PCR) Experimental Detection of HIF-1a Expression; Compared with the Model Group, *p < .05.
Discussion
In China, lung cancer incidence is showing a rising trend. The radiotherapy and chemotherapy drugs used by patients with advanced lung cancer can control the occurrence and development of tumors, but there are some side effects. The commonly used chemotherapy drug cisplatin has bone marrow. In addition, acquired drug resistance will also have a certain degree of impact on patient compliance and treatment efficacy (He et al., 2021). At present, more effective drugs for the treatment of lung cancer are being actively sought clinically. With the deepening of research, allicin, as an active substance with anti-tumor properties, has become an anti-tumor drug to improve the effect of tumor suppression.
In this study, a mouse model of lung cancer was successfully prepared. By measuring and calculating the lung tumor weight of each group of mice, our results showed that tumor inhibition rate was significantly increased in the allicin group and cisplatin group, and the allicin combined with the cisplatin group inhibited tumor growth. The tumor rate was the highest, which further illustrated that the combination of allicin and cisplatin can significantly enhance the anti-tumor effect. Further cell experiments found that the proliferation rate, migration, and invasion numbers of Lewis cells in lung cancer mice in allicin group were lower than in the model group, and apoptosis was higher, which further confirmed that allicin has an inhibitory effect on lung cancer. Several clinical trials and studies found that (Ossama et al., 2021), allicin can affect the activity of signaling pathways and then affect the progression of tumors. The mammalian target of rapamycin (mTOR) pathway is one of the signaling pathways regulated by allicin. Allicin can regulate the expression of mTOR, induce autophagy, and promote cell apoptosis. In addition, the results of an animal experiment also showed that (Pandey et al., 2020), allicin can inhibit the growth of tumor tissue by increasing the content of intracellular calcium ions and cyclic adenosine monophosphate. Sarvizadeh et al. also pointed out (Rosas-Gonzalez et al., 2020; Sarvizadeh et al., 2021) that allicin can inhibit the migration, invasion, and adhesion of lung adenocarcinoma cells. The above research content confirmed the speculation of this study that allicin inhibits lung cancer. Combination medication has become a common method to reduce drug side effects and improve therapeutic effects in the clinic. Traditional Chinese medicine and its extracts are often used in combination with anti-neoplastic drugs because of their high safety and low side effects. At present, studies have pointed out (Rosas-Gonzalez et al., 2020; Sarvizadeh et al., 2021) that the combined application of allicin and anti-tumor drugs can enhance the anti-tumor efficacy of drugs. Schultz et al. (2020) pointed out that allicin can reduce the level of mobile-related protein-1 and, combined with 5-fluorouracil, can more effectively induce the apoptosis of liver cancer cells. Further experiments in this study found that the combination of allicin and cisplatin has a more obvious inhibitory effect on lung cancer. In this study, compared with other groups, allicin combined with the cisplatin group had the lowest proliferation rate and number of migration and invasion of Lewis cells in lung cancer mice, and the highest apoptosis rate of Lewis cells. According to the analysis, allicin and cisplatin can achieve a better tumor suppression effect, which is consistent with Sultan et al. (2020), the research results show that the application of allicin can reduce the resistance of human breast cancer cells to cisplatin and improve the anti-tumor effect. On the one hand, when allicin is combined with cisplatin, the effect of down-regulating cyclin-dependent kinase 2 (CDK2), BCL2-associated X (Bax), and other proteins are more significant, and at the same time, the ratio of Bax/B-cell lymphoma 2 (Bcl-2) is significantly increased, so the anti-tumor effect is more significant than that of cisplatin alone. On the other hand, while cisplatin is anti-tumor, it may lead to a decrease in the level of antioxidant substances in the patient’s body, thereby reducing organ damage. Since allicin has antioxidant activity and can remove free radicals in the body, when combined with cisplatin, allicin can reduce the side effects caused by cisplatin. In addition, this study found that compared with the model group, HIF-1a levels after intervention were lower. As a key transcription factor of hypoxia response, HIF-1a can promote the proliferation of tumors, increase the activities of tumor cells, and promote tumors. It is highly expressed in various malignant tumors. Tigu et al. (2020) pointed out that the lower level of HIF-1a indicates that the prognosis of breast cancer patients is better. The study by Wu et al. (2020) also showed that HIF-1a can be used as a risk factor for the prognosis of liver cancer, and the prognosis and survival of poorly differentiated HCC patients with high HIF-1a expression are worse. The expression of HIF-1a in lung cancer is also of great significance. A previous study found (Xiang et al., 2018) that the content of HIF-1a in non-small cell lung cancer is higher. Zhang and Yang (2019) also proposed HIF-1a as a predictor of the prognosis of lung cancer patients. Allicin can inhibit VEGF expression in cancer, which also affects the activities of tumor cells, and inhibition of HIF-1a will also reduce VEGF level, so we speculate the mechanism of allicin’s inhibition of proliferation and induction of apoptosis in lung cancer A549 cells may be related to its inhibition of HIF-1a expression. In order to assess the effect of allicin on HIF-1a, we administered HIF-1a agonists and HIF-1a inhibitors on the basis of allicin. We showed lower HIF-1a level in the allicin group than allicin combined with HIF-1a agonist group, and it was lowest after HIF-1a inhibitor combined with allicin treatment, which fully proved the inhibitory effect of allicin on HIF-1a. Further results showed that after using HIF-1a inhibitors, the proliferation inhibition and apoptosis induction of mouse Lewis cells in each group were significantly enhanced. The tumor inhibitory effect of HIF-1a inhibitors applied on the basis of platinum is most significant, which suggests that allicin combined with cisplatin can inhibit tumors by inhibiting HIF-1a.
Conclusion
In summary, after the intervention of allicin combined with cisplatin in lung cancer, it can inhibit the growth and metastasis of Lewis cells by inhibiting the expression of HIF-1a and has an inhibitory effect on the malignant expression shape of lung cancer cells. It can provide a new direction for lung cancer treatment. However, this study also has some shortcomings. The molecular mechanism between the inhibition of HIF-1a and tumors needs further study. In addition, how allicin mediates the expression of HIF-1a to inhibit Lewis cell growth in lung cancer mice still needs a lot of research to be confirmed. Whether allicin combined with cisplatin may affect cell growth and metastasis through other channels is worthy of further exploration.
Footnotes
Abbreviations
AIF: Apoptosis-inducing factor; Bax: BCL2-associated X; BCA: Bicinchoninic acid; Bcl-2: B-cell lymphoma 2; EP: Eppendorf; cDNA: Complementary deoxyribonucleic acid; CDK2: Cyclin-dependent kinase 2; DMEM: Dulbecco’s modified Eagle medium; DKK1: Dickkopf WNT signaling pathway inhibitor 1; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; GREM1: Gremlin 1; HIF-1a: Hypoxia-inducible factor 1a; IL-8: Interleukin 8; MDR1: Multidrug resistance protein 1; mTOR: Mammalian target of rapamycin; MTT: Methyl thiazolyl tetrazole; NF-κB: Nuclear factor kappa B; PARP-1: Poly (ADP-ribose) polymerase 1; PBS: Phosphate buffered saline; PCR: Polymerase chain reaction; PI: Propidium iodide; RNA: Ribonucleic acid; ROS: Reactive oxygen species; THBS2: Thrombospondin 2; TMEM16A: Transmembrane protein 16A; TNF-α: Tumor necrosis factor alpha; VEGF: Vascular endothelial growth factor; WNT5A: Wnt family member 5A.
Acknowledgments
We gratefully acknowledge the Hubei No. 3 People’s Hospital of Jianghan University for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethnic committee of Hubei No. 3 People’s Hospital of Jianghan University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
