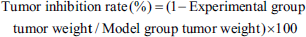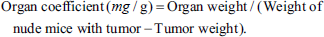Abstract
Background
Cordyceps sinensis is a unique and precious Traditional Chinese Medicine. The fermented mycelia of C. sinensis are obtained from the medicinal strains isolated from C. sinensis by liquid-submerged fermentation, which have similar pharmacological effects to natural C. sinensis. Polysaccharide is one of the main components of natural and artificial Cordyceps, which has anti-tumor effects and is currently used in the adjuvant treatment of various cancers. However, the anti-hepatoma efficacy of C. sinensis fermented mycelia polysaccharides and the underlying mechanism have not been reported, and the synergistic effect of combined treatment with cyclophosphamide (CTX) remains to be explored.
Objectives
Investigating the effects of the refined polysaccharide Cordyceps polysaccharide 1 (CP1) from C. sinensis mycelium on the PI3K/AKT/mTOR pathway in HepG2 hepatocellular carcinoma cells, and its detoxifying effect when combined with CTX.
Materials and Methods
The subcutaneous transplantation tumor model of human hepatocellular carcinoma HepG2 in nude mice was established. The tumor volume and weight were recorded, and the tumor inhibition rates were calculated. Enzyme-linked immunosorbent assay (ELISA) was used to examine the levels of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), interleukin-1β (IL-1β), and vascular endothelial growth factor (VEGF) secretions in cancerous tissues. Immunohistochemical methods were used to identify the expression rates of BAX and BCL-2 genes associated with apoptosis in cancerous tissues. Western blotting was used to identify the levels of protein expression in the phosphoinositide 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) signaling pathway. Mice had their peripheral blood collected for standard blood testing.
Results
Tumor suppression percentages for the CP1 high-, medium-, and low-dosage categories stood at 46.60%, 45.18%, and 36.84%, respectively. CP1 increased the secretion of TNF-α and decreased the secretion of VEGF, IL-6, and IL-1β in tumor tissues. There was a decrease in the expression of the anti-apoptotic protein B-cell lymphoma-2 (BCL-2) and an increase in the expression of the pro-apoptotic protein BCL2-associated X (BAX). There was a decrease in the phosphorylation levels of PI3K, AKT, and mTOR proteins within tumor tissues. Tumor suppression percentages for CTX, CP1 high, medium, and low doses in conjunction with CTX groups stood at 71.29%, 77.63%, 76.98%, and 75.24%, respectively. In groups receiving high, medium, and low doses of CP1 along with CTX, there was an elevation in the spleen coefficient and the count of leukocytes, neutrophils, and lymphocytes in peripheral blood.
Conclusion
CP1 can significantly and effectively inhibit the growth of hepatocellular carcinoma, and its mechanism may be related to the inhibition of protein expression of the PI3K/AKT/mTOR signaling pathway. The combination of CP1 and CTX can increase the anti-tumor effect and alleviate the bone marrow suppression of CTX.
Introduction
Hepatocellular carcinoma (HCC) frequently occurs as a primary malignant tumor in the liver. The occurrence rate of HCC is on the rise annually (Perugorria et al., 2019). HCC poses a significant risk to human health, with the 5-year survival rate for those suffering from advanced HCC being under 20% (Zhao & Gao, 2023). At present, cyclophosphamide (CTX) is the main chemotherapy drug for HCC, but it is often accompanied by side effects such as hepatotoxicity, hematotoxicity, and immunosuppression during the chemotherapy process, which seriously affects the tolerance and prognosis of patients (Briseño-Bugarín et al., 2021). Therefore, it is urgent to find anti-hepatoma drugs with high efficiency and low toxicity.
Studies have found that many traditional Chinese medicine polysaccharides combined with chemotherapy drugs can play a role in enhancing efficacy and reducing toxicity. Cordyceps sinensis is a precious traditional Chinese medicine unique to China. The pharmacological effects of C. sinensis include anti-oxidation, anti-inflammation, regulating immune balance, and preventing metabolic diseases (Wang, 2021). The fermented mycelia of C. sinensis are obtained from the medicinal strains isolated from C. sinensis by liquid-submerged fermentation, which have similar pharmacological effects to natural C. sinensis (Qian et al., 2021). Polysaccharide is one of the main components of natural and artificial Cordyceps. Studies have shown that Cordyceps polysaccharide can promote autophagy and apoptosis by increasing AMPKα expression and ULK1 phosphorylation and significantly inhibit the proliferation of colon cancer cell HCT116 (Qi et al., 2020). However, so far, the anti-hepatoma efficacy of C. sinensis fermented mycelia polysaccharides (CP1) and its related mechanism have not been reported, and the toxicity-reducing and synergistic effect of CP1 combined with CTX remain to be explored.
The phosphoinositide 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway is a traditional intracellular signal route significantly influencing tumor formation and emergence (Heng & Shunxiang, 2024). Studies have shown that the PI3K/AKT/mTOR pathway is activated in HCC tissues, and blocking the excessive activation of the PI3K/AKT/mTOR pathway may be a new approach for the treatment of HCC (Luo et al., 2024). The purpose of this study is to explore the effect of CP1 on HCC by regulating the PI3K/AKT/mTOR signaling pathway and the synergistic effect of CP1 combined with CTX in vivo in order to provide a research basis for its application as an anti-tumor adjuvant drug.
Materials and Methods
Material and Reagents
BALB/c nude mice were produced in Hangzhou Medical College (Animal Center), 4–5 weeks old, male, certificate number 20221114Abzz0100018804.
Human HCC cell line HepG2 was purchased from Wuhan Punosai Life Science and Technology Co., Ltd. (Wuhan, China).
The crude extract of C. sinensis mycelium polysaccharide was prepared in the laboratory. Cyclophosphamide injection (200 mg/branch) was purchased from Jiangsu Hengrui Pharmaceutical Co., Ltd. (Jiangsu, China). High glucose Dulbecco’s Modified Eagle Medium (DMEM) medium, fetal bovine serum, penicillin, and streptomycin were purchased from Gibco (Grand Island, NY, USA). Enzyme-linked immunosorbent assay (ELISA) kit was purchased from Hangzhou Jinhenuo Biotechnology Co., Ltd. (Hangzhou, China). Immunohistochemistry (IHC) kit was purchased from Yeasen Biotechnology Co., Ltd. (Shanghai, China). Phospho-mammalian target of rapamycin (p-mTOR) antibody (#5536), mTOR antibody (#2983), phospho-PI3K (p-PI3K) antibody (#4228), phosphoinositide 3-kinase (PI3K) antibody (#4257), phospho-AKT (p-AKT) antibody (#4060), AKT antibody (#9271), B-cell lymphoma-2 (BCL-2) antibody (#3498), BCL2-associated X (BAX) antibody (#2772), and β-actin antibody (#4970) were purchased from Cell Signaling Technology (Danvers, MA, USA).
EVOS M7000 fluorescence inverted microscope (Thermo Fisher Scientific, USA), CLM-170B-8-NF cell incubator (ESCO, USA), multi-function microplate reader (Tecan Spark, Switzerland), Amersham ImageQuant™ 500 chemiluminescence developer (Cytiva, USA), MEK-7222K automatic blood cell analyzer (Japan Photoelectric Industry Co., Ltd., Japan).
Separation of Refined Polysaccharides
According to literature, the crude polysaccharide extract of C. sinensis mycelium prepared by the research group (Zhuge et al., 2016) was treated by a DEAE Fast Flow ion exchange chromatography column (Wang, 2019). The eluent was 0.6 mmol/L NaCl solution, the flow rate was 1 mL/min, and 2 mL/tube was collected by the automatic collector. The polysaccharide content of each tube was determined by sulfuric acid-phenol method. There were two obvious elution peaks, CP1 and CP2, which were combined with CP1 and CP2, respectively. The polysaccharide content in each tube was determined using the sulfuric acid-phenol method. It was found that there were two distinct elution peaks, CP1 and CP2. The fractions corresponding to CP1 and CP2 were combined separately. The combined fractions were dialyzed using a 7000 D dialysis bag to remove salts, followed by freeze-drying. The polysaccharide content was then measured using the sulfuric acid-phenol method. The polysaccharide content of CP1 was 84.48%, and that of CP2 was 55.83%. The preliminary experiment showed that CP2 had no anti-tumor effect and CP1 had an obvious anti-tumor effect. CP1 was taken for subsequent experiments.
Cell Lines and Culture Conditions
Throughout the experiment, the HepG2 human HCC cell line, in its logarithmic growth stage, was gathered and modified to a concentration of 1.1 × 107 cells/mL for the cell suspension. Cultivation of HepG2 cells took place in DMEM medium, enriched with 10% fetal bovine serum, 1% streptomycin and penicillin, under conditions of 5% CO2, 37°C, and 95% relative humidity.
Establishment of HepG2 Xenograft Tumor Model in Nude Mice
Two separate experiments were set up to study the anti-HCC effect of CP1 and the synergistic effect of CP1 combined with CTX. After HepG2 cells were expanded to a sufficient number, the original medium was discarded, and the adherent cells were digested with trypsin. After centrifugation, the cells were resuspended in a serum-free medium, and the cell concentration was adjusted to 1.1 × 107 cells/mL. 0.2 mL cells were subcutaneously inoculated in the armpit of nude mice, and the tumor volume was increased to about 100 mm3. Randomly grouped as follows:
Single administration test: Nude mice were divided into normal group, model group, CTX group (50 mg/kg), and CP1 high-, medium-, and low-dose (160, 80, and 40 mg/kg) group, with six rats in each group. CP1 was given by tail vein injection every other day, and CTX was given by tail vein injection twice a week for a total of 13 days.
Combined administration test: The nude mice were divided into normal group, model group, CTX group (100 mg/kg), CTX and CP1 high-, medium-, and low-dose (160, 80, and 40 mg/kg) combined group, with six rats in each group. CP1 was given by tail vein injection every other day, and CTX was given by intraperitoneal injection twice a week for 13 days.
General Observation Index, Tumor Inhibition Rate Calculation, and Specimen Processing
After the drug intervention, the general condition of nude mice was observed, and the body weight of nude mice and the length and diameter of the tumor were recorded every 3 days. When the tumor volume of the model group reached about 2,000 mm3, the peripheral blood of mice was taken, anticoagulant treatment was performed, and blood routine was measured by an automatic blood cell analyzer. The tumor, liver, kidney, and spleen tissues were weighed, and the tumor inhibition rate and organ coefficient were calculated. The calculation formula can be referred to:
ELISA Analysis
The tumor tissue stored at –80°C was taken out, and an appropriate amount of tumor tissue was weighed. The tumor tissue and phosphate-buffered saline (PBS) were added to the homogenate tube. After homogenization, the supernatant was centrifuged, and the protein concentration was determined with a BCA kit. Tumor necrosis factor-α (TNF-α), angiogenesis factor (vascular endothelial growth factor (VEGF)), interleukin-6 (IL-6), and interleukin-1β (IL-1β) in the tumor tissue were measured according to the kit instructions.
Immunohistochemistry
The tumor tissue fixed in 4% paraformaldehyde reagent was embedded in paraffin to make paraffin sections, followed by dewaxing, hydration, and antigen repair. The next steps followed the instructions of the IHC kit. The sections were dehydrated with gradient alcohol, finally mounted, photographed under a microscope, and the protein expression position of BAX and BCL-2 was observed. The average optical density value was analyzed by using ImageJ.
Immunoblotting
20 mg of tumor tissue was weighed into a homogenizer tube. Subsequently, RIPA lysate was added, and the mixture was homogenized at 4,500 rpm for 30 s. The homogenate was allowed to stand at 4°C for 20 min to ensure complete lysis. Following this, low-temperature centrifugation was performed at 4°C and 15,000 rpm for 10 min to separate the components. The supernatant was taken, and the protein concentration was determined by a BCA kit. After adjusting to a uniform concentration, the same volume of equal protein was added to the corresponding concentration of sodium dodecyl sulfate (SDS)–polyacrylamide gel for electrophoresis and transferred to a 0.45 µm polyvinylidene fluoride (PVDF) membrane. The 5% skim milk was blocked at room temperature for 1 h, and the bands were placed in a primary antibody (1:1,000) solution overnight at 4°C. Protein bands were incubated with horseradish peroxidase (1:10,000) for 1 h at 25°C. ECL development and ImageJ image processing system were used to analyze the gray value of Western blot bands.
Statistical Analysis
Data analysis was conducted using GraphPad Prism software, with outcomes presented as the average ± standard deviation (x ± s). Comparisons between the two groups were conducted using the t-test, while one-way analysis of variance (ANOVA) was employed for contrasting various groups. p < .05 was deemed to hold statistical significance.
Results
Effects of CP1 on HepG2 Transplanted Tumor in Nude Mice
For investigating CP1’s tumor-fighting effectiveness, 0.9% NaCl saline solution was administered to the tail vein of the test cohort, while polysaccharides at dosages of 160, 80, and 40 mg/kg were given bi-daily to the tail vein in each CP1 group. The CTX group was injected with CTX 50 mg/kg twice a week, and the volume of administration was 10 mL/kg. Starting the 7th day of treatment, the tumor expansion rate in mice receiving high, medium, and low doses markedly decelerated in comparison to the control group. By the 13th day after administration, the variance reached statistical significance (p < .001) (Figure 1A). After 13 days of tail vein injection, the size of the tumor mass in each group was photographed (Figure 1B). Compared with the model group, the tumor weight of CP1 high-, medium-, and low-dose groups was greatly reduced, and the differences were statistically significant (***p < .001) (Figure 1C). The tumor inhibition rates of CP1 high-, medium-, and low-dose groups were 46.60%, 45.18%, and 36.84% (Figure 1D), respectively. The effect of tumor suppression varied with dosage, and the rate of tumor inhibition in the high-, medium-, and low-dosage CP1 groups surpassed that in the CTX group (32.68%) (Figure 1D).
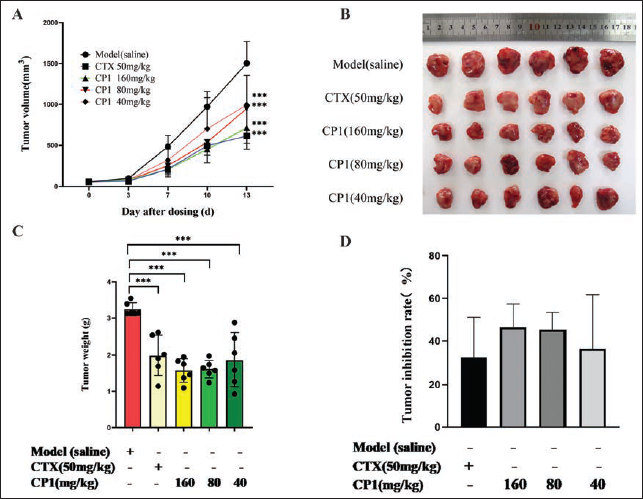
(A) During the 13-day Administration Period, the Tumor Growth Volume Change Line Chart. After 7 Days of Administration, the Tumor Volume Growth Rate of Each Polysaccharide Administration Group was Significantly Reduced.
The Effect of CP1 on the Expression of TNF-α, VEGF, IL-6, and IL-1β in Tumor Tissues of Mice
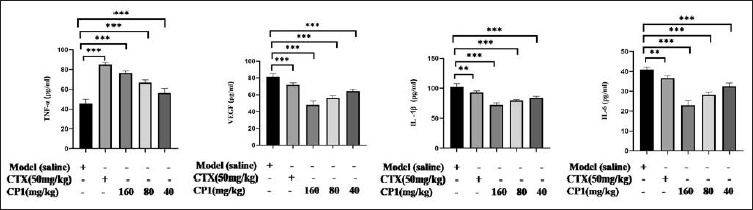
The levels of IL-6, IL-1β, TNF-α, and angiogenic factor VEGF were measured by us. Compared with the model group, CP1 markedly raised the levels of TNF-α (p < .001), while reducing the amounts of VEGF, IL-6, and IL-1β (p < .001) (Figure 2).
The Secretion Levels of Interleukin-1β (IL-1β), Interleukin-6 (IL-6) and Vascular Endothelial Growth Factor (VEGF) in Cordyceps Polysaccharide 1 (CP1) 160, 80, and 40 mg/kg Groups were Significantly Decreased, and the Content of Tumor Necrosis Factor-α (TNF-α) in CP1 Groups was Significantly Increased in a Dose-dependent Manner, Indicating that CP1 can Inhibit the Angiogenesis of Tumor Tissue, Promote the Apoptosis of Tumor Cells, Inhibit the Formation of Tumor, and Reduce Inflammation In Vivo (Compared with the Model Group, *p < .05, **p < .01, ***p < .001, n = 6).
Effect of CP1 on the Apoptosis-related Proteins in Tumor Tissues
Additionally, we observed the presence of proteins BAX and BCL-2, which are linked to apoptosis, in cancerous tissues to investigate their apoptotic processes. The expression levels of apoptosis-related proteins BAX and BCL-2 in tumor tissues were detected by IHC (Figure 3A). There was an increase in the expression of the pro-apoptotic protein BAX, while the expression of the anti-apoptotic protein BCL-2 was reduced. BCL-2/BAX reflects the trend of apoptosis. With the increase of polysaccharide concentration, the ratio of BCL-2/BAX decreased and promoted apoptosis, showing a dose-dependent manner (Figure 3B).
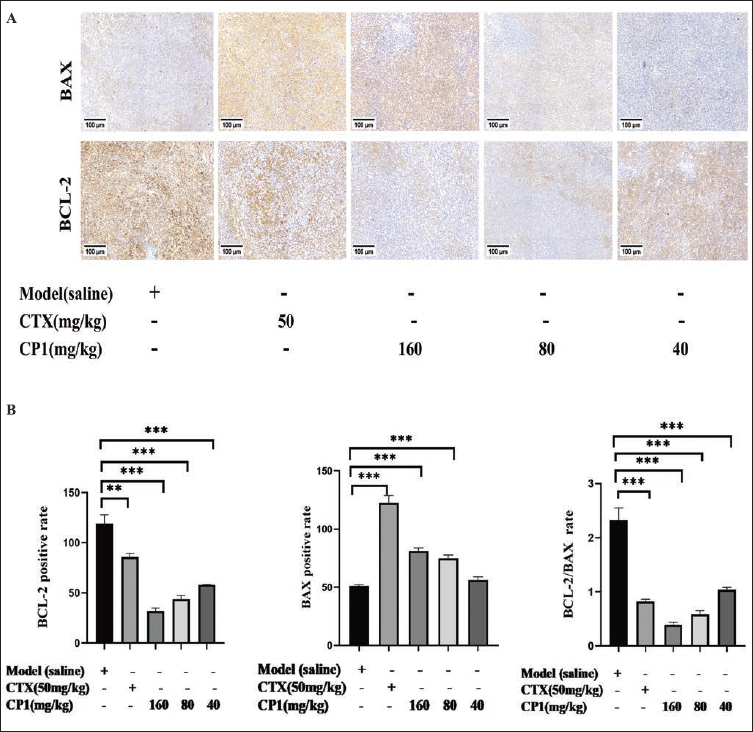
(A) Microscopic Imaging was Conducted on the Protein Expression Areas of BCL2-associated X (BAX) and B-cell Lymphoma-2 (BCL-2), Using a Scale Bar of 100 µm. The Positive Expression was Yellowish Brown and the Negative Expression was Blue-purple.
Effects of CP1 on the Expression of PI3K/AKT/mTOR Protein in Tumor Tissues of Mice
The expression of protein phosphorylation in the PI3K/AKT/mTOR signaling pathway was identified using Western blotting (Figure 4A). Compared with the model group, the protein expression of mTOR, p-mTOR, and p-PI3K decreased in the CTX group (**p < .01, ***p < .001, ***p < .001). The phosphorylation of PI3K and AKT protein in tumor tissues of CP1 high-, medium-, and low-dose groups decreased (*p < .05, **p < .01, ***p < .001) in a dose-dependent manner. The phosphorylation of mTOR protein in the CP1 high-dose group was markedly decreased (p < .001) (Figure 4B).
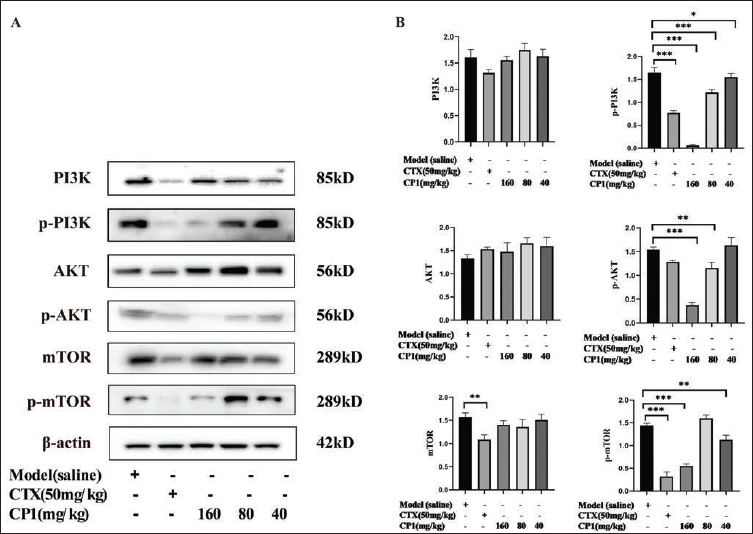
(A) Protein was Extracted from Tumor Tissue and Detected by Western Blotting to Obtain Protein Expression Bands.
Effects of CP1 Combined with CTX on the Tumor Growth of HCC in Nude Mice
In order to further verify the synergistic effect of CP1 combined with CTX, the model group was injected with 0.9% NaCl saline through the tail vein; the CP1 administration group was injected through the tail vein once every other day; the CTX group was intraperitoneally injected with CTX twice a week, with a volume of 10 mL/kg. After 3 days of administration, compared with the model group, the tumor growth rate of the CTX group was obviously slowed down, and tumor volume decreased significantly. During the 13-day administration period, compared with the CTX group, the tumor growth rate of the CTX with CP1 different dose combined group was further slowed down, and the tumor volume was further reduced. The difference between the CP1 high-dose combined group was statistically significant (p < .05) (Figure 5A). Anatomy after the end of administration, compared with the model group, the tumor weight of the CTX group was vastly reduced (p < .001) (Figure 5C). Compared with the CTX group, the tumor weight of the CTX and CP1 high-, medium-, and low-dose combination groups was further reduced. The effect was dose-dependent, and the difference in the CP1 high-dose combination group had statistical significance (p < .05) (Figure 5C). The tumor inhibition rates of CP1 high-, medium-, and low-dose combination groups were 77.63%, 76.98%, and 75.24% (Figure 5D), which were higher than that of the CTX group (71.29%) (Figure 5D).
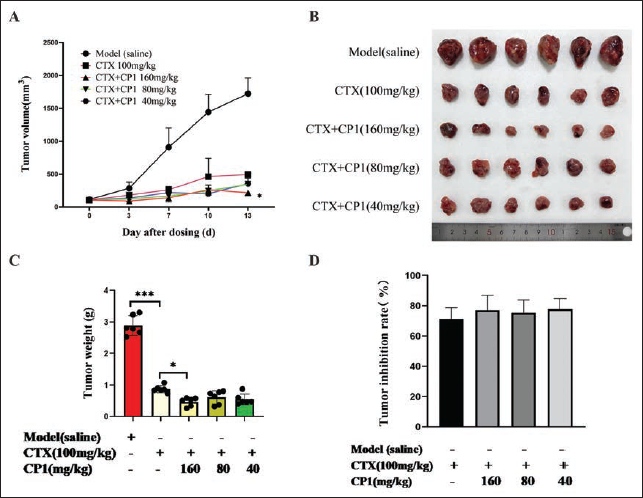
(A) During the 13-day Administration Period, The Tumor Growth Volume Change Line Chart.
Effects of CP1 Combined with CTX on the Main Organ Coefficients of HepG2 Tumor-bearing Mice
Objective to investigate the effect of CTX toxic dose on the liver, kidney, and spleen, and the attenuation and synergistic effect of CTX after the combined application of CP1, we measured the organ coefficients of the liver, kidney, and spleen. The results in Table 1 show that, contrasted with the standard group, the hepatic, spleen, and renal functions in the test group drastically escalated (p < .01). In relation to the model group, there was a reduction in the liver and spleen coefficients in the CTX group (p < .01). Relative to the CTX group, a significant surge was observed in the liver and spleen coefficients for CP1 high, medium, and low doses when combined with the CTX group (p < .01), with the increase being dose-responsive. It may be due to the inflammatory reaction of the tumor-bearing mice in the model group, which leads to the inflammatory enlargement of the main organs, or due to the excessive tumor volume and mass. The weight of the tumor-bearing mice after tumor resection is relatively light, which makes the coefficients of the liver, spleen, kidney, and other organs larger. Compared with the model control group, the weight and organ coefficient of the spleen in the CTX group were noticeably reduced, which may be due to the fact that CTX is an immunosuppressant, which reduces the spleen and affects the normal immune function of the spleen. Compared with the CTX group, the spleen weight and organ coefficient of CP1 high, medium, and low doses combined with the CTX group were significantly increased, indicating that CP1 combined with CTX could alleviate the inhibitory effect of CTX on the spleen.
Effects of Cordyceps Polysaccharide 1 (CP1) Combined with Cyclophosphamide (CTX) on the Main Organ Coefficients of the Liver, Kidney, and Spleen in HepG2 Tumor-bearing Mice (n = 6).
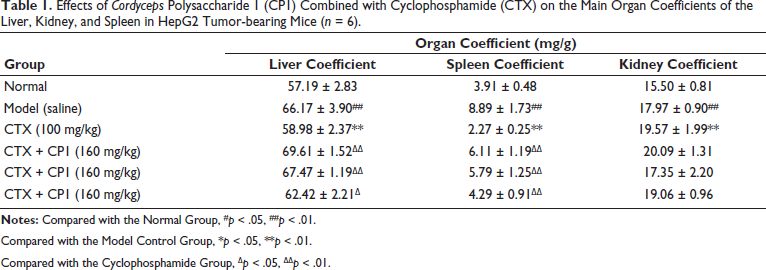
Compared with the Model Control Group, *p < .05, **p < .01.
Compared with the Cyclophosphamide Group, ∇p < .05, ∇∇p < .01.
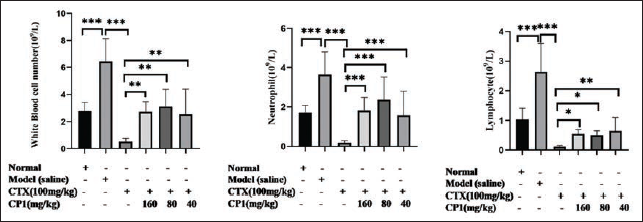
Effect of CP1 Combined with CTX on Blood Routine Indexes of Nude Mice
CTX exhibits toxicity that inhibits bone marrow activity, impacting the count of white blood cells in peripheral blood cells. The number of white blood cells in blood routine, including the number of neutrophils and lymphocytes, was detected to explore whether the combined application of CP1 can alleviate the bone marrow suppression of CTX. Blood routine examination of peripheral blood showed that compared with the normal group, the number of white blood cells in the model group was increased markedly (p < .001), and neutrophils and lymphocytes were mainly increased (p < .001 and p < .001). Compared with the model group, the CTX group considerably reduced the number of white blood cells (p < .001), especially the number of neutrophils and lymphocytes (p < .001 and p < .001). Compared with the CTX group, the number of white blood cells (including neutrophils and lymphocytes) in CTX combined with CP1 high-, medium-, and low-dose groups was dramatically increased (Figure 6).
The Impact of Cordyceps Polysaccharide 1 (CP1) in Conjunction with Cyclophosphamide (CTX) on the Blood Standard Indices of Mice with HepG2 Tumors was Measured by the Count of White Blood Cells, Encompassing Neutrophils and Lymphocytes (*p < .05, **p < .01, ***p < .001, n = 6).
Discussion
The etiology of HCC is complex, and the prognosis is extremely poor. Most patients are diagnosed at an advanced stage, thus losing the best time for radical surgical resection (Chuang et al., 2024). CTX is a classic clinical chemotherapy drug, but it can cause bone marrow suppression, hematotoxicity, gastrointestinal reactions, and other serious adverse reactions (Voelcker, 2020).
A lot of research findings have discovered that many natural polysaccharides can enhance the body’s immunity, inhibit the malignant growth, proliferation, and invasive migration of tumor cells, and antagonize or partially antagonize the toxic side effects of chemotherapy drugs (Sohretoglu & Huang, 2018). They are considered to be ideal adjuvants for reducing toxicity and enhancing the efficacy of chemotherapy drugs (Liu et al., 2024). For example, Phellinus igniarius polysaccharides and CTX synergistically inhibited the growth of liver cancer tumors and alleviated the immune function decline caused by CTX (Zhao et al., 2020). Polygonatum cyrtonema Hua polysaccharides can enhance the inhibitory effect of CTX on H22 hepatoma cells, improve CTX-induced immunosuppression, and antagonize liver and kidney injury induced by CTX (Peng, 2021). C. sinensis mycelium polysaccharide is extracted from Cordyceps fungus mycelium or mycelium fermentation broth. Our previous studies have found that oral administration of crude polysaccharides from mycelium is basically ineffective, but intraperitoneal injection can regulate the immune status of the body and inhibit tumor growth in mice (Zhuge et al., 2016). However, the underlying mechanism of its anti-tumor effect and the synergistic effect of its combination with chemotherapy drugs are still unclear.
In this study, a CP1 separate administration test was established to further explore the efficacy and mechanism of CP1 against HepG2 liver cancer. The results reveal that CP1 could effectively inhibit the malignant proliferation of tumor cells, reduce the expression of anti-apoptotic protein BCL-2 in solid tumors, increase the expression of pro-apoptotic protein BAX in solid tumors, and to a large extent, reduce the ratio of BCL-2/BAX, suggesting that CP1 could promote tumor cell apoptosis.
TNF-α is a cytokine mainly produced by macrophages and monocytes, which can promote the killing of tumor cells by immune cells such as T cells and can act on vascular endothelial cells to cause vascular injury, resulting in local blood flow occlusion of tumor tissue and bleeding, hypoxia, and necrosis (Huang et al., 2024). VEGF is an effective mediator of angiogenesis in the body. It begins to be produced in large quantities during the transformation of tumor cell clusters into solid tumors. It can be used as a tumor marker for tumor screening index (Mukherjee et al., 2024). IL-6 and IL-1β are cellular immune factors synthesized and released by immune cells, which are related to the body’s protective immune response (Ma et al., 2024). Studies have found that tumor-derived IL-1β and IL-6 can promote tumor growth and angiogenesis, and their expression levels are positively correlated with the malignancy of tumor phenotypes (Chen et al., 2024). In this study, we found that CP1 could increase the secretion level of TNF-α in mouse tumor tissues and reduce the secretion levels of VEGF, IL-6, and IL-1β, suggesting that CP1 can reduce the formation of tumor blood vessels, increase tumor cell necrosis, and inhibit tumor growth.
During tumor growth, the activation of the PI3K/AKT/mTOR signaling pathway promotes tumor cell growth, inhibits tumor cell apoptosis, and increases angiogenesis (Afrashteh et al., 2024). PI3K is phosphorylated under the stimulation of upstream signal factors. As a downstream effector of PI3K, p-PI3K can transfer AKT to the cell membrane for phosphorylation (Lin et al., 2024). Phosphorylated AKT can inhibit apoptosis and promote tumor cell differentiation and survival by regulating the expression of apoptosis-related proteins. p-AKT can induce phosphorylation of mTOR, and p-mTOR can regulate the cell cycle and promote cancer cell proliferation (Alzahrani, 2019). Therefore, targeting the PI3K/AKT/mTOR signaling pathway is a promising tumor treatment strategy (Cao et al., 2024). This study found that CP1 could reduce the phosphorylation of PI3K, AKT, and mTOR proteins in tumor tissues, suggesting that CP1 may play an anti-tumor role by regulating the PI3K/AKT/mTOR signaling pathway, promoting tumor cell apoptosis, and reducing tumor angiogenesis.
In order to study the attenuation and synergistic effect of CP1-assisted CTX, the administration test of CP1 combined with CTX was established. The results of this study showed that the tumor inhibition rate of the combined administration group was higher than that of the CTX group, indicating that the combined administration of CP1 could increase the efficacy of CTX in inhibiting tumor growth.
The calculation of the main organ coefficient can reflect the damage of the organ. The increase of the organ coefficient indicates congestion, edema, or hypertrophy of the organ. The decrease of organ coefficient indicates organ atrophy and other degenerative changes (Ying et al., 2023). Leukocytes can be divided into granulocytes, monocytes, and lymphocytes, which are produced by hematopoietic stem cells of bone marrow. The spleen is an important immune organ, where mature lymphocytes settle and has an immune response function (Pizzi et al., 2023). CTX is an immunosuppressant and has bone marrow suppression. After treatment with CTX, the spleen of mice was significantly reduced, the spleen coefficient was strongly reduced, and the number of white blood cells in peripheral blood, including neutrophils and lymphocytes, was significantly reduced, indicating that CTX produced bone marrow toxicity in mice, thereby destroying immune function. The spleen coefficient of mice combined with CP1 was freely increased, and the number of white blood cells in peripheral blood, including neutrophils and lymphocytes, was significantly increased, indicating that CP1 alleviated the myelosuppression of CTX and enhanced the immune function of the body. In summary, CP1 has a significant anti-HepG2 liver cancer effect in vivo, which can inhibit tumor angiogenesis and promote tumor cell apoptosis. The mechanism may be related to the PI3K/AKT/mTOR signaling pathway. Combined with CTX, it can increase the anti-tumor effect and alleviate the bone marrow suppression caused by CTX. This study provides a theoretical basis for the clinical application of CP1 as an adjuvant drug in chemotherapy.
Conclusion
In conclusion, our study found that C. sinensis mycelial polysaccharide CP1 has anti-hepatoma effect in vivo, which may be achieved by inhibiting the expression of the PI3K/AKT/mTOR signaling pathway, and CP1 combined with CTX can improve therapeutic efficacy and alleviate immune suppression caused by CTX in the treatment of HCC.
Footnotes
Abbreviations
BAX: BCL2-associated X; BCL-2: B-cell lymphoma-2; CP1: Cordyceps polysaccharide 1; CTX: Cyclophosphamide; HCC: Hepatocellular carcinoma; IL-1: Interleukin-1; IL-1β: Interleukin-1β; IL-6: Interleukin-6; mTOR: Mammalian target of rapamycin; p-AKT: Phospho-AKT; p-mTOR: Phospho-mammalian target of rapamycin; PI3K: Phosphoinositide 3-kinase; p-PI3K: Phospho-PI3K; TNF-α: Tumor necrosis factor-α; VEGF: Vascular endothelial growth factor.
Authors’ Contributions
Guochen Sui: Article writing, experimental operation, experimental design.
Zhuoran Zhang: Experimental operation, article polishing.
Xuejun Tian: Experimental operation, article polishing.
Yunping Miao: Experimental operation.
Min Cheng: Experimental operation.
Wei Pan: Experimental operation.
Gaoli Zheng: Corresponding author, experimental guidance, article polishing.
Xiaodi Ye: Corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal experimental processes were approved by the Ethics Committee of Hangzhou Medical College and conducted in strict accordance with the standard of the Guide for the Care and Use of Laboratory Animals published by the Ministry of Science and Technology of the People’s Republic of China in 2006.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hangzhou Medical College Institute Special Plan Project (YS2021005) and Zhejiang Chinese Medicine Science and Technology Project (2024ZF057).