Abstract
Background
Myocardial ischemia/reperfusion (I/R) injury refers to myocardial tissue damage caused by blood reperfusion after acute heart ischemia caused by coronary artery thrombosis and others. This process is closely related to inflammation and endoplasmic reticulum (ER) stress, and there is currently no ideal prevention means. In recent years, some traditional natural plant products with anti-inflammatory and anti-ER stress properties have been used to counteract myocardial I/R injury. Eupatilin (EPT), a pharmacologically active flavone derived from the Artemisia plant species, possesses significant anti-inflammatory and anti-ER stress activity. Still, its protective effect against myocardial I/R injury in vivo has not been revealed.
Objectives
This study aimed to evaluate EPT’s effect and potential mechanism against myocardial I/R injury.
Materials and Methods
The rat myocardial I/R injury model was prepared and treated with EPT. Then, the levels of myocardial injury markers and hematoxylin and eosin staining were used to evaluate the pathological damage of the myocardial tissue. Western blotting was used to detect the expression of key proteins in the potential signaling pathways.
Results
It was found that EPT could significantly reduce the levels of myocardial injury markers lactate dehydrogenase and creatine kinase, decrease the levels of inflammatory factors, and reduce apoptosis and pathological damage in myocardial tissue. In addition, the expression of key proteins in purinergic P2x7 receptor (P2X7R)/NOD-, LRR- and pyrin domain-containing protein 3 (NLRP3), Janus kinase 2 (JAK2)/signal transducer and activator of transcription 3 (STAT3)/suppressor of cytokine signaling 3 (SOCS3) pro-inflammatory pathways, and pancreatic endoplasmic reticulum eIF2alpha kinase (PERK/eIF2α)/activating transcription factor 4 (ATF4)/C/EBP homologous protein (CHOP) ER stress pathway was significantly downregulated in the EPT-treated group. Molecular docking simulations showed that this may be associated with the fact that EPT can bind to P2X7R, PERK, and other proteins in the aforementioned pathways.
Conclusion
This study demonstrated that EPT could attenuate myocardial I/R injury in rats, and the protective mechanism was related to the ability of EPT to bind to the initiating proteins of the P2X7R/NLRP3 pro-inflammatory pathway and the PERK/eIF2α/ATF4/CHOP ER stress pathway, which inhibited the inflammatory response and ER stress, thus reducing cardiomyocyte apoptosis. Our findings provide some valuable references for the future application of EPT in treating myocardial I/R injury.
Keywords
Introduction
With the improvement of living standards and the aging of the population, the incidence of coronary heart disease is on the rise (Syme & Guralnik, 2022), and acute myocardial infarction is its most serious clinical manifestation, which often causes sudden death (Bui & Waks, 2018). At present, the treatment of acute myocardial infarction of coronary heart disease is mainly through arteriovenous thrombolysis, percutaneous coronary angioplasty, coronary artery bypass grafting, coronary artery bypass grafting surgery, and cardiac surgery (Reddy et al., 2015), which can prevent myocardial death due to sustained ischemia and hypoxia, and reduce the infarct area. However, it has been found that as the blood vessels are reopened when the myocardium regains blood supply after a sustained period of ischemia, additional cell death occurs due to pH alterations, oxidative stress, cellular inflammation, endoplasmic reticulum (ER) stress, and other factors that cause further damage to myocardial tissues, a pathologic process known as myocardial ischemia/reperfusion (I/R) injury (Turer & Hill, 2010).
There is still no effective therapy for myocardial I/R injury (Hausenloy & Yellon, 2013), and additive cardioprotective interventions are advocated, including some mechanical and pharmacological approaches. Recent studies indicated that some traditional natural plant products showed obvious protective effects against myocardial I/R injury, benefitting from their excellent anti-inflammatory and anti-apoptotic properties, such as curcumin (Mokhtari-Zaer et al., 2019), polysaccharide fucoidan (Omata et al., 1997), quercetin (Zhang, Zhang et al., 2020), and ginsenosides (Wu et al., 2011). Eupatilin (EPT), a natural product extracted from Artemisia Linn. plant species (Nageen et al., 2018), has efficient anti-inflammatory (Choi et al., 2011) and anti-ER stress properties (Wang et al., 2024). It has shown a favorable potential to protect against I/R injury in other organs, such as the liver (Lee et al., 2016) and kidney (Jeong et al., 2015), but its protective effect on myocardial I/R injury has not been revealed. Therefore, the preliminary aim of this study is to verify the protective effect of EPT against myocardial I/R injury.
In addition, in order to reveal this potential role of EPT more clearly, this study also intends to continue to explore the underlying mechanisms in depth. Previous studies have shown that the activation of NOD-, LRR- and pyrin domain-containing protein 3 (NLRP3) inflammasome can exacerbate myocardial I/R injury (Wu et al., 2020), and targeting and inhibiting this inflammatory response is a potential target pathway to attenuate myocardial I/R injury (Ghafouri-Fard et al., 2022; Shen et al., 2022; Wang et al., 2020). Zhang, Huang et al. (2020) found that metformin could counteract myocardial I/R injury by inhibiting the NLRP3 pathway. Consistently, betulin (Yu et al., 2021) and luteolin (Zhang et al., 2017) showed similar mechanisms in counteracting myocardial I/R injury. This suggests that evaluating the regulatory role of EPT on the NLRP3 inflammatory pathway is a vital link to revealing its potential protective mechanism against myocardial I/R injury. In addition, ER stress is another important process that contributes to cardiomyocyte apoptosis and causes I/R injury; therefore, it is also one of the biological response processes that need to be targeted and inhibited when mitigating myocardial injury (Wu et al., 2016; Zhu & Zhou, 2021). Therefore, exploring the role of EPT in regulating the ER stress pathway will also be helpful in clarifying its protective effect against myocardial I/R injury.
Therefore, on the basis of the above previous studies and the above hypothesis, the present study was proposed to conduct the myocardial I/R injury rat models and treat them with EPT, subsequently evaluating the protective effect of EPT against myocardial I/R injury by detecting cardiomyocyte injury markers, myocardial histopathological changes, and cardiomyocyte apoptosis. Furthermore, the study also intended to reveal the potential mechanism of the protective effect of EPT against myocardial I/R injury by exploring its regulatory effect on the pro-inflammatory pathways, such as purinergic P2x7 receptor (P2X7R)/NLRP3, and ER stress pathway, such as pancreatic endoplasmic reticulum eIF2alpha kinase (PERK/eIF2α)/activating transcription factor 4 (ATF4)/C/EBP homologous protein (CHOP), in myocardial tissues, and by exploring the regulatory effects of EPT on ER stress signaling pathway such as PERK/eIF2α/ATF4/CHOP in myocardial tissues. In addition, in order to further explore the regulation mechanism of EPT on the above pathways, this study also used the molecular docking method to simulate the binding of EPT to key proteins in P2X7R/NLRP3 and PERK/eIF2α/ATF4/CHOP pathways; it will also contribute to revealing the protective mechanism of EPT on myocardial I/R injury.
Materials and Methods
Ethics Statement
The animal experiments were strictly adhered to the international regulations on the welfare of experimental animals and carried out under the approval of the Animal Care and Use Committee.
Materials and Reagents
EPT was obtained from Yuanye Biotechnology Company (Shanghai, China), No. B21568. Enzyme-linked immunosorbent assay (ELISA) kits used for the measurement of serum biochemical parameters (lactate dehydrogenase (LDH), creatine kinase (CK)-MB, tumor necrosis factor alpha (TNF-α), interleukin (IL)-1β, IL-6, monocyte chemoattractant protein-1 (MCP-1)) were obtained from Jiancheng Bioengineering Company (Nanjing, China). The antibodies against p-JAK2, Janus kinase 2 (JAK2), Caspase-1, p-PERK, p-eIF2α, CHOP, glycolytic glyceraldehyde-3-phosphate dehydrogenase (GAPDH) proteins were obtained from Cell Signaling Technology (Danvers, MA, USA); antibodies against suppressor of cytokine signaling 3 (SOCS3), P2X7R, NLRP3, IL-18, Bax, Bcl-2, GRP78, ATF4 proteins were obtained from Abcam Company (Cambridge, UK); antibodies against STAT3, IL-1β were obtained from BOSTER Biological Technology Co., Ltd. (Wuhan, China).
I/R Rat Model Preparation and EPT Treatment
Male Wistar rats (180–210 g) were kept in an animal house at 23 ± 2°C, with free access to food and water and 12 h of light exposure daily. Sixty rats were randomly divided into four groups: the sham operation group (Sham), the IR model group, the 10 mg/kg low-dose EPT-treated group (I/R + EPT 10), and the 20 mg/kg high-dose EPT-treated group (I/R + EPT 20), 15 each. EPT dissolved in 0.5% CMC-Na. Rats in the I/R + EPT 10 and I/R + EPT 20 groups were gavaged 10 mg/kg or 20 mg/kg of EPT daily for 1 week, while rats in the I/R group and Sham group were gavaged with an equal amount of 0.5% CMC-Na solution.
Then the I/R rat model was made according to the previous research, 1 h after the last administration (Lv et al., 2021). Briefly, rats were anesthetized with 2.5% sodium pentobarbital. Then all rats were fixed in the supine position. Rats in the I/R, I/R + EPT 10, and I/R + EPT 20 groups underwent I/R surgery: the left anterior descending coronary artery (LAD) was ligated with a 6-0 silk for 30 min, and ischemia was confirmed by discoloration of the cardiac surface and elevation of the ST segments recorded by electrocardiogram (ECG). The LAD ligature was then loosened and reperfused for 180 min, and reperfusion was confirmed by reddening of the cardiac surface and ST lowering recorded by ECG, and the I/R rat model was completed. Rats in the Sham group received the same surgical procedure treatment, but the LAD was threaded but not ligated. Then, blood was collected from the abdominal aorta of each group, rats were sacrificed by cervical dislocation, and heart tissue was collected.
Measurement of Serum Biochemical Parameters
The blood samples collected in section “I/R Rat Model Preparation and EPT Treatment” were centrifuged, and the levels of serum cardiac enzyme (LDH, CK-MB) and inflammatory cytokines (TNF-α, IL-1β, IL-6, MCP-1) were measured according to the requirements of the ELISA kits for LDH, CK-MB, TNF-α, IL-1β, IL-6, and MCP-1, respectively.
Histopathological Analysis of Myocardial Tissue
Hematoxylin and eosin (H&E) staining was used to analyze the histopathological changes in the myocardial tissue. The left ventricular region of the heart tissue obtained in section “I/R Rat Model Preparation and EPT Treatment” was fixed in 4% paraformaldehyde for 24 h. The tissue was sequentially dehydrated, transparent, and embedded, and then 5 µM sequential slices were cut using a continuous slicer. The slices were sequentially placed into hematoxylin staining solution and eosin staining solution for staining, and the pathological changes of myocardial tissues were observed with an inverted fluorescence microscope.
Measurement of the Cell Apoptosis in Myocardial Tissue
The terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay was used to measure the apoptosis rate of cardiomyocytes. Briefly, after myocardial tissue sections were deparaffinized, they were incubated in the working solution containing proteinase K, followed by membrane rupture using a membrane-breaking working solution. Then they were incubated for 60 min using a buffer solution containing TDTase and dUTP. The nuclei of the cells were re-stained with 4’,6-diamidino-2-phenylindole (DAPI), and the results of the cell apoptosis in myocardial tissue were observed under a fluorescence microscope.
Western Blot Analysis
Myocardial tissue from section “I/R Rat Model Preparation and EPT Treatment” was homogenized, and the total protein was obtained by centrifugation of the homogenate with RAPI lysate. The protein concentration was measured by the bicinchoninic acid (BCA) method. Then the protein samples were sequentially separated by SDS-PAGE, and the sample further underwent membrane transfer and membrane sealing treatment. The protein membranes were then incubated in an incubation solution containing the primary antibody against the corresponding proteins (including GAPDH) at 4°C overnight. Then the membranes were incubated in an incubation solution containing the secondary antibody, and the optical density of the proteins was analyzed by Image Lab software (Bio-Rad, Richmond, CA, USA).
Molecular Docking
The procedure of molecular docking was carried out according to earlier research (Muhammed & Aki-Yalcin, 2024; Yang et al., 2023). Briefly, the structure of EPT was obtained from the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP) database (
Statistical Analysis
The experimental data were analyzed statistically using SPSS 25.0 software (SPSS Inc., Chicago, IL, USA), and Tukey’s method in one-way analysis of variance (ANOVA) was used to analyze the significance, and the difference was considered to be statistically significant when p < .05.
Results
EPT Attenuated Myocardium Damage Caused by I/R in Rats
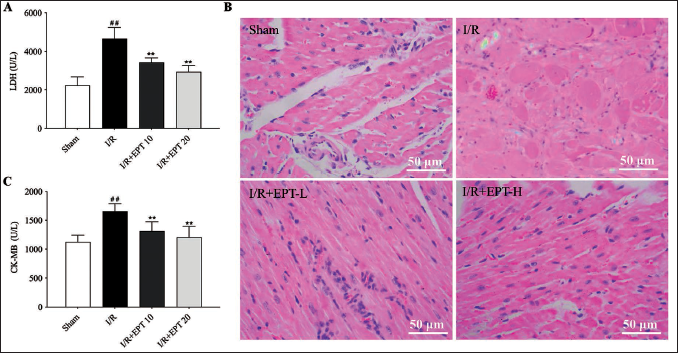
As shown in Figure 1, the levels of LDH and CK, the biomarkers of myocardial injury, were remarkably increased in the I/R group. As expected, EPT treatment could inhibit the upregulation of these two biomarkers caused by I/R (Figure 1A–B, p < .01). H&E staining showed that the myocardial tissue of rats was obviously damaged, as the myocardial cells were swollen, disorganized, and infiltrated with a large number of inflammatory cells. Whereas the myocardial tissue damage of rats in the I/R + EPT groups was obviously reduced by EPT treatment, and the effect was more obvious as the dose of EPT increased (Figure 1C). These observations indicated that EPT could attenuate myocardium damage caused by I/R in rats.
EPT Attenuated Inflammation Response Caused by I/R in Rats
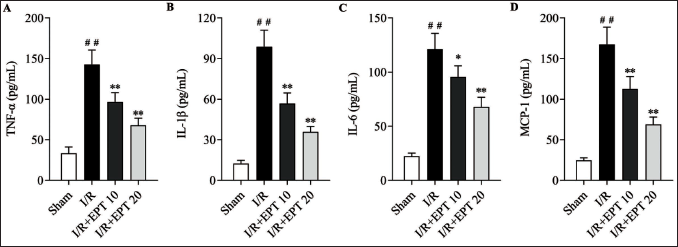
Figure 2 showed that the serum levels of inflammation factors were remarkably increased in the I/R group. As expected, the levels of TNF-α, IL-1β, IL-6, and MCP-1 were remarkably decreased in the I/R + EPT groups (Figure 2A–D, p < .01). These measurements indicated that EPT could attenuate the inflammation response caused by I/R in rats.
The Inhibition of P2X7R/NLRP3 and JAK2/STAT3/SOCS3 Pro-inflammation Pathway by EPT in the Myocardial Tissue of I/R Rats
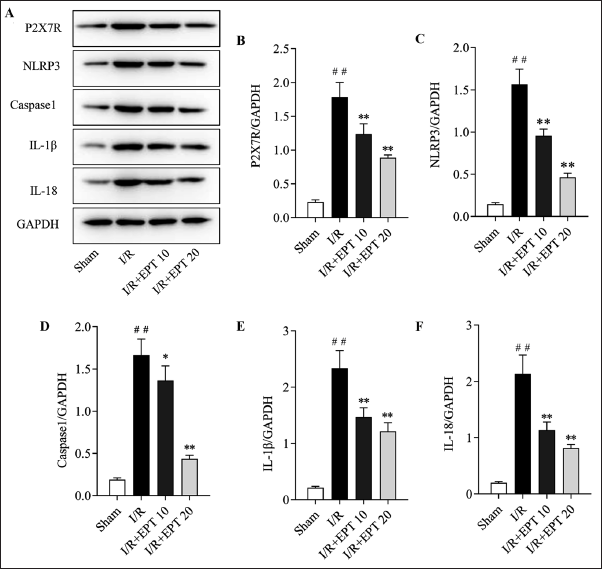
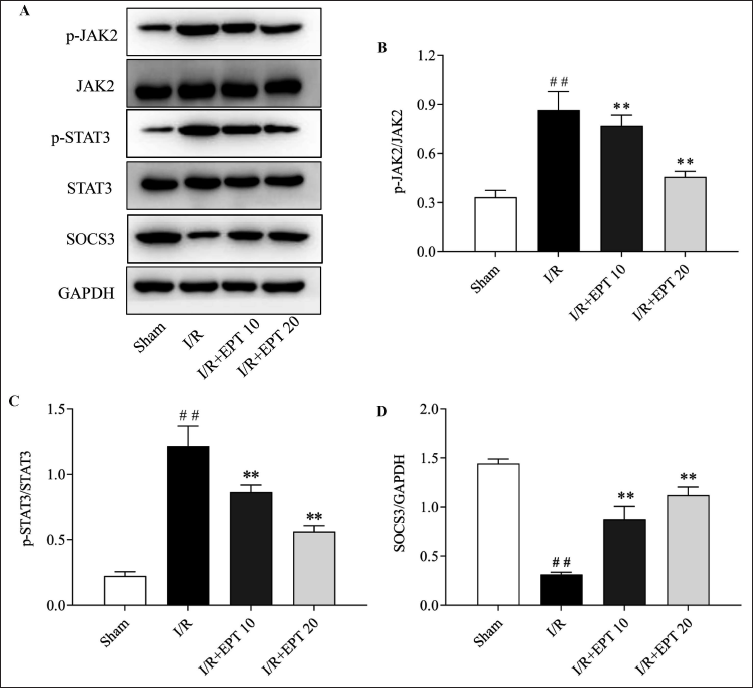
As shown in Figures 3 and 4, the two inflammation-related pathways, P2X7R/NLRP3 and JAK2/STAT3/SOCS3, were obviously activated in the I/R model group, as the key proteins in these pathways were remarkably upregulated (Figures 3 and 4, p < .01). As indicated, EPT suppressed the expression of the key proteins in the pro-inflammation pathways, as the levels of P2X7R, NLRP3, Caspase-1, or the ratio of the activated (phosphorylation-modified) JAK2 and STAT3, were significantly downregulated in the I/R + EPT groups (Figures 3B–F and 4B–D, p < .05). These measurements indicated that EPT could inhibit the P2X7R/NLRP3 and JAK2/STAT3/SOCS3 pro-inflammation pathway in the myocardial tissue of I/R rats.
EPT Reduced Cell Apoptosis in the Myocardial Tissue of I/R Rats
Figure 5 showed that the expression of apoptosis-related proteins changed significantly, as the levels of Bax and activated Caspase-3 (cleaved Caspase-3), the upregulation of which is often associated with increased cell apoptosis, were remarkably upregulated (Figure 5A–D, p < .05). As expected, the drastic changes in the levels of these apoptosis-related proteins are partially reversed in the I/R + EPT groups (Figure 5A–D, p < .05). The TUNEL assay also indicated that EPT could reduce cell apoptosis in the myocardial tissue of I/R rats. As shown in Figure 5E–F, the rate of cell apoptosis was significantly decreased in the I/R + EPT groups (p < .05).
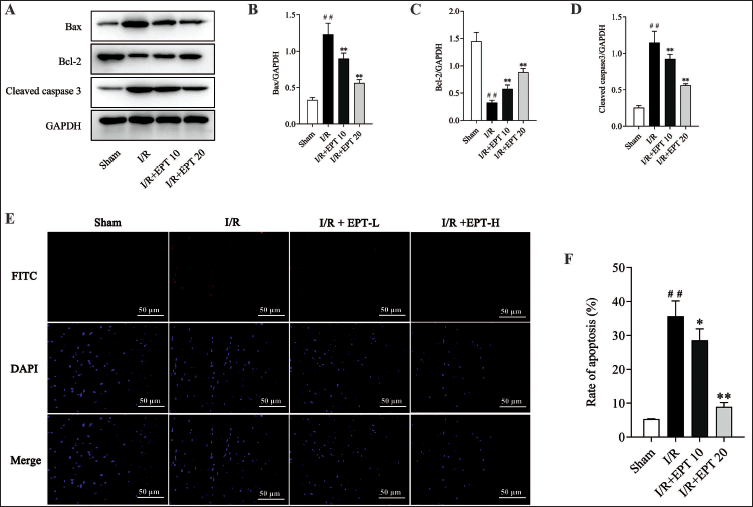
The Inhibition of PERK/eIF2α/ATF4/CHOP ER Stress Signaling Pathway by EPT in the Myocardial Tissue of I/R Rats
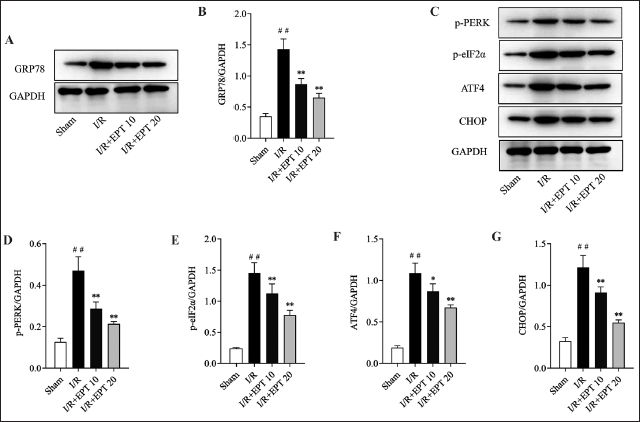
Figure 6A–B showed that the level of GRP78, the marker of ER stress, was remarkably increased in the I/R group, but such upregulation was partly reversed when treated with EPT (Figure 6A–B, p < .01). This suppressive effect is associated with the inhibition of the activated PERK/eIF2α/ATF4/CHOP ER stress pathways. As shown in Figure 6C–G, the levels of PERK, EIF2α, ATF4, and CHOP proteins were significantly decreased in the I/R + EPT groups (Figure 6C–G, p < .05). These results indicated that EPT could inhibit the PERK/eIF2α/ATF4/CHOP ER stress signaling pathway in the myocardial tissue of I/R rats.
The Molecular Docking Simulation of EPT with Key Proteins in the Pro-inflammation and ER Stress Signaling Pathway
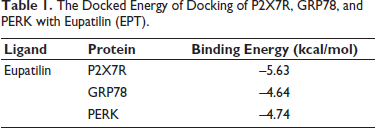
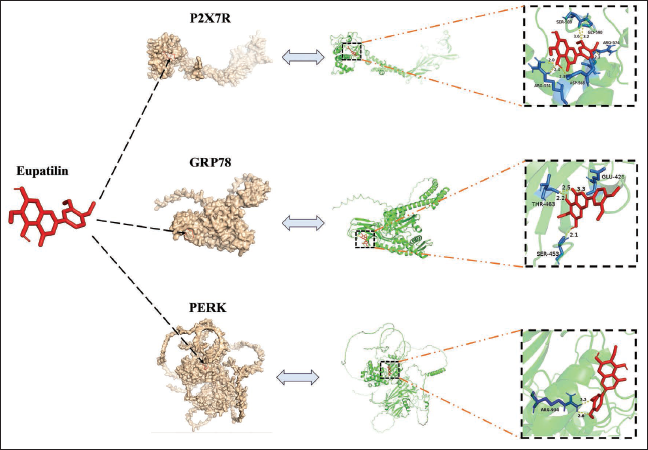
Molecular docking simulation can reflect the interaction between the small molecule ligands with macromolecules. Figure 7 showed that EPT could closely bind to P2X7R, GRP78, and PERK proteins (Figure 7), and the lowest binding energies were –5.63, –4.64, and –4.74 kcal/mol (Table 1). As shown in Figure 1, the binding energies of the 10 molecular docking models calculated for each protein were all less than –1.2 kcal/mol. According to the autodocking software, all of these bindings were relatively stable, suggesting that EPT can form stable bindings with key proteins in the P2X7R/NLRP3 pathway and PERK/eIF2α/ATF4/CHOP pathway. This may be a potential mechanism by which EPT inhibits the P2X7R/NLRP3 pro-inflammation pathway and the PERK/eIF2α/ATF4/CHOP ER stress signaling pathway.
The Docked Energy of Docking of P2X7R, GRP78, and PERK with Eupatilin (EPT).
The Molecular Docking Simulation of Eupatilin with Key Proteins in the Pro-inflammation and Endoplasmic Reticulum (ER) Stress Signaling Pathway.
Discussion
Consistent with our hypothesis, the results of the present study showed that EPT had a noteworthy protective effect against myocardial I/R injury in rats. This perspective was supported by several pieces of evidence: first, H&E staining and myocardial injury marker measurements showed that the pathological changes in myocardial tissues were significantly attenuated, and myocardial injury marker levels were obviously decreased after EPT treatment. Second, the inflammatory response, ER stress, and apoptosis rate were significantly reduced in the myocardial tissues of the EPT-treated group. In addition, EPT could inhibit the P2X7R/NLRP3 pro-inflammatory signaling pathway and the PERK/eIF2α/ATF4/CHOP pro-ER stress signaling pathway in cardiomyocytes. This protective effect of EPT against myocardial I/R injury, as clarified in the present study, is a piece of well-complementary and corroborating evidence for its visceral protective function. It has been shown that EPT has a significant damage-protective effect on both liver (Lee et al., 2016) and kidney (Xu et al., 2024). In fact, it is worth noting that such aforementioned effect of EPT is not unique to traditional herbal extracts, as icariin exerts (Meng et al., 2015), celastrol (Li et al., 2017), and ginsenosides (Wu et al., 2011) have shown some potential to counteract myocardial I/R injury. However, it should be noted that EPT is mainly derived from Artemisia Linn. leaves, which have obvious advantages of wide source, easy cultivation, and low raw material price compared to ginseng and other valuable herbs. Therefore, it may be cheaper and more readily available for subsequent applications, which is more conducive to EPT’s widespread use.
In addition to differences in medicinal value, there are some differences in the mechanisms by which EPT and other natural plant products protect against myocardial I/R injury. For example, although botulin (Yu et al., 2021), luteolin (Zhang et al., 2017), and ginsenosides (Qi et al., 2022) can inhibit inflammatory responses in the myocardium by targeting NLRP3 inflammasome activity, there are some subtle differences in their specific mechanisms. Such differences may be due to the different structures of the small molecules, which result in variations in the types of target proteins they bind to in the inflammatory pathway (An et al., 2004). In addition, there are also discrepancies in the binding ability of different small molecules to the same protein (Gilson & Zhou, 2007). In this study, we simulated by molecular docking that EPT could stabilize binding to P2X7R proteins, which may be the direct cause of its inhibition of the P2X7R/NLRP3 pro-inflammatory pathway. In contrast, ginsenosides and luteolin may bind to other key proteins in the inflammatory pathway, but the relevant information has yet to be verified by subsequent experiments such as molecular docking simulations.
In addition, molecular docking simulations in this study also showed that EPT could stably bind to PERK, a key protein in the PERK/eIF2α/ATF4/CHOP pathway that mediates ER stress (Liu et al., 2013), as well as to the ER stress marker protein GRP78 (Rao et al., 2002). These may be the potential mechanism by which EPT could modulate ER stress and thus inhibit apoptosis of cardiomyocytes. Again, this theory has yet to be verified with the assistance of cryo-electron microscopy (Murata & Wolf, 2018), which could allow researchers to observe the actual molecular structure of EPT-target proteins complex. In addition, when exploring the specific mechanisms of EPT’s regulation of the above pathways, specific inhibitors of the pathway (Das et al., 2021) could be used to verify whether the pathway plays a key role in the protective effects of EPT.
In addition to the lack of practical validation of the molecular docking simulation results, the present study also did not conduct long-term follow-up measurements of myocardial function and organ damage (Suwaidi et al., 2000) in the EPT-treated group, which is an important reference for evaluating the effectiveness of a drug in the clinic, and therefore, the relevant data should be supplemented in the subsequent study. In addition, given that the respiratory system is closely related to cardiovascular function (Koepchen, 1983), it should also be observed in subsequent studies whether EPT improves lung function while alleviating myocardial I/R injury and explore whether this potential improvement effect is related to the modulation of the respiratory chain by EPT.
Conclusion
In summary, this study demonstrated that EPT could attenuate myocardial I/R injury in rats and that this protective mechanism may be related to the binding of EPT to P2X7R protein, which, in turn, inhibits the P2X7R/NLRP3 pro-inflammatory pathway, as well as the binding of EPT to PERK protein, which, in turn, inhibits the PERK/eIF2α/ATF4/CHOP pro-ER stress signaling pathway. These findings provide some valuable references for the clinical application of EPT for coronary heart disease protection and attenuation of myocardial IR injury in the future.
Footnotes
Abbreviations
ANOVA: Analysis of variance; ATF4: Activating transcription factor 4; BCA: Bicinchoninic acid; CHOP: C/EBP homologous protein; CK: Creatine kinase; DAPI: 4′,6-Diamidino-2-phenylindole; ECG: Electrocardiogram; ELISA: Enzyme-linked immunosorbent assay; EPT: Eupatilin; ER: Endoplasmic reticulum; H&E: Hematoxylin and eosin; I/R: Ischemia/Reperfusion; JAK2: Janus kinase 2; LAD: Left anterior descending coronary artery; LDH: Lactate dehydrogenase; NLRP3: NOD-, LRR- and pyrin domain-containing protein 3; P2X7R: Purinergic P2x7 receptor; PERK/eIF2α: Pancreatic endoplasmic reticulum eIF2alpha kinase; RCSB: The Research Collaboratory for Structural Bioinformatics Protein Data Bank; SOCS3: Suppressor of cytokine signaling 3; STAT3: Signal transducer and activator of transcription 3; TCMSP: The Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform; TUNEL: Terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling.
Data Availability Statement
Data will be made available upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal experiments were strictly adhered to international regulations on the welfare of experimental animals and carried out with the approval of the Animal Care and Use Committee of Yanbian University Hospital.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (82060052).
